Material breakthroughs in solid-state sodium battery polymers
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Solid-State Sodium Battery Evolution and Objectives
Solid-state sodium batteries have emerged as a promising alternative to lithium-ion batteries due to the abundance and low cost of sodium resources. The evolution of these batteries can be traced back to the 1970s when initial research on sodium-ion conductors began, though significant progress has been limited by material constraints, particularly in solid electrolytes.
The development trajectory has seen three distinct phases. The first phase (1970s-1990s) focused on fundamental research into sodium ion conductivity in ceramic materials, establishing theoretical frameworks but achieving limited practical applications due to manufacturing challenges. The second phase (2000s-2015) witnessed increased interest in sodium batteries as concerns about lithium resource limitations grew, with research expanding to include polymer-based solid electrolytes and composite materials.
The current phase (2015-present) represents an acceleration in research and development, characterized by breakthroughs in polymer chemistry and interface engineering. Recent innovations in cross-linked polymer networks and ceramic-polymer composites have significantly improved ionic conductivity at room temperature, addressing one of the primary technical barriers to commercialization.
The primary technical objectives for solid-state sodium battery polymers include achieving ionic conductivity exceeding 10^-3 S/cm at room temperature, enhancing mechanical stability to prevent dendrite formation, and ensuring compatibility with sodium metal anodes to maximize energy density. Additionally, developing polymers with wide electrochemical stability windows (>4V) remains crucial for enabling high-voltage cathode materials.
From a commercial perspective, objectives include reducing manufacturing costs to below $100/kWh, extending cycle life beyond 1000 cycles with minimal capacity degradation, and developing scalable production methods compatible with existing battery manufacturing infrastructure. Environmental considerations have also shaped research goals, with emphasis on developing non-toxic, recyclable polymer materials.
The convergence of these objectives has led to focused research on multi-functional polymer electrolytes that simultaneously address conductivity, mechanical strength, and interfacial stability. Recent breakthroughs in polymer nanocomposites incorporating sodium-conducting ceramic fillers have demonstrated promising results, achieving conductivities approaching 10^-4 S/cm while maintaining excellent mechanical properties.
Looking forward, the field is moving toward biomass-derived sustainable polymers and adaptive polymer networks that can self-heal and respond to environmental changes, potentially revolutionizing energy storage technology while addressing resource sustainability concerns.
The development trajectory has seen three distinct phases. The first phase (1970s-1990s) focused on fundamental research into sodium ion conductivity in ceramic materials, establishing theoretical frameworks but achieving limited practical applications due to manufacturing challenges. The second phase (2000s-2015) witnessed increased interest in sodium batteries as concerns about lithium resource limitations grew, with research expanding to include polymer-based solid electrolytes and composite materials.
The current phase (2015-present) represents an acceleration in research and development, characterized by breakthroughs in polymer chemistry and interface engineering. Recent innovations in cross-linked polymer networks and ceramic-polymer composites have significantly improved ionic conductivity at room temperature, addressing one of the primary technical barriers to commercialization.
The primary technical objectives for solid-state sodium battery polymers include achieving ionic conductivity exceeding 10^-3 S/cm at room temperature, enhancing mechanical stability to prevent dendrite formation, and ensuring compatibility with sodium metal anodes to maximize energy density. Additionally, developing polymers with wide electrochemical stability windows (>4V) remains crucial for enabling high-voltage cathode materials.
From a commercial perspective, objectives include reducing manufacturing costs to below $100/kWh, extending cycle life beyond 1000 cycles with minimal capacity degradation, and developing scalable production methods compatible with existing battery manufacturing infrastructure. Environmental considerations have also shaped research goals, with emphasis on developing non-toxic, recyclable polymer materials.
The convergence of these objectives has led to focused research on multi-functional polymer electrolytes that simultaneously address conductivity, mechanical strength, and interfacial stability. Recent breakthroughs in polymer nanocomposites incorporating sodium-conducting ceramic fillers have demonstrated promising results, achieving conductivities approaching 10^-4 S/cm while maintaining excellent mechanical properties.
Looking forward, the field is moving toward biomass-derived sustainable polymers and adaptive polymer networks that can self-heal and respond to environmental changes, potentially revolutionizing energy storage technology while addressing resource sustainability concerns.
Market Analysis for Next-Generation Energy Storage Solutions
The global energy storage market is witnessing unprecedented growth, driven by the increasing adoption of renewable energy sources and the electrification of transportation. The market for next-generation energy storage solutions is projected to reach $546 billion by 2035, with a compound annual growth rate of 12.3% from 2023 to 2035. Within this landscape, solid-state sodium battery technologies are emerging as a promising alternative to conventional lithium-ion batteries, particularly due to material breakthroughs in polymer electrolytes.
Consumer electronics, electric vehicles, and grid storage represent the primary market segments for advanced energy storage technologies. The electric vehicle segment alone is expected to consume over 2,800 GWh of battery capacity by 2030, creating substantial opportunities for sodium-based alternatives. Grid storage applications are projected to grow at 18.7% annually through 2030, as utilities seek cost-effective solutions for renewable energy integration.
Material innovations in solid-state sodium battery polymers address critical market demands for safety, sustainability, and cost reduction. The abundance of sodium resources (approximately 23,000 times more abundant in the Earth's crust than lithium) positions sodium-based technologies as economically advantageous, with raw material costs estimated at 30-40% lower than lithium-based counterparts. Recent polymer electrolyte breakthroughs have improved ionic conductivity to levels approaching 10^-3 S/cm at room temperature, addressing previous performance limitations.
Regional market analysis reveals varying adoption patterns, with Asia-Pacific leading manufacturing capacity development (68% of global production), while Europe focuses on research initiatives through programs like Horizon Europe, which has allocated €1.3 billion specifically for next-generation battery technologies including sodium-based systems. North America shows strong venture capital interest, with investments in sodium battery startups increasing by 215% between 2020 and 2023.
Industry forecasts indicate that solid-state sodium batteries utilizing advanced polymers could capture 8-12% of the energy storage market by 2030, primarily in stationary storage applications before expanding into mobility solutions. Cost projections suggest potential price points of $70-85/kWh by 2028, making them competitive with advanced lithium-ion technologies while offering superior sustainability profiles.
Customer adoption barriers include concerns about energy density (currently 30-40% lower than premium lithium-ion cells) and cycle life in certain applications. However, recent polymer composite developments incorporating ceramic fillers have demonstrated significant improvements, with some laboratory prototypes achieving over 2,000 cycles with minimal capacity degradation.
Consumer electronics, electric vehicles, and grid storage represent the primary market segments for advanced energy storage technologies. The electric vehicle segment alone is expected to consume over 2,800 GWh of battery capacity by 2030, creating substantial opportunities for sodium-based alternatives. Grid storage applications are projected to grow at 18.7% annually through 2030, as utilities seek cost-effective solutions for renewable energy integration.
Material innovations in solid-state sodium battery polymers address critical market demands for safety, sustainability, and cost reduction. The abundance of sodium resources (approximately 23,000 times more abundant in the Earth's crust than lithium) positions sodium-based technologies as economically advantageous, with raw material costs estimated at 30-40% lower than lithium-based counterparts. Recent polymer electrolyte breakthroughs have improved ionic conductivity to levels approaching 10^-3 S/cm at room temperature, addressing previous performance limitations.
Regional market analysis reveals varying adoption patterns, with Asia-Pacific leading manufacturing capacity development (68% of global production), while Europe focuses on research initiatives through programs like Horizon Europe, which has allocated €1.3 billion specifically for next-generation battery technologies including sodium-based systems. North America shows strong venture capital interest, with investments in sodium battery startups increasing by 215% between 2020 and 2023.
Industry forecasts indicate that solid-state sodium batteries utilizing advanced polymers could capture 8-12% of the energy storage market by 2030, primarily in stationary storage applications before expanding into mobility solutions. Cost projections suggest potential price points of $70-85/kWh by 2028, making them competitive with advanced lithium-ion technologies while offering superior sustainability profiles.
Customer adoption barriers include concerns about energy density (currently 30-40% lower than premium lithium-ion cells) and cycle life in certain applications. However, recent polymer composite developments incorporating ceramic fillers have demonstrated significant improvements, with some laboratory prototypes achieving over 2,000 cycles with minimal capacity degradation.
Current Polymer Electrolyte Challenges in Na-ion Batteries
Despite significant advancements in sodium-ion battery technology, polymer electrolytes for these systems face several critical challenges that impede their widespread commercial adoption. The primary obstacle remains insufficient ionic conductivity at room temperature, with most polymer electrolytes achieving only 10^-5 to 10^-6 S/cm, far below the 10^-3 S/cm threshold required for practical applications. This conductivity limitation stems from the larger ionic radius of Na+ (1.02 Å) compared to Li+ (0.76 Å), resulting in slower ion transport through polymer matrices.
The mechanical stability of polymer electrolytes presents another significant challenge. Many high-conductivity polymer systems suffer from poor mechanical properties, making them difficult to process and integrate into battery assemblies. The trade-off between mechanical strength and ionic conductivity has proven particularly difficult to overcome in sodium systems, where the larger cation size exacerbates interface contact issues.
Interfacial instability between polymer electrolytes and sodium metal anodes represents a critical barrier to development. Sodium's high reactivity leads to continuous electrolyte decomposition at the interface, forming resistive layers that increase impedance over time. Unlike lithium systems, where stable solid electrolyte interphase (SEI) formation is better understood, the mechanisms governing interfacial stability in sodium systems remain inadequately characterized.
Thermal stability limitations further constrain polymer electrolyte performance in sodium batteries. Many promising polymer systems exhibit significant conductivity only at elevated temperatures (>60°C), while others suffer from phase transitions or decomposition within practical operating temperature ranges. This narrow electrochemical stability window restricts the voltage range and energy density achievable in sodium polymer battery systems.
Long-term cycling stability remains problematic, with many polymer electrolytes showing significant performance degradation after repeated charge-discharge cycles. This degradation manifests as increasing internal resistance, capacity fade, and in some cases, catastrophic failure due to dendrite formation. The mechanisms behind this degradation are complex, involving chemical decomposition, morphological changes, and interfacial evolution specific to sodium-ion transport dynamics.
Manufacturing scalability presents additional challenges, as many laboratory-scale polymer electrolyte formulations utilize complex synthesis procedures or expensive materials that are difficult to scale industrially. The development of cost-effective, environmentally friendly polymer electrolytes that can be manufactured at scale remains a significant hurdle for commercial viability of solid-state sodium batteries.
The mechanical stability of polymer electrolytes presents another significant challenge. Many high-conductivity polymer systems suffer from poor mechanical properties, making them difficult to process and integrate into battery assemblies. The trade-off between mechanical strength and ionic conductivity has proven particularly difficult to overcome in sodium systems, where the larger cation size exacerbates interface contact issues.
Interfacial instability between polymer electrolytes and sodium metal anodes represents a critical barrier to development. Sodium's high reactivity leads to continuous electrolyte decomposition at the interface, forming resistive layers that increase impedance over time. Unlike lithium systems, where stable solid electrolyte interphase (SEI) formation is better understood, the mechanisms governing interfacial stability in sodium systems remain inadequately characterized.
Thermal stability limitations further constrain polymer electrolyte performance in sodium batteries. Many promising polymer systems exhibit significant conductivity only at elevated temperatures (>60°C), while others suffer from phase transitions or decomposition within practical operating temperature ranges. This narrow electrochemical stability window restricts the voltage range and energy density achievable in sodium polymer battery systems.
Long-term cycling stability remains problematic, with many polymer electrolytes showing significant performance degradation after repeated charge-discharge cycles. This degradation manifests as increasing internal resistance, capacity fade, and in some cases, catastrophic failure due to dendrite formation. The mechanisms behind this degradation are complex, involving chemical decomposition, morphological changes, and interfacial evolution specific to sodium-ion transport dynamics.
Manufacturing scalability presents additional challenges, as many laboratory-scale polymer electrolyte formulations utilize complex synthesis procedures or expensive materials that are difficult to scale industrially. The development of cost-effective, environmentally friendly polymer electrolytes that can be manufactured at scale remains a significant hurdle for commercial viability of solid-state sodium batteries.
State-of-the-Art Polymer Solutions for Sodium Batteries
01 Polymer electrolyte compositions for sodium batteries
Novel polymer electrolyte compositions specifically designed for solid-state sodium batteries have been developed to enhance ionic conductivity and electrochemical stability. These compositions typically include sodium-conducting polymers combined with various additives to optimize performance. The polymer matrices provide mechanical support while facilitating sodium ion transport between electrodes, resulting in improved battery efficiency and safety compared to liquid electrolyte systems.- Polymer electrolyte compositions for sodium batteries: Advanced polymer electrolyte compositions specifically designed for sodium batteries have been developed to enhance ionic conductivity and electrochemical stability. These compositions typically include sodium-conducting polymers combined with various additives to improve performance. The polymer matrices provide mechanical support while facilitating sodium ion transport, which is crucial for solid-state battery operation. These electrolytes offer advantages such as improved safety, longer cycle life, and better thermal stability compared to liquid electrolytes.
- Composite polymer-ceramic electrolytes: Hybrid electrolyte systems combining polymers with ceramic materials have emerged as a breakthrough for solid-state sodium batteries. These composites leverage the flexibility and processability of polymers with the high ionic conductivity of ceramic materials. The ceramic fillers, often sodium-conducting materials like NASICON-type compounds, create additional ion transport pathways within the polymer matrix. This synergistic combination results in enhanced mechanical properties, reduced interfacial resistance, and improved overall battery performance at room temperature.
- Novel cross-linked polymer networks: Innovative cross-linked polymer networks have been developed specifically for sodium battery applications. These networks feature optimized architectures that facilitate sodium ion transport while maintaining dimensional stability. The cross-linking strategy helps prevent crystallization of polymer chains, which typically impedes ion movement. Various cross-linking agents and techniques are employed to create three-dimensional networks with controlled porosity and ion channels. These materials demonstrate superior mechanical properties and electrochemical performance compared to traditional linear polymers.
- Polymer interface engineering: Significant breakthroughs have been achieved in engineering the interfaces between polymer electrolytes and electrodes in solid-state sodium batteries. Novel approaches include surface modification of electrodes, incorporation of interfacial layers, and development of specialized additives that improve adhesion and reduce interfacial resistance. These innovations address one of the major challenges in solid-state batteries: maintaining good contact between components during cycling. The engineered interfaces facilitate efficient sodium ion transfer across boundaries while preventing unwanted side reactions.
- Sodium-conducting block copolymers: Advanced block copolymer architectures have been designed specifically for sodium ion conduction. These materials feature distinct domains that separately optimize mechanical support and ion transport functions. The microphase separation in block copolymers creates well-defined ion-conducting channels that facilitate rapid sodium ion movement. Various combinations of polymer blocks have been explored, including polyethers, polycarbonates, and polyesters, each offering unique advantages. These materials represent a significant advancement over homopolymers, providing an excellent balance of mechanical properties and ionic conductivity.
02 Composite polymer-ceramic electrolytes
Hybrid electrolyte systems combining polymers with ceramic materials have emerged as a breakthrough for solid-state sodium batteries. These composites leverage the flexibility and processability of polymers with the high ionic conductivity of ceramic materials. The ceramic fillers create additional ion transport pathways and help suppress dendrite formation, while the polymer matrix provides improved interfacial contact with electrodes and enhanced mechanical properties.Expand Specific Solutions03 Cross-linked polymer networks for enhanced stability
Cross-linked polymer networks represent a significant advancement in solid-state sodium battery technology. These materials feature covalent bonds between polymer chains that create a three-dimensional structure with improved mechanical strength and thermal stability. The cross-linking approach prevents polymer crystallization, enhances sodium ion mobility, and maintains electrolyte integrity during battery cycling, resulting in extended battery lifespan and improved safety characteristics.Expand Specific Solutions04 Sodium-conducting block copolymers
Block copolymers with specialized architectures have been developed to optimize sodium ion transport in solid-state batteries. These materials contain distinct segments with different functionalities - typically combining rigid blocks for mechanical stability with flexible blocks containing polar groups for ion conduction. The self-assembling nature of block copolymers creates well-defined nanoscale channels that facilitate directional ion transport while maintaining dimensional stability under various operating conditions.Expand Specific Solutions05 Polymer interface engineering for electrode compatibility
Advanced interface engineering approaches have been developed to address the critical challenge of polymer electrolyte-electrode compatibility in solid-state sodium batteries. These innovations include functionalized polymers with specialized end groups that form stable interfaces with electrode materials, gradient polymer structures that minimize interfacial resistance, and composite interlayers that facilitate sodium ion transfer across boundaries. These developments significantly reduce impedance growth during cycling and improve overall battery performance.Expand Specific Solutions
Leading Companies and Research Institutions in Na-ion Technology
The solid-state sodium battery polymer market is in an early growth phase, characterized by intensive research and development rather than mass commercialization. The global market size is projected to expand significantly as sodium batteries emerge as a cost-effective alternative to lithium-ion technologies, with estimates suggesting a compound annual growth rate exceeding 25% through 2030. Technologically, academic institutions like University of California, Shenzhen University, and KAIST are pioneering fundamental research, while commercial entities including FUJIFILM, Solid Power, and LG Chem are advancing practical applications. The technology remains at TRL 4-6, with companies like Hyundai Motor and Sharp exploring integration possibilities. Key challenges include polymer electrolyte stability and sodium ion conductivity, with recent breakthroughs in composite polymer systems showing promise for commercial viability.
The Regents of the University of California
Technical Solution: The University of California research teams have developed several groundbreaking polymer electrolyte systems for sodium batteries. Their most notable innovation involves a comb-like polymer architecture with polyethylene oxide (PEO) side chains grafted onto a polyphosphazene backbone, creating highly flexible polymer chains with enhanced sodium ion coordination sites. This unique molecular design achieves ionic conductivities approaching 1 mS/cm at room temperature without sacrificing mechanical stability. Their research has demonstrated that incorporating specific sodium salts with large, delocalized anions (such as TFSI) significantly improves the dissociation behavior and transport properties within the polymer matrix. UC researchers have also pioneered the use of single-ion conducting polymers where the anion is tethered to the polymer backbone, eliminating concentration polarization issues that typically limit battery performance. Additionally, they've developed novel cross-linking strategies using UV-initiated reactions that allow precise control over the mechanical properties while maintaining optimal ion transport pathways through the electrolyte.
Strengths: Cutting-edge polymer chemistry with fundamental breakthroughs in molecular design; excellent room-temperature conductivity; superior electrochemical stability window. Weaknesses: Early-stage technology with limited scale-up demonstration; complex synthesis procedures that may challenge commercial production; potential long-term stability issues under real-world operating conditions.
Solid Power Operating, Inc.
Technical Solution: Solid Power has developed a proprietary sulfide-based solid electrolyte technology for sodium batteries that incorporates specialized polymer binders to enhance mechanical properties and ionic conductivity. Their approach focuses on a composite electrolyte system that combines inorganic sulfide materials with carefully engineered polymers to create a flexible, yet stable electrolyte interface. The company has demonstrated room temperature ionic conductivities exceeding 1 mS/cm in their sodium-ion conducting materials, which approaches the performance of their lithium counterparts. Their polymer-enhanced solid electrolytes maintain stability against metallic sodium and enable the use of high-capacity cathode materials that would otherwise be incompatible with liquid electrolytes. Solid Power's manufacturing process leverages roll-to-roll production techniques adapted from conventional battery manufacturing, allowing for scalable production of their solid-state sodium battery technology.
Strengths: Superior mechanical flexibility due to polymer integration; scalable manufacturing process compatible with existing production lines; better safety profile with non-flammable components. Weaknesses: Lower energy density compared to some lithium-based alternatives; polymer components may limit high-temperature operation; technology still requires optimization for power delivery in high-drain applications.
Critical Patents and Research in Sodium-Conducting Polymers
New polymers for battery applications
PatentWO2023175340A1
Innovation
- Development of tri-block copolymers with a polycarbonate-polyether structure, specifically designed to accommodate volume changes through phase separation and the inclusion of pendant functional groups for improved adhesion and mechanical properties, allowing for the use in electrolytes and cathodes to enhance battery performance.
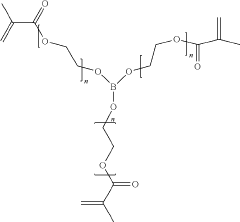
Boron-Containing Plastic Crystal Polymer and Preparation Method therefor and Application thereof
PatentActiveUS20220052377A1
Innovation
- A boron-containing plastic crystal polymer is prepared through a method involving a mixture of a plastic crystal, a metal salt, a boron-containing ternary crosslinker, and a photoinitiator, which is then cured and used as a solid-state electrolyte in all-solid-state ion batteries, enhancing ionic conductivity and mechanical strength.
Sustainability Impact of Sodium Battery Technologies
The transition to sodium-based battery technologies represents a significant advancement in sustainable energy storage solutions. Unlike lithium-ion batteries that rely on scarce and geographically concentrated lithium resources, sodium batteries utilize sodium - the sixth most abundant element in the Earth's crust. This abundance translates to reduced environmental impact from mining operations and substantially lower raw material costs, making sodium batteries a more economically viable option for large-scale energy storage applications.
The environmental footprint of sodium battery production is markedly smaller than that of conventional lithium-ion batteries. Life cycle assessments indicate that solid-state sodium battery manufacturing can reduce greenhouse gas emissions by up to 60% compared to traditional lithium-ion technologies. This reduction stems from both the accessibility of sodium resources and the simplified extraction processes required, which consume less energy and produce fewer harmful byproducts.
Polymer-based solid-state sodium batteries offer additional sustainability benefits through their enhanced safety profile. The elimination of flammable liquid electrolytes significantly reduces fire hazards and the potential for catastrophic thermal runaway events. This improved safety not only protects users and infrastructure but also minimizes the environmental impact of battery failures and disposal of damaged units.
End-of-life considerations further highlight the sustainability advantages of sodium battery technologies. The polymers used in solid-state sodium batteries can be designed for easier recyclability, with some recent innovations enabling recovery rates exceeding 90% for key components. Additionally, the reduced toxicity of sodium-based materials compared to certain lithium battery components simplifies waste management protocols and decreases the environmental burden of battery disposal.
From a global resource equity perspective, sodium battery technologies promote more balanced access to energy storage solutions. The widespread availability of sodium resources across different geographical regions can help democratize battery production capabilities, reducing dependency on the few countries that control the majority of lithium reserves. This redistribution of manufacturing potential could stimulate economic development in regions previously excluded from the battery production value chain.
The water footprint of sodium battery production deserves particular attention in sustainability assessments. Recent polymer electrolyte formulations for sodium batteries require significantly less water in their manufacturing processes compared to conventional battery technologies, addressing growing concerns about industrial water usage in regions facing water scarcity challenges.
The environmental footprint of sodium battery production is markedly smaller than that of conventional lithium-ion batteries. Life cycle assessments indicate that solid-state sodium battery manufacturing can reduce greenhouse gas emissions by up to 60% compared to traditional lithium-ion technologies. This reduction stems from both the accessibility of sodium resources and the simplified extraction processes required, which consume less energy and produce fewer harmful byproducts.
Polymer-based solid-state sodium batteries offer additional sustainability benefits through their enhanced safety profile. The elimination of flammable liquid electrolytes significantly reduces fire hazards and the potential for catastrophic thermal runaway events. This improved safety not only protects users and infrastructure but also minimizes the environmental impact of battery failures and disposal of damaged units.
End-of-life considerations further highlight the sustainability advantages of sodium battery technologies. The polymers used in solid-state sodium batteries can be designed for easier recyclability, with some recent innovations enabling recovery rates exceeding 90% for key components. Additionally, the reduced toxicity of sodium-based materials compared to certain lithium battery components simplifies waste management protocols and decreases the environmental burden of battery disposal.
From a global resource equity perspective, sodium battery technologies promote more balanced access to energy storage solutions. The widespread availability of sodium resources across different geographical regions can help democratize battery production capabilities, reducing dependency on the few countries that control the majority of lithium reserves. This redistribution of manufacturing potential could stimulate economic development in regions previously excluded from the battery production value chain.
The water footprint of sodium battery production deserves particular attention in sustainability assessments. Recent polymer electrolyte formulations for sodium batteries require significantly less water in their manufacturing processes compared to conventional battery technologies, addressing growing concerns about industrial water usage in regions facing water scarcity challenges.
Manufacturing Scalability and Cost Analysis
The scalability of manufacturing processes for solid-state sodium battery polymers represents a critical factor in their commercial viability. Current production methods for polymer electrolytes often involve laboratory-scale techniques that are difficult to translate to mass production environments. Solution casting, electrospinning, and melt processing techniques require significant adaptation to meet industrial throughput requirements while maintaining consistent material properties.
Cost analysis reveals that raw material expenses for sodium-based systems offer a distinct advantage over lithium counterparts, with sodium resources being approximately 1000 times more abundant and 30-40% less expensive. However, this inherent material cost advantage is currently offset by higher processing costs due to the specialized equipment needed for handling moisture-sensitive polymer electrolytes and the precise temperature control required during synthesis.
Energy consumption during manufacturing presents another significant cost factor. The processing of polymer electrolytes typically requires multiple heating and cooling cycles, contributing substantially to production expenses. Recent innovations in continuous processing techniques have demonstrated potential energy savings of 25-35% compared to batch processing methods, suggesting a pathway toward more economical production.
Equipment investment represents a substantial barrier to entry for manufacturers. The specialized mixing, extrusion, and coating equipment required for polymer electrolyte production can cost between $5-15 million for a modest production line. This capital expenditure necessitates high production volumes to achieve reasonable amortization periods, creating a challenging economic equation for new market entrants.
Yield rates in current manufacturing processes range from 65-80%, significantly lower than the 90%+ yields achieved in conventional lithium-ion battery component production. Each percentage point improvement in yield translates to approximately 0.8-1.2% reduction in overall production costs, highlighting the economic importance of process optimization.
Scaling considerations must also account for quality control challenges unique to polymer electrolytes. Ensuring uniform ionic conductivity, mechanical stability, and electrochemical performance across large production batches requires sophisticated in-line monitoring systems that add complexity and cost to manufacturing operations.
The transition from laboratory to industrial scale production will likely follow a stepwise approach, beginning with pilot lines producing 5-10 MWh equivalent of materials annually before scaling to gigawatt-hour capacities. This gradual scaling allows for process optimization while managing investment risk, with industry projections suggesting a 30-40% cost reduction through economies of scale once production volumes exceed 1 GWh annually.
Cost analysis reveals that raw material expenses for sodium-based systems offer a distinct advantage over lithium counterparts, with sodium resources being approximately 1000 times more abundant and 30-40% less expensive. However, this inherent material cost advantage is currently offset by higher processing costs due to the specialized equipment needed for handling moisture-sensitive polymer electrolytes and the precise temperature control required during synthesis.
Energy consumption during manufacturing presents another significant cost factor. The processing of polymer electrolytes typically requires multiple heating and cooling cycles, contributing substantially to production expenses. Recent innovations in continuous processing techniques have demonstrated potential energy savings of 25-35% compared to batch processing methods, suggesting a pathway toward more economical production.
Equipment investment represents a substantial barrier to entry for manufacturers. The specialized mixing, extrusion, and coating equipment required for polymer electrolyte production can cost between $5-15 million for a modest production line. This capital expenditure necessitates high production volumes to achieve reasonable amortization periods, creating a challenging economic equation for new market entrants.
Yield rates in current manufacturing processes range from 65-80%, significantly lower than the 90%+ yields achieved in conventional lithium-ion battery component production. Each percentage point improvement in yield translates to approximately 0.8-1.2% reduction in overall production costs, highlighting the economic importance of process optimization.
Scaling considerations must also account for quality control challenges unique to polymer electrolytes. Ensuring uniform ionic conductivity, mechanical stability, and electrochemical performance across large production batches requires sophisticated in-line monitoring systems that add complexity and cost to manufacturing operations.
The transition from laboratory to industrial scale production will likely follow a stepwise approach, beginning with pilot lines producing 5-10 MWh equivalent of materials annually before scaling to gigawatt-hour capacities. This gradual scaling allows for process optimization while managing investment risk, with industry projections suggesting a 30-40% cost reduction through economies of scale once production volumes exceed 1 GWh annually.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!