Sequencing via HPLC: Automating High-Performance Goals
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Sequencing Evolution and Objectives
High-Performance Liquid Chromatography (HPLC) has evolved significantly since its inception in the late 1960s, transforming from a specialized analytical technique to an essential tool in modern laboratories. The journey began with the development of packed columns and basic pumping systems, which laid the foundation for what would become a revolutionary separation method. By the 1980s, HPLC had established itself as a standard analytical technique across pharmaceutical, environmental, and biochemical industries.
The evolution of HPLC sequencing technology has been marked by continuous improvements in column technology, detection methods, and automation capabilities. Early systems required significant manual intervention, with scientists physically injecting samples and collecting data through chart recorders. The introduction of microprocessor-controlled systems in the 1990s represented a significant leap forward, enabling more precise control over separation parameters and data acquisition.
Recent advancements have focused on miniaturization, increased sensitivity, and enhanced automation. Ultra-High Performance Liquid Chromatography (UHPLC) emerged as a significant innovation, utilizing sub-2-micron particles and higher pressures to achieve faster separations without sacrificing resolution. This development has dramatically reduced analysis times from hours to minutes, increasing laboratory throughput exponentially.
The integration of HPLC with mass spectrometry (LC-MS) has further expanded its capabilities, allowing for more definitive identification of compounds in complex mixtures. This hyphenated technique has become indispensable in proteomics, metabolomics, and pharmaceutical development, enabling researchers to sequence peptides and analyze complex biological samples with unprecedented accuracy.
Current technological objectives in HPLC sequencing automation center around several key areas. First is the development of fully autonomous systems capable of sample preparation, injection, analysis, and data interpretation with minimal human intervention. These systems aim to eliminate human error, increase reproducibility, and enable 24/7 operation in high-throughput environments.
Second is the enhancement of real-time decision-making capabilities through artificial intelligence and machine learning algorithms. These technologies enable systems to adapt separation parameters based on initial results, optimizing conditions for challenging samples without analyst intervention.
Third is the pursuit of green chemistry principles through reduced solvent consumption, lower energy requirements, and miniaturized systems. Nano-HPLC and chip-based separations represent promising directions in this regard, offering comparable analytical performance with significantly reduced environmental impact.
The ultimate goal of modern HPLC sequencing automation is to create intelligent analytical platforms that seamlessly integrate with laboratory information management systems (LIMS) and other analytical instruments, creating a comprehensive analytical ecosystem. This vision includes predictive maintenance capabilities, remote operation possibilities, and adaptive workflows that respond to changing analytical needs in real-time.
The evolution of HPLC sequencing technology has been marked by continuous improvements in column technology, detection methods, and automation capabilities. Early systems required significant manual intervention, with scientists physically injecting samples and collecting data through chart recorders. The introduction of microprocessor-controlled systems in the 1990s represented a significant leap forward, enabling more precise control over separation parameters and data acquisition.
Recent advancements have focused on miniaturization, increased sensitivity, and enhanced automation. Ultra-High Performance Liquid Chromatography (UHPLC) emerged as a significant innovation, utilizing sub-2-micron particles and higher pressures to achieve faster separations without sacrificing resolution. This development has dramatically reduced analysis times from hours to minutes, increasing laboratory throughput exponentially.
The integration of HPLC with mass spectrometry (LC-MS) has further expanded its capabilities, allowing for more definitive identification of compounds in complex mixtures. This hyphenated technique has become indispensable in proteomics, metabolomics, and pharmaceutical development, enabling researchers to sequence peptides and analyze complex biological samples with unprecedented accuracy.
Current technological objectives in HPLC sequencing automation center around several key areas. First is the development of fully autonomous systems capable of sample preparation, injection, analysis, and data interpretation with minimal human intervention. These systems aim to eliminate human error, increase reproducibility, and enable 24/7 operation in high-throughput environments.
Second is the enhancement of real-time decision-making capabilities through artificial intelligence and machine learning algorithms. These technologies enable systems to adapt separation parameters based on initial results, optimizing conditions for challenging samples without analyst intervention.
Third is the pursuit of green chemistry principles through reduced solvent consumption, lower energy requirements, and miniaturized systems. Nano-HPLC and chip-based separations represent promising directions in this regard, offering comparable analytical performance with significantly reduced environmental impact.
The ultimate goal of modern HPLC sequencing automation is to create intelligent analytical platforms that seamlessly integrate with laboratory information management systems (LIMS) and other analytical instruments, creating a comprehensive analytical ecosystem. This vision includes predictive maintenance capabilities, remote operation possibilities, and adaptive workflows that respond to changing analytical needs in real-time.
Market Demand Analysis for Automated HPLC Systems
The global market for automated High-Performance Liquid Chromatography (HPLC) systems has experienced robust growth in recent years, driven primarily by increasing demand in pharmaceutical research, clinical diagnostics, and environmental testing sectors. Current market valuations indicate that the automated HPLC systems market reached approximately 4.2 billion USD in 2022, with projections suggesting a compound annual growth rate of 5.8% through 2028.
Pharmaceutical and biotechnology industries remain the largest consumers of automated HPLC technology, accounting for nearly 45% of the total market share. This dominance stems from stringent regulatory requirements for drug development and quality control processes, where precision and reproducibility are paramount. The rise in biopharmaceutical development has further accelerated demand for advanced automated sequencing capabilities.
Clinical laboratories represent the fastest-growing segment for automated HPLC systems, with an estimated growth rate of 7.3% annually. This surge is attributed to increasing adoption of HPLC techniques in clinical diagnostics, particularly for therapeutic drug monitoring, vitamin D testing, and hemoglobin A1c analysis. Healthcare facilities are increasingly recognizing the value of automation in reducing human error and improving throughput.
Regional analysis reveals North America as the leading market for automated HPLC systems, holding approximately 38% of the global market share. However, Asia-Pacific regions, particularly China and India, are demonstrating the highest growth potential, with market expansion rates exceeding 9% annually. This growth is fueled by increasing investments in pharmaceutical research infrastructure and the expansion of contract research organizations in these regions.
End-user preferences are shifting decisively toward fully integrated automation solutions that offer seamless sample preparation, injection, separation, and data analysis capabilities. Market surveys indicate that 73% of laboratory managers prioritize systems with comprehensive automation features when making purchasing decisions, citing labor cost reduction and improved workflow efficiency as primary motivators.
The COVID-19 pandemic has accelerated automation trends across laboratory environments, with 67% of facilities reporting increased interest in reducing manual handling through automated systems. This shift reflects both safety concerns and the need to maintain operations with reduced on-site personnel during public health emergencies.
Market forecasts suggest particular growth in modular automated HPLC systems that allow laboratories to customize automation levels according to their specific needs and budgets. This trend toward flexibility is expected to expand the market to smaller laboratories and academic institutions previously priced out of fully automated solutions.
Pharmaceutical and biotechnology industries remain the largest consumers of automated HPLC technology, accounting for nearly 45% of the total market share. This dominance stems from stringent regulatory requirements for drug development and quality control processes, where precision and reproducibility are paramount. The rise in biopharmaceutical development has further accelerated demand for advanced automated sequencing capabilities.
Clinical laboratories represent the fastest-growing segment for automated HPLC systems, with an estimated growth rate of 7.3% annually. This surge is attributed to increasing adoption of HPLC techniques in clinical diagnostics, particularly for therapeutic drug monitoring, vitamin D testing, and hemoglobin A1c analysis. Healthcare facilities are increasingly recognizing the value of automation in reducing human error and improving throughput.
Regional analysis reveals North America as the leading market for automated HPLC systems, holding approximately 38% of the global market share. However, Asia-Pacific regions, particularly China and India, are demonstrating the highest growth potential, with market expansion rates exceeding 9% annually. This growth is fueled by increasing investments in pharmaceutical research infrastructure and the expansion of contract research organizations in these regions.
End-user preferences are shifting decisively toward fully integrated automation solutions that offer seamless sample preparation, injection, separation, and data analysis capabilities. Market surveys indicate that 73% of laboratory managers prioritize systems with comprehensive automation features when making purchasing decisions, citing labor cost reduction and improved workflow efficiency as primary motivators.
The COVID-19 pandemic has accelerated automation trends across laboratory environments, with 67% of facilities reporting increased interest in reducing manual handling through automated systems. This shift reflects both safety concerns and the need to maintain operations with reduced on-site personnel during public health emergencies.
Market forecasts suggest particular growth in modular automated HPLC systems that allow laboratories to customize automation levels according to their specific needs and budgets. This trend toward flexibility is expected to expand the market to smaller laboratories and academic institutions previously priced out of fully automated solutions.
Current HPLC Automation Challenges
Despite significant advancements in HPLC technology, current automation systems face several persistent challenges that limit their full potential in sequencing applications. Sample preparation remains a major bottleneck, with many laboratories still relying on manual processes for sample filtration, dilution, and derivatization. These manual interventions introduce variability, increase the risk of contamination, and significantly extend analysis time. Even in systems with automated sample preparation, the integration between sample handling and the analytical workflow often lacks seamless coordination.
Method development and optimization continue to require substantial human expertise and intervention. While some platforms offer automated method development tools, these typically explore a limited parameter space and may miss optimal conditions that experienced chromatographers could identify. The translation of methods between different instruments and laboratories also presents challenges, with subtle variations in hardware configurations leading to reproducibility issues.
Data management and integration represent another significant hurdle. Current HPLC automation systems generate enormous volumes of data, but the infrastructure for processing, storing, and extracting meaningful insights from this data remains inadequate. Many laboratories struggle with fragmented software ecosystems where sample tracking, instrument control, data analysis, and reporting tools operate as separate entities rather than as an integrated platform.
System maintenance and troubleshooting continue to demand specialized expertise. Automated diagnostics have improved but still cannot address many common issues without human intervention. Consumable management, including monitoring column performance degradation and predicting maintenance needs, remains largely reactive rather than proactive in most systems.
Cross-platform compatibility presents ongoing challenges, with proprietary interfaces limiting the integration of components from different manufacturers. This vendor lock-in restricts laboratories' ability to assemble optimal systems from best-in-class components and increases dependency on single suppliers for critical workflow elements.
Resource utilization efficiency represents another area for improvement. Current systems often operate with significant idle time between analyses or utilize instruments at full capacity for simple separations that could be accomplished with less sophisticated equipment. This inefficient resource allocation increases operational costs and reduces overall laboratory throughput.
Finally, regulatory compliance and validation processes for automated HPLC systems remain cumbersome. Documentation requirements, system suitability testing, and performance verification procedures have not kept pace with automation capabilities, creating administrative bottlenecks that diminish the productivity gains offered by technological advancements.
Method development and optimization continue to require substantial human expertise and intervention. While some platforms offer automated method development tools, these typically explore a limited parameter space and may miss optimal conditions that experienced chromatographers could identify. The translation of methods between different instruments and laboratories also presents challenges, with subtle variations in hardware configurations leading to reproducibility issues.
Data management and integration represent another significant hurdle. Current HPLC automation systems generate enormous volumes of data, but the infrastructure for processing, storing, and extracting meaningful insights from this data remains inadequate. Many laboratories struggle with fragmented software ecosystems where sample tracking, instrument control, data analysis, and reporting tools operate as separate entities rather than as an integrated platform.
System maintenance and troubleshooting continue to demand specialized expertise. Automated diagnostics have improved but still cannot address many common issues without human intervention. Consumable management, including monitoring column performance degradation and predicting maintenance needs, remains largely reactive rather than proactive in most systems.
Cross-platform compatibility presents ongoing challenges, with proprietary interfaces limiting the integration of components from different manufacturers. This vendor lock-in restricts laboratories' ability to assemble optimal systems from best-in-class components and increases dependency on single suppliers for critical workflow elements.
Resource utilization efficiency represents another area for improvement. Current systems often operate with significant idle time between analyses or utilize instruments at full capacity for simple separations that could be accomplished with less sophisticated equipment. This inefficient resource allocation increases operational costs and reduces overall laboratory throughput.
Finally, regulatory compliance and validation processes for automated HPLC systems remain cumbersome. Documentation requirements, system suitability testing, and performance verification procedures have not kept pace with automation capabilities, creating administrative bottlenecks that diminish the productivity gains offered by technological advancements.
Current Automated HPLC Sequencing Solutions
01 Automated sample preparation systems for HPLC
Automated systems for preparing samples prior to HPLC analysis, including features such as automated pipetting, sample dilution, filtration, and derivatization. These systems improve efficiency by reducing manual handling, minimizing human error, and increasing throughput in analytical laboratories. The automation of sample preparation steps ensures consistency in sample processing and helps maintain the integrity of analytical results.- Automated sample preparation and injection systems: Automated systems for HPLC that handle sample preparation and injection processes, improving efficiency and reducing manual intervention. These systems include robotic sample handlers, automated dilution, filtration, and precise injection mechanisms that ensure consistent sample delivery to the chromatography column. The automation of these pre-analysis steps significantly reduces human error and increases throughput in analytical laboratories.
- Intelligent control and monitoring systems: Advanced control systems that manage HPLC operations through intelligent software interfaces. These systems provide real-time monitoring of chromatographic parameters, automated method development, and adaptive control mechanisms that respond to changing analytical conditions. The integration of artificial intelligence and machine learning algorithms enables predictive maintenance and optimization of separation parameters for improved analytical performance.
- Integrated data processing and analysis solutions: Comprehensive software solutions that automate data acquisition, processing, and interpretation of HPLC results. These systems feature automated peak detection, integration, quantification, and reporting capabilities. Advanced algorithms enable automated data validation, outlier detection, and trend analysis, significantly reducing the time required for post-run data processing while improving the reliability and consistency of analytical results.
- High-throughput multi-sample analysis systems: Specialized HPLC automation platforms designed for high-throughput environments that can process multiple samples simultaneously. These systems incorporate parallel processing capabilities, multi-column configurations, and rapid gradient generation for accelerated analysis. Automated column switching, multi-detector arrangements, and synchronized sample preparation enable continuous operation with minimal downtime, making them ideal for industrial quality control and large-scale research applications.
- Modular and integrated HPLC automation platforms: Flexible automation platforms that combine various HPLC components into integrated systems with modular design. These platforms allow for customization based on specific analytical needs while maintaining full automation capabilities. The modular approach enables easy upgrades, maintenance, and reconfiguration of the system to adapt to changing analytical requirements. Integration with laboratory information management systems (LIMS) and other analytical instruments creates comprehensive automated workflows for enhanced laboratory efficiency.
02 Integrated HPLC automation platforms
Comprehensive automation platforms that integrate multiple aspects of HPLC workflows, including sample injection, column switching, mobile phase preparation, and data analysis. These integrated systems provide end-to-end automation solutions that can be controlled through centralized software interfaces. The platforms enable unattended operation of HPLC systems, allowing for continuous analysis of large sample batches and improving laboratory productivity.Expand Specific Solutions03 Robotic sample handling for HPLC
Robotic systems designed specifically for handling samples in HPLC workflows, featuring automated arm movements, precision positioning, and specialized grippers for vials and plates. These robotic handlers can transfer samples between different instruments or preparation stations, load samples into autosamplers, and manage sample storage. The implementation of robotics in HPLC workflows reduces manual intervention and enables continuous operation of analytical systems.Expand Specific Solutions04 Intelligent control systems for HPLC automation
Advanced control systems that incorporate artificial intelligence, machine learning, and sophisticated algorithms to optimize HPLC operations. These intelligent systems can automatically adjust chromatographic parameters, predict maintenance needs, detect anomalies in system performance, and optimize separation conditions. The implementation of intelligent control enhances the reliability of HPLC analysis, reduces method development time, and enables adaptive responses to changing analytical conditions.Expand Specific Solutions05 High-throughput HPLC automation solutions
Specialized automation solutions designed to maximize sample throughput in HPLC analysis, featuring parallel processing capabilities, rapid column switching, fast gradient systems, and multiplexed detection. These high-throughput systems enable the analysis of large sample sets in minimal time while maintaining analytical quality. The implementation of these solutions is particularly valuable in applications such as pharmaceutical quality control, environmental monitoring, and clinical diagnostics where large numbers of samples require timely analysis.Expand Specific Solutions
Key Industry Players in HPLC Automation
The HPLC sequencing automation market is currently in a growth phase, with increasing demand driven by pharmaceutical, biotechnology, and research applications. The market size is expanding rapidly as laboratories seek higher throughput and precision in analytical workflows. Technologically, the field is evolving from semi-automated to fully integrated systems, with companies at varying maturity levels. Industry leaders like Illumina and Thermo Fisher (not listed) have established mature platforms, while companies such as Edico Genome are advancing computational acceleration for sequence analysis. FANUC and Beckhoff Automation contribute robotics expertise, while semiconductor players including Intel, TSMC, and GlobalFoundries provide essential computing infrastructure. Research institutions like King Abdullah University and Michigan State University are driving innovation through academic-industrial partnerships, creating a competitive landscape balanced between established players and emerging specialized solution providers.
Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
Technical Solution: Mindray has developed an advanced HPLC-based sequencing automation platform specifically designed for clinical diagnostics and point-of-care applications. Their system integrates compact, high-precision liquid handling robotics with miniaturized HPLC components to enable rapid sequencing in resource-constrained environments. The platform features a novel thermal regulation system that maintains optimal separation conditions across varying ambient temperatures, ensuring consistent results in diverse clinical settings. Their proprietary "SmartFlow" technology incorporates pressure-adaptive algorithms that automatically optimize flow rates and gradient profiles based on sample viscosity and column conditions, reducing analysis time by approximately 25% compared to conventional systems[3]. The platform includes automated calibration routines that compensate for instrument drift, maintaining accuracy over extended operational periods without manual intervention. Mindray's solution also incorporates comprehensive data security features with role-based access controls and audit trails to meet regulatory requirements in clinical environments.
Strengths: Exceptional reliability in clinical settings with minimal maintenance requirements; compact footprint suitable for space-constrained laboratories; intuitive user interface requiring minimal specialized training. Weaknesses: More limited throughput compared to research-focused platforms; fewer customization options for specialized research applications; more focused on clinical applications than basic research capabilities.
Chengdu Olymvax Biopharmaceuticals, Inc.
Technical Solution: Olymvax has developed a specialized HPLC-based sequencing automation platform focused on vaccine development and quality control applications. Their system integrates high-precision chromatography with automated sample preparation to enable rapid characterization of nucleic acid-based vaccines and therapeutics. The platform features proprietary separation columns specifically optimized for the analysis of modified nucleotides commonly used in vaccine formulations. Their "VaxSeq" technology incorporates automated impurity profiling capabilities that can detect contaminants at concentrations below 0.05%, exceeding regulatory requirements for vaccine manufacturing[5]. The system includes integrated stability testing protocols that automatically analyze samples at predetermined intervals, generating comprehensive degradation profiles without manual intervention. Olymvax's platform also features automated method development capabilities that systematically evaluate multiple chromatographic conditions to identify optimal separation parameters for novel vaccine candidates, reducing development timelines by approximately 40% compared to traditional approaches.
Strengths: Specialized optimization for vaccine development and quality control; exceptional sensitivity for impurity detection; comprehensive regulatory documentation generation for compliance purposes. Weaknesses: Limited application scope outside pharmaceutical/vaccine development; less flexible for general research applications; higher specialization requires more specific technical support.
Breakthrough Patents in HPLC Automation
Automated analysis systems
PatentWO2014168865A1
Innovation
- An automated system controlled by a computer that manages the flow of samples and solvents through capture and analysis columns, ensuring precise control over flow rates, volumes, and timing to trap, elute, and separate components, thereby reducing human intervention and enhancing consistency and reproducibility.
Parallel high-performance liquid chromatography with post-separation treatment
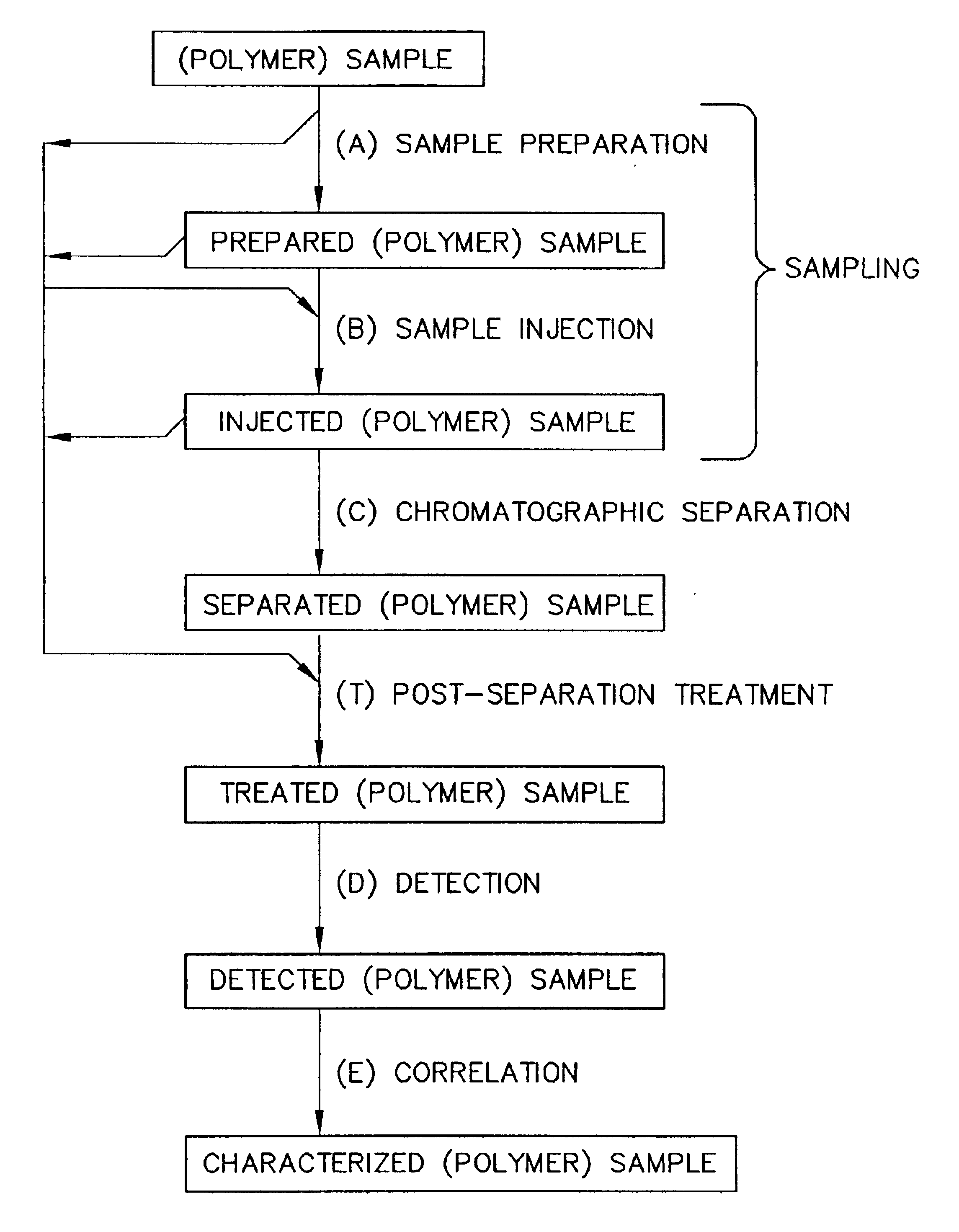
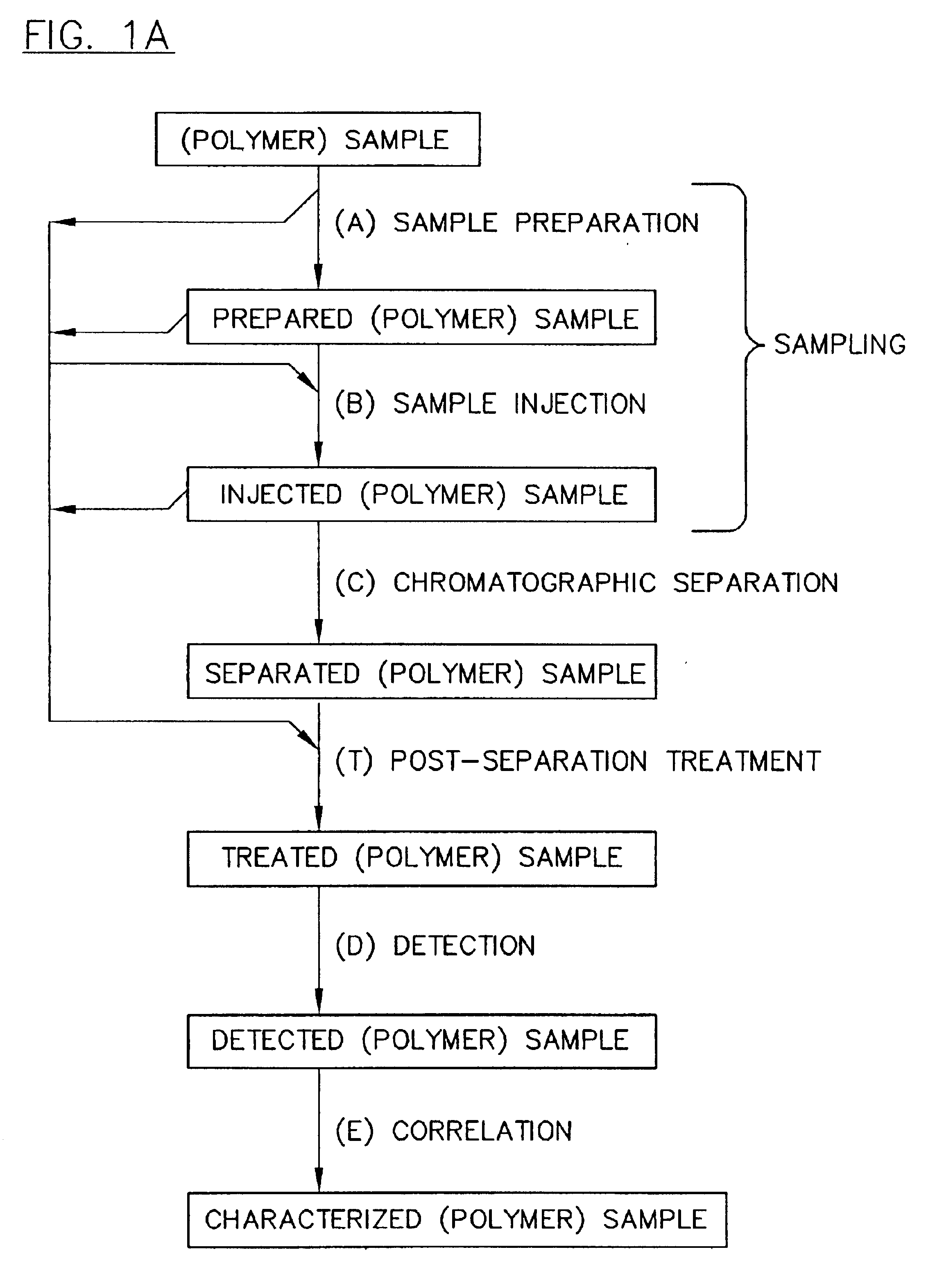
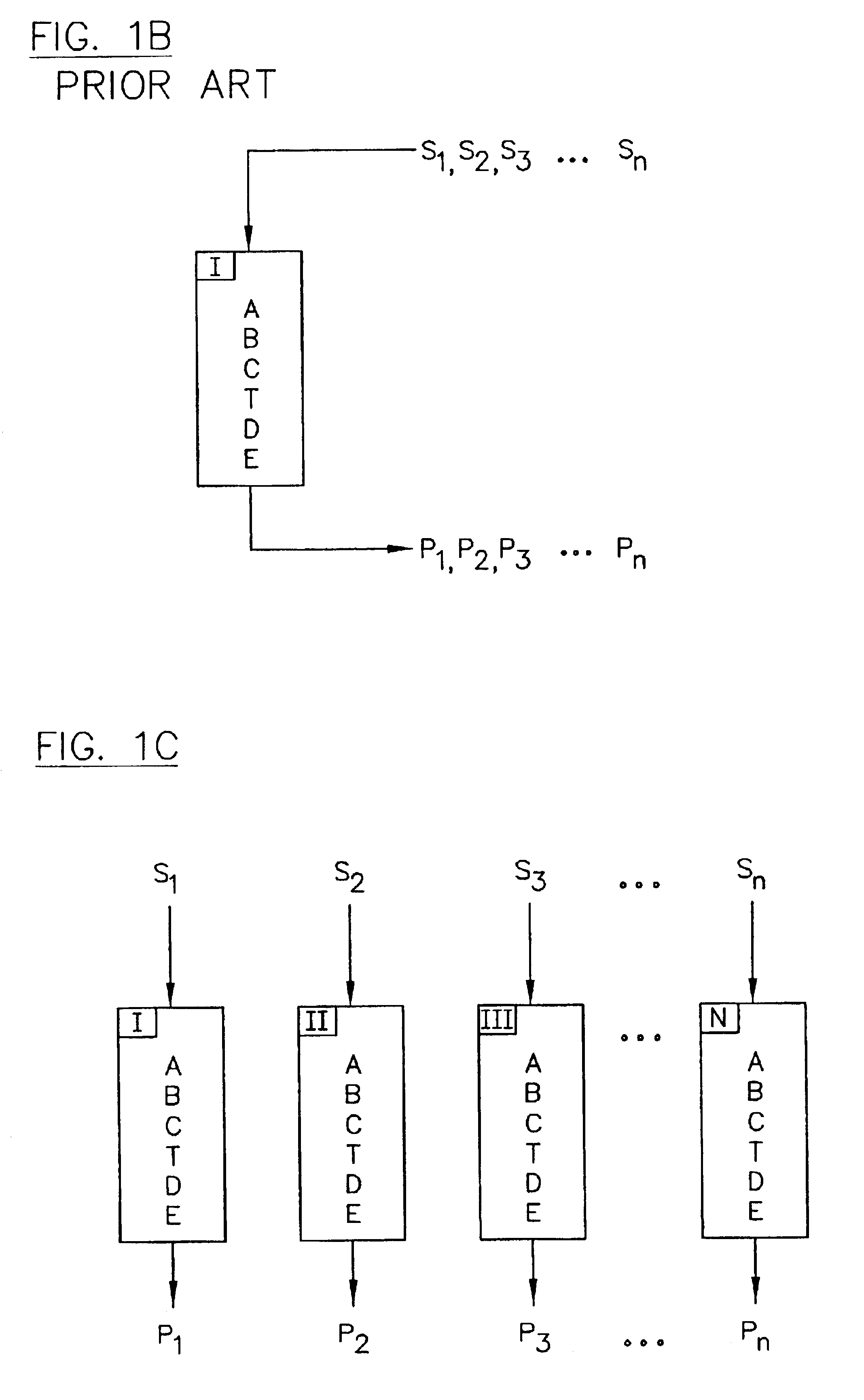
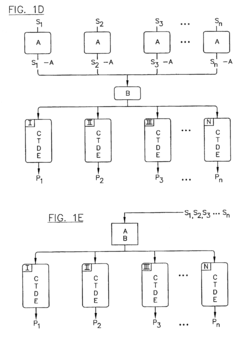
PatentInactiveUS6776902B2
Innovation
- The implementation of parallel HPLC systems with post-separation treatment protocols, where samples are separated in parallel across multiple chromatographic columns and then treated with agents like precipitants or derivatizing agents to enhance detectability, particularly using optical detectors for cost-effective and high-throughput analysis.
Regulatory Compliance for Automated HPLC Systems
Automated HPLC systems must adhere to stringent regulatory frameworks across different jurisdictions to ensure compliance with quality standards and data integrity requirements. In the pharmaceutical and clinical diagnostics sectors, regulatory bodies such as the FDA, EMA, and PMDA have established comprehensive guidelines that directly impact the implementation and operation of automated HPLC sequencing technologies. These regulations primarily focus on system validation, data security, audit trails, and quality assurance protocols.
The FDA's 21 CFR Part 11 represents a cornerstone regulation for automated HPLC systems, mandating electronic records and signatures that are trustworthy, reliable, and equivalent to paper records. This necessitates robust authentication mechanisms, comprehensive audit trails, and secure data storage solutions within automated HPLC platforms. Similarly, the EU GMP Annex 11 provides parallel requirements for computerized systems in regulated environments across European markets.
Compliance with ISO 17025 standards becomes essential for laboratories utilizing automated HPLC sequencing, particularly when test results may impact public health decisions or regulatory submissions. This standard emphasizes technical competence, impartial operation, and consistent valid results through proper calibration and maintenance procedures.
The implementation of GAMP 5 (Good Automated Manufacturing Practice) guidelines offers a risk-based approach to computerized system validation for automated HPLC platforms. This framework helps organizations categorize system components based on risk levels and apply appropriate validation strategies, reducing unnecessary documentation while maintaining compliance.
Data integrity remains a critical regulatory concern, with regulatory bodies increasingly scrutinizing the ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available) in automated systems. Modern HPLC automation must incorporate features that prevent data manipulation, maintain complete audit trails, and ensure traceability of all analytical processes.
Regulatory compliance also extends to software validation requirements, where automated HPLC systems must demonstrate that their controlling software consistently performs as intended. This includes verification of algorithm accuracy, system suitability tests, and performance qualification under actual operating conditions.
For global deployment, automated HPLC sequencing technologies must navigate the complex landscape of regional regulatory variations. While harmonization efforts through initiatives like ICH (International Council for Harmonisation) have made progress, significant differences remain in implementation details across major markets, necessitating adaptable compliance strategies.
The FDA's 21 CFR Part 11 represents a cornerstone regulation for automated HPLC systems, mandating electronic records and signatures that are trustworthy, reliable, and equivalent to paper records. This necessitates robust authentication mechanisms, comprehensive audit trails, and secure data storage solutions within automated HPLC platforms. Similarly, the EU GMP Annex 11 provides parallel requirements for computerized systems in regulated environments across European markets.
Compliance with ISO 17025 standards becomes essential for laboratories utilizing automated HPLC sequencing, particularly when test results may impact public health decisions or regulatory submissions. This standard emphasizes technical competence, impartial operation, and consistent valid results through proper calibration and maintenance procedures.
The implementation of GAMP 5 (Good Automated Manufacturing Practice) guidelines offers a risk-based approach to computerized system validation for automated HPLC platforms. This framework helps organizations categorize system components based on risk levels and apply appropriate validation strategies, reducing unnecessary documentation while maintaining compliance.
Data integrity remains a critical regulatory concern, with regulatory bodies increasingly scrutinizing the ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available) in automated systems. Modern HPLC automation must incorporate features that prevent data manipulation, maintain complete audit trails, and ensure traceability of all analytical processes.
Regulatory compliance also extends to software validation requirements, where automated HPLC systems must demonstrate that their controlling software consistently performs as intended. This includes verification of algorithm accuracy, system suitability tests, and performance qualification under actual operating conditions.
For global deployment, automated HPLC sequencing technologies must navigate the complex landscape of regional regulatory variations. While harmonization efforts through initiatives like ICH (International Council for Harmonisation) have made progress, significant differences remain in implementation details across major markets, necessitating adaptable compliance strategies.
Cost-Benefit Analysis of HPLC Automation Implementation
Implementing automation in High-Performance Liquid Chromatography (HPLC) sequencing systems requires substantial initial investment but offers significant long-term financial benefits. The initial capital expenditure typically ranges from $150,000 to $500,000 depending on the scale and sophistication of the automation solution, including robotic sample handlers, automated injection systems, and integrated data management software.
Labor cost reduction represents one of the most quantifiable benefits, with automated HPLC systems reducing manual operation time by 65-80%. For a laboratory processing 100 samples daily, this translates to approximately 25-30 labor hours saved per week, or $75,000-$90,000 annually based on average laboratory technician salaries.
Throughput enhancement delivers additional economic value, with automated systems capable of operating continuously, including overnight and weekend runs without supervision. This capability increases sample processing capacity by 150-200% compared to manual operations, potentially doubling revenue generation without proportional cost increases.
Error reduction constitutes another significant economic benefit. Manual HPLC operations typically experience error rates of 2-5%, requiring costly repeat analyses. Automation reduces this to below 0.5%, saving approximately $20,000-$40,000 annually for mid-sized laboratories in wasted materials, reagents, and labor hours dedicated to error correction.
Reagent consumption optimization through precise dispensing mechanisms in automated systems reduces chemical usage by 15-25% compared to manual operations. For laboratories with annual reagent budgets of $100,000, this represents $15,000-$25,000 in direct savings, with additional benefits from reduced waste disposal costs.
Return on investment (ROI) calculations indicate that most HPLC automation implementations achieve break-even within 18-36 months, depending on laboratory throughput and utilization rates. Facilities operating at high capacity typically recoup investments faster, sometimes within 12-18 months.
Maintenance costs must be factored into long-term financial planning, typically adding 8-12% of the initial system cost annually. However, these are offset by reduced downtime and extended equipment lifespan, as automated systems operate under more consistent conditions than manually operated equipment.
Scalability considerations reveal that incremental automation expansions often deliver better ROI than complete system overhauls. Laboratories can strategically implement automation in phases, targeting high-volume or error-prone processes first to maximize early returns while distributing capital expenditures over longer periods.
Labor cost reduction represents one of the most quantifiable benefits, with automated HPLC systems reducing manual operation time by 65-80%. For a laboratory processing 100 samples daily, this translates to approximately 25-30 labor hours saved per week, or $75,000-$90,000 annually based on average laboratory technician salaries.
Throughput enhancement delivers additional economic value, with automated systems capable of operating continuously, including overnight and weekend runs without supervision. This capability increases sample processing capacity by 150-200% compared to manual operations, potentially doubling revenue generation without proportional cost increases.
Error reduction constitutes another significant economic benefit. Manual HPLC operations typically experience error rates of 2-5%, requiring costly repeat analyses. Automation reduces this to below 0.5%, saving approximately $20,000-$40,000 annually for mid-sized laboratories in wasted materials, reagents, and labor hours dedicated to error correction.
Reagent consumption optimization through precise dispensing mechanisms in automated systems reduces chemical usage by 15-25% compared to manual operations. For laboratories with annual reagent budgets of $100,000, this represents $15,000-$25,000 in direct savings, with additional benefits from reduced waste disposal costs.
Return on investment (ROI) calculations indicate that most HPLC automation implementations achieve break-even within 18-36 months, depending on laboratory throughput and utilization rates. Facilities operating at high capacity typically recoup investments faster, sometimes within 12-18 months.
Maintenance costs must be factored into long-term financial planning, typically adding 8-12% of the initial system cost annually. However, these are offset by reduced downtime and extended equipment lifespan, as automated systems operate under more consistent conditions than manually operated equipment.
Scalability considerations reveal that incremental automation expansions often deliver better ROI than complete system overhauls. Laboratories can strategically implement automation in phases, targeting high-volume or error-prone processes first to maximize early returns while distributing capital expenditures over longer periods.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!