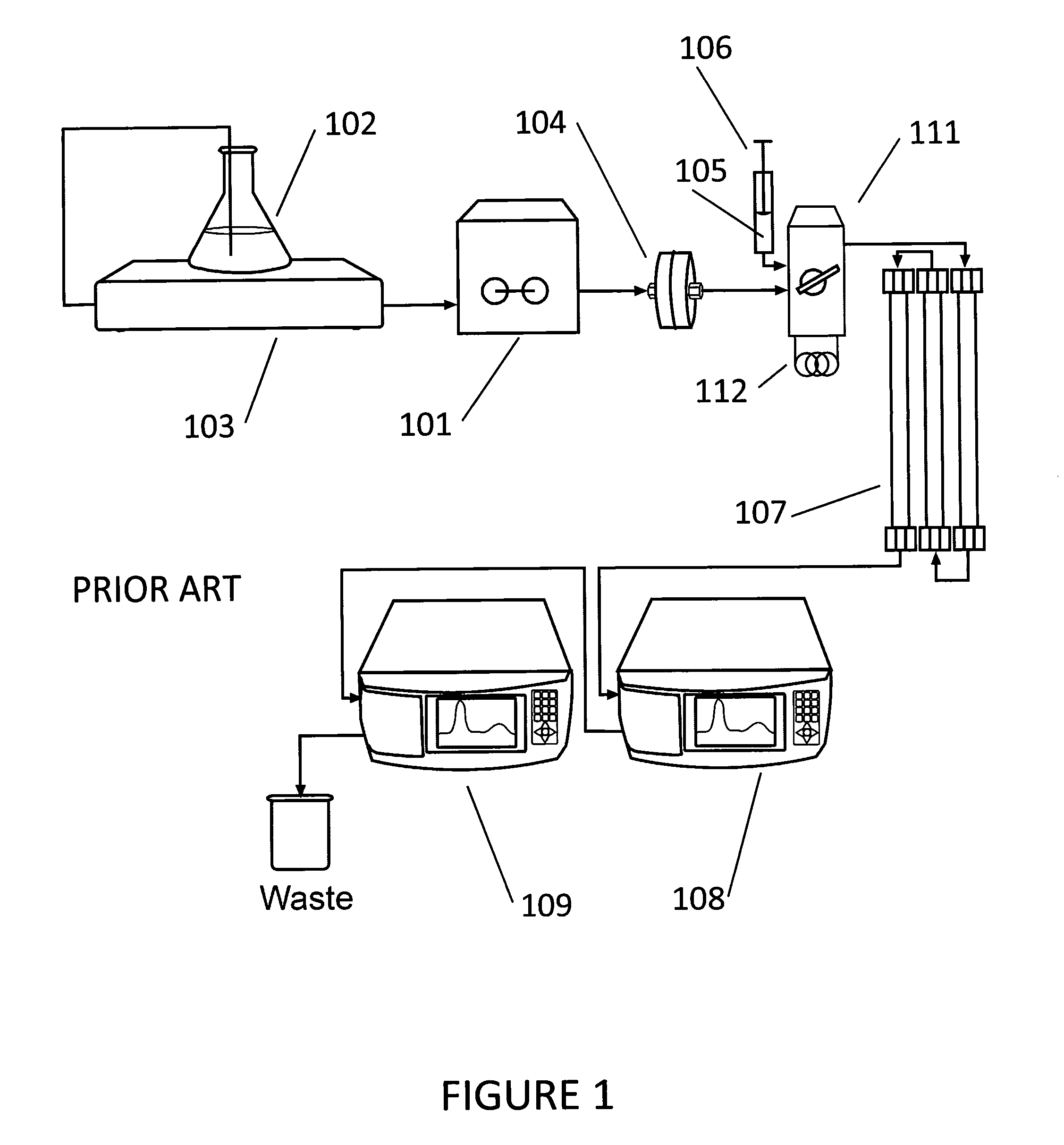
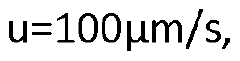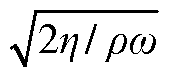Enhancing Peak Detection in HPLC: Parameters and Techniques
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Peak Detection Background and Objectives
High-performance liquid chromatography (HPLC) has evolved significantly since its inception in the 1960s, becoming an indispensable analytical technique in pharmaceutical, environmental, food, and clinical laboratories. Peak detection, as a fundamental aspect of chromatographic analysis, represents the critical process of identifying and quantifying compounds separated during the chromatographic run. The historical trajectory of HPLC peak detection has progressed from manual interpretation of paper chromatograms to sophisticated computerized systems employing advanced algorithms.
The evolution of peak detection technology has been driven by increasing demands for sensitivity, accuracy, and throughput in analytical laboratories. Early detection methods relied heavily on UV-visible spectroscopy with limited sensitivity and specificity. Modern systems incorporate multiple detection technologies including diode array detection (DAD), fluorescence, mass spectrometry, and electrochemical detection, significantly enhancing the capability to detect and characterize complex mixtures.
Current technological trends in HPLC peak detection focus on overcoming persistent challenges such as baseline drift, peak overlap, and detection of trace components in complex matrices. Machine learning and artificial intelligence approaches are emerging as powerful tools for automated peak detection and integration, particularly for handling complex chromatograms where traditional algorithms struggle. Additionally, there is a growing emphasis on real-time peak detection and analysis to support process analytical technology (PAT) applications in pharmaceutical manufacturing.
The primary objectives of enhancing peak detection in HPLC encompass several dimensions. First, improving detection sensitivity to identify trace components in complex matrices, which is particularly crucial in environmental monitoring and bioanalytical applications. Second, enhancing peak resolution capabilities to distinguish closely eluting compounds, addressing a persistent challenge in metabolomics and natural product analysis. Third, developing robust algorithms that can accurately detect and integrate peaks across varying chromatographic conditions, reducing the need for manual intervention.
Furthermore, there is a strategic focus on developing adaptive peak detection systems that can automatically optimize detection parameters based on sample characteristics and separation conditions. This adaptability is essential for handling diverse sample types and achieving consistent analytical performance across different laboratories and instruments. The ultimate goal is to establish standardized approaches that ensure reproducible peak detection results, facilitating data comparison and method transfer between different analytical platforms.
The evolution of peak detection technology has been driven by increasing demands for sensitivity, accuracy, and throughput in analytical laboratories. Early detection methods relied heavily on UV-visible spectroscopy with limited sensitivity and specificity. Modern systems incorporate multiple detection technologies including diode array detection (DAD), fluorescence, mass spectrometry, and electrochemical detection, significantly enhancing the capability to detect and characterize complex mixtures.
Current technological trends in HPLC peak detection focus on overcoming persistent challenges such as baseline drift, peak overlap, and detection of trace components in complex matrices. Machine learning and artificial intelligence approaches are emerging as powerful tools for automated peak detection and integration, particularly for handling complex chromatograms where traditional algorithms struggle. Additionally, there is a growing emphasis on real-time peak detection and analysis to support process analytical technology (PAT) applications in pharmaceutical manufacturing.
The primary objectives of enhancing peak detection in HPLC encompass several dimensions. First, improving detection sensitivity to identify trace components in complex matrices, which is particularly crucial in environmental monitoring and bioanalytical applications. Second, enhancing peak resolution capabilities to distinguish closely eluting compounds, addressing a persistent challenge in metabolomics and natural product analysis. Third, developing robust algorithms that can accurately detect and integrate peaks across varying chromatographic conditions, reducing the need for manual intervention.
Furthermore, there is a strategic focus on developing adaptive peak detection systems that can automatically optimize detection parameters based on sample characteristics and separation conditions. This adaptability is essential for handling diverse sample types and achieving consistent analytical performance across different laboratories and instruments. The ultimate goal is to establish standardized approaches that ensure reproducible peak detection results, facilitating data comparison and method transfer between different analytical platforms.
Market Analysis for Advanced HPLC Technologies
The High-Performance Liquid Chromatography (HPLC) market continues to demonstrate robust growth, driven by increasing demand across pharmaceutical, biotechnology, food safety, environmental monitoring, and clinical diagnostic sectors. The global HPLC market was valued at approximately 4.5 billion USD in 2022 and is projected to reach 6.8 billion USD by 2028, growing at a CAGR of 5.8% during the forecast period.
Peak detection technologies represent a critical segment within the broader HPLC market, with specialized software and hardware solutions commanding premium pricing due to their direct impact on analytical accuracy and efficiency. This segment is estimated to account for about 18% of the total HPLC market value, with particularly strong growth in pharmaceutical quality control applications.
Regional analysis reveals North America maintains the largest market share at 38%, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region is experiencing the fastest growth rate at 7.2% annually, primarily driven by expanding pharmaceutical manufacturing capabilities in China and India, coupled with increasing regulatory requirements for analytical testing.
The pharmaceutical and biotechnology sectors collectively represent the largest end-user segment, accounting for approximately 62% of the advanced HPLC technologies market. This dominance stems from stringent regulatory requirements for drug development and quality control, where precise peak detection is essential for compound identification and quantification.
Market trends indicate a growing preference for integrated systems that combine advanced peak detection algorithms with automated data processing capabilities. End-users are increasingly prioritizing solutions that minimize manual intervention and reduce analysis time while maintaining or improving detection sensitivity and accuracy.
Pricing analysis shows premium positioning for advanced peak detection technologies, with comprehensive software packages commanding 15-25% higher prices compared to standard HPLC software. The return on investment is justified through improved laboratory efficiency, reduced analysis time, and enhanced data quality.
Customer surveys indicate that key purchasing factors include detection sensitivity (ranked highest by 68% of respondents), integration with existing laboratory information management systems (57%), and technical support availability (52%). These insights suggest that successful market penetration requires not only superior technical performance but also emphasis on system compatibility and customer support infrastructure.
The competitive landscape features both established analytical instrument manufacturers and specialized software developers, with recent market consolidation through strategic acquisitions aimed at combining hardware expertise with advanced algorithm development capabilities.
Peak detection technologies represent a critical segment within the broader HPLC market, with specialized software and hardware solutions commanding premium pricing due to their direct impact on analytical accuracy and efficiency. This segment is estimated to account for about 18% of the total HPLC market value, with particularly strong growth in pharmaceutical quality control applications.
Regional analysis reveals North America maintains the largest market share at 38%, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region is experiencing the fastest growth rate at 7.2% annually, primarily driven by expanding pharmaceutical manufacturing capabilities in China and India, coupled with increasing regulatory requirements for analytical testing.
The pharmaceutical and biotechnology sectors collectively represent the largest end-user segment, accounting for approximately 62% of the advanced HPLC technologies market. This dominance stems from stringent regulatory requirements for drug development and quality control, where precise peak detection is essential for compound identification and quantification.
Market trends indicate a growing preference for integrated systems that combine advanced peak detection algorithms with automated data processing capabilities. End-users are increasingly prioritizing solutions that minimize manual intervention and reduce analysis time while maintaining or improving detection sensitivity and accuracy.
Pricing analysis shows premium positioning for advanced peak detection technologies, with comprehensive software packages commanding 15-25% higher prices compared to standard HPLC software. The return on investment is justified through improved laboratory efficiency, reduced analysis time, and enhanced data quality.
Customer surveys indicate that key purchasing factors include detection sensitivity (ranked highest by 68% of respondents), integration with existing laboratory information management systems (57%), and technical support availability (52%). These insights suggest that successful market penetration requires not only superior technical performance but also emphasis on system compatibility and customer support infrastructure.
The competitive landscape features both established analytical instrument manufacturers and specialized software developers, with recent market consolidation through strategic acquisitions aimed at combining hardware expertise with advanced algorithm development capabilities.
Current Challenges in HPLC Peak Detection
High-performance liquid chromatography (HPLC) peak detection faces several significant challenges that impede accurate analysis and reliable results. One primary obstacle is baseline drift, which occurs due to changes in mobile phase composition during gradient elution. This phenomenon creates an unstable baseline that complicates the identification of small peaks and leads to integration errors, particularly in complex samples with numerous analytes.
Signal-to-noise ratio limitations represent another critical challenge, especially when detecting trace compounds in environmental or biological samples. Instrument noise from pumps, detectors, and electronic components can mask small peaks, while matrix interference from sample components can create false positives or negatives, compromising analytical integrity.
Peak overlap presents a persistent problem in HPLC analysis, particularly in complex mixtures where compounds with similar retention times elute simultaneously. This co-elution creates merged or shouldered peaks that traditional detection algorithms struggle to resolve accurately, leading to quantification errors and misidentification of compounds.
Current detection software also exhibits limitations in handling real-world analytical challenges. Many algorithms employ fixed thresholds that fail to adapt to changing baseline conditions or varying peak shapes across a chromatogram. This inflexibility results in missed peaks or false detections, requiring time-consuming manual review and adjustment by analysts.
The variability in peak shapes further complicates detection efforts. Factors such as column aging, mobile phase composition, temperature fluctuations, and sample matrix effects can cause peak tailing, fronting, or broadening. These irregular peak profiles deviate from the Gaussian models assumed by many detection algorithms, leading to integration inaccuracies.
Data processing speed remains a bottleneck in high-throughput environments. As analytical methods generate increasingly large datasets, particularly in multi-dimensional chromatography or when coupled with mass spectrometry, processing demands can overwhelm computational resources, creating delays in result reporting and decision-making.
Method transferability issues also plague peak detection across different instruments or laboratories. Detection parameters optimized for one system often perform poorly when transferred to another, necessitating extensive revalidation and adjustment. This lack of robustness hampers standardization efforts and creates inconsistencies in multi-site studies or regulatory submissions.
Signal-to-noise ratio limitations represent another critical challenge, especially when detecting trace compounds in environmental or biological samples. Instrument noise from pumps, detectors, and electronic components can mask small peaks, while matrix interference from sample components can create false positives or negatives, compromising analytical integrity.
Peak overlap presents a persistent problem in HPLC analysis, particularly in complex mixtures where compounds with similar retention times elute simultaneously. This co-elution creates merged or shouldered peaks that traditional detection algorithms struggle to resolve accurately, leading to quantification errors and misidentification of compounds.
Current detection software also exhibits limitations in handling real-world analytical challenges. Many algorithms employ fixed thresholds that fail to adapt to changing baseline conditions or varying peak shapes across a chromatogram. This inflexibility results in missed peaks or false detections, requiring time-consuming manual review and adjustment by analysts.
The variability in peak shapes further complicates detection efforts. Factors such as column aging, mobile phase composition, temperature fluctuations, and sample matrix effects can cause peak tailing, fronting, or broadening. These irregular peak profiles deviate from the Gaussian models assumed by many detection algorithms, leading to integration inaccuracies.
Data processing speed remains a bottleneck in high-throughput environments. As analytical methods generate increasingly large datasets, particularly in multi-dimensional chromatography or when coupled with mass spectrometry, processing demands can overwhelm computational resources, creating delays in result reporting and decision-making.
Method transferability issues also plague peak detection across different instruments or laboratories. Detection parameters optimized for one system often perform poorly when transferred to another, necessitating extensive revalidation and adjustment. This lack of robustness hampers standardization efforts and creates inconsistencies in multi-site studies or regulatory submissions.
Current Parameter Optimization Techniques
01 Signal processing algorithms for peak detection
Advanced signal processing algorithms can significantly enhance peak detection in HPLC analysis. These methods include mathematical transformations, filtering techniques, and statistical approaches that improve signal-to-noise ratio and peak resolution. By implementing specialized algorithms, chromatographic data can be processed to identify peaks more accurately, even in complex samples with overlapping signals or low concentration analytes.- Signal processing algorithms for peak detection: Advanced signal processing algorithms can significantly enhance peak detection in HPLC analysis. These methods include mathematical transformations, filtering techniques, and statistical approaches that improve signal-to-noise ratio and peak resolution. By applying these algorithms to chromatographic data, analysts can detect peaks that might otherwise be missed due to baseline noise or overlapping signals, thereby increasing the sensitivity and accuracy of HPLC detection.
- Hardware improvements for enhanced detection sensitivity: Hardware modifications and improvements can enhance HPLC peak detection capabilities. These include advanced detector designs, improved flow cell configurations, and optimized optical systems that increase signal strength while minimizing noise. Such hardware enhancements enable detection of lower concentration analytes and improve the overall sensitivity of HPLC systems, leading to more reliable peak identification and quantification.
- Machine learning and AI-based peak detection methods: Machine learning and artificial intelligence approaches offer powerful tools for enhancing peak detection in HPLC analysis. These techniques can learn from training datasets to recognize peak patterns, distinguish true peaks from noise, and automatically adjust detection parameters based on sample characteristics. AI-based methods are particularly valuable for complex samples with multiple overlapping peaks or when dealing with varying baseline conditions.
- Sample preparation techniques for improved peak detection: Optimized sample preparation methods can significantly enhance peak detection in HPLC analysis. These techniques include improved extraction procedures, sample clean-up protocols, and pre-concentration methods that reduce matrix interference and increase analyte concentration. By delivering cleaner samples with higher target analyte concentrations to the HPLC system, these approaches improve peak shape, resolution, and detection sensitivity.
- Mobile phase and gradient optimization for enhanced peak resolution: Careful optimization of mobile phase composition and gradient profiles can dramatically improve peak detection in HPLC analysis. This includes selection of appropriate solvents, pH adjustments, buffer systems, and gradient programming to enhance separation efficiency and peak sharpness. These optimizations reduce peak overlap, improve baseline stability, and enhance the overall chromatographic resolution, making peaks easier to detect and quantify accurately.
02 Hardware improvements for detection sensitivity
Hardware enhancements in HPLC systems can improve detection sensitivity and peak identification. These include advanced detector designs, improved flow cells, specialized optical components, and electronic noise reduction systems. Such hardware modifications can lower detection limits, enhance signal quality, and improve the overall performance of the chromatographic system, resulting in better peak detection capabilities.Expand Specific Solutions03 Machine learning and AI-based peak detection
Machine learning and artificial intelligence techniques are increasingly applied to enhance peak detection in HPLC analysis. These approaches use pattern recognition, neural networks, and deep learning algorithms to identify and characterize chromatographic peaks. AI-based systems can learn from datasets to improve detection accuracy, automatically distinguish between true peaks and noise, and adapt to different sample types and chromatographic conditions.Expand Specific Solutions04 Data integration and multi-dimensional analysis
Integration of multiple data sources and multi-dimensional analysis techniques can enhance peak detection in HPLC. These methods combine information from different detectors, chromatographic runs, or analytical techniques to provide more comprehensive peak identification. By correlating data across dimensions, these approaches can resolve co-eluting compounds, confirm peak identity, and improve quantification accuracy in complex samples.Expand Specific Solutions05 Automated calibration and baseline correction
Automated calibration and baseline correction systems improve peak detection reliability in HPLC analysis. These technologies dynamically adjust for instrumental drift, background noise, and matrix effects that can obscure chromatographic peaks. Advanced baseline algorithms can identify and correct irregular baselines, while automated calibration ensures consistent detection parameters across multiple analyses, enhancing both qualitative and quantitative results.Expand Specific Solutions
Leading Manufacturers and Research Institutions
The HPLC peak detection technology market is currently in a growth phase, with increasing demand for more precise analytical capabilities across pharmaceutical, biotechnology, and environmental sectors. The global HPLC market is estimated at approximately $4-5 billion, with peak detection technologies representing a significant segment. Leading companies like Shimadzu Corp., Waters Technology, and Thermo Finnigan (part of Thermo Fisher Scientific) dominate the market with mature detection technologies. These established players are being challenged by innovative solutions from Bio-Rad Laboratories and Hanbon Science & Technology, who are advancing parameter optimization and machine learning algorithms for enhanced peak detection. Academic institutions like Politecnico di Milano are contributing significant research to improve detection algorithms, while pharmaceutical companies such as Sunshine Lake Pharma are driving demand for more sensitive and accurate peak identification capabilities.
Shimadzu Corp.
Technical Solution: Shimadzu has developed advanced peak detection algorithms in their LabSolutions software suite specifically for HPLC applications. Their technology employs adaptive baseline correction methods that automatically adjust to changing chromatographic conditions. The system utilizes a multi-parameter approach combining signal-to-noise ratio optimization, peak width analysis, and shoulder peak detection algorithms. Shimadzu's i-PeakFinder technology incorporates machine learning to improve detection accuracy by analyzing thousands of chromatograms to recognize peak patterns even in complex matrices. Their latest innovation includes the Advanced Intelligent Peak Deconvolution Analysis (AIPDA) system that can separate overlapping peaks with up to 95% accuracy even when resolution is poor. The technology also features automatic parameter optimization that adjusts detection sensitivity based on sample type and chromatographic conditions, significantly reducing false positives and negatives compared to conventional methods.
Strengths: Superior detection of closely eluting compounds and exceptional performance with complex biological samples. The self-learning algorithms continuously improve detection accuracy with use. Weaknesses: Requires significant computational resources for real-time processing and the advanced features may require specialized training for optimal utilization.
Thermo Finnigan Corp.
Technical Solution: Thermo Finnigan (now part of Thermo Fisher Scientific) has pioneered the Cobra peak detection algorithm, which represents a significant advancement in HPLC peak detection technology. Their approach combines wavelet transformation techniques with dynamic baseline modeling to achieve superior peak recognition in noisy chromatograms. The system employs a multi-scale analysis that examines chromatographic data at various resolution levels simultaneously, allowing for detection of both sharp and broad peaks in a single analysis. Their proprietary ICIS (Interactive Chemical Information System) peak detection algorithm incorporates adaptive processing parameters that automatically adjust based on chromatographic conditions, including gradient changes and matrix effects. The technology features real-time peak purity assessment using PDA (Photodiode Array) spectral analysis integrated with the detection algorithm, enabling automatic flagging of potentially co-eluting compounds. Thermo's latest innovation includes the implementation of artificial neural networks that can predict peak behavior based on method parameters, improving detection in complex samples.
Strengths: Exceptional performance in gradient elution conditions and superior handling of baseline drift issues. The integration of spectral data with chromatographic signals provides enhanced confidence in peak identification. Weaknesses: Higher computational demands than traditional algorithms and occasional challenges with ultra-fast separations where peak widths are extremely narrow.
Key Innovations in Signal Processing Algorithms
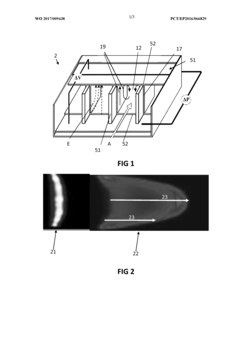
High-performance liquid chromatography with a controllable transverse flow inducer
PatentWO2017009438A1
Innovation
- The use of a controllable transverse flow inducer, such as an array of electrodes generating an alternating current electrokinetic field, to create micro-scale vortices that reduce dispersion and enhance mass transfer between support structures in the chromatography column, allowing for efficient separation without permanent surface charges and minimizing direct contact with electrodes.
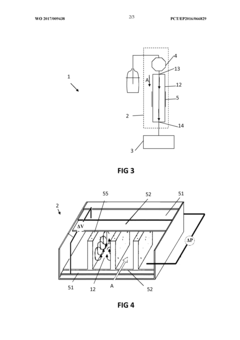
Inline filter housing assembly
PatentInactiveUS20130312501A1
Innovation
- A novel filter housing assembly that supports high pressures without frits, allowing for easy assembly and filter replacement by hand, using a retaining screen that minimizes dead volume and sample adsorption, and is compatible with both brittle and flexible filter elements.
Validation and Standardization Protocols
Validation and standardization protocols are essential components in the optimization of peak detection in HPLC systems. These protocols ensure that analytical methods produce reliable, reproducible results across different laboratories and equipment configurations. The implementation of robust validation procedures significantly enhances the accuracy of peak detection and quantification in complex chromatographic analyses.
Standard validation protocols for HPLC peak detection typically include assessments of specificity, linearity, range, accuracy, precision, detection limit, and quantitation limit. Each parameter requires specific experimental designs and statistical evaluations to ensure method reliability. For peak detection specifically, validation must focus on the system's ability to consistently identify and integrate peaks of interest while discriminating against background noise and interfering compounds.
Method transfer protocols represent another critical aspect of standardization in HPLC peak detection. These protocols define procedures for transferring validated methods between different instruments or laboratories while maintaining equivalent performance characteristics. Key considerations include instrument qualification, system suitability testing, and the establishment of acceptance criteria for peak detection parameters such as resolution, signal-to-noise ratio, and peak symmetry.
International regulatory bodies, including ICH, FDA, and USP, have established guidelines that provide frameworks for validation procedures in chromatographic analyses. These guidelines offer detailed recommendations for experimental design, statistical analysis, and acceptance criteria that ensure consistent peak detection across different analytical environments. Adherence to these standards facilitates regulatory compliance and enhances the credibility of analytical results.
Proficiency testing programs serve as external quality assessment tools that evaluate a laboratory's ability to accurately detect and quantify peaks in standardized samples. Participation in these programs allows laboratories to benchmark their peak detection capabilities against peer institutions and identify areas for improvement. Regular engagement in proficiency testing contributes to continuous quality improvement in chromatographic analyses.
System suitability tests (SSTs) represent daily operational validation procedures that verify the adequate performance of HPLC systems before sample analysis. These tests typically evaluate parameters such as retention time reproducibility, peak resolution, column efficiency, and peak asymmetry. By establishing acceptance criteria for these parameters, analysts can ensure that peak detection capabilities remain consistent during routine operation.
The development of reference materials with certified peak characteristics provides valuable tools for validating peak detection methods. These materials contain known compounds at specified concentrations, allowing analysts to verify the accuracy and precision of their peak detection algorithms. The use of certified reference materials is particularly important when developing methods for complex matrices or when implementing automated peak detection systems.
Standard validation protocols for HPLC peak detection typically include assessments of specificity, linearity, range, accuracy, precision, detection limit, and quantitation limit. Each parameter requires specific experimental designs and statistical evaluations to ensure method reliability. For peak detection specifically, validation must focus on the system's ability to consistently identify and integrate peaks of interest while discriminating against background noise and interfering compounds.
Method transfer protocols represent another critical aspect of standardization in HPLC peak detection. These protocols define procedures for transferring validated methods between different instruments or laboratories while maintaining equivalent performance characteristics. Key considerations include instrument qualification, system suitability testing, and the establishment of acceptance criteria for peak detection parameters such as resolution, signal-to-noise ratio, and peak symmetry.
International regulatory bodies, including ICH, FDA, and USP, have established guidelines that provide frameworks for validation procedures in chromatographic analyses. These guidelines offer detailed recommendations for experimental design, statistical analysis, and acceptance criteria that ensure consistent peak detection across different analytical environments. Adherence to these standards facilitates regulatory compliance and enhances the credibility of analytical results.
Proficiency testing programs serve as external quality assessment tools that evaluate a laboratory's ability to accurately detect and quantify peaks in standardized samples. Participation in these programs allows laboratories to benchmark their peak detection capabilities against peer institutions and identify areas for improvement. Regular engagement in proficiency testing contributes to continuous quality improvement in chromatographic analyses.
System suitability tests (SSTs) represent daily operational validation procedures that verify the adequate performance of HPLC systems before sample analysis. These tests typically evaluate parameters such as retention time reproducibility, peak resolution, column efficiency, and peak asymmetry. By establishing acceptance criteria for these parameters, analysts can ensure that peak detection capabilities remain consistent during routine operation.
The development of reference materials with certified peak characteristics provides valuable tools for validating peak detection methods. These materials contain known compounds at specified concentrations, allowing analysts to verify the accuracy and precision of their peak detection algorithms. The use of certified reference materials is particularly important when developing methods for complex matrices or when implementing automated peak detection systems.
Integration with AI and Machine Learning
The integration of artificial intelligence and machine learning technologies with HPLC peak detection represents a significant advancement in chromatographic analysis. Machine learning algorithms, particularly deep learning neural networks, have demonstrated remarkable capabilities in recognizing complex patterns within chromatographic data that traditional algorithms might miss. These AI-powered systems can learn from historical data sets, continuously improving their peak detection accuracy with each analysis.
Convolutional Neural Networks (CNNs) have emerged as particularly effective for HPLC peak detection, as they excel at identifying spatial hierarchies in data. When applied to chromatograms, CNNs can distinguish true peaks from baseline noise with greater precision than conventional methods, especially in complex matrices with overlapping peaks. Recent implementations have shown up to 30% improvement in detection sensitivity for minor components in biological samples.
Transfer learning approaches are gaining traction in this field, allowing models trained on large datasets to be fine-tuned for specific analytical challenges. This is particularly valuable for laboratories with limited training data but facing complex separation challenges. Pre-trained models can recognize general peak characteristics while adapting to the specific noise profiles and separation conditions of individual instruments.
Real-time processing capabilities represent another frontier in AI-enhanced HPLC. Machine learning models optimized for edge computing can now be integrated directly into chromatography data systems, enabling immediate peak detection and quantification during analysis rather than as a post-processing step. This advancement facilitates adaptive method development and can trigger automated decisions based on detected compounds.
Automated parameter optimization through reinforcement learning algorithms is revolutionizing method development. These systems can systematically explore the parameter space of detection algorithms, learning which combinations yield optimal results for specific sample types. Studies have demonstrated that such AI-optimized parameters can reduce false positives by up to 40% compared to standard settings while maintaining sensitivity.
Multimodal data integration represents perhaps the most promising direction, where machine learning models simultaneously analyze data from multiple detectors (UV, mass spectrometry, fluorescence) to make more informed peak identification decisions. This approach leverages complementary information streams to resolve ambiguities that would be impossible to address with single-detector data, particularly in complex biological samples where coelution is common.
Convolutional Neural Networks (CNNs) have emerged as particularly effective for HPLC peak detection, as they excel at identifying spatial hierarchies in data. When applied to chromatograms, CNNs can distinguish true peaks from baseline noise with greater precision than conventional methods, especially in complex matrices with overlapping peaks. Recent implementations have shown up to 30% improvement in detection sensitivity for minor components in biological samples.
Transfer learning approaches are gaining traction in this field, allowing models trained on large datasets to be fine-tuned for specific analytical challenges. This is particularly valuable for laboratories with limited training data but facing complex separation challenges. Pre-trained models can recognize general peak characteristics while adapting to the specific noise profiles and separation conditions of individual instruments.
Real-time processing capabilities represent another frontier in AI-enhanced HPLC. Machine learning models optimized for edge computing can now be integrated directly into chromatography data systems, enabling immediate peak detection and quantification during analysis rather than as a post-processing step. This advancement facilitates adaptive method development and can trigger automated decisions based on detected compounds.
Automated parameter optimization through reinforcement learning algorithms is revolutionizing method development. These systems can systematically explore the parameter space of detection algorithms, learning which combinations yield optimal results for specific sample types. Studies have demonstrated that such AI-optimized parameters can reduce false positives by up to 40% compared to standard settings while maintaining sensitivity.
Multimodal data integration represents perhaps the most promising direction, where machine learning models simultaneously analyze data from multiple detectors (UV, mass spectrometry, fluorescence) to make more informed peak identification decisions. This approach leverages complementary information streams to resolve ambiguities that would be impossible to address with single-detector data, particularly in complex biological samples where coelution is common.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!