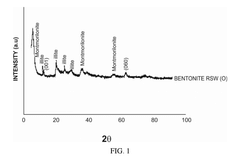
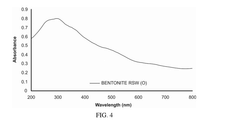
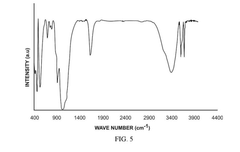
Solid-state sodium battery technology in pharmaceutical applications
OCT 27, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sodium Battery Evolution and Pharmaceutical Integration Goals
Sodium-ion battery technology has evolved significantly over the past three decades, transitioning from theoretical concepts to practical applications. Initially developed as an alternative to lithium-ion batteries due to sodium's greater abundance and lower cost, these batteries faced substantial challenges in energy density and cycle life. The evolution began with liquid electrolyte systems in the 1990s, progressing through various electrode material innovations in the 2000s, and culminating in recent breakthroughs in solid-state configurations that have dramatically improved safety profiles and operational stability.
The pharmaceutical industry's interest in sodium battery technology has grown in parallel with these advancements, driven by increasing needs for reliable power sources in drug delivery systems, portable diagnostic equipment, and remote patient monitoring devices. Traditional battery technologies have presented limitations in these applications, particularly regarding biocompatibility, size constraints, and operational longevity in biological environments.
Solid-state sodium batteries represent a significant technological leap, offering enhanced safety through elimination of flammable liquid electrolytes—a critical consideration for pharmaceutical applications where patient safety is paramount. These batteries also demonstrate improved stability at varying temperatures, addressing the strict environmental control requirements of pharmaceutical products and processes.
The integration goals for sodium battery technology in pharmaceuticals encompass several dimensions. Primary objectives include developing miniaturized power sources for implantable drug delivery systems that can operate reliably within the human body for extended periods without replacement. This requires batteries with high energy density, minimal self-discharge, and biocompatible components that won't trigger immune responses or tissue damage.
Another key goal involves creating flexible battery configurations that can conform to the complex geometries of pharmaceutical devices, enabling new form factors for both internal and external medical applications. This flexibility would allow for more patient-friendly designs and potentially expand treatment options for conditions requiring consistent medication delivery.
Temperature stability represents another critical integration target, as pharmaceutical applications often demand consistent performance across varying environmental conditions, from cold chain storage to body temperature operation. Solid-state sodium batteries show promising characteristics in this regard, maintaining performance across wider temperature ranges than conventional alternatives.
Long-term development aims include creating self-charging capabilities through integration with energy harvesting technologies, potentially allowing pharmaceutical devices to generate power from body movement, temperature differentials, or biochemical processes. This would represent a transformative advancement for chronic condition management, potentially enabling lifetime implantable drug delivery systems without battery replacement requirements.
The pharmaceutical industry's interest in sodium battery technology has grown in parallel with these advancements, driven by increasing needs for reliable power sources in drug delivery systems, portable diagnostic equipment, and remote patient monitoring devices. Traditional battery technologies have presented limitations in these applications, particularly regarding biocompatibility, size constraints, and operational longevity in biological environments.
Solid-state sodium batteries represent a significant technological leap, offering enhanced safety through elimination of flammable liquid electrolytes—a critical consideration for pharmaceutical applications where patient safety is paramount. These batteries also demonstrate improved stability at varying temperatures, addressing the strict environmental control requirements of pharmaceutical products and processes.
The integration goals for sodium battery technology in pharmaceuticals encompass several dimensions. Primary objectives include developing miniaturized power sources for implantable drug delivery systems that can operate reliably within the human body for extended periods without replacement. This requires batteries with high energy density, minimal self-discharge, and biocompatible components that won't trigger immune responses or tissue damage.
Another key goal involves creating flexible battery configurations that can conform to the complex geometries of pharmaceutical devices, enabling new form factors for both internal and external medical applications. This flexibility would allow for more patient-friendly designs and potentially expand treatment options for conditions requiring consistent medication delivery.
Temperature stability represents another critical integration target, as pharmaceutical applications often demand consistent performance across varying environmental conditions, from cold chain storage to body temperature operation. Solid-state sodium batteries show promising characteristics in this regard, maintaining performance across wider temperature ranges than conventional alternatives.
Long-term development aims include creating self-charging capabilities through integration with energy harvesting technologies, potentially allowing pharmaceutical devices to generate power from body movement, temperature differentials, or biochemical processes. This would represent a transformative advancement for chronic condition management, potentially enabling lifetime implantable drug delivery systems without battery replacement requirements.
Market Demand Analysis for Pharmaceutical Energy Solutions
The pharmaceutical industry is experiencing a significant shift in its energy requirements, driven by the need for more reliable, efficient, and sustainable power solutions. Current market analysis reveals a growing demand for advanced energy storage technologies that can address the unique challenges faced by pharmaceutical manufacturing, research facilities, and medical device applications. The global pharmaceutical energy market, particularly for specialized battery technologies, is projected to grow substantially as companies seek to enhance operational efficiency while reducing environmental impact.
Solid-state sodium battery technology represents a promising solution to meet these evolving needs. Market research indicates that pharmaceutical companies are increasingly prioritizing energy solutions that offer enhanced safety profiles, longer operational lifespans, and reduced maintenance requirements. Traditional lithium-ion batteries, while widely used, present concerns regarding thermal stability and potential fire hazards—issues particularly critical in pharmaceutical environments where contamination risks must be minimized and regulatory compliance is paramount.
The demand for portable and reliable power sources in pharmaceutical applications continues to expand across several key segments. Laboratory equipment, particularly sensitive analytical instruments, requires stable power supplies that can maintain consistent performance without interference. Similarly, the growing field of portable medical devices and point-of-care diagnostics necessitates compact energy solutions with extended operational capabilities. Market surveys indicate that pharmaceutical companies are willing to invest in premium energy technologies that can ensure uninterrupted operations and protect valuable research assets.
Temperature-sensitive pharmaceutical manufacturing and storage represent another significant market opportunity. Cold chain logistics and ultra-low temperature storage facilities for vaccines and biologics require dependable backup power systems that can maintain critical temperatures during grid failures. The recent global pandemic has further highlighted this need, accelerating investment in resilient energy infrastructure throughout the pharmaceutical supply chain.
Sustainability considerations are increasingly driving market demand as pharmaceutical companies establish ambitious carbon reduction targets. Energy solutions that offer improved environmental profiles, reduced resource consumption, and alignment with circular economy principles are gaining traction. Market analysis shows that companies are increasingly factoring total lifecycle environmental impact into procurement decisions, creating opportunities for technologies like sodium batteries that utilize more abundant and less environmentally problematic materials than conventional lithium-ion alternatives.
Regulatory factors are also shaping market dynamics, with stricter safety standards and sustainability requirements influencing technology adoption decisions. The pharmaceutical industry's highly regulated nature means that energy solutions must meet rigorous validation requirements, creating entry barriers but also premium opportunities for technologies that can demonstrate compliance advantages.
Solid-state sodium battery technology represents a promising solution to meet these evolving needs. Market research indicates that pharmaceutical companies are increasingly prioritizing energy solutions that offer enhanced safety profiles, longer operational lifespans, and reduced maintenance requirements. Traditional lithium-ion batteries, while widely used, present concerns regarding thermal stability and potential fire hazards—issues particularly critical in pharmaceutical environments where contamination risks must be minimized and regulatory compliance is paramount.
The demand for portable and reliable power sources in pharmaceutical applications continues to expand across several key segments. Laboratory equipment, particularly sensitive analytical instruments, requires stable power supplies that can maintain consistent performance without interference. Similarly, the growing field of portable medical devices and point-of-care diagnostics necessitates compact energy solutions with extended operational capabilities. Market surveys indicate that pharmaceutical companies are willing to invest in premium energy technologies that can ensure uninterrupted operations and protect valuable research assets.
Temperature-sensitive pharmaceutical manufacturing and storage represent another significant market opportunity. Cold chain logistics and ultra-low temperature storage facilities for vaccines and biologics require dependable backup power systems that can maintain critical temperatures during grid failures. The recent global pandemic has further highlighted this need, accelerating investment in resilient energy infrastructure throughout the pharmaceutical supply chain.
Sustainability considerations are increasingly driving market demand as pharmaceutical companies establish ambitious carbon reduction targets. Energy solutions that offer improved environmental profiles, reduced resource consumption, and alignment with circular economy principles are gaining traction. Market analysis shows that companies are increasingly factoring total lifecycle environmental impact into procurement decisions, creating opportunities for technologies like sodium batteries that utilize more abundant and less environmentally problematic materials than conventional lithium-ion alternatives.
Regulatory factors are also shaping market dynamics, with stricter safety standards and sustainability requirements influencing technology adoption decisions. The pharmaceutical industry's highly regulated nature means that energy solutions must meet rigorous validation requirements, creating entry barriers but also premium opportunities for technologies that can demonstrate compliance advantages.
Solid-State Sodium Battery Technology: Current Status and Barriers
Solid-state sodium batteries represent a significant advancement in energy storage technology, offering potential advantages over conventional lithium-ion batteries. Currently, these batteries are at various stages of development globally, with research institutions and companies in Asia, particularly Japan, South Korea, and China, leading many breakthrough initiatives. European and North American research centers are also making substantial contributions to the field.
The fundamental technology relies on solid electrolytes that conduct sodium ions between electrodes, eliminating the need for liquid components. This architecture addresses several critical issues in battery technology, including safety concerns related to flammable liquid electrolytes and the potential for higher energy density. However, despite promising theoretical advantages, several significant barriers impede widespread commercialization.
Interface stability remains one of the most challenging technical hurdles. The solid-solid interfaces between electrodes and electrolytes often develop high resistance over time, leading to capacity fade and shortened battery life. This issue is particularly pronounced in pharmaceutical applications where long-term stability is essential for devices like implantable drug delivery systems or portable diagnostic equipment.
Ionic conductivity presents another major barrier. While some solid electrolytes show promising conductivity at elevated temperatures, achieving comparable performance to liquid electrolytes at room temperature remains difficult. This limitation affects charging rates and power delivery capabilities, restricting potential pharmaceutical applications that require rapid response times.
Manufacturing scalability constitutes a significant industrial challenge. Current laboratory-scale production methods for solid electrolytes and electrode materials are often complex and expensive, involving specialized equipment and precise environmental controls. Transitioning these processes to mass production while maintaining quality and performance consistency has proven difficult.
Material costs and availability also present economic barriers. Some promising solid electrolytes contain elements with limited availability or geopolitical supply concerns. For pharmaceutical applications, where regulatory approval processes are rigorous, the stability of material supply chains becomes particularly critical.
Regulatory hurdles specific to pharmaceutical applications add another layer of complexity. Battery technologies intended for medical devices or drug delivery systems must meet stringent safety, reliability, and biocompatibility standards. The novel nature of solid-state sodium batteries means that regulatory frameworks for their evaluation in pharmaceutical contexts are still evolving.
Integration challenges with existing pharmaceutical technologies and infrastructure further complicate adoption. Current pharmaceutical devices and systems are designed around conventional battery technologies, requiring significant redesign to accommodate the different form factors, voltage profiles, and operational characteristics of solid-state sodium batteries.
The fundamental technology relies on solid electrolytes that conduct sodium ions between electrodes, eliminating the need for liquid components. This architecture addresses several critical issues in battery technology, including safety concerns related to flammable liquid electrolytes and the potential for higher energy density. However, despite promising theoretical advantages, several significant barriers impede widespread commercialization.
Interface stability remains one of the most challenging technical hurdles. The solid-solid interfaces between electrodes and electrolytes often develop high resistance over time, leading to capacity fade and shortened battery life. This issue is particularly pronounced in pharmaceutical applications where long-term stability is essential for devices like implantable drug delivery systems or portable diagnostic equipment.
Ionic conductivity presents another major barrier. While some solid electrolytes show promising conductivity at elevated temperatures, achieving comparable performance to liquid electrolytes at room temperature remains difficult. This limitation affects charging rates and power delivery capabilities, restricting potential pharmaceutical applications that require rapid response times.
Manufacturing scalability constitutes a significant industrial challenge. Current laboratory-scale production methods for solid electrolytes and electrode materials are often complex and expensive, involving specialized equipment and precise environmental controls. Transitioning these processes to mass production while maintaining quality and performance consistency has proven difficult.
Material costs and availability also present economic barriers. Some promising solid electrolytes contain elements with limited availability or geopolitical supply concerns. For pharmaceutical applications, where regulatory approval processes are rigorous, the stability of material supply chains becomes particularly critical.
Regulatory hurdles specific to pharmaceutical applications add another layer of complexity. Battery technologies intended for medical devices or drug delivery systems must meet stringent safety, reliability, and biocompatibility standards. The novel nature of solid-state sodium batteries means that regulatory frameworks for their evaluation in pharmaceutical contexts are still evolving.
Integration challenges with existing pharmaceutical technologies and infrastructure further complicate adoption. Current pharmaceutical devices and systems are designed around conventional battery technologies, requiring significant redesign to accommodate the different form factors, voltage profiles, and operational characteristics of solid-state sodium batteries.
Current Implementation Approaches in Pharmaceutical Applications
01 Electrolyte materials for solid-state sodium batteries
Various electrolyte materials can be used in solid-state sodium batteries to enhance ionic conductivity and battery performance. These materials include sodium-based solid electrolytes, polymer electrolytes, and composite electrolytes. The selection of appropriate electrolyte materials is crucial for achieving high ionic conductivity, good electrochemical stability, and improved interface compatibility between the electrolyte and electrodes in solid-state sodium batteries.- Solid-state electrolyte materials for sodium batteries: Various solid-state electrolyte materials are being developed specifically for sodium batteries to improve safety and performance. These materials include sodium-ion conducting ceramics, polymer electrolytes, and composite materials that offer high ionic conductivity while preventing dendrite formation. The solid electrolytes enable the elimination of flammable liquid components, enhancing the overall safety profile of sodium batteries while maintaining efficient sodium ion transport.
- Electrode materials and interfaces for solid-state sodium batteries: Advanced electrode materials and interface engineering are critical for solid-state sodium battery performance. Researchers are developing specialized cathode and anode materials compatible with solid electrolytes, focusing on reducing interfacial resistance and improving electrochemical stability. These developments include structured electrodes, buffer layers, and surface modifications that enhance the contact between electrodes and solid electrolytes, leading to better cycling performance and energy density.
- Manufacturing processes for solid-state sodium batteries: Novel manufacturing techniques are being developed to address the challenges of producing solid-state sodium batteries at scale. These processes include specialized sintering methods, cold pressing techniques, and advanced deposition approaches that enable the creation of thin, uniform layers with good interfacial contact. Innovations in manufacturing aim to reduce production costs while ensuring consistent quality and performance of the solid-state sodium battery components.
- Sodium battery architectures and designs: Innovative architectural designs for solid-state sodium batteries focus on optimizing cell configuration for improved performance and manufacturability. These designs include layered structures, 3D architectures, and novel cell geometries that maximize active material utilization while minimizing internal resistance. The battery architectures are engineered to accommodate the volume changes during cycling and to enhance thermal management, resulting in batteries with higher energy density and longer cycle life.
- Performance enhancement and stability solutions: Various approaches are being implemented to enhance the performance and stability of solid-state sodium batteries. These include additives that improve ionic conductivity, protective coatings that prevent side reactions, and composite structures that maintain mechanical integrity during cycling. Researchers are also developing strategies to mitigate issues such as volume expansion, interfacial degradation, and capacity fading, resulting in sodium batteries with improved cycle life, rate capability, and temperature stability.
02 Electrode materials and structures for sodium batteries
Advanced electrode materials and structures are essential for solid-state sodium batteries to achieve high energy density and long cycle life. These include various cathode materials (such as sodium transition metal oxides), anode materials (such as hard carbon, sodium metal, and alloys), and electrode architectures designed to accommodate sodium ion insertion/extraction. The development of electrode materials with high capacity, good rate capability, and structural stability is critical for improving the overall performance of solid-state sodium batteries.Expand Specific Solutions03 Interface engineering in solid-state sodium batteries
Interface engineering focuses on improving the contact between electrodes and solid electrolytes to reduce interfacial resistance and enhance electrochemical performance. This includes surface modifications, buffer layers, and interface stabilization techniques to mitigate issues such as chemical incompatibility and mechanical stress at interfaces. Effective interface engineering is crucial for achieving stable cycling performance and preventing dendrite formation in solid-state sodium batteries.Expand Specific Solutions04 Manufacturing processes for solid-state sodium batteries
Various manufacturing processes are employed to fabricate solid-state sodium batteries, including dry pressing, tape casting, solution processing, and advanced deposition techniques. These processes aim to achieve uniform component distribution, good interfacial contact, and high density of the battery components. The development of scalable and cost-effective manufacturing methods is essential for the commercialization of solid-state sodium batteries.Expand Specific Solutions05 Safety and performance enhancement strategies
Various strategies are employed to enhance the safety and performance of solid-state sodium batteries, including thermal management systems, protective coatings, and additives. These approaches aim to prevent thermal runaway, improve cycling stability, and extend battery life. The development of inherently safe battery designs with high energy density and long cycle life is a key focus area for advancing solid-state sodium battery technology.Expand Specific Solutions
Key Industry Players and Competitive Landscape
The solid-state sodium battery technology in pharmaceutical applications is in an early development stage, with market growth driven by increasing demand for safer, more sustainable energy storage solutions. The global market is expanding as pharmaceutical companies seek alternatives to traditional lithium-ion batteries. Key players demonstrate varying levels of technological maturity: academic institutions like Sichuan University and Beijing Institute of Technology are conducting foundational research, while commercial entities such as Broadbit Batteries Oy and Farasis Energy are advancing toward practical applications. Honda Motor, Murata Manufacturing, and PowerCo SE are leveraging their manufacturing expertise to scale production, while research organizations like Shanghai Institute of Ceramics and Forschungszentrum Jülich are focusing on material innovations to overcome current limitations in energy density and cycle life.
Shanghai Institute of Ceramics, Chinese Academy of Sciences
Technical Solution: The Shanghai Institute of Ceramics has developed an innovative solid-state sodium battery technology optimized for pharmaceutical applications. Their approach centers on a NASICON-type (Na3Zr2Si2PO12) ceramic electrolyte with proprietary dopants that enhance ionic conductivity to 3.2 mS/cm at body temperature while maintaining structural integrity. The institute's technology incorporates specialized interface engineering using thin polymer interlayers (PEO-based) that improve electrode-electrolyte contact and accommodate volume changes during cycling. Their batteries utilize a P2-type layered oxide cathode (Na0.7Fe0.5Mn0.5O2) that delivers stable voltage profiles essential for medical device operation. The manufacturing process employs low-temperature sintering techniques (700-800°C) that preserve the nanostructured electrode materials while enabling complex form factors suitable for pharmaceutical delivery systems. The resulting batteries demonstrate exceptional cycling stability (>1000 cycles at 80% capacity retention) and operate effectively across temperature ranges relevant to pharmaceutical storage and transport (-10°C to 50°C).
Strengths: Excellent thermal stability suitable for sterilization processes in pharmaceutical manufacturing; non-toxic components ensuring safety in medical applications; flexible form factors enabling integration into various drug delivery systems. Weaknesses: Higher manufacturing complexity compared to conventional batteries; limited production scale currently available; requires specialized expertise for integration with pharmaceutical systems.
Uchicago Argonne LLC
Technical Solution: Argonne National Laboratory has developed advanced solid-state sodium battery technology specifically tailored for pharmaceutical applications. Their approach utilizes a sodium-ion conducting ceramic electrolyte (Na3Zr2Si2PO12) that eliminates flammable liquid components, making it inherently safer for medical environments. The technology incorporates specialized polymer interfaces that enhance ion transport between electrodes and electrolyte while maintaining stability during temperature fluctuations common in pharmaceutical storage and transport. Argonne's batteries feature proprietary cathode materials optimized for stable voltage profiles required in drug delivery systems and medical devices. Their design achieves energy densities of 160-180 Wh/kg while operating effectively across a wide temperature range (-20°C to 60°C), addressing pharmaceutical cold chain requirements.
Strengths: Superior safety profile with non-flammable components ideal for medical settings; excellent thermal stability supporting pharmaceutical cold chain requirements; longer cycle life (2000+ cycles) reducing replacement frequency in implantable devices. Weaknesses: Higher manufacturing costs compared to conventional batteries; lower energy density than some lithium alternatives; more limited track record in commercial pharmaceutical applications.
Critical Patents and Technical Innovations Analysis
All solid-state sodium-sulfur or lithium-sulfur battery prepared using cast-annealing method
PatentActiveUS20210028440A1
Innovation
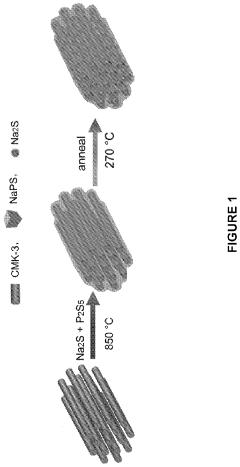
- A melting-casting process followed by a stress-release annealing-precipitation process is used to fabricate sodium and lithium nanocomposites, incorporating ordered mesoporous carbon with Na2S or Li2S and Na3PS4 or Li3PS4, which reduces interfacial resistance and eliminates stress, enhancing electrochemical performance by ensuring close contact between the solid electrolyte and electron conductive agent without residential stress.
Solid-state sodium silicate battery (SSSB) employing sodium and calcium rich electrolyte enriched with sodium
PatentActiveIN202241033884A
Innovation
- A solid-state sodium silicate battery design featuring a sodium silicate cathode, carbon black anode, and a solid-state-electrolyte formed by coating a naturally Sodium and Calcium rich material, enriched with sodium, on a Polypropylene separator membrane, which enhances ionic conductivity and stability.
Regulatory Framework for Medical Device Power Solutions
The regulatory landscape for solid-state sodium battery technology in pharmaceutical applications presents a complex framework that manufacturers must navigate carefully. Medical devices powered by these innovative batteries are subject to stringent oversight from multiple regulatory bodies, with the FDA in the United States and the EMA in Europe serving as primary gatekeepers. These agencies have established specific requirements for battery-powered medical devices, focusing on safety, reliability, and performance characteristics.
FDA regulations, particularly 21 CFR Part 820 for quality system requirements, mandate comprehensive documentation of battery performance, safety testing, and risk assessment. For solid-state sodium batteries integrated into pharmaceutical delivery systems, additional considerations under combination product regulations (21 CFR Part 4) apply, requiring manufacturers to demonstrate both drug and device compliance.
In Europe, the Medical Device Regulation (MDR 2017/745) has significantly increased requirements for technical documentation and post-market surveillance of battery-powered medical devices. Manufacturers must provide detailed evidence of battery safety, including thermal stability assessments particularly relevant to sodium-based technologies, which have different thermal profiles compared to traditional lithium-ion systems.
International standards play a crucial role in regulatory compliance, with IEC 60601-1 serving as the cornerstone for electrical medical device safety. For solid-state sodium batteries specifically, emerging standards like IEC 62133-5 (under development) will address safety requirements for sodium-ion technologies in portable applications, though pharmaceutical-specific adaptations remain in early stages.
Environmental regulations also impact the development pathway, with the EU's Restriction of Hazardous Substances (RoHS) Directive and Battery Directive establishing parameters for material selection and end-of-life management. The reduced environmental impact of sodium compared to lithium presents potential regulatory advantages, though full lifecycle assessment requirements are increasingly stringent.
Regulatory pathways for novel battery technologies in pharmaceutical applications typically require extensive pre-market testing, including biocompatibility assessment (ISO 10993 series) when batteries are incorporated into implantable or wearable drug delivery systems. The FDA's breakthrough device designation and EMA's innovation pathways offer potential accelerated routes for truly innovative solid-state sodium battery technologies that demonstrate significant advantages over existing solutions.
Ongoing regulatory developments suggest a trend toward harmonization of international requirements for medical device power solutions, with increasing emphasis on cybersecurity for connected devices and sustainability metrics throughout the product lifecycle. Manufacturers pursuing solid-state sodium battery implementation must maintain vigilance regarding these evolving regulatory frameworks to ensure successful market access.
FDA regulations, particularly 21 CFR Part 820 for quality system requirements, mandate comprehensive documentation of battery performance, safety testing, and risk assessment. For solid-state sodium batteries integrated into pharmaceutical delivery systems, additional considerations under combination product regulations (21 CFR Part 4) apply, requiring manufacturers to demonstrate both drug and device compliance.
In Europe, the Medical Device Regulation (MDR 2017/745) has significantly increased requirements for technical documentation and post-market surveillance of battery-powered medical devices. Manufacturers must provide detailed evidence of battery safety, including thermal stability assessments particularly relevant to sodium-based technologies, which have different thermal profiles compared to traditional lithium-ion systems.
International standards play a crucial role in regulatory compliance, with IEC 60601-1 serving as the cornerstone for electrical medical device safety. For solid-state sodium batteries specifically, emerging standards like IEC 62133-5 (under development) will address safety requirements for sodium-ion technologies in portable applications, though pharmaceutical-specific adaptations remain in early stages.
Environmental regulations also impact the development pathway, with the EU's Restriction of Hazardous Substances (RoHS) Directive and Battery Directive establishing parameters for material selection and end-of-life management. The reduced environmental impact of sodium compared to lithium presents potential regulatory advantages, though full lifecycle assessment requirements are increasingly stringent.
Regulatory pathways for novel battery technologies in pharmaceutical applications typically require extensive pre-market testing, including biocompatibility assessment (ISO 10993 series) when batteries are incorporated into implantable or wearable drug delivery systems. The FDA's breakthrough device designation and EMA's innovation pathways offer potential accelerated routes for truly innovative solid-state sodium battery technologies that demonstrate significant advantages over existing solutions.
Ongoing regulatory developments suggest a trend toward harmonization of international requirements for medical device power solutions, with increasing emphasis on cybersecurity for connected devices and sustainability metrics throughout the product lifecycle. Manufacturers pursuing solid-state sodium battery implementation must maintain vigilance regarding these evolving regulatory frameworks to ensure successful market access.
Sustainability and Environmental Impact Assessment
The integration of solid-state sodium battery technology in pharmaceutical applications presents significant sustainability advantages and environmental benefits compared to conventional battery technologies. These batteries eliminate the need for toxic and flammable liquid electrolytes, substantially reducing environmental hazards associated with production, usage, and disposal phases. The abundant nature of sodium resources—approximately 1000 times more plentiful than lithium in the Earth's crust—offers a more sustainable alternative to lithium-ion batteries, alleviating concerns about resource depletion and geopolitical supply chain vulnerabilities.
From a life cycle assessment perspective, solid-state sodium batteries demonstrate reduced carbon footprints during manufacturing processes. The elimination of organic solvents and simplified production methods contribute to lower energy consumption and decreased greenhouse gas emissions. Additionally, these batteries typically require less energy-intensive extraction methods compared to lithium mining operations, which often involve substantial water usage and habitat disruption.
In pharmaceutical applications specifically, the environmental benefits become even more pronounced. Temperature-controlled pharmaceutical storage and transportation systems powered by solid-state sodium batteries can maintain consistent conditions with minimal environmental impact. The extended cycle life of these batteries—potentially reaching 3000-5000 cycles compared to 500-1000 cycles for conventional batteries—significantly reduces waste generation and resource consumption over time.
The end-of-life management of solid-state sodium batteries also presents environmental advantages. Their components are generally less toxic and more recyclable than conventional battery materials. Preliminary research indicates recovery rates of up to 90% for sodium and other key materials, creating opportunities for closed-loop recycling systems within pharmaceutical supply chains.
However, certain environmental challenges remain unaddressed. The production of solid electrolytes sometimes involves energy-intensive high-temperature processes, and some formulations utilize rare earth elements that carry their own sustainability concerns. Comprehensive environmental impact assessments must consider these factors alongside the benefits when evaluating implementation in pharmaceutical contexts.
Regulatory frameworks are evolving to address these emerging technologies, with organizations like the EPA and European Chemicals Agency developing specific guidelines for sodium battery production and disposal. Pharmaceutical companies adopting this technology will need to align with these evolving standards while demonstrating environmental stewardship through transparent sustainability reporting and responsible material sourcing practices.
From a life cycle assessment perspective, solid-state sodium batteries demonstrate reduced carbon footprints during manufacturing processes. The elimination of organic solvents and simplified production methods contribute to lower energy consumption and decreased greenhouse gas emissions. Additionally, these batteries typically require less energy-intensive extraction methods compared to lithium mining operations, which often involve substantial water usage and habitat disruption.
In pharmaceutical applications specifically, the environmental benefits become even more pronounced. Temperature-controlled pharmaceutical storage and transportation systems powered by solid-state sodium batteries can maintain consistent conditions with minimal environmental impact. The extended cycle life of these batteries—potentially reaching 3000-5000 cycles compared to 500-1000 cycles for conventional batteries—significantly reduces waste generation and resource consumption over time.
The end-of-life management of solid-state sodium batteries also presents environmental advantages. Their components are generally less toxic and more recyclable than conventional battery materials. Preliminary research indicates recovery rates of up to 90% for sodium and other key materials, creating opportunities for closed-loop recycling systems within pharmaceutical supply chains.
However, certain environmental challenges remain unaddressed. The production of solid electrolytes sometimes involves energy-intensive high-temperature processes, and some formulations utilize rare earth elements that carry their own sustainability concerns. Comprehensive environmental impact assessments must consider these factors alongside the benefits when evaluating implementation in pharmaceutical contexts.
Regulatory frameworks are evolving to address these emerging technologies, with organizations like the EPA and European Chemicals Agency developing specific guidelines for sodium battery production and disposal. Pharmaceutical companies adopting this technology will need to align with these evolving standards while demonstrating environmental stewardship through transparent sustainability reporting and responsible material sourcing practices.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!