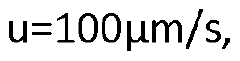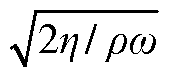Optimizing Data Collection in HPLC for Bioanalytical Results
SEP 19, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Bioanalytical Evolution and Objectives
High-performance liquid chromatography (HPLC) has undergone significant evolution since its inception in the 1960s, transforming from a rudimentary analytical technique to a sophisticated bioanalytical tool essential for pharmaceutical development, clinical diagnostics, and biological research. The trajectory of HPLC development has been characterized by continuous improvements in column technology, detection methods, automation capabilities, and data processing algorithms, all contributing to enhanced sensitivity, specificity, and throughput.
Early HPLC systems faced limitations in resolution, detection limits, and analysis speed, making comprehensive bioanalytical applications challenging. The introduction of reversed-phase chromatography in the 1970s marked a pivotal advancement, enabling more effective separation of biomolecules. Subsequent innovations in stationary phase chemistry, including the development of sub-2-micron particles and monolithic columns, have dramatically improved separation efficiency and reduced analysis time.
Detection technology has similarly progressed from simple UV-visible spectrophotometry to more sophisticated methods including fluorescence, electrochemical detection, and mass spectrometry coupling. The integration of HPLC with tandem mass spectrometry (LC-MS/MS) represents perhaps the most significant technological leap, enabling unprecedented sensitivity and selectivity for complex biological matrices analysis.
The current technological landscape is increasingly focused on miniaturization, automation, and integration with other analytical platforms. Ultra-high-performance liquid chromatography (UHPLC) systems operating at pressures exceeding 15,000 psi have become standard in many bioanalytical laboratories, offering superior resolution and faster analysis times compared to conventional HPLC.
Data collection optimization in bioanalytical HPLC aims to address several critical objectives. Primary among these is enhancing analytical sensitivity to detect increasingly lower concentrations of analytes in complex biological matrices. This is particularly crucial for pharmacokinetic studies, therapeutic drug monitoring, and biomarker discovery where trace analysis is often required.
Another key objective is improving throughput without compromising data quality, addressing the growing demand for high-volume sample analysis in clinical and research settings. This necessitates optimized sampling rates, detector response times, and data acquisition parameters tailored to specific analytical challenges.
Data integrity and reliability represent additional critical goals, requiring robust validation protocols, appropriate calibration procedures, and effective quality control measures. The optimization process must also consider regulatory compliance, particularly for methods used in drug development and clinical diagnostics where adherence to GLP/GMP standards is mandatory.
Looking forward, the field is trending toward intelligent data collection systems incorporating machine learning algorithms for real-time method optimization, predictive maintenance, and automated data interpretation. These developments align with broader industry movements toward laboratory digitalization and the implementation of artificial intelligence in analytical workflows.
Early HPLC systems faced limitations in resolution, detection limits, and analysis speed, making comprehensive bioanalytical applications challenging. The introduction of reversed-phase chromatography in the 1970s marked a pivotal advancement, enabling more effective separation of biomolecules. Subsequent innovations in stationary phase chemistry, including the development of sub-2-micron particles and monolithic columns, have dramatically improved separation efficiency and reduced analysis time.
Detection technology has similarly progressed from simple UV-visible spectrophotometry to more sophisticated methods including fluorescence, electrochemical detection, and mass spectrometry coupling. The integration of HPLC with tandem mass spectrometry (LC-MS/MS) represents perhaps the most significant technological leap, enabling unprecedented sensitivity and selectivity for complex biological matrices analysis.
The current technological landscape is increasingly focused on miniaturization, automation, and integration with other analytical platforms. Ultra-high-performance liquid chromatography (UHPLC) systems operating at pressures exceeding 15,000 psi have become standard in many bioanalytical laboratories, offering superior resolution and faster analysis times compared to conventional HPLC.
Data collection optimization in bioanalytical HPLC aims to address several critical objectives. Primary among these is enhancing analytical sensitivity to detect increasingly lower concentrations of analytes in complex biological matrices. This is particularly crucial for pharmacokinetic studies, therapeutic drug monitoring, and biomarker discovery where trace analysis is often required.
Another key objective is improving throughput without compromising data quality, addressing the growing demand for high-volume sample analysis in clinical and research settings. This necessitates optimized sampling rates, detector response times, and data acquisition parameters tailored to specific analytical challenges.
Data integrity and reliability represent additional critical goals, requiring robust validation protocols, appropriate calibration procedures, and effective quality control measures. The optimization process must also consider regulatory compliance, particularly for methods used in drug development and clinical diagnostics where adherence to GLP/GMP standards is mandatory.
Looking forward, the field is trending toward intelligent data collection systems incorporating machine learning algorithms for real-time method optimization, predictive maintenance, and automated data interpretation. These developments align with broader industry movements toward laboratory digitalization and the implementation of artificial intelligence in analytical workflows.
Market Demand for Precise Bioanalytical Data
The bioanalytical market has witnessed substantial growth in recent years, driven primarily by increasing demand for precise and reliable analytical data across pharmaceutical, biotechnology, and clinical research sectors. High-Performance Liquid Chromatography (HPLC) remains a cornerstone technology in this domain, with market analysts projecting the global HPLC market to reach $5.7 billion by 2025, growing at a CAGR of approximately 5.2% from 2020.
The pharmaceutical industry represents the largest consumer segment for bioanalytical HPLC applications, accounting for nearly 40% of the total market share. This dominance stems from stringent regulatory requirements for drug development and quality control processes, where precise bioanalytical data is non-negotiable for regulatory submissions and product approvals.
Clinical diagnostics constitutes another rapidly expanding segment, with hospitals and diagnostic laboratories increasingly adopting advanced HPLC techniques for therapeutic drug monitoring, vitamin analysis, and biomarker detection. The market in this sector is expected to grow at 6.8% annually through 2025, reflecting the critical importance of accurate bioanalytical results in clinical decision-making.
Contract Research Organizations (CROs) have emerged as significant market players, with many pharmaceutical companies outsourcing their bioanalytical testing needs. This trend has created a specialized market segment valued at approximately $1.2 billion in 2021, with projections indicating continued robust growth as drug development pipelines expand globally.
Geographically, North America leads the market with approximately 38% share, followed by Europe (30%) and Asia-Pacific (22%). However, the Asia-Pacific region demonstrates the fastest growth rate at 7.5% annually, driven by increasing R&D investments in China, India, and South Korea.
The demand for optimized data collection in HPLC is further intensified by the rise of personalized medicine approaches, which require highly sensitive and specific bioanalytical methods. Industry surveys indicate that 78% of pharmaceutical companies consider improved data quality and reproducibility as their top priorities when investing in new HPLC technologies.
Regulatory agencies worldwide have also raised their expectations regarding data integrity and analytical precision. The FDA, EMA, and other regulatory bodies now require more comprehensive validation data and stricter adherence to Good Laboratory Practices (GLP), directly influencing market demand for advanced HPLC data collection and management solutions.
Environmental analysis represents an emerging application area, with growing requirements for detecting pharmaceutical residues and other bioactive compounds in environmental samples, creating new market opportunities for specialized HPLC applications and data collection methodologies.
The pharmaceutical industry represents the largest consumer segment for bioanalytical HPLC applications, accounting for nearly 40% of the total market share. This dominance stems from stringent regulatory requirements for drug development and quality control processes, where precise bioanalytical data is non-negotiable for regulatory submissions and product approvals.
Clinical diagnostics constitutes another rapidly expanding segment, with hospitals and diagnostic laboratories increasingly adopting advanced HPLC techniques for therapeutic drug monitoring, vitamin analysis, and biomarker detection. The market in this sector is expected to grow at 6.8% annually through 2025, reflecting the critical importance of accurate bioanalytical results in clinical decision-making.
Contract Research Organizations (CROs) have emerged as significant market players, with many pharmaceutical companies outsourcing their bioanalytical testing needs. This trend has created a specialized market segment valued at approximately $1.2 billion in 2021, with projections indicating continued robust growth as drug development pipelines expand globally.
Geographically, North America leads the market with approximately 38% share, followed by Europe (30%) and Asia-Pacific (22%). However, the Asia-Pacific region demonstrates the fastest growth rate at 7.5% annually, driven by increasing R&D investments in China, India, and South Korea.
The demand for optimized data collection in HPLC is further intensified by the rise of personalized medicine approaches, which require highly sensitive and specific bioanalytical methods. Industry surveys indicate that 78% of pharmaceutical companies consider improved data quality and reproducibility as their top priorities when investing in new HPLC technologies.
Regulatory agencies worldwide have also raised their expectations regarding data integrity and analytical precision. The FDA, EMA, and other regulatory bodies now require more comprehensive validation data and stricter adherence to Good Laboratory Practices (GLP), directly influencing market demand for advanced HPLC data collection and management solutions.
Environmental analysis represents an emerging application area, with growing requirements for detecting pharmaceutical residues and other bioactive compounds in environmental samples, creating new market opportunities for specialized HPLC applications and data collection methodologies.
Current HPLC Data Collection Challenges
High-performance liquid chromatography (HPLC) has become an indispensable analytical technique in bioanalytical laboratories, yet data collection processes face significant challenges that impact result quality and reliability. Current HPLC systems generate massive volumes of data during bioanalytical testing, often overwhelming traditional data management infrastructures. A typical bioanalytical laboratory may produce terabytes of chromatographic data annually, creating substantial storage and processing demands.
Integration errors represent a persistent challenge in HPLC data collection. Peak integration algorithms frequently struggle with complex biological matrices, leading to inconsistent baseline determinations and peak boundary definitions. Studies indicate that integration variability can contribute up to 15% of total analytical error in bioanalytical methods, particularly when analyzing samples with low analyte concentrations or significant matrix interference.
Data integrity concerns have intensified with regulatory bodies implementing more stringent requirements. The FDA, EMA, and other regulatory agencies now demand complete data traceability, audit trails, and electronic signatures for all bioanalytical data. Many existing HPLC data collection systems lack robust compliance features, creating validation gaps that can jeopardize regulatory submissions.
Interoperability issues between different vendors' instruments and software platforms create significant workflow inefficiencies. Laboratories utilizing multiple HPLC systems often resort to manual data transfer and reformatting, introducing opportunities for transcription errors and data corruption. The absence of standardized data formats across the industry compounds this challenge, with an estimated 30% of analyst time spent on data manipulation rather than analysis.
Signal-to-noise optimization remains problematic, particularly for complex biological samples. Current detector technologies and data collection parameters often struggle to distinguish true analyte signals from background noise in challenging matrices like plasma, urine, or tissue homogenates. This limitation directly impacts method sensitivity and quantification limits, constraining bioanalytical capabilities.
Real-time data monitoring capabilities are frequently inadequate, preventing timely intervention during analytical runs. Most systems only allow comprehensive data evaluation after run completion, resulting in wasted resources when problems could have been identified and addressed earlier. This reactive rather than proactive approach extends analysis timelines and increases operational costs.
Data security vulnerabilities persist in many HPLC data collection systems. With increasing cybersecurity threats targeting scientific and healthcare organizations, inadequate encryption, access controls, and backup protocols pose significant risks to valuable bioanalytical data. Recent industry surveys indicate that over 40% of laboratories have experienced some form of data compromise or loss within the past five years.
Integration errors represent a persistent challenge in HPLC data collection. Peak integration algorithms frequently struggle with complex biological matrices, leading to inconsistent baseline determinations and peak boundary definitions. Studies indicate that integration variability can contribute up to 15% of total analytical error in bioanalytical methods, particularly when analyzing samples with low analyte concentrations or significant matrix interference.
Data integrity concerns have intensified with regulatory bodies implementing more stringent requirements. The FDA, EMA, and other regulatory agencies now demand complete data traceability, audit trails, and electronic signatures for all bioanalytical data. Many existing HPLC data collection systems lack robust compliance features, creating validation gaps that can jeopardize regulatory submissions.
Interoperability issues between different vendors' instruments and software platforms create significant workflow inefficiencies. Laboratories utilizing multiple HPLC systems often resort to manual data transfer and reformatting, introducing opportunities for transcription errors and data corruption. The absence of standardized data formats across the industry compounds this challenge, with an estimated 30% of analyst time spent on data manipulation rather than analysis.
Signal-to-noise optimization remains problematic, particularly for complex biological samples. Current detector technologies and data collection parameters often struggle to distinguish true analyte signals from background noise in challenging matrices like plasma, urine, or tissue homogenates. This limitation directly impacts method sensitivity and quantification limits, constraining bioanalytical capabilities.
Real-time data monitoring capabilities are frequently inadequate, preventing timely intervention during analytical runs. Most systems only allow comprehensive data evaluation after run completion, resulting in wasted resources when problems could have been identified and addressed earlier. This reactive rather than proactive approach extends analysis timelines and increases operational costs.
Data security vulnerabilities persist in many HPLC data collection systems. With increasing cybersecurity threats targeting scientific and healthcare organizations, inadequate encryption, access controls, and backup protocols pose significant risks to valuable bioanalytical data. Recent industry surveys indicate that over 40% of laboratories have experienced some form of data compromise or loss within the past five years.
Modern HPLC Data Collection Methodologies
01 HPLC system design and hardware components
Various hardware components and system designs for HPLC data collection, including specialized detectors, pumps, and column configurations. These systems are designed to improve detection sensitivity, data accuracy, and operational efficiency. Some designs incorporate automated sample handling and multiple detection channels for comprehensive analysis of complex samples.- HPLC system design and hardware components: Various hardware components and system designs for HPLC data collection, including specialized detectors, pumps, and column configurations. These systems are designed to optimize data acquisition accuracy, sensitivity, and throughput for different analytical applications. Innovations include integrated modules, automated sample handling, and specialized detection systems that enhance the quality of chromatographic data collection.
- Data acquisition and processing methods: Methods for acquiring, processing, and analyzing HPLC data, including signal processing algorithms, peak integration techniques, and data transformation approaches. These methods focus on improving signal-to-noise ratios, enhancing peak detection sensitivity, and providing more accurate quantitative analysis. Advanced mathematical models and algorithms are employed to extract meaningful information from chromatographic data and address common analytical challenges.
- Automation and high-throughput analysis: Automated systems and methods for high-throughput HPLC data collection, enabling efficient analysis of multiple samples with minimal human intervention. These innovations include robotic sample preparation, automated injection systems, parallel processing capabilities, and intelligent scheduling algorithms. The focus is on increasing analytical throughput while maintaining data quality and reducing operator-dependent variability in the analytical process.
- Quality control and validation techniques: Methods for ensuring the quality, reliability, and validity of HPLC data collection processes. These include system suitability testing, calibration procedures, internal standard methodologies, and statistical approaches for data validation. Techniques focus on identifying and minimizing sources of error, ensuring instrument performance consistency, and establishing traceability in analytical measurements to meet regulatory requirements and scientific standards.
- Specialized applications and method development: Specialized HPLC data collection methods developed for specific analytical challenges or sample types, including complex matrices, trace analysis, and challenging compound separations. These innovations include modified mobile phase compositions, gradient optimization strategies, and specialized detection techniques tailored to particular analytes or sample characteristics. Method development approaches focus on optimizing chromatographic parameters to achieve desired separation efficiency and data quality for specific analytical goals.
02 Data acquisition and processing methods
Methods for acquiring and processing HPLC data, including signal processing algorithms, peak detection techniques, and data integration approaches. These methods aim to improve the accuracy of quantitative analysis, reduce noise, and enhance the detection of trace components. Advanced software solutions enable real-time data processing and automated analysis of chromatographic results.Expand Specific Solutions03 Automated HPLC analysis and high-throughput screening
Automated systems and methods for high-throughput HPLC analysis, enabling rapid screening of multiple samples. These approaches incorporate robotics, parallel processing, and intelligent scheduling algorithms to maximize analytical efficiency. Such systems are particularly valuable in pharmaceutical development, quality control, and large-scale screening applications where numerous samples must be analyzed quickly.Expand Specific Solutions04 Mobile and cloud-based HPLC data collection
Mobile and cloud-based solutions for HPLC data collection, storage, and analysis. These technologies enable remote monitoring, real-time data sharing, and collaborative analysis across different locations. Cloud platforms provide secure data storage, advanced analytics capabilities, and integration with laboratory information management systems, enhancing workflow efficiency and data accessibility.Expand Specific Solutions05 Specialized HPLC applications and detection methods
Specialized HPLC applications and detection methods for specific analytical challenges, including bioanalysis, environmental monitoring, and pharmaceutical quality control. These approaches involve tailored sample preparation techniques, specialized column chemistries, and detection methods optimized for particular compound classes. Advanced detection technologies such as mass spectrometry coupling and multi-wavelength detection enhance the specificity and sensitivity of analysis.Expand Specific Solutions
Leading HPLC Instrument and Software Providers
The HPLC bioanalytical data collection optimization market is in a growth phase, with increasing demand driven by pharmaceutical and biotechnology research advancements. Major players include established analytical instrumentation companies like Waters Technology, Agilent Technologies, and Merck, alongside specialized biotech firms such as Biogen and Amgen. The technology landscape shows varying maturity levels, with companies like DH Technologies and Hitachi High-Tech focusing on innovative hardware solutions, while BASF and Samsung contribute advanced materials and semiconductor technologies. Academic institutions like New Jersey Institute of Technology and Jinan University provide research support. The market is characterized by increasing integration of AI and IoT capabilities, with competition intensifying between traditional analytical equipment manufacturers and emerging technology-focused entrants.
DH Technologies Development Pte Ltd.
Technical Solution: DH Technologies (parent company of SCIEX) has developed the SCIEX OS platform specifically optimized for bioanalytical HPLC-MS data collection. Their approach focuses on intelligent data acquisition strategies that dynamically adjust mass spectrometer parameters based on chromatographic conditions. The system implements scheduled MRM algorithms that optimize dwell times for each analyte based on retention time windows, maximizing sensitivity while maintaining adequate sampling across all compounds. Their technology incorporates automated calibration curve assessment during acquisition, flagging potential quantitation issues in real-time. SCIEX's platform features advanced peak integration algorithms specifically designed for bioanalytical applications, with automatic adjustment capabilities for challenging matrix interferences. The system also includes specialized data reduction protocols that optimize file sizes while preserving critical analytical information, addressing the challenges of managing large bioanalytical datasets.
Strengths: Exceptional mass spectrometry integration; industry-leading sensitivity for complex biological matrices; advanced scheduling algorithms for multi-analyte methods. Weaknesses: Steeper learning curve compared to some competitors; higher cost for full implementation; optimization primarily focused on MS detection rather than UV or other detection methods.
Merck Sharp & Dohme Corp.
Technical Solution: Merck has developed proprietary HPLC data collection optimization protocols specifically for bioanalytical applications in pharmaceutical development. Their approach integrates automated method development with multivariate statistical process control to ensure optimal data collection parameters for each specific bioanalytical assay. Merck's system implements adaptive sampling rate technology that automatically adjusts data acquisition frequency based on chromatographic peak characteristics, optimizing data density while minimizing file size. Their platform incorporates specialized calibration routines for bioanalytical matrices that compensate for matrix effects in real-time during data acquisition. Merck has also pioneered integrated internal standard monitoring algorithms that continuously evaluate internal standard response throughout analytical runs, automatically flagging potential extraction or ionization issues that could compromise data quality before batch completion.
Strengths: Pharmaceutical-specific optimization protocols; excellent matrix effect compensation; robust regulatory compliance features. Weaknesses: Solutions primarily optimized for pharmaceutical applications; less flexible for academic research applications; higher implementation complexity requiring specialized expertise.
Key Patents in HPLC Data Optimization
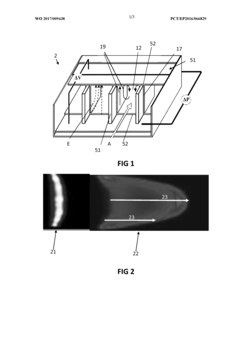
High-performance liquid chromatography with a controllable transverse flow inducer
PatentWO2017009438A1
Innovation
- The use of a controllable transverse flow inducer, such as an array of electrodes generating an alternating current electrokinetic field, to create micro-scale vortices that reduce dispersion and enhance mass transfer between support structures in the chromatography column, allowing for efficient separation without permanent surface charges and minimizing direct contact with electrodes.
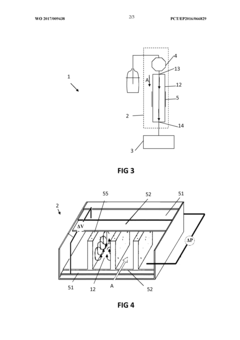
High-performance liquid chromatography
PatentActiveUS20180202981A1
Innovation
- The use of alternating current (AC) electrokinetics to generate micro-scale vortices that suppress dispersion by inducing a flow orthogonal to the axial direction, allowing for efficient separation without permanent surface charges and reducing undesired analyte interactions, combined with pressure flow to enhance mass transport beyond diffusion rates.
Regulatory Compliance in Bioanalytical Methods
Regulatory compliance in bioanalytical methods represents a critical framework governing the implementation of High-Performance Liquid Chromatography (HPLC) techniques in pharmaceutical and clinical research. The optimization of data collection in HPLC must adhere to stringent regulatory guidelines established by international bodies such as the FDA, EMA, and ICH. These regulations ensure the reliability, reproducibility, and integrity of bioanalytical results across different laboratories and research institutions.
The FDA's Guidance for Industry on Bioanalytical Method Validation (2018) specifically addresses HPLC data collection requirements, emphasizing the importance of system suitability tests, calibration standards, and quality control samples. Compliance with these guidelines necessitates comprehensive documentation of all analytical procedures, including detailed records of sample preparation, chromatographic conditions, and data processing algorithms.
EMA's Guideline on Bioanalytical Method Validation (2011) further elaborates on the validation parameters essential for HPLC methods, including selectivity, carry-over, lower limit of quantification (LLOQ), calibration curve, accuracy, precision, and stability. These parameters must be rigorously evaluated and documented to ensure regulatory acceptance of bioanalytical data.
The implementation of Good Laboratory Practice (GLP) and Good Clinical Practice (GCP) standards is mandatory for bioanalytical laboratories conducting HPLC analyses. These standards encompass requirements for personnel training, equipment qualification, and standard operating procedures (SOPs) that directly impact data collection quality and compliance.
Data integrity represents a paramount concern in regulatory compliance for HPLC bioanalytical methods. The ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available) must be applied to all electronic and paper records generated during HPLC analyses. Modern HPLC systems must incorporate features that prevent unauthorized data manipulation and maintain comprehensive audit trails.
Method transfer and cross-validation procedures are essential regulatory considerations when implementing optimized HPLC methods across different laboratories or instruments. These procedures ensure consistency in data collection and interpretation, regardless of the specific analytical platform or operator involved.
Regulatory bodies increasingly emphasize risk-based approaches to method development and validation, requiring laboratories to identify critical method parameters that may impact data quality. This approach necessitates thorough risk assessments and the implementation of appropriate control strategies to mitigate identified risks in HPLC data collection processes.
Compliance with evolving regulatory expectations requires continuous monitoring of guideline updates and implementation of appropriate changes to existing HPLC methods. This dynamic regulatory landscape necessitates flexible yet robust data collection strategies that can adapt to changing compliance requirements while maintaining scientific validity and operational efficiency.
The FDA's Guidance for Industry on Bioanalytical Method Validation (2018) specifically addresses HPLC data collection requirements, emphasizing the importance of system suitability tests, calibration standards, and quality control samples. Compliance with these guidelines necessitates comprehensive documentation of all analytical procedures, including detailed records of sample preparation, chromatographic conditions, and data processing algorithms.
EMA's Guideline on Bioanalytical Method Validation (2011) further elaborates on the validation parameters essential for HPLC methods, including selectivity, carry-over, lower limit of quantification (LLOQ), calibration curve, accuracy, precision, and stability. These parameters must be rigorously evaluated and documented to ensure regulatory acceptance of bioanalytical data.
The implementation of Good Laboratory Practice (GLP) and Good Clinical Practice (GCP) standards is mandatory for bioanalytical laboratories conducting HPLC analyses. These standards encompass requirements for personnel training, equipment qualification, and standard operating procedures (SOPs) that directly impact data collection quality and compliance.
Data integrity represents a paramount concern in regulatory compliance for HPLC bioanalytical methods. The ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available) must be applied to all electronic and paper records generated during HPLC analyses. Modern HPLC systems must incorporate features that prevent unauthorized data manipulation and maintain comprehensive audit trails.
Method transfer and cross-validation procedures are essential regulatory considerations when implementing optimized HPLC methods across different laboratories or instruments. These procedures ensure consistency in data collection and interpretation, regardless of the specific analytical platform or operator involved.
Regulatory bodies increasingly emphasize risk-based approaches to method development and validation, requiring laboratories to identify critical method parameters that may impact data quality. This approach necessitates thorough risk assessments and the implementation of appropriate control strategies to mitigate identified risks in HPLC data collection processes.
Compliance with evolving regulatory expectations requires continuous monitoring of guideline updates and implementation of appropriate changes to existing HPLC methods. This dynamic regulatory landscape necessitates flexible yet robust data collection strategies that can adapt to changing compliance requirements while maintaining scientific validity and operational efficiency.
Data Integrity and Validation Strategies
Data integrity and validation strategies are fundamental pillars in optimizing HPLC data collection for bioanalytical results. The implementation of robust data integrity measures ensures that collected data remains accurate, complete, consistent, and reliable throughout its lifecycle. This begins with proper system validation according to regulatory guidelines such as FDA 21 CFR Part 11 and EMA's guidance on data integrity.
A comprehensive data integrity framework for HPLC bioanalytical methods must incorporate both technical and procedural controls. Technical controls include system access restrictions, audit trails, electronic signatures, and data encryption mechanisms that prevent unauthorized modifications. These systems must be configured to automatically log all data manipulations, ensuring complete traceability from raw data acquisition through final reporting.
Procedural controls complement technical measures through standard operating procedures (SOPs) that govern data handling practices. These SOPs should clearly define responsibilities for data review, approval workflows, and documentation requirements. Regular training programs ensure that laboratory personnel understand and adhere to these procedures, minimizing human error and intentional data manipulation risks.
Validation strategies for HPLC bioanalytical methods must follow a lifecycle approach, beginning with user requirements specification and continuing through installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ). Method validation parameters including accuracy, precision, selectivity, sensitivity, reproducibility, and stability must be rigorously tested and documented according to ICH Q2(R1) guidelines.
Data review processes represent another critical component, implementing a multi-tiered approach where raw data undergoes technical, scientific, and quality assurance reviews. Automated data verification tools can supplement manual reviews by flagging potential anomalies or deviations from expected results, enhancing detection of data integrity issues.
Risk-based approaches to validation and data integrity have gained prominence, allowing organizations to allocate resources proportionally to critical data points that directly impact patient safety and product quality. This approach focuses validation efforts on high-risk areas while maintaining appropriate controls across all systems.
Continuous monitoring through periodic system audits and data integrity assessments ensures ongoing compliance and identifies opportunities for improvement. These assessments should evaluate both technical system performance and procedural adherence, with findings incorporated into corrective and preventive action plans.
A comprehensive data integrity framework for HPLC bioanalytical methods must incorporate both technical and procedural controls. Technical controls include system access restrictions, audit trails, electronic signatures, and data encryption mechanisms that prevent unauthorized modifications. These systems must be configured to automatically log all data manipulations, ensuring complete traceability from raw data acquisition through final reporting.
Procedural controls complement technical measures through standard operating procedures (SOPs) that govern data handling practices. These SOPs should clearly define responsibilities for data review, approval workflows, and documentation requirements. Regular training programs ensure that laboratory personnel understand and adhere to these procedures, minimizing human error and intentional data manipulation risks.
Validation strategies for HPLC bioanalytical methods must follow a lifecycle approach, beginning with user requirements specification and continuing through installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ). Method validation parameters including accuracy, precision, selectivity, sensitivity, reproducibility, and stability must be rigorously tested and documented according to ICH Q2(R1) guidelines.
Data review processes represent another critical component, implementing a multi-tiered approach where raw data undergoes technical, scientific, and quality assurance reviews. Automated data verification tools can supplement manual reviews by flagging potential anomalies or deviations from expected results, enhancing detection of data integrity issues.
Risk-based approaches to validation and data integrity have gained prominence, allowing organizations to allocate resources proportionally to critical data points that directly impact patient safety and product quality. This approach focuses validation efforts on high-risk areas while maintaining appropriate controls across all systems.
Continuous monitoring through periodic system audits and data integrity assessments ensures ongoing compliance and identifies opportunities for improvement. These assessments should evaluate both technical system performance and procedural adherence, with findings incorporated into corrective and preventive action plans.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!