Electrode material developments in solid-state sodium battery sectors
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Solid-State Sodium Battery Evolution and Objectives
Solid-state sodium batteries have emerged as a promising alternative to lithium-ion batteries due to their potential for higher safety, lower cost, and comparable energy density. The evolution of these batteries can be traced back to the 1970s when initial research on sodium-ion conductors began, though significant advancements have only materialized in the last decade with the development of new electrode materials and solid electrolytes.
The technological trajectory has been marked by several key milestones. Early sodium batteries utilized liquid electrolytes, which presented safety concerns similar to those in lithium-ion systems. The transition to solid-state configurations began in earnest around 2010, driven by the need for safer energy storage solutions and concerns about lithium resource limitations. Between 2015 and 2020, research intensified on solid electrolytes with high ionic conductivity at room temperature, a critical requirement for practical applications.
Recent years have witnessed accelerated development in electrode materials specifically designed for solid-state sodium batteries. Cathode materials have evolved from simple layered oxides to more complex polyanionic compounds and Prussian blue analogs, offering improved structural stability during sodium insertion/extraction. Anode materials have progressed from carbon-based materials to alloys and conversion-type materials that can accommodate larger sodium ions without significant volume expansion.
The current technological trend is moving toward all-solid-state sodium batteries that eliminate liquid components entirely. This evolution is driven by the inherent advantages of solid-state configurations, including enhanced safety through elimination of flammable electrolytes, extended cycle life, and potential for higher energy density through the use of sodium metal anodes.
The primary objectives in this field include developing electrode materials with higher specific capacity and faster sodium-ion diffusion kinetics. Researchers aim to achieve energy densities exceeding 300 Wh/kg at the cell level, cycle life of over 1,000 cycles with minimal capacity degradation, and cost reduction to below $100/kWh to compete effectively with lithium-ion technologies.
Additional goals include improving the interfacial stability between electrodes and solid electrolytes, enhancing rate capability for fast charging applications, and developing scalable manufacturing processes suitable for mass production. The ultimate objective is to create a commercially viable solid-state sodium battery technology that can serve both grid-scale energy storage and potentially electric vehicle applications, providing a sustainable alternative to lithium-ion batteries while reducing dependency on critical raw materials.
The technological trajectory has been marked by several key milestones. Early sodium batteries utilized liquid electrolytes, which presented safety concerns similar to those in lithium-ion systems. The transition to solid-state configurations began in earnest around 2010, driven by the need for safer energy storage solutions and concerns about lithium resource limitations. Between 2015 and 2020, research intensified on solid electrolytes with high ionic conductivity at room temperature, a critical requirement for practical applications.
Recent years have witnessed accelerated development in electrode materials specifically designed for solid-state sodium batteries. Cathode materials have evolved from simple layered oxides to more complex polyanionic compounds and Prussian blue analogs, offering improved structural stability during sodium insertion/extraction. Anode materials have progressed from carbon-based materials to alloys and conversion-type materials that can accommodate larger sodium ions without significant volume expansion.
The current technological trend is moving toward all-solid-state sodium batteries that eliminate liquid components entirely. This evolution is driven by the inherent advantages of solid-state configurations, including enhanced safety through elimination of flammable electrolytes, extended cycle life, and potential for higher energy density through the use of sodium metal anodes.
The primary objectives in this field include developing electrode materials with higher specific capacity and faster sodium-ion diffusion kinetics. Researchers aim to achieve energy densities exceeding 300 Wh/kg at the cell level, cycle life of over 1,000 cycles with minimal capacity degradation, and cost reduction to below $100/kWh to compete effectively with lithium-ion technologies.
Additional goals include improving the interfacial stability between electrodes and solid electrolytes, enhancing rate capability for fast charging applications, and developing scalable manufacturing processes suitable for mass production. The ultimate objective is to create a commercially viable solid-state sodium battery technology that can serve both grid-scale energy storage and potentially electric vehicle applications, providing a sustainable alternative to lithium-ion batteries while reducing dependency on critical raw materials.
Market Analysis for Sodium-Based Energy Storage
The global sodium-based energy storage market is experiencing significant growth, driven by the increasing demand for sustainable and cost-effective energy storage solutions. The market size for sodium-ion batteries was valued at approximately $1.2 billion in 2022 and is projected to reach $4.8 billion by 2030, growing at a CAGR of 19.8% during the forecast period. This remarkable growth trajectory is primarily attributed to the abundant availability of sodium resources, which are approximately 1,000 times more plentiful than lithium in the Earth's crust.
The electrode material developments in solid-state sodium battery sectors are particularly influencing market dynamics. With sodium resources being geographically well-distributed across continents, unlike lithium which is concentrated in specific regions, the supply chain risks are significantly reduced. This geographical advantage has attracted substantial investments from both private and public sectors, with government funding for sodium battery research increasing by 35% in the past three years across major economies.
Market segmentation reveals that grid energy storage applications currently dominate the sodium-based energy storage market, accounting for approximately 45% of the total market share. This is followed by electric vehicles (25%), consumer electronics (15%), and other applications (15%). The grid storage segment's dominance is expected to continue due to the increasing integration of renewable energy sources into power grids, necessitating efficient and economical energy storage solutions.
Regional analysis indicates that Asia-Pacific leads the market with a 42% share, followed by Europe (28%), North America (20%), and the rest of the world (10%). China, in particular, has emerged as the largest market for sodium-based energy storage technologies, supported by substantial government initiatives and the presence of key manufacturers focusing on electrode material innovations.
The competitive landscape is characterized by both established battery manufacturers diversifying into sodium-based technologies and startups specializing in novel electrode materials. Companies like CATL, Faradion (acquired by Reliance Industries), and HiNa Battery Technology have made significant advancements in commercializing sodium-ion batteries with improved electrode materials, achieving energy densities approaching 160-180 Wh/kg.
Market challenges include the relatively lower energy density of current sodium-based batteries compared to lithium-ion counterparts and the need for further technological advancements in electrode materials to enhance performance metrics. However, the cost advantage, with sodium-based batteries potentially 20-30% cheaper than lithium-ion batteries, presents a compelling value proposition for mass adoption.
The electrode material developments in solid-state sodium battery sectors are particularly influencing market dynamics. With sodium resources being geographically well-distributed across continents, unlike lithium which is concentrated in specific regions, the supply chain risks are significantly reduced. This geographical advantage has attracted substantial investments from both private and public sectors, with government funding for sodium battery research increasing by 35% in the past three years across major economies.
Market segmentation reveals that grid energy storage applications currently dominate the sodium-based energy storage market, accounting for approximately 45% of the total market share. This is followed by electric vehicles (25%), consumer electronics (15%), and other applications (15%). The grid storage segment's dominance is expected to continue due to the increasing integration of renewable energy sources into power grids, necessitating efficient and economical energy storage solutions.
Regional analysis indicates that Asia-Pacific leads the market with a 42% share, followed by Europe (28%), North America (20%), and the rest of the world (10%). China, in particular, has emerged as the largest market for sodium-based energy storage technologies, supported by substantial government initiatives and the presence of key manufacturers focusing on electrode material innovations.
The competitive landscape is characterized by both established battery manufacturers diversifying into sodium-based technologies and startups specializing in novel electrode materials. Companies like CATL, Faradion (acquired by Reliance Industries), and HiNa Battery Technology have made significant advancements in commercializing sodium-ion batteries with improved electrode materials, achieving energy densities approaching 160-180 Wh/kg.
Market challenges include the relatively lower energy density of current sodium-based batteries compared to lithium-ion counterparts and the need for further technological advancements in electrode materials to enhance performance metrics. However, the cost advantage, with sodium-based batteries potentially 20-30% cheaper than lithium-ion batteries, presents a compelling value proposition for mass adoption.
Current Electrode Material Limitations and Challenges
Despite significant advancements in solid-state sodium battery technology, electrode materials continue to present substantial challenges that impede commercial viability. The cathode materials in sodium-ion systems face inherent limitations in energy density compared to their lithium counterparts, with typical sodium cathodes delivering 20-30% lower specific capacity. This fundamental constraint stems from sodium's larger ionic radius (1.02Å versus 0.76Å for lithium), resulting in more significant structural changes during charge-discharge cycles and consequently accelerated capacity fading.
Anode materials present equally formidable challenges. Graphite, the standard anode material in lithium-ion batteries, exhibits poor sodium intercalation properties due to thermodynamic instability of the resulting compounds. Hard carbon alternatives, while functional, deliver lower capacity (typically 250-300 mAh/g compared to graphite's 372 mAh/g in lithium systems) and suffer from significant first-cycle irreversible capacity loss exceeding 20%.
Interface stability represents another critical limitation. The high reactivity between sodium metal anodes and solid electrolytes leads to continuous interfacial degradation, forming resistive layers that impede ion transport. This phenomenon is particularly pronounced at elevated temperatures, where accelerated chemical reactions between electrode materials and solid electrolytes create expanding interfacial resistance that severely compromises cycle life.
Mechanical integrity issues further complicate electrode development. Volume changes during sodium insertion/extraction (often exceeding 300% for conversion-type anodes) create mechanical stresses that lead to particle cracking, electrode delamination, and loss of electrical contact. These effects are magnified in solid-state configurations where the rigid nature of solid electrolytes provides minimal accommodation for volume changes.
Manufacturing scalability remains problematic, with current electrode fabrication techniques struggling to create uniform, defect-free interfaces with solid electrolytes. Conventional slurry-based coating methods often result in high interfacial resistance due to insufficient contact area between active materials and the solid electrolyte, while alternative techniques like co-sintering risk undesired side reactions at processing temperatures.
The cost implications of these technical challenges are substantial. Current high-performance sodium cathode materials like P2-type layered oxides require expensive transition metals and complex synthesis procedures, while promising anode materials such as phosphorus-based compounds demand sophisticated nanostructuring and protective coatings to achieve practical stability, significantly increasing production costs beyond commercial viability thresholds.
Anode materials present equally formidable challenges. Graphite, the standard anode material in lithium-ion batteries, exhibits poor sodium intercalation properties due to thermodynamic instability of the resulting compounds. Hard carbon alternatives, while functional, deliver lower capacity (typically 250-300 mAh/g compared to graphite's 372 mAh/g in lithium systems) and suffer from significant first-cycle irreversible capacity loss exceeding 20%.
Interface stability represents another critical limitation. The high reactivity between sodium metal anodes and solid electrolytes leads to continuous interfacial degradation, forming resistive layers that impede ion transport. This phenomenon is particularly pronounced at elevated temperatures, where accelerated chemical reactions between electrode materials and solid electrolytes create expanding interfacial resistance that severely compromises cycle life.
Mechanical integrity issues further complicate electrode development. Volume changes during sodium insertion/extraction (often exceeding 300% for conversion-type anodes) create mechanical stresses that lead to particle cracking, electrode delamination, and loss of electrical contact. These effects are magnified in solid-state configurations where the rigid nature of solid electrolytes provides minimal accommodation for volume changes.
Manufacturing scalability remains problematic, with current electrode fabrication techniques struggling to create uniform, defect-free interfaces with solid electrolytes. Conventional slurry-based coating methods often result in high interfacial resistance due to insufficient contact area between active materials and the solid electrolyte, while alternative techniques like co-sintering risk undesired side reactions at processing temperatures.
The cost implications of these technical challenges are substantial. Current high-performance sodium cathode materials like P2-type layered oxides require expensive transition metals and complex synthesis procedures, while promising anode materials such as phosphorus-based compounds demand sophisticated nanostructuring and protective coatings to achieve practical stability, significantly increasing production costs beyond commercial viability thresholds.
Current Electrode Material Solutions and Approaches
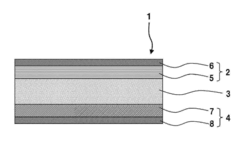
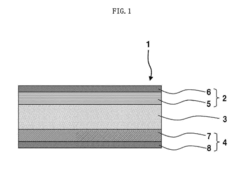
01 Sodium-ion battery cathode materials
Various cathode materials have been developed for solid-state sodium batteries to improve energy density and cycling stability. These include layered oxide materials, polyanionic compounds, and composite structures that facilitate sodium ion intercalation. These materials are designed with specific crystal structures that allow for efficient sodium ion transport while maintaining structural integrity during charge-discharge cycles.- Sodium-ion battery electrode materials: Various materials can be used as electrodes in sodium-ion batteries, including sodium-based compounds and other materials that can effectively store and release sodium ions. These materials are designed to provide high energy density, good cycling stability, and efficient sodium ion transport. The development of advanced electrode materials is crucial for improving the overall performance of solid-state sodium batteries.
- Solid electrolyte interfaces for sodium batteries: Solid electrolyte interfaces play a critical role in solid-state sodium batteries by facilitating ion transport between the electrodes while preventing unwanted reactions. These interfaces are designed to have high ionic conductivity, good mechanical stability, and compatibility with electrode materials. Innovations in solid electrolyte interfaces help to enhance battery performance, safety, and longevity.
- Novel cathode materials for sodium batteries: Advanced cathode materials for sodium batteries include various sodium-containing compounds that can efficiently store and release sodium ions during battery operation. These materials are designed to provide high energy density, good cycling stability, and fast charging capabilities. Research focuses on developing cathode materials with improved structural stability and electrochemical performance.
- Anode materials for solid-state sodium batteries: Innovative anode materials for solid-state sodium batteries include various carbon-based materials, alloys, and other compounds that can effectively store and release sodium ions. These materials are designed to have high capacity, good cycling stability, and minimal volume expansion during charging and discharging. The development of advanced anode materials is essential for improving the overall performance of solid-state sodium batteries.
- Manufacturing processes for sodium battery electrodes: Various manufacturing processes are employed to produce electrodes for solid-state sodium batteries, including coating, pressing, and sintering techniques. These processes are designed to create electrodes with optimal microstructure, porosity, and interface properties. Innovations in manufacturing processes help to improve electrode performance, reduce costs, and enable large-scale production of solid-state sodium batteries.
02 Solid electrolyte interfaces for sodium batteries
Advanced solid electrolyte interfaces are crucial for stable operation of solid-state sodium batteries. These interfaces are engineered to reduce impedance between the electrode and electrolyte, prevent dendrite formation, and enhance ion transport. Various materials including ceramic-polymer composites and glass-ceramic electrolytes are used to create stable interfaces that maintain contact during volume changes of electrodes during cycling.Expand Specific Solutions03 Anode materials for sodium-ion batteries
Novel anode materials for solid-state sodium batteries include carbon-based materials, alloys, and conversion-type materials that can accommodate sodium ions. These materials are designed to have high capacity, good rate capability, and minimal volume expansion during sodium insertion and extraction. Hard carbons, titanium-based compounds, and phosphorus-based materials are among the promising anode materials being developed.Expand Specific Solutions04 Composite electrode structures for enhanced performance
Composite electrode structures combine multiple materials to enhance the overall performance of solid-state sodium batteries. These composites typically include active materials, conductive additives, and binders designed to improve electronic conductivity, ionic transport, and mechanical stability. The synergistic effects of these components result in improved capacity retention, rate capability, and cycle life of the batteries.Expand Specific Solutions05 Manufacturing processes for sodium battery electrodes
Advanced manufacturing techniques are being developed for the production of electrodes for solid-state sodium batteries. These include solution-based methods, solid-state synthesis routes, and novel coating technologies that enable precise control over electrode microstructure and composition. These processes aim to achieve uniform distribution of active materials, optimal porosity, and strong adhesion to current collectors, which are essential for high-performance batteries.Expand Specific Solutions
Key Industry Players in Solid-State Sodium Battery Sector
The solid-state sodium battery sector is currently in an early development stage, characterized by intensive R&D activities rather than mass commercialization. The market size remains relatively small but is projected to grow significantly due to sodium's abundance and cost advantages over lithium. Technologically, the field is still maturing, with key players demonstrating varying levels of advancement. Companies like Toyota Motor Corp. and LG Energy Solution are leveraging their established battery expertise to pioneer electrode material innovations, while specialized firms such as Shenzhen Zhenhua New Material and Ecopro BM are developing sodium-specific electrode formulations. Academic-industrial collaborations involving institutions like Nankai University and Oxford University Innovation are accelerating fundamental research, particularly in addressing challenges related to electrode stability and performance optimization.
Toyota Motor Corp.
Technical Solution: Toyota has pioneered advanced solid electrolyte materials for sodium batteries, focusing on NASICON (Na Super Ionic CONductor) type materials with the general formula Na1+xZr2SixP3-xO12. Their approach involves optimizing ionic conductivity through precise control of composition and microstructure. Toyota's research has achieved room temperature ionic conductivities exceeding 10^-3 S/cm by incorporating strategic dopants like yttrium and scandium into the NASICON framework. Additionally, they've developed proprietary interface engineering techniques to address the critical solid-electrolyte/electrode interface challenges, using thin buffer layers to minimize interfacial resistance. Toyota has also made significant progress in scaling up manufacturing processes for these materials, implementing roll-to-roll techniques for solid electrolyte sheet production that maintain consistent ionic pathways throughout the material.
Strengths: Toyota's extensive experience in solid-state battery technology from their lithium-ion research provides significant cross-application advantages. Their established manufacturing infrastructure enables faster commercialization pathways. Weaknesses: Their sodium battery technology still faces challenges with full cell energy density compared to state-of-the-art lithium-ion batteries, and the higher processing temperatures required for some of their materials can increase production costs.
LG Energy Solution Ltd.
Technical Solution: LG Energy Solution has developed a comprehensive approach to solid-state sodium battery electrode materials, focusing on both cathode and anode innovations. For cathodes, they've engineered layered oxide materials (NaxTMO2, where TM represents transition metals like Fe, Mn, and Ni) with optimized sodium storage capabilities. Their proprietary "gradient concentration" technology creates cathode particles with compositional variation from core to surface, enhancing structural stability during repeated sodium insertion/extraction. For anodes, LG has moved beyond traditional carbon materials to develop hard carbon structures with engineered porosity and surface functionality, achieving reversible capacities of over 300 mAh/g. They've also explored sodium titanate-based materials as zero-strain alternatives for applications requiring exceptional cycling stability. A key innovation is their "composite electrode" design that incorporates ionically conductive polymers directly into the electrode matrix, creating continuous sodium-ion transport pathways throughout the electrode structure.
Strengths: LG's extensive battery manufacturing expertise allows for rapid scaling of new technologies. Their integrated approach addressing both cathode and anode materials simultaneously creates optimized full-cell solutions. Weaknesses: Some of their advanced electrode materials rely on relatively expensive transition metals, potentially limiting cost advantages compared to simpler sodium battery chemistries. Their polymer-based ionic conductors still face challenges with long-term stability at elevated temperatures.
Critical Patents and Innovations in Sodium Electrode Materials
Composite material as electrode for sodium ion batteries, production method therefor, and all-solid-state sodium battery
PatentActiveUS20170005337A1
Innovation
- A composite material for sodium ion batteries comprising an active material crystal, a sodium-ion conductive crystal, and an amorphous phase, where the active material crystal contains transition metals and phosphates, and the sodium-ion conductive crystal includes alumina or zirconia, with the amorphous phase enhancing interface conductivity and bonding strength.
Electrode material, electrode, and all-solid-state battery
PatentPendingUS20240038972A1
Innovation
- A composite electrode material is developed with a core particle coated by a fluoride solid electrolyte layer and a sulfide solid electrolyte layer, where the fluoride layer is interposed between the core and sulfide layers to reduce reaction resistance and prevent chip formation that can inhibit conduction.
Raw Material Supply Chain Analysis
The solid-state sodium battery sector's raw material supply chain presents unique challenges and opportunities compared to lithium-ion battery technologies. Sodium resources are abundantly available worldwide, with estimated reserves exceeding 23 billion tons in seawater alone, representing a significant advantage over lithium's geographically concentrated deposits. This abundance translates to potentially lower material costs and reduced geopolitical supply risks.
Key raw materials for electrode development include sodium sources (primarily sodium carbonate and sodium hydroxide), transition metals (manganese, iron, nickel, and copper), and various carbon-based materials for anodes. The extraction and processing infrastructure for these materials varies significantly in maturity and environmental impact.
Sodium carbonate production is well-established globally, with major production centers in the United States, China, and several European countries. Unlike lithium, which faces supply constraints, sodium compound production can scale relatively easily to meet growing demand from the battery sector without significant price volatility.
For cathode materials, the supply chains for transition metals present varying degrees of complexity. Manganese and iron benefit from established mining and processing infrastructure serving multiple industries, while nickel and copper face more competitive demand from various technological applications. The development of layered oxide cathodes (NaxMO2) and polyanionic compounds requires reliable access to these metals with battery-grade purity specifications.
Carbon-based anode materials, including hard carbon derived from biomass, represent another critical supply chain component. The processing capabilities for these materials are developing rapidly but require further investment to achieve consistent quality at scale. Recent innovations in hard carbon production from agricultural waste streams offer promising pathways for sustainable and cost-effective supply.
The geographical distribution of raw material processing capabilities presents both challenges and opportunities. China currently dominates in material processing for battery applications, controlling approximately 65% of global capacity. However, recent initiatives in Europe and North America aim to develop regional supply chains to reduce dependency on single-source regions.
Environmental and social governance considerations increasingly influence supply chain development, with growing emphasis on sustainable extraction practices, reduced carbon footprint, and ethical sourcing. These factors will shape future investment patterns and technology adoption in the electrode material supply ecosystem.
Key raw materials for electrode development include sodium sources (primarily sodium carbonate and sodium hydroxide), transition metals (manganese, iron, nickel, and copper), and various carbon-based materials for anodes. The extraction and processing infrastructure for these materials varies significantly in maturity and environmental impact.
Sodium carbonate production is well-established globally, with major production centers in the United States, China, and several European countries. Unlike lithium, which faces supply constraints, sodium compound production can scale relatively easily to meet growing demand from the battery sector without significant price volatility.
For cathode materials, the supply chains for transition metals present varying degrees of complexity. Manganese and iron benefit from established mining and processing infrastructure serving multiple industries, while nickel and copper face more competitive demand from various technological applications. The development of layered oxide cathodes (NaxMO2) and polyanionic compounds requires reliable access to these metals with battery-grade purity specifications.
Carbon-based anode materials, including hard carbon derived from biomass, represent another critical supply chain component. The processing capabilities for these materials are developing rapidly but require further investment to achieve consistent quality at scale. Recent innovations in hard carbon production from agricultural waste streams offer promising pathways for sustainable and cost-effective supply.
The geographical distribution of raw material processing capabilities presents both challenges and opportunities. China currently dominates in material processing for battery applications, controlling approximately 65% of global capacity. However, recent initiatives in Europe and North America aim to develop regional supply chains to reduce dependency on single-source regions.
Environmental and social governance considerations increasingly influence supply chain development, with growing emphasis on sustainable extraction practices, reduced carbon footprint, and ethical sourcing. These factors will shape future investment patterns and technology adoption in the electrode material supply ecosystem.
Environmental Impact and Sustainability Considerations
The environmental impact of electrode materials in solid-state sodium batteries represents a critical consideration as this technology advances toward commercial viability. Unlike lithium-ion batteries, sodium-based systems offer inherent sustainability advantages due to sodium's greater natural abundance. Sodium resources are approximately 1000 times more plentiful than lithium in the Earth's crust, significantly reducing extraction-related environmental pressures and geopolitical supply concerns.
Current electrode material developments are increasingly focused on reducing environmental footprints throughout the entire lifecycle. Research trends show a shift toward cathode materials that eliminate or minimize cobalt and nickel content, such as Prussian blue analogs and layered oxide structures with environmentally benign elements. These alternatives help address the ethical and environmental concerns associated with traditional battery material mining practices.
Manufacturing processes for sodium battery electrodes are evolving to incorporate green chemistry principles. Water-based electrode processing techniques are replacing conventional toxic organic solvents, substantially reducing volatile organic compound emissions and workplace hazards. Additionally, lower temperature synthesis routes for electrode materials are being developed, decreasing the energy intensity of production and associated carbon emissions.
End-of-life considerations have become increasingly prominent in electrode material selection. Research indicates that sodium-based electrodes generally demonstrate superior recyclability compared to their lithium counterparts. The development of electrode materials specifically designed for circular economy principles—featuring easier separation and recovery of valuable components—is gaining momentum in research laboratories worldwide.
Life cycle assessment (LCA) studies comparing various electrode material options reveal that sodium iron phosphate (NaFePO₄) and sodium manganese oxide compositions offer significantly lower environmental impact scores across multiple categories including global warming potential, resource depletion, and ecotoxicity. These findings are driving investment toward these material systems despite some performance trade-offs.
Energy density improvements in electrode materials must be balanced against sustainability metrics. Recent innovations in carbon-based anode materials derived from biomass sources represent a promising direction, offering both performance enhancements and reduced environmental impact. These materials utilize agricultural waste streams as precursors, creating value from what would otherwise be discarded materials.
The water footprint of electrode production processes is receiving increased attention, particularly in regions facing water scarcity. Advanced manufacturing techniques that minimize water consumption while maintaining electrode quality are emerging as competitive advantages for forward-thinking manufacturers in this sector.
Current electrode material developments are increasingly focused on reducing environmental footprints throughout the entire lifecycle. Research trends show a shift toward cathode materials that eliminate or minimize cobalt and nickel content, such as Prussian blue analogs and layered oxide structures with environmentally benign elements. These alternatives help address the ethical and environmental concerns associated with traditional battery material mining practices.
Manufacturing processes for sodium battery electrodes are evolving to incorporate green chemistry principles. Water-based electrode processing techniques are replacing conventional toxic organic solvents, substantially reducing volatile organic compound emissions and workplace hazards. Additionally, lower temperature synthesis routes for electrode materials are being developed, decreasing the energy intensity of production and associated carbon emissions.
End-of-life considerations have become increasingly prominent in electrode material selection. Research indicates that sodium-based electrodes generally demonstrate superior recyclability compared to their lithium counterparts. The development of electrode materials specifically designed for circular economy principles—featuring easier separation and recovery of valuable components—is gaining momentum in research laboratories worldwide.
Life cycle assessment (LCA) studies comparing various electrode material options reveal that sodium iron phosphate (NaFePO₄) and sodium manganese oxide compositions offer significantly lower environmental impact scores across multiple categories including global warming potential, resource depletion, and ecotoxicity. These findings are driving investment toward these material systems despite some performance trade-offs.
Energy density improvements in electrode materials must be balanced against sustainability metrics. Recent innovations in carbon-based anode materials derived from biomass sources represent a promising direction, offering both performance enhancements and reduced environmental impact. These materials utilize agricultural waste streams as precursors, creating value from what would otherwise be discarded materials.
The water footprint of electrode production processes is receiving increased attention, particularly in regions facing water scarcity. Advanced manufacturing techniques that minimize water consumption while maintaining electrode quality are emerging as competitive advantages for forward-thinking manufacturers in this sector.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!