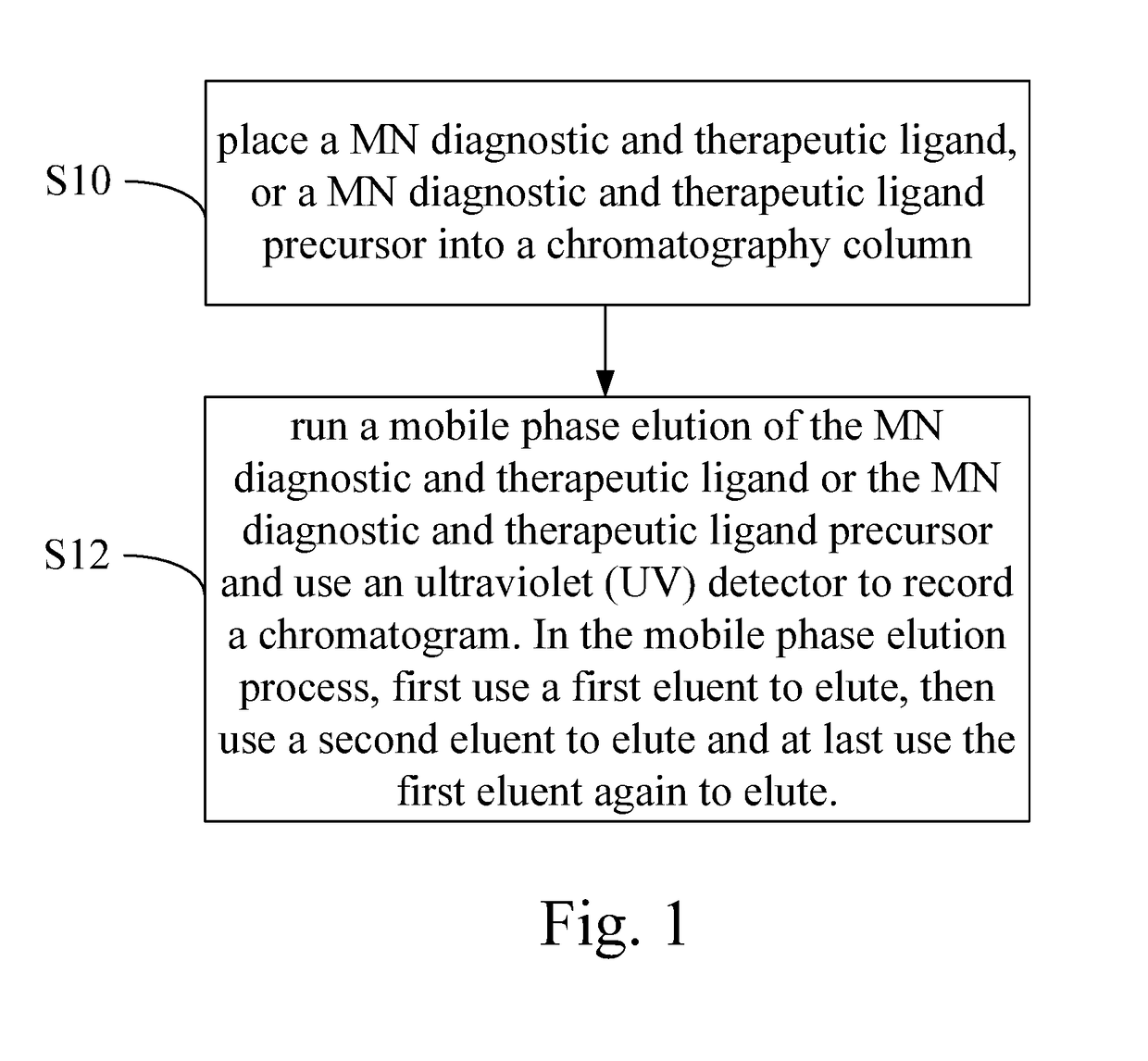
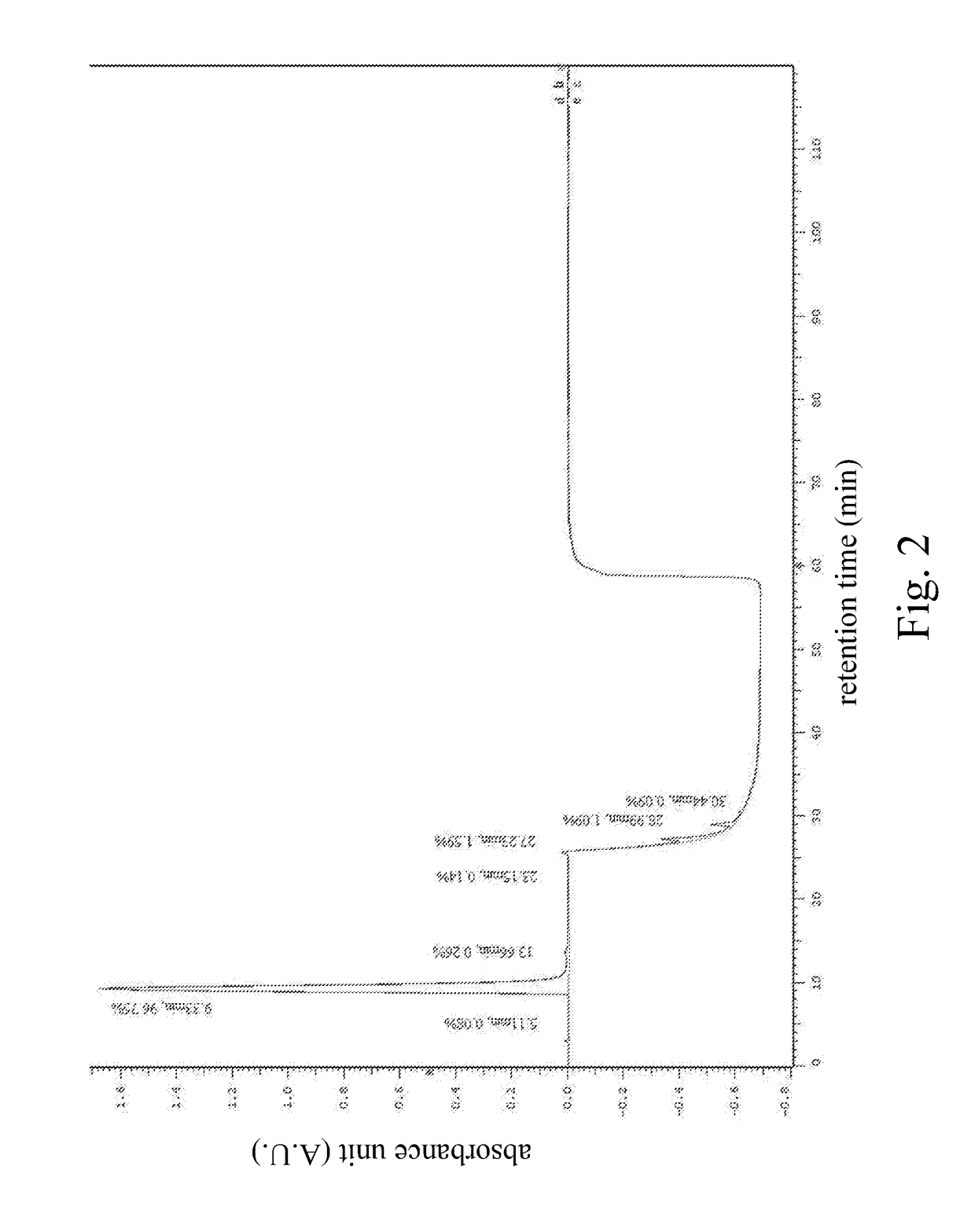
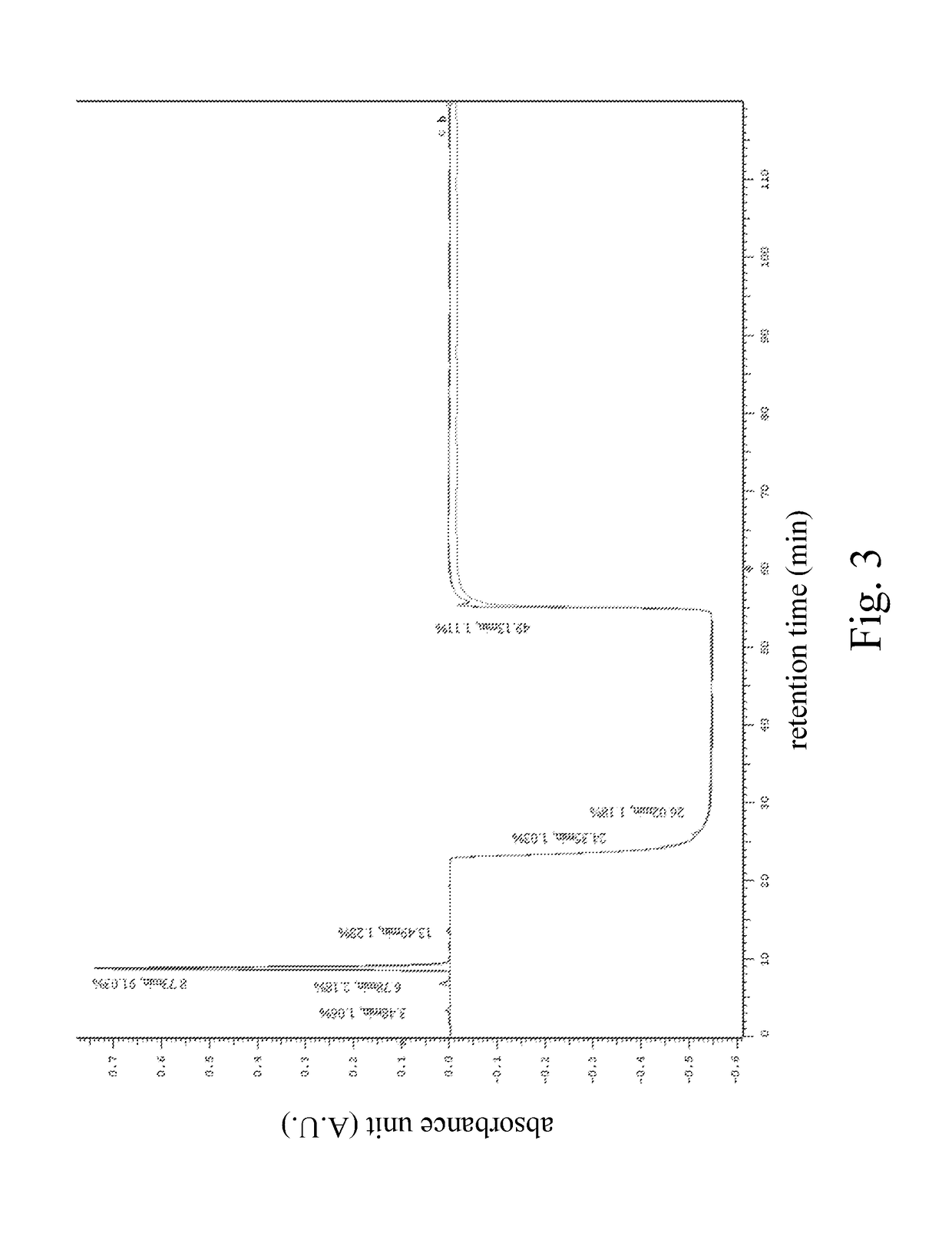
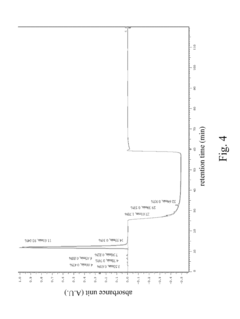
HPLC Operational Compatibility: Industry Best Practices
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Technology Evolution and Objectives
High-Performance Liquid Chromatography (HPLC) has evolved significantly since its inception in the late 1960s, transforming from rudimentary separation techniques to sophisticated analytical instrumentation essential across multiple industries. The evolution of HPLC technology represents one of the most impactful developments in analytical chemistry, with continuous improvements in sensitivity, resolution, and operational efficiency.
The initial development phase of HPLC (1970s-1980s) focused on fundamental improvements in column technology and pumping systems. During this period, silica-based stationary phases became standardized, and the introduction of microparticulate packing materials (5-10 μm) significantly enhanced separation efficiency. Mechanical pumping systems evolved to provide more stable flow rates, which was critical for reproducible analyses.
The 1990s marked a significant advancement with the introduction of smaller particle sizes (3-5 μm) and the development of more robust HPLC systems capable of operating at higher pressures. This period also saw the integration of computerized data systems, allowing for automated method development and more sophisticated data analysis capabilities.
The early 2000s witnessed the emergence of Ultra-High Performance Liquid Chromatography (UHPLC), utilizing sub-2 μm particles and systems capable of operating at pressures exceeding 15,000 psi. This technological leap dramatically improved resolution, sensitivity, and analysis speed, reducing typical run times from 30-60 minutes to under 5 minutes for many applications.
Recent technological trends include the development of core-shell particle technology, which offers efficiency comparable to sub-2 μm fully porous particles but with lower backpressure requirements. Additionally, advances in column chemistry have expanded the range of compounds that can be effectively separated, particularly in challenging areas such as chiral separations and the analysis of biomolecules.
The primary objectives of modern HPLC operational compatibility focus on several key areas: enhancing system robustness to minimize downtime, improving method transferability across different instruments and laboratories, reducing environmental impact through decreased solvent consumption, and integrating with other analytical techniques for comprehensive sample characterization.
Future development trajectories aim to address persistent challenges in HPLC technology, including further miniaturization for reduced sample and solvent requirements, enhanced automation capabilities for unattended operation, improved detection sensitivity for trace analysis, and the development of "green" chromatography approaches utilizing environmentally friendly mobile phases and reduced energy consumption.
The ultimate goal of HPLC technological evolution is to establish standardized operational practices that ensure consistent, reliable analytical results while maximizing instrument uptime and minimizing the expertise required for routine operation, thereby supporting critical applications across pharmaceutical, environmental, food safety, and clinical diagnostic sectors.
The initial development phase of HPLC (1970s-1980s) focused on fundamental improvements in column technology and pumping systems. During this period, silica-based stationary phases became standardized, and the introduction of microparticulate packing materials (5-10 μm) significantly enhanced separation efficiency. Mechanical pumping systems evolved to provide more stable flow rates, which was critical for reproducible analyses.
The 1990s marked a significant advancement with the introduction of smaller particle sizes (3-5 μm) and the development of more robust HPLC systems capable of operating at higher pressures. This period also saw the integration of computerized data systems, allowing for automated method development and more sophisticated data analysis capabilities.
The early 2000s witnessed the emergence of Ultra-High Performance Liquid Chromatography (UHPLC), utilizing sub-2 μm particles and systems capable of operating at pressures exceeding 15,000 psi. This technological leap dramatically improved resolution, sensitivity, and analysis speed, reducing typical run times from 30-60 minutes to under 5 minutes for many applications.
Recent technological trends include the development of core-shell particle technology, which offers efficiency comparable to sub-2 μm fully porous particles but with lower backpressure requirements. Additionally, advances in column chemistry have expanded the range of compounds that can be effectively separated, particularly in challenging areas such as chiral separations and the analysis of biomolecules.
The primary objectives of modern HPLC operational compatibility focus on several key areas: enhancing system robustness to minimize downtime, improving method transferability across different instruments and laboratories, reducing environmental impact through decreased solvent consumption, and integrating with other analytical techniques for comprehensive sample characterization.
Future development trajectories aim to address persistent challenges in HPLC technology, including further miniaturization for reduced sample and solvent requirements, enhanced automation capabilities for unattended operation, improved detection sensitivity for trace analysis, and the development of "green" chromatography approaches utilizing environmentally friendly mobile phases and reduced energy consumption.
The ultimate goal of HPLC technological evolution is to establish standardized operational practices that ensure consistent, reliable analytical results while maximizing instrument uptime and minimizing the expertise required for routine operation, thereby supporting critical applications across pharmaceutical, environmental, food safety, and clinical diagnostic sectors.
Market Demand Analysis for HPLC Systems
The global High-Performance Liquid Chromatography (HPLC) market continues to demonstrate robust growth, with current valuations exceeding $4 billion and projected to reach $6 billion by 2027, representing a compound annual growth rate of approximately 5.2%. This growth is primarily driven by increasing applications across pharmaceutical, biotechnology, food safety, environmental monitoring, and clinical diagnostics sectors.
Pharmaceutical and biotechnology industries remain the dominant consumers of HPLC systems, accounting for nearly 60% of the total market share. These sectors require highly reliable and compatible HPLC systems for drug development, quality control, and regulatory compliance. The stringent regulatory environment, particularly FDA and EMA requirements for analytical method validation, has significantly increased demand for HPLC systems with enhanced operational compatibility and standardized protocols.
Market research indicates a growing preference for integrated HPLC systems that offer seamless compatibility with various columns, detectors, and software platforms. End-users increasingly demand systems that can easily integrate into existing laboratory workflows while maintaining compliance with industry standards. This trend is reflected in the 15% annual increase in sales of modular HPLC systems that offer enhanced interoperability.
The academic and research segment represents another significant market, with universities and research institutions accounting for approximately 20% of HPLC system purchases. These institutions typically seek cost-effective solutions with flexibility to accommodate diverse research applications, creating demand for systems with broad operational compatibility across different experimental conditions.
Geographically, North America leads the HPLC market with 40% share, followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is experiencing the fastest growth rate at 7.8% annually, driven by expanding pharmaceutical manufacturing, contract research organizations, and increasing regulatory requirements in countries like China and India.
Customer surveys reveal that operational compatibility ranks among the top three purchasing criteria for HPLC systems, alongside analytical performance and cost-effectiveness. Specifically, 78% of laboratory managers cite compatibility with existing infrastructure as "very important" or "critical" in their procurement decisions. This has prompted manufacturers to focus on developing systems with broader compatibility features and standardized interfaces.
The market also shows increasing demand for HPLC systems with enhanced data integrity features, automated compliance documentation, and compatibility with cloud-based laboratory information management systems. This trend aligns with the broader industry movement toward digital transformation and reflects the growing importance of data management in analytical workflows.
Pharmaceutical and biotechnology industries remain the dominant consumers of HPLC systems, accounting for nearly 60% of the total market share. These sectors require highly reliable and compatible HPLC systems for drug development, quality control, and regulatory compliance. The stringent regulatory environment, particularly FDA and EMA requirements for analytical method validation, has significantly increased demand for HPLC systems with enhanced operational compatibility and standardized protocols.
Market research indicates a growing preference for integrated HPLC systems that offer seamless compatibility with various columns, detectors, and software platforms. End-users increasingly demand systems that can easily integrate into existing laboratory workflows while maintaining compliance with industry standards. This trend is reflected in the 15% annual increase in sales of modular HPLC systems that offer enhanced interoperability.
The academic and research segment represents another significant market, with universities and research institutions accounting for approximately 20% of HPLC system purchases. These institutions typically seek cost-effective solutions with flexibility to accommodate diverse research applications, creating demand for systems with broad operational compatibility across different experimental conditions.
Geographically, North America leads the HPLC market with 40% share, followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is experiencing the fastest growth rate at 7.8% annually, driven by expanding pharmaceutical manufacturing, contract research organizations, and increasing regulatory requirements in countries like China and India.
Customer surveys reveal that operational compatibility ranks among the top three purchasing criteria for HPLC systems, alongside analytical performance and cost-effectiveness. Specifically, 78% of laboratory managers cite compatibility with existing infrastructure as "very important" or "critical" in their procurement decisions. This has prompted manufacturers to focus on developing systems with broader compatibility features and standardized interfaces.
The market also shows increasing demand for HPLC systems with enhanced data integrity features, automated compliance documentation, and compatibility with cloud-based laboratory information management systems. This trend aligns with the broader industry movement toward digital transformation and reflects the growing importance of data management in analytical workflows.
Current HPLC Compatibility Challenges
High-performance liquid chromatography (HPLC) systems face several significant operational compatibility challenges in today's laboratory environments. The integration of HPLC equipment from different manufacturers remains problematic, with inconsistent communication protocols and proprietary interfaces limiting seamless operation. This fragmentation creates workflow inefficiencies and increases operational costs as laboratories must maintain multiple software platforms and training programs.
Column compatibility presents another major challenge, as variations in connection designs, pressure ratings, and chemical compatibilities across manufacturers necessitate careful matching of columns with specific instruments. The lack of standardization in column dimensions and connection types frequently leads to leakage, system damage, and compromised analytical results when mismatched components are used.
Mobile phase compatibility issues continue to plague HPLC operations, particularly when transitioning between different solvent systems. Buffer precipitation, immiscibility problems, and unexpected chemical interactions can damage expensive equipment and compromise analytical integrity. These challenges are exacerbated by the increasing complexity of modern sample matrices and the demand for multi-residue analytical methods.
Data format incompatibility represents a growing concern as laboratories implement data management systems. The proliferation of proprietary data formats hampers data transfer between different platforms, complicating multi-instrument method validation and regulatory compliance. This challenge is particularly acute in regulated environments where data integrity and traceability are paramount requirements.
Automation compatibility limitations restrict the full potential of laboratory efficiency gains. Many HPLC systems struggle to integrate effectively with robotic sample preparation systems, laboratory information management systems (LIMS), and enterprise resource planning (ERP) software. These integration gaps create manual intervention points that introduce error risks and reduce throughput.
Detector compatibility issues arise as laboratories incorporate advanced detection technologies. Signal processing differences, timing synchronization problems, and data scaling inconsistencies between detectors and chromatography data systems lead to interpretation challenges and method transfer difficulties across different laboratory sites.
Maintenance compatibility challenges emerge as organizations attempt to standardize service protocols across diverse instrument fleets. Variations in maintenance requirements, consumable specifications, and troubleshooting procedures increase the complexity of laboratory operations and technical training needs, ultimately impacting instrument uptime and analytical productivity.
Column compatibility presents another major challenge, as variations in connection designs, pressure ratings, and chemical compatibilities across manufacturers necessitate careful matching of columns with specific instruments. The lack of standardization in column dimensions and connection types frequently leads to leakage, system damage, and compromised analytical results when mismatched components are used.
Mobile phase compatibility issues continue to plague HPLC operations, particularly when transitioning between different solvent systems. Buffer precipitation, immiscibility problems, and unexpected chemical interactions can damage expensive equipment and compromise analytical integrity. These challenges are exacerbated by the increasing complexity of modern sample matrices and the demand for multi-residue analytical methods.
Data format incompatibility represents a growing concern as laboratories implement data management systems. The proliferation of proprietary data formats hampers data transfer between different platforms, complicating multi-instrument method validation and regulatory compliance. This challenge is particularly acute in regulated environments where data integrity and traceability are paramount requirements.
Automation compatibility limitations restrict the full potential of laboratory efficiency gains. Many HPLC systems struggle to integrate effectively with robotic sample preparation systems, laboratory information management systems (LIMS), and enterprise resource planning (ERP) software. These integration gaps create manual intervention points that introduce error risks and reduce throughput.
Detector compatibility issues arise as laboratories incorporate advanced detection technologies. Signal processing differences, timing synchronization problems, and data scaling inconsistencies between detectors and chromatography data systems lead to interpretation challenges and method transfer difficulties across different laboratory sites.
Maintenance compatibility challenges emerge as organizations attempt to standardize service protocols across diverse instrument fleets. Variations in maintenance requirements, consumable specifications, and troubleshooting procedures increase the complexity of laboratory operations and technical training needs, ultimately impacting instrument uptime and analytical productivity.
Industry Standard HPLC Compatibility Solutions
01 Mobile phase compatibility and optimization
The selection and optimization of mobile phases is critical for HPLC operational compatibility. This includes considerations for solvent miscibility, pH stability, buffer compatibility, and gradient elution parameters. Proper mobile phase selection ensures optimal separation efficiency, peak resolution, and system stability during chromatographic analysis. Factors such as solvent polarity, ionic strength, and additives must be carefully balanced to achieve reproducible results while maintaining instrument performance.- Mobile phase compatibility and optimization: The composition and properties of the mobile phase are critical for HPLC operational compatibility. Optimization involves selecting appropriate solvents, adjusting pH levels, and incorporating additives to enhance separation efficiency. Compatibility between the mobile phase and sample components must be ensured to prevent precipitation or degradation. Gradient elution techniques can be employed to improve separation of complex mixtures while maintaining system compatibility.
- Column selection and maintenance for compatibility: Selecting the appropriate HPLC column based on stationary phase chemistry is essential for operational compatibility with specific analytes. Regular maintenance procedures, including proper conditioning, equilibration, and cleaning protocols, extend column life and ensure consistent performance. Column temperature control can improve separation reproducibility and prevent compatibility issues. Specialized columns may be required for particular applications to maintain compatibility with challenging sample matrices.
- Sample preparation techniques for HPLC compatibility: Effective sample preparation is crucial for HPLC operational compatibility, involving filtration, extraction, and clean-up procedures to remove particulates and interfering compounds. Sample solubility in the mobile phase must be ensured to prevent precipitation and system clogging. Derivatization techniques may be employed to enhance detection sensitivity or improve chromatographic behavior of certain analytes. Proper sample storage conditions help maintain sample integrity and compatibility with the HPLC system.
- Detection system compatibility and optimization: Selection of appropriate detection systems (UV-Vis, fluorescence, mass spectrometry) based on analyte properties ensures optimal sensitivity and selectivity. Detector settings must be compatible with mobile phase composition and flow rates to minimize baseline noise and maximize signal response. Interface compatibility between HPLC and detection systems, particularly in hyphenated techniques like LC-MS, requires careful optimization of parameters such as ionization conditions and transfer line temperatures. Signal processing and data acquisition systems must be compatible with the detection method to ensure accurate quantification.
- Instrument and system integration compatibility: HPLC system components (pumps, injectors, detectors) must be compatible with each other and with the analytical method requirements. Software integration enables seamless operation and data management across different system components. Automation compatibility allows for high-throughput analysis while maintaining consistent performance. Temperature control systems throughout the HPLC setup ensure stable operating conditions and prevent compatibility issues related to temperature fluctuations. Pressure compatibility across system components is essential to prevent leaks and ensure safe operation at required pressures.
02 Column selection and maintenance for operational efficiency
Selecting appropriate HPLC columns and implementing proper maintenance protocols significantly impacts operational compatibility. This includes considerations for stationary phase chemistry, particle size, column dimensions, and temperature stability. Regular column maintenance procedures such as washing, regeneration, and storage protocols help extend column life and maintain separation efficiency. Compatibility between sample matrices, mobile phases, and column chemistry must be evaluated to prevent damage and ensure consistent chromatographic performance.Expand Specific Solutions03 Sample preparation techniques for HPLC compatibility
Effective sample preparation is essential for HPLC operational compatibility. This includes filtration, extraction, dilution, and derivatization methods that ensure samples are compatible with both the column chemistry and detection system. Proper sample preparation prevents system contamination, column clogging, and detector interference while improving reproducibility and accuracy. Techniques must be optimized based on sample matrix complexity, target analyte properties, and the specific requirements of the chromatographic method.Expand Specific Solutions04 Detection system compatibility and optimization
Optimizing detector compatibility is crucial for successful HPLC operations. This includes considerations for detector type selection (UV-Vis, fluorescence, mass spectrometry, etc.), signal-to-noise ratio optimization, and interface compatibility between chromatographic and detection systems. Parameters such as wavelength selection, flow cell design, and data acquisition rates must be optimized based on analyte properties and separation conditions. Proper detector maintenance and calibration ensure consistent sensitivity and linearity across analytical runs.Expand Specific Solutions05 System integration and automation for enhanced compatibility
Integration of HPLC systems with automation technologies improves operational compatibility and efficiency. This includes autosampler compatibility, software integration, method transfer protocols, and system suitability testing. Automated systems can monitor critical parameters, perform self-diagnostics, and adjust conditions to maintain optimal performance. Standardized interfaces and communication protocols enable seamless integration between components from different manufacturers, enhancing overall system reliability and reducing operator intervention requirements.Expand Specific Solutions
Major HPLC Manufacturers and Competitors
The HPLC operational compatibility market is currently in a growth phase, with increasing demand for standardized practices across pharmaceutical, biotechnology, and chemical industries. The global HPLC market is estimated to exceed $5 billion, driven by rising analytical testing requirements and quality control standards. Technologically, the field shows varying maturity levels with companies like Agilent Technologies and Waters Corporation leading innovation in instrument compatibility and method transferability. Microsoft Technology Licensing and Hewlett Packard Enterprise are advancing data management solutions, while pharmaceutical players like Vertex Pharmaceuticals are developing application-specific protocols. Emerging players such as TEAC Corp and Augury Systems are introducing predictive maintenance technologies to enhance operational reliability, indicating a trend toward smart, integrated HPLC systems with improved cross-platform compatibility.
Hewlett Packard Enterprise Development LP
Technical Solution: Hewlett Packard Enterprise (HPE) has leveraged its legacy in analytical instrumentation (from its historical connection to Agilent, which was spun off from HP) to develop advanced IT infrastructure solutions specifically for HPLC laboratory environments. Their approach focuses on data management and computational aspects of HPLC operational compatibility. HPE's Laboratory Enterprise solution integrates chromatography data systems with enterprise IT infrastructure through specialized middleware that enables seamless data transfer between analytical instruments and laboratory information management systems (LIMS). Their platform incorporates machine learning algorithms that analyze operational patterns across multiple HPLC systems to identify compatibility issues before they impact analytical results. HPE has developed specialized edge computing solutions for HPLC laboratories that process chromatographic data at the instrument level, reducing network bandwidth requirements while maintaining centralized data management. Their cloud-based chromatography data storage solutions implement industry-standard data formats and APIs that ensure long-term accessibility and compatibility with evolving software platforms.
Strengths: Enterprise-level data management solutions; strong IT infrastructure integration capabilities; advanced predictive analytics for system compatibility. Weaknesses: Less focus on hardware-level compatibility issues; requires significant IT expertise to implement effectively; solutions may be oversized for smaller laboratory operations.
Agilent Technologies, Inc.
Technical Solution: Agilent Technologies has developed comprehensive HPLC operational compatibility solutions centered around their InfinityLab LC series. Their approach focuses on instrument-to-instrument and method transfer compatibility across different laboratory environments. The InfinityLab system incorporates intelligent system emulation technology (ISET) that allows precise replication of chromatographic conditions between different HPLC instruments, regardless of manufacturer or model. This technology addresses one of the most significant challenges in analytical laboratories - method transfer between instruments with different delay volumes, mixing behaviors, and detector characteristics. Agilent's OpenLAB CDS software platform further enhances operational compatibility by providing a unified control interface for multiple instrument types and brands, enabling seamless integration of legacy equipment with modern systems. Their compatibility framework also includes standardized column technology with intelligent identification chips that automatically configure optimal operating parameters, reducing human error and improving reproducibility across different laboratory settings.
Strengths: Industry-leading method transfer technology allowing cross-platform compatibility; comprehensive software integration capabilities; intelligent consumables with auto-identification features. Weaknesses: Premium pricing structure may be prohibitive for smaller laboratories; proprietary aspects of their ecosystem can create vendor lock-in; complex implementation may require specialized training.
Key Patents and Innovations in HPLC Compatibility
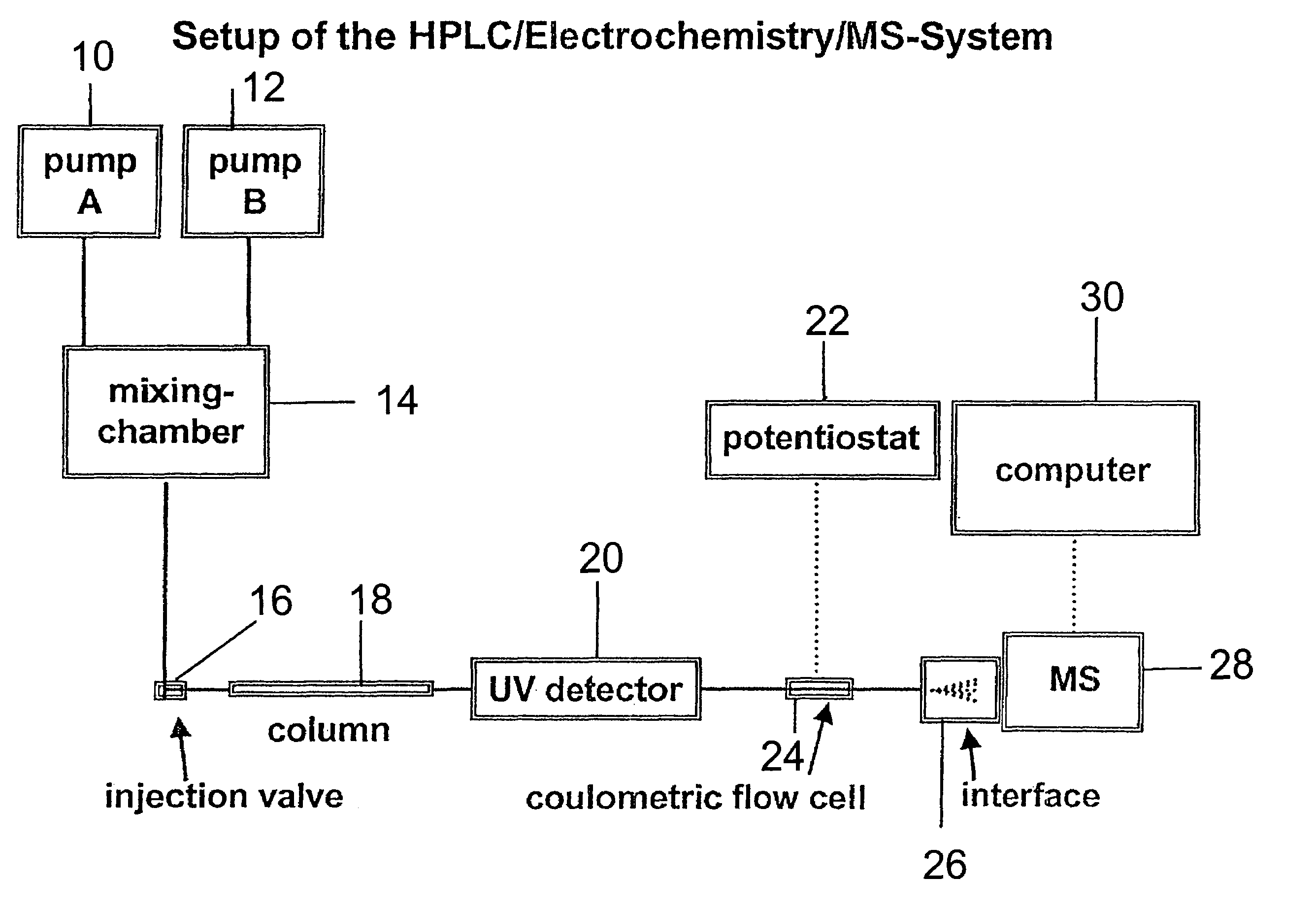
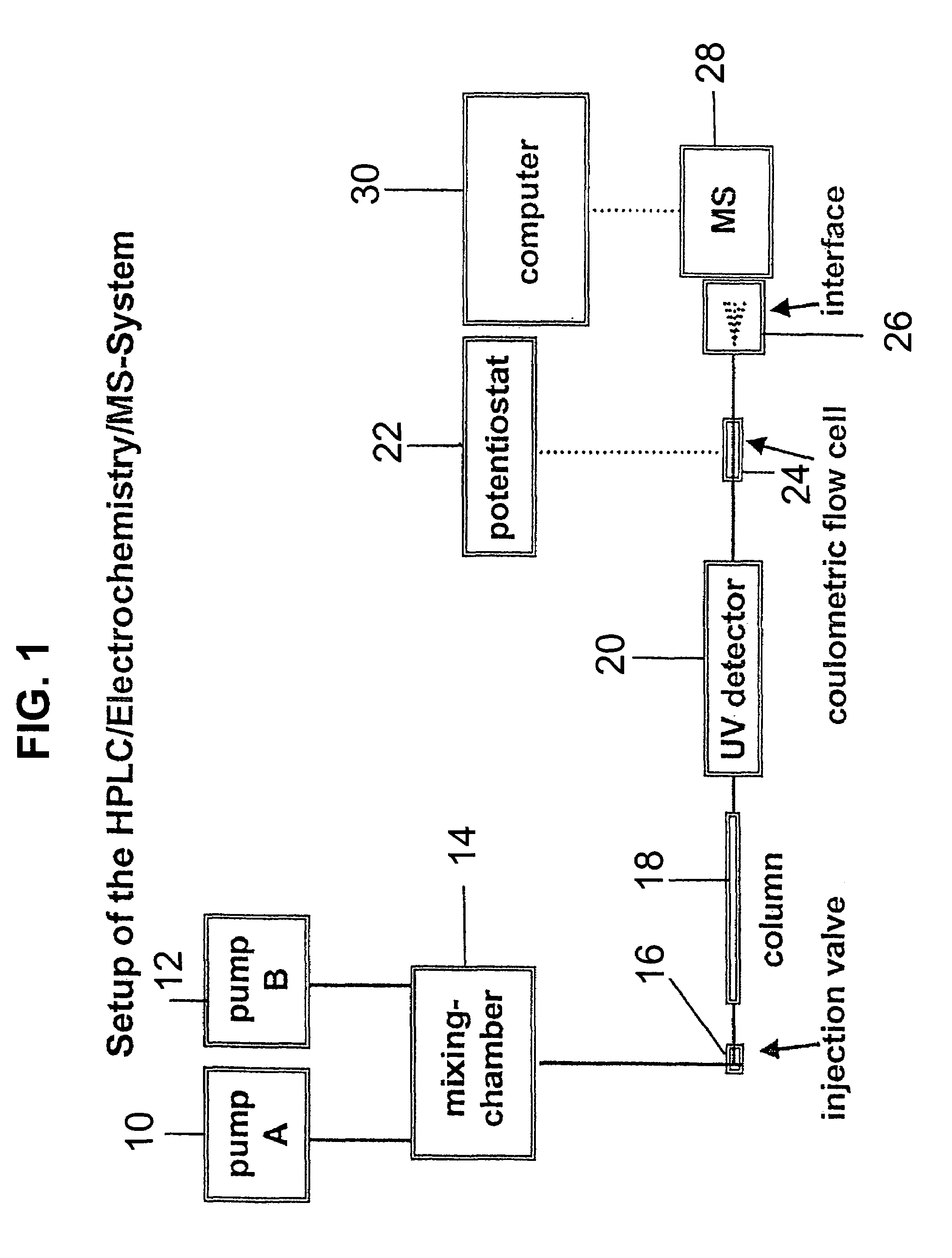
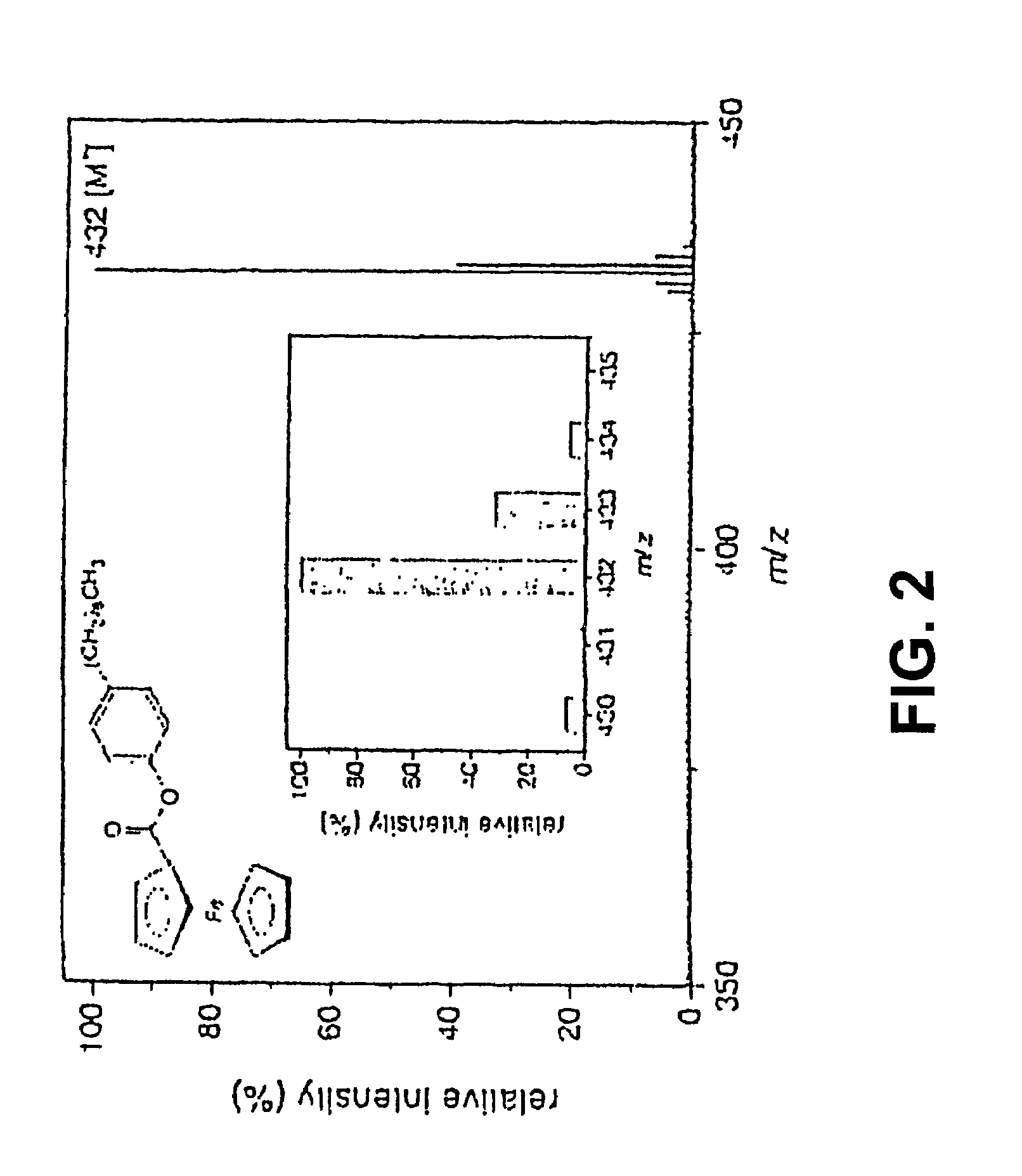
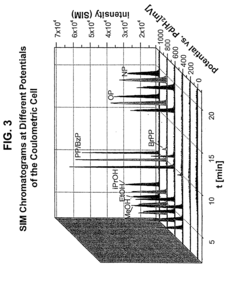
Coupling electrochemistry to mass spectrometry and high performance liquid chromatography
PatentInactiveUS7028537B2
Innovation
- A new HPLC-electrochemistry-MS technique is developed, where a coulometric three-electrode electrochemical cell is inserted between the HPLC column and mass spectrometer, enabling post-column electrochemical oxidation or reduction of analytes, forming charged or strongly polar products compatible with ESI or APCI mass spectrometry, thereby improving ionization efficiency.
High performance liquid chromatography method for analysis of MN diagnostic and therapeutic ligand and precursor
PatentInactiveUS20180059072A1
Innovation
- A HPLC method utilizing a high ratio of acetonitrile in the mobile phase to wash out low-polarity impurities and employing gradient elution with a UV detector at 210 nm to improve detection accuracy, ensuring accurate analysis and reducing residual impurities in the column.
Regulatory Compliance in HPLC Operations
Regulatory compliance represents a critical dimension of HPLC operations across pharmaceutical, food safety, environmental monitoring, and clinical diagnostics industries. Organizations must navigate a complex landscape of regulations established by authorities such as the FDA, EMA, ICH, and ISO to ensure the validity and reliability of analytical results.
The FDA's 21 CFR Part 11 establishes requirements for electronic records and signatures, mandating comprehensive audit trails, system validation, and data integrity controls for HPLC systems. Similarly, the EMA's Annex 11 provides guidance on computerized systems in GMP environments, emphasizing risk management approaches to data handling and system validation.
ICH Q2(R1) guidelines specifically address validation of analytical procedures, requiring HPLC methods to demonstrate specificity, linearity, accuracy, precision, and robustness. These parameters must be thoroughly documented through qualification protocols that verify system suitability and performance.
Data integrity compliance has emerged as a paramount concern, with regulatory bodies increasingly scrutinizing laboratory practices. The ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available) serve as the foundation for proper data management in HPLC operations.
Implementation of effective compliance strategies requires a multi-faceted approach. Organizations must establish comprehensive standard operating procedures (SOPs) that detail all aspects of HPLC operation, from sample preparation to data analysis and reporting. These SOPs should incorporate regular system suitability testing, calibration verification, and preventive maintenance schedules.
Training programs represent another essential component of regulatory compliance. Personnel must receive thorough instruction on both technical operation of HPLC systems and regulatory requirements governing their use. Documentation of training completion and competency assessment is mandatory for regulatory inspections.
Audit readiness demands meticulous record-keeping practices. Laboratories must maintain complete documentation of instrument qualification (IQ/OQ/PQ), method validation, system suitability tests, maintenance records, and calibration certificates. Electronic data management systems must incorporate appropriate access controls, audit trails, and data backup procedures to prevent unauthorized modifications.
The evolving regulatory landscape necessitates continuous monitoring of guideline updates and emerging compliance expectations. Organizations that implement robust quality management systems, conduct regular internal audits, and foster a culture of compliance will be best positioned to meet regulatory requirements while maintaining operational efficiency in their HPLC operations.
The FDA's 21 CFR Part 11 establishes requirements for electronic records and signatures, mandating comprehensive audit trails, system validation, and data integrity controls for HPLC systems. Similarly, the EMA's Annex 11 provides guidance on computerized systems in GMP environments, emphasizing risk management approaches to data handling and system validation.
ICH Q2(R1) guidelines specifically address validation of analytical procedures, requiring HPLC methods to demonstrate specificity, linearity, accuracy, precision, and robustness. These parameters must be thoroughly documented through qualification protocols that verify system suitability and performance.
Data integrity compliance has emerged as a paramount concern, with regulatory bodies increasingly scrutinizing laboratory practices. The ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available) serve as the foundation for proper data management in HPLC operations.
Implementation of effective compliance strategies requires a multi-faceted approach. Organizations must establish comprehensive standard operating procedures (SOPs) that detail all aspects of HPLC operation, from sample preparation to data analysis and reporting. These SOPs should incorporate regular system suitability testing, calibration verification, and preventive maintenance schedules.
Training programs represent another essential component of regulatory compliance. Personnel must receive thorough instruction on both technical operation of HPLC systems and regulatory requirements governing their use. Documentation of training completion and competency assessment is mandatory for regulatory inspections.
Audit readiness demands meticulous record-keeping practices. Laboratories must maintain complete documentation of instrument qualification (IQ/OQ/PQ), method validation, system suitability tests, maintenance records, and calibration certificates. Electronic data management systems must incorporate appropriate access controls, audit trails, and data backup procedures to prevent unauthorized modifications.
The evolving regulatory landscape necessitates continuous monitoring of guideline updates and emerging compliance expectations. Organizations that implement robust quality management systems, conduct regular internal audits, and foster a culture of compliance will be best positioned to meet regulatory requirements while maintaining operational efficiency in their HPLC operations.
Method Transfer Strategies Between HPLC Systems
Method transfer between HPLC systems represents a critical process in analytical laboratories, particularly when transitioning methods between different instruments, laboratories, or during collaborative multi-site studies. Successful transfer strategies must account for potential variations in instrument design, detector sensitivity, and column technologies that can significantly impact chromatographic performance.
The foundation of effective method transfer begins with comprehensive documentation of the source method parameters, including detailed specifications of column dimensions, particle size, stationary phase chemistry, mobile phase composition, gradient profiles, and detection parameters. This documentation serves as the baseline reference for the transfer process and facilitates troubleshooting if discrepancies arise during implementation.
Risk assessment constitutes an essential preliminary step, where critical method attributes are identified and potential variations between systems are evaluated. Parameters such as dwell volume differences, detector response factors, and pressure limitations require particular attention as they frequently contribute to method transfer challenges. Laboratories implementing a systematic risk-based approach report significantly higher success rates in method transfer projects.
Adjustment strategies for system-specific parameters form the technical core of method transfer. Gradient delay volume compensation techniques, for instance, involve modifications to initial hold times or implementation of mathematical corrections to maintain chromatographic separation patterns. Similarly, flow rate and injection volume scaling may be necessary when transferring between systems with different column dimensions or detection sensitivities.
Verification protocols represent another crucial component, typically involving the analysis of system suitability samples on both source and target instruments to confirm equivalent performance. Statistical tools such as equivalence testing provide objective measures for assessing the success of method transfer, with acceptance criteria commonly established for retention time reproducibility, resolution between critical pairs, and quantitative accuracy.
Regulatory considerations must guide transfer strategies in GMP environments, with particular attention to ICH guidelines and FDA recommendations regarding analytical method validation. Documentation requirements typically include transfer protocols, equivalence data, and formal approval processes that demonstrate the transferred method's continued fitness for purpose.
Modern approaches increasingly incorporate software solutions that can predict chromatographic behavior across different instrument platforms, allowing for virtual method transfer simulations before physical implementation. These tools have demonstrated significant time and resource savings by identifying potential issues before experimental work begins.
The foundation of effective method transfer begins with comprehensive documentation of the source method parameters, including detailed specifications of column dimensions, particle size, stationary phase chemistry, mobile phase composition, gradient profiles, and detection parameters. This documentation serves as the baseline reference for the transfer process and facilitates troubleshooting if discrepancies arise during implementation.
Risk assessment constitutes an essential preliminary step, where critical method attributes are identified and potential variations between systems are evaluated. Parameters such as dwell volume differences, detector response factors, and pressure limitations require particular attention as they frequently contribute to method transfer challenges. Laboratories implementing a systematic risk-based approach report significantly higher success rates in method transfer projects.
Adjustment strategies for system-specific parameters form the technical core of method transfer. Gradient delay volume compensation techniques, for instance, involve modifications to initial hold times or implementation of mathematical corrections to maintain chromatographic separation patterns. Similarly, flow rate and injection volume scaling may be necessary when transferring between systems with different column dimensions or detection sensitivities.
Verification protocols represent another crucial component, typically involving the analysis of system suitability samples on both source and target instruments to confirm equivalent performance. Statistical tools such as equivalence testing provide objective measures for assessing the success of method transfer, with acceptance criteria commonly established for retention time reproducibility, resolution between critical pairs, and quantitative accuracy.
Regulatory considerations must guide transfer strategies in GMP environments, with particular attention to ICH guidelines and FDA recommendations regarding analytical method validation. Documentation requirements typically include transfer protocols, equivalence data, and formal approval processes that demonstrate the transferred method's continued fitness for purpose.
Modern approaches increasingly incorporate software solutions that can predict chromatographic behavior across different instrument platforms, allowing for virtual method transfer simulations before physical implementation. These tools have demonstrated significant time and resource savings by identifying potential issues before experimental work begins.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!