Navigating Complex Buffers in HPLC for Compatibility
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Buffer Compatibility Background and Objectives
High-performance liquid chromatography (HPLC) has evolved significantly since its inception in the 1960s, becoming an indispensable analytical technique in pharmaceutical, biotechnology, environmental, and food industries. The development of buffer systems for HPLC has paralleled this evolution, with increasing complexity to meet the demands of separating diverse analytes under various conditions. Buffer compatibility represents a critical aspect of HPLC methodology that directly impacts separation efficiency, column longevity, and instrument performance.
The historical progression of HPLC buffer systems began with simple aqueous-organic mobile phases and has advanced to sophisticated buffer formulations designed to control pH precisely, enhance selectivity, and maintain sample stability. This evolution has been driven by the need to analyze increasingly complex molecules, including proteins, peptides, nucleic acids, and small molecule pharmaceuticals with varying physicochemical properties.
Current trends in HPLC buffer technology focus on developing robust buffer systems that maintain compatibility across different detection methods, column chemistries, and sample matrices. The industry is moving toward buffer formulations that minimize salt precipitation, reduce corrosion of instrument components, and prevent microbial growth while maintaining chromatographic performance.
The primary objective of navigating complex buffers in HPLC is to establish systematic approaches for selecting, preparing, and utilizing buffer systems that optimize separation while avoiding common compatibility issues. This includes understanding the chemical interactions between buffer components, mobile phase modifiers, analytes, and stationary phases under various operational conditions.
Specific technical goals include developing predictive models for buffer compatibility, creating standardized protocols for buffer preparation and storage, and identifying novel buffer formulations that extend the application range of HPLC. Additionally, there is a growing emphasis on environmentally friendly buffer systems that reduce waste and toxicity while maintaining analytical performance.
The advancement of HPLC buffer technology intersects with emerging trends in analytical chemistry, including miniaturization, automation, and integration with mass spectrometry and other detection techniques. This convergence necessitates buffer systems that function optimally across multiple analytical platforms while maintaining chemical compatibility with diverse sample types.
Understanding the fundamental principles governing buffer behavior in chromatographic systems remains essential for addressing current limitations and developing next-generation solutions. This includes elucidating the molecular mechanisms of buffer-analyte interactions, buffer-stationary phase interactions, and the impact of temperature, pressure, and organic modifiers on buffer performance.
The historical progression of HPLC buffer systems began with simple aqueous-organic mobile phases and has advanced to sophisticated buffer formulations designed to control pH precisely, enhance selectivity, and maintain sample stability. This evolution has been driven by the need to analyze increasingly complex molecules, including proteins, peptides, nucleic acids, and small molecule pharmaceuticals with varying physicochemical properties.
Current trends in HPLC buffer technology focus on developing robust buffer systems that maintain compatibility across different detection methods, column chemistries, and sample matrices. The industry is moving toward buffer formulations that minimize salt precipitation, reduce corrosion of instrument components, and prevent microbial growth while maintaining chromatographic performance.
The primary objective of navigating complex buffers in HPLC is to establish systematic approaches for selecting, preparing, and utilizing buffer systems that optimize separation while avoiding common compatibility issues. This includes understanding the chemical interactions between buffer components, mobile phase modifiers, analytes, and stationary phases under various operational conditions.
Specific technical goals include developing predictive models for buffer compatibility, creating standardized protocols for buffer preparation and storage, and identifying novel buffer formulations that extend the application range of HPLC. Additionally, there is a growing emphasis on environmentally friendly buffer systems that reduce waste and toxicity while maintaining analytical performance.
The advancement of HPLC buffer technology intersects with emerging trends in analytical chemistry, including miniaturization, automation, and integration with mass spectrometry and other detection techniques. This convergence necessitates buffer systems that function optimally across multiple analytical platforms while maintaining chemical compatibility with diverse sample types.
Understanding the fundamental principles governing buffer behavior in chromatographic systems remains essential for addressing current limitations and developing next-generation solutions. This includes elucidating the molecular mechanisms of buffer-analyte interactions, buffer-stationary phase interactions, and the impact of temperature, pressure, and organic modifiers on buffer performance.
Market Demand Analysis for Compatible HPLC Buffer Systems
The global HPLC (High-Performance Liquid Chromatography) buffer market has been experiencing robust growth, driven by expanding applications in pharmaceutical research, biotechnology, and clinical diagnostics. Current market assessments value the HPLC reagents and consumables sector at approximately USD 4.5 billion, with buffer systems representing a significant segment showing annual growth rates of 6-7%.
The pharmaceutical industry remains the largest consumer of HPLC buffer systems, accounting for nearly 45% of market demand. This dominance stems from stringent regulatory requirements for drug development and quality control processes, where buffer compatibility and stability are critical factors. Research institutions and academic laboratories constitute the second-largest market segment, contributing roughly 25% of the demand.
A notable market trend is the increasing preference for ready-to-use buffer solutions over traditional self-prepared buffers. This shift is evidenced by the 15% year-over-year growth in pre-packaged buffer sales, reflecting laboratories' focus on efficiency, reproducibility, and minimizing preparation errors. The premium pricing of these solutions, typically 30-40% higher than raw materials, has not deterred adoption due to their convenience and reliability benefits.
Geographically, North America leads the market with approximately 38% share, followed by Europe (30%) and Asia-Pacific (25%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth trajectory with rates exceeding 9% annually, driven by expanding pharmaceutical manufacturing capabilities and increasing R&D investments.
Customer surveys reveal that buffer compatibility issues represent a significant pain point, with 67% of HPLC users reporting challenges related to buffer interactions with analytes, columns, or detection systems. This has created substantial demand for buffer systems specifically designed to address compatibility concerns across diverse analytical conditions.
The market is witnessing increased demand for specialized buffer systems that offer extended stability, broader pH ranges, and enhanced compatibility with various detection methods, particularly mass spectrometry. Mass spectrometry-compatible volatile buffers have seen demand growth of approximately 12% annually, outpacing the overall market.
Environmental considerations are also shaping market dynamics, with growing interest in biodegradable and less toxic buffer components. This trend is particularly evident in academic and government research settings, where 38% of procurement decisions now incorporate sustainability criteria alongside performance metrics.
The pharmaceutical industry remains the largest consumer of HPLC buffer systems, accounting for nearly 45% of market demand. This dominance stems from stringent regulatory requirements for drug development and quality control processes, where buffer compatibility and stability are critical factors. Research institutions and academic laboratories constitute the second-largest market segment, contributing roughly 25% of the demand.
A notable market trend is the increasing preference for ready-to-use buffer solutions over traditional self-prepared buffers. This shift is evidenced by the 15% year-over-year growth in pre-packaged buffer sales, reflecting laboratories' focus on efficiency, reproducibility, and minimizing preparation errors. The premium pricing of these solutions, typically 30-40% higher than raw materials, has not deterred adoption due to their convenience and reliability benefits.
Geographically, North America leads the market with approximately 38% share, followed by Europe (30%) and Asia-Pacific (25%). The Asia-Pacific region, particularly China and India, demonstrates the fastest growth trajectory with rates exceeding 9% annually, driven by expanding pharmaceutical manufacturing capabilities and increasing R&D investments.
Customer surveys reveal that buffer compatibility issues represent a significant pain point, with 67% of HPLC users reporting challenges related to buffer interactions with analytes, columns, or detection systems. This has created substantial demand for buffer systems specifically designed to address compatibility concerns across diverse analytical conditions.
The market is witnessing increased demand for specialized buffer systems that offer extended stability, broader pH ranges, and enhanced compatibility with various detection methods, particularly mass spectrometry. Mass spectrometry-compatible volatile buffers have seen demand growth of approximately 12% annually, outpacing the overall market.
Environmental considerations are also shaping market dynamics, with growing interest in biodegradable and less toxic buffer components. This trend is particularly evident in academic and government research settings, where 38% of procurement decisions now incorporate sustainability criteria alongside performance metrics.
Current Challenges in Complex Buffer Management
Despite significant advancements in HPLC technology, complex buffer management remains one of the most challenging aspects for analytical chemists and researchers. The primary challenge lies in ensuring buffer compatibility with both the analyte and the chromatographic system while maintaining separation efficiency. Buffer-related issues account for approximately 35% of method failures in pharmaceutical analysis, highlighting the critical nature of this challenge.
Buffer precipitation presents a significant obstacle, particularly when mixing buffers with organic solvents. This phenomenon can damage expensive equipment, clog columns, and produce inconsistent results. For instance, phosphate buffers, widely used for their excellent buffering capacity, frequently precipitate at high organic solvent concentrations (typically above 50% acetonitrile), creating operational difficulties and reducing column lifespan.
pH stability across gradient elution represents another major challenge. As the proportion of organic solvent increases during gradient runs, the effective pH of the mobile phase can shift significantly, sometimes by as much as 2 pH units. This shift alters analyte ionization states, leading to peak broadening, retention time shifts, and compromised resolution. The challenge intensifies when analyzing compounds with multiple ionizable groups, where small pH changes can dramatically affect separation profiles.
Buffer-induced metal leaching from stainless steel components poses an underappreciated but serious challenge. Halide-containing buffers (particularly chloride) and those with chelating properties can promote corrosion of system components, introducing metal contaminants that may interfere with analysis or damage sensitive detectors like mass spectrometers. This issue is particularly problematic in trace analysis applications where even minimal contamination can compromise results.
Microbial growth in buffer solutions represents a persistent challenge, especially in methods requiring extended run times or when instruments operate continuously. Phosphate and acetate buffers at neutral pH provide ideal conditions for microbial proliferation, leading to column contamination, pressure fluctuations, and baseline instability. Current preventive measures often involve addition of organic solvents or antimicrobial agents, which may themselves introduce compatibility issues.
The increasing complexity of biological samples analyzed by HPLC compounds these challenges. Proteins, peptides, and other biomolecules often require specialized buffer systems that maintain sample integrity while enabling effective separation. These buffers must balance ionic strength, pH stability, and solubility considerations while avoiding detrimental interactions with both the sample and the chromatographic system. The need for MS-compatible buffers further restricts options, as traditional buffers like phosphates and borates are generally incompatible with mass spectrometric detection.
Buffer precipitation presents a significant obstacle, particularly when mixing buffers with organic solvents. This phenomenon can damage expensive equipment, clog columns, and produce inconsistent results. For instance, phosphate buffers, widely used for their excellent buffering capacity, frequently precipitate at high organic solvent concentrations (typically above 50% acetonitrile), creating operational difficulties and reducing column lifespan.
pH stability across gradient elution represents another major challenge. As the proportion of organic solvent increases during gradient runs, the effective pH of the mobile phase can shift significantly, sometimes by as much as 2 pH units. This shift alters analyte ionization states, leading to peak broadening, retention time shifts, and compromised resolution. The challenge intensifies when analyzing compounds with multiple ionizable groups, where small pH changes can dramatically affect separation profiles.
Buffer-induced metal leaching from stainless steel components poses an underappreciated but serious challenge. Halide-containing buffers (particularly chloride) and those with chelating properties can promote corrosion of system components, introducing metal contaminants that may interfere with analysis or damage sensitive detectors like mass spectrometers. This issue is particularly problematic in trace analysis applications where even minimal contamination can compromise results.
Microbial growth in buffer solutions represents a persistent challenge, especially in methods requiring extended run times or when instruments operate continuously. Phosphate and acetate buffers at neutral pH provide ideal conditions for microbial proliferation, leading to column contamination, pressure fluctuations, and baseline instability. Current preventive measures often involve addition of organic solvents or antimicrobial agents, which may themselves introduce compatibility issues.
The increasing complexity of biological samples analyzed by HPLC compounds these challenges. Proteins, peptides, and other biomolecules often require specialized buffer systems that maintain sample integrity while enabling effective separation. These buffers must balance ionic strength, pH stability, and solubility considerations while avoiding detrimental interactions with both the sample and the chromatographic system. The need for MS-compatible buffers further restricts options, as traditional buffers like phosphates and borates are generally incompatible with mass spectrometric detection.
Current Buffer Compatibility Solutions and Approaches
01 Buffer composition for HPLC mobile phases
Specific buffer compositions are critical for HPLC analysis to maintain proper pH control and ensure compatibility with various analytes. Complex buffer systems can include phosphate, acetate, or citrate buffers that provide stable pH environments while minimizing interactions with stationary phases. The selection of appropriate buffer components affects retention time reproducibility, peak shape, and overall chromatographic performance.- Buffer composition for HPLC mobile phases: Complex buffer systems in HPLC mobile phases can significantly impact chromatographic performance. These buffers typically consist of acid-base pairs that maintain a stable pH during separation. The composition of these buffers affects retention time, peak shape, and resolution. Properly formulated buffers can enhance the compatibility between the analyte and the stationary phase, leading to improved separation efficiency and reproducibility in analytical methods.
- pH control and stability in HPLC systems: Maintaining precise pH control is critical for HPLC analysis, especially when dealing with ionizable compounds. Complex buffer systems provide enhanced pH stability across varying conditions, preventing pH drift during separation. The selection of appropriate buffer capacity and concentration is essential to ensure consistent chromatographic performance. Stable pH conditions contribute to reproducible retention times and improved peak symmetry, particularly important for method validation and transfer between different HPLC systems.
- Compatibility of buffers with mass spectrometry: When coupling HPLC with mass spectrometry detection, buffer compatibility becomes a critical consideration. Volatile buffers such as ammonium acetate, ammonium formate, and their acid counterparts are preferred as they do not form non-volatile residues that could contaminate the ion source. The concentration of these buffers must be optimized to provide adequate chromatographic performance while minimizing ion suppression effects in the mass spectrometer, ensuring sensitive and reliable detection of analytes.
- Buffer interactions with stationary phases: Complex buffers can interact with HPLC stationary phases in ways that affect separation performance. These interactions may include ion-exchange mechanisms, surface adsorption, or modification of the stationary phase characteristics. Understanding these interactions is crucial for method development, particularly when working with specialty columns such as HILIC, ion-exchange, or mixed-mode phases. Proper buffer selection can enhance column lifetime and maintain consistent chromatographic behavior across multiple injections.
- Temperature effects on buffer performance: Temperature significantly impacts the behavior of complex buffers in HPLC systems. As temperature changes, buffer pH, solubility, and interaction with analytes can all be affected. This is particularly important for temperature-controlled separations where buffer capacity must remain consistent across the operational temperature range. Optimizing buffer composition for specific temperature conditions can improve method robustness and ensure consistent chromatographic performance across different laboratory environments.
02 Compatibility of buffers with mass spectrometry detection
When coupling HPLC with mass spectrometry, buffer selection requires special consideration as many traditional buffers can cause ion suppression or contamination of the ion source. Volatile buffers such as ammonium acetate, ammonium formate, and ammonium bicarbonate are preferred for LC-MS applications. The concentration and pH of these buffers must be optimized to maintain chromatographic separation while ensuring MS compatibility.Expand Specific Solutions03 pH stability and gradient compatibility of complex buffers
Complex buffer systems must maintain pH stability throughout gradient elution conditions in HPLC. As organic solvent concentration changes during gradient runs, buffer capacity can be affected, potentially leading to pH shifts. Properly designed buffer systems with adequate capacity and appropriate pKa values relative to the target pH range ensure consistent chromatographic conditions throughout the analysis.Expand Specific Solutions04 Buffer interactions with stationary phases and analytes
Complex buffers can interact with both stationary phases and analytes in HPLC systems. These interactions may include ion-exchange effects, metal chelation, or competitive binding. Understanding and controlling these interactions is essential for developing robust methods, particularly for analyzing biomolecules, pharmaceuticals, and other complex samples. Buffer components must be selected to minimize unwanted interactions while enhancing desired separation mechanisms.Expand Specific Solutions05 Buffer preparation and stability considerations
Preparation and storage of complex buffer solutions for HPLC require specific protocols to ensure reproducibility and stability. Factors such as dissolution order of components, filtration requirements, and storage conditions affect buffer performance. Some buffer systems are prone to microbial growth or precipitation over time, necessitating fresh preparation or addition of preservatives. Proper documentation and standardization of buffer preparation procedures are essential for method validation and transfer.Expand Specific Solutions
Key Industry Players in HPLC Buffer Solutions
The HPLC buffer compatibility landscape is currently in a mature growth phase, with an estimated market size of $3-4 billion and steady annual growth of 5-7%. The competitive environment features established analytical instrumentation leaders like IBM and Quest Diagnostics providing comprehensive buffer solutions, alongside specialized innovators such as Merlin Biomedical and SambaNova Systems developing novel compatibility technologies. Academic-industry partnerships with institutions like Anhui University and Hunan University are accelerating technological advancements. The market shows regional segmentation with Sharp, ZTE, and China Telecom dominating Asian markets, while QUALCOMM and Microsoft lead Western markets with integrated buffer systems that address increasingly complex separation challenges in pharmaceutical and biomedical applications.
Merlin Biomedical (Xiamen) Co., Ltd.
Technical Solution: Merlin Biomedical has pioneered the BioCompat HPLC Buffer System specifically designed for navigating complex buffer challenges in pharmaceutical and biomedical applications. Their approach integrates multiple technologies: 1) Adaptive pH gradient systems that maintain separation efficiency across diverse compound classes; 2) Novel zwitterionic buffer additives that enhance compatibility with both hydrophilic and hydrophobic analytes; 3) Proprietary buffer stabilization technology that extends buffer shelf-life by preventing microbial growth and chemical degradation; and 4) Intelligent buffer selection software that recommends optimal buffer compositions based on analyte properties. The system features their patented BufferShield technology, which incorporates molecular crowding agents to protect sensitive biomolecules during analysis. Their buffers are specifically engineered to minimize metal ion leaching from HPLC components, critical for accurate analysis of metal-sensitive compounds. Merlin's system also includes specialized buffer formulations for compatibility with various detection methods, including UV, fluorescence, and mass spectrometry[3].
Strengths: Comprehensive approach addressing multiple buffer compatibility challenges; excellent versatility across different analyte types; strong focus on buffer stability and shelf-life. Weaknesses: Relatively new technology with limited long-term performance data; complex buffer systems may require more extensive method development; higher cost compared to conventional buffer solutions.
Quest Diagnostics Investments LLC
Technical Solution: Quest Diagnostics has developed advanced buffer compatibility systems for HPLC analysis in clinical diagnostics. Their proprietary BufferIQ technology addresses complex buffer challenges through a multi-component approach that includes: 1) Automated buffer selection algorithms that analyze sample composition and recommend optimal buffer systems; 2) Specialized buffer formulations with enhanced stability across varying pH ranges (4.0-8.0); 3) Predictive modeling software that anticipates potential buffer-analyte interactions; and 4) Real-time buffer adjustment capabilities during analysis runs. The system incorporates machine learning to continuously improve buffer selection based on historical performance data, particularly valuable for complex biological matrices in clinical testing. Quest's buffer systems are specifically engineered to maintain compatibility with mass spectrometry detection methods, reducing ion suppression effects commonly encountered with traditional buffers[1].
Strengths: Specialized expertise in clinical diagnostic applications; integrated approach combining software and chemistry solutions; proven effectiveness with complex biological matrices. Weaknesses: Solutions primarily optimized for clinical diagnostics rather than broader applications; proprietary nature may limit flexibility for custom research applications; potentially higher cost compared to standard buffer systems.
Critical Buffer Interaction Mechanisms and Patents
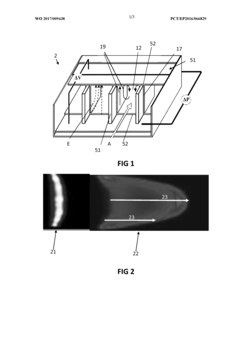
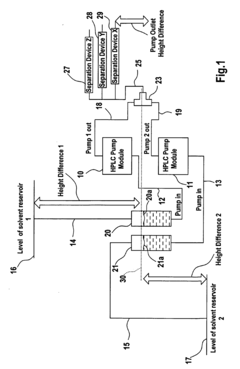
High-performance liquid chromatography with a controllable transverse flow inducer
PatentWO2017009438A1
Innovation
- The use of a controllable transverse flow inducer, such as an array of electrodes generating an alternating current electrokinetic field, to create micro-scale vortices that reduce dispersion and enhance mass transfer between support structures in the chromatography column, allowing for efficient separation without permanent surface charges and minimizing direct contact with electrodes.
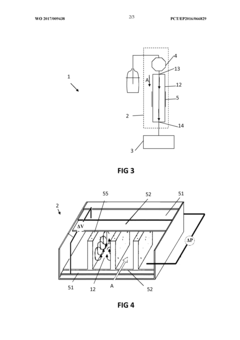
Pumping arrangement
PatentInactiveEP1376121A1
Innovation
- A pumping arrangement with filtration devices comprising a porous inner member within a borosilicate glass outer chamber, allowing two-phase systems to handle non-degassed solvents and prevent backflow, featuring reusable and sterilizable design to manage high flow rates and level differences between reservoirs.
Regulatory Compliance for HPLC Buffer Systems
Regulatory compliance for HPLC buffer systems represents a critical consideration in analytical method development across pharmaceutical, biotechnology, and clinical laboratory settings. The regulatory landscape governing HPLC buffer preparation and usage is multifaceted, encompassing guidelines from various international bodies including the FDA, EMA, ICH, and USP. These regulations establish stringent requirements for buffer quality, documentation, and validation processes.
The FDA's current Good Manufacturing Practices (cGMP) mandates comprehensive documentation of buffer preparation procedures, including detailed records of components, concentrations, pH adjustments, and filtration processes. Similarly, the EMA's guidelines emphasize traceability of all buffer components and validation of buffer stability under various storage conditions.
ICH Q2(R1) guidelines specifically address the validation of analytical procedures, requiring demonstration that HPLC buffer systems maintain consistent performance throughout their intended use period. This includes stability-indicating studies to verify that buffer degradation does not compromise analytical results.
Pharmacopeial standards, particularly those from USP and Ph. Eur., provide specific requirements for water quality used in buffer preparation. USP <1058> on Analytical Instrument Qualification outlines expectations for system suitability testing that directly relates to buffer performance and compatibility.
Environmental regulations also impact HPLC buffer systems, with increasing scrutiny on disposal practices for buffers containing hazardous components such as acetonitrile, methanol, and phosphate salts. Many jurisdictions now require documented waste management protocols for these materials.
Quality by Design (QbD) principles, endorsed by regulatory agencies, encourage systematic evaluation of buffer variables during method development. This approach necessitates understanding the design space within which buffer composition can vary while maintaining method performance, thereby establishing a regulatory-compliant operating range.
For complex buffer systems involving multiple ionic species or organic modifiers, additional validation may be required to demonstrate robustness across manufacturing lots and storage conditions. Regulatory bodies increasingly expect risk assessments that identify potential failure modes related to buffer incompatibility or instability.
Compliance documentation must include certificates of analysis for buffer components, validation of preparation equipment, and verification of personnel training. Electronic records pertaining to buffer preparation must comply with 21 CFR Part 11 requirements for data integrity and security.
As analytical methods become more sophisticated, regulatory expectations continue to evolve, with increasing emphasis on understanding the fundamental chemistry of buffer interactions rather than relying solely on empirical testing. This trend underscores the importance of scientific rigor in developing compatible buffer systems that meet both analytical performance requirements and regulatory compliance standards.
The FDA's current Good Manufacturing Practices (cGMP) mandates comprehensive documentation of buffer preparation procedures, including detailed records of components, concentrations, pH adjustments, and filtration processes. Similarly, the EMA's guidelines emphasize traceability of all buffer components and validation of buffer stability under various storage conditions.
ICH Q2(R1) guidelines specifically address the validation of analytical procedures, requiring demonstration that HPLC buffer systems maintain consistent performance throughout their intended use period. This includes stability-indicating studies to verify that buffer degradation does not compromise analytical results.
Pharmacopeial standards, particularly those from USP and Ph. Eur., provide specific requirements for water quality used in buffer preparation. USP <1058> on Analytical Instrument Qualification outlines expectations for system suitability testing that directly relates to buffer performance and compatibility.
Environmental regulations also impact HPLC buffer systems, with increasing scrutiny on disposal practices for buffers containing hazardous components such as acetonitrile, methanol, and phosphate salts. Many jurisdictions now require documented waste management protocols for these materials.
Quality by Design (QbD) principles, endorsed by regulatory agencies, encourage systematic evaluation of buffer variables during method development. This approach necessitates understanding the design space within which buffer composition can vary while maintaining method performance, thereby establishing a regulatory-compliant operating range.
For complex buffer systems involving multiple ionic species or organic modifiers, additional validation may be required to demonstrate robustness across manufacturing lots and storage conditions. Regulatory bodies increasingly expect risk assessments that identify potential failure modes related to buffer incompatibility or instability.
Compliance documentation must include certificates of analysis for buffer components, validation of preparation equipment, and verification of personnel training. Electronic records pertaining to buffer preparation must comply with 21 CFR Part 11 requirements for data integrity and security.
As analytical methods become more sophisticated, regulatory expectations continue to evolve, with increasing emphasis on understanding the fundamental chemistry of buffer interactions rather than relying solely on empirical testing. This trend underscores the importance of scientific rigor in developing compatible buffer systems that meet both analytical performance requirements and regulatory compliance standards.
Environmental Impact of HPLC Buffer Disposal
The disposal of HPLC buffers represents a significant environmental concern within analytical laboratories worldwide. These buffers, containing various salts, organic modifiers, and pH adjusters, can contribute to environmental pollution when improperly discharged. Most HPLC buffers contain phosphates, acetates, and other chemical compounds that may disrupt aquatic ecosystems by altering pH levels and introducing excessive nutrients, potentially leading to eutrophication in water bodies.
Traditional disposal methods often involve direct discharge into wastewater systems, which presents challenges for municipal treatment facilities not specifically designed to handle laboratory chemicals. Studies indicate that approximately 60-70% of HPLC buffer components pass through conventional wastewater treatment processes unchanged, eventually reaching natural water systems.
Organic modifiers in HPLC buffers, such as acetonitrile and methanol, pose particular environmental hazards due to their toxicity to aquatic organisms and persistence in the environment. Research has demonstrated that even low concentrations of these solvents (5-10 ppm) can adversely affect fish reproduction and development. Additionally, volatile organic compounds from these modifiers contribute to air pollution when evaporated during disposal processes.
Buffer salts containing heavy metals or toxic elements present another layer of environmental concern. These components can accumulate in sediments and bioaccumulate through food chains, potentially reaching concentrations harmful to wildlife and humans. Long-term studies have documented increased heavy metal concentrations in watersheds surrounding areas with high densities of analytical laboratories.
Regulatory frameworks addressing HPLC waste disposal vary significantly across regions, creating inconsistent environmental protection standards. The European Union's REACH regulations and the United States EPA guidelines provide frameworks for chemical waste management, but enforcement and compliance remain challenging, particularly in developing nations where analytical chemistry adoption is rapidly growing.
Sustainable alternatives for HPLC buffer disposal are emerging, including on-site treatment systems that neutralize pH, remove organic content, and precipitate harmful salts before discharge. Advanced oxidation processes and activated carbon filtration systems have demonstrated 85-95% reduction in environmental impact when properly implemented. Buffer recycling technologies are also gaining traction, with closed-loop systems capable of purifying and reusing certain buffer components.
Cost-benefit analyses indicate that while implementing environmentally responsible disposal methods increases operational costs by 15-20% initially, long-term benefits include regulatory compliance, reduced environmental liability, and potential cost savings through buffer recycling and reduced waste management fees.
Traditional disposal methods often involve direct discharge into wastewater systems, which presents challenges for municipal treatment facilities not specifically designed to handle laboratory chemicals. Studies indicate that approximately 60-70% of HPLC buffer components pass through conventional wastewater treatment processes unchanged, eventually reaching natural water systems.
Organic modifiers in HPLC buffers, such as acetonitrile and methanol, pose particular environmental hazards due to their toxicity to aquatic organisms and persistence in the environment. Research has demonstrated that even low concentrations of these solvents (5-10 ppm) can adversely affect fish reproduction and development. Additionally, volatile organic compounds from these modifiers contribute to air pollution when evaporated during disposal processes.
Buffer salts containing heavy metals or toxic elements present another layer of environmental concern. These components can accumulate in sediments and bioaccumulate through food chains, potentially reaching concentrations harmful to wildlife and humans. Long-term studies have documented increased heavy metal concentrations in watersheds surrounding areas with high densities of analytical laboratories.
Regulatory frameworks addressing HPLC waste disposal vary significantly across regions, creating inconsistent environmental protection standards. The European Union's REACH regulations and the United States EPA guidelines provide frameworks for chemical waste management, but enforcement and compliance remain challenging, particularly in developing nations where analytical chemistry adoption is rapidly growing.
Sustainable alternatives for HPLC buffer disposal are emerging, including on-site treatment systems that neutralize pH, remove organic content, and precipitate harmful salts before discharge. Advanced oxidation processes and activated carbon filtration systems have demonstrated 85-95% reduction in environmental impact when properly implemented. Buffer recycling technologies are also gaining traction, with closed-loop systems capable of purifying and reusing certain buffer components.
Cost-benefit analyses indicate that while implementing environmentally responsible disposal methods increases operational costs by 15-20% initially, long-term benefits include regulatory compliance, reduced environmental liability, and potential cost savings through buffer recycling and reduced waste management fees.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!