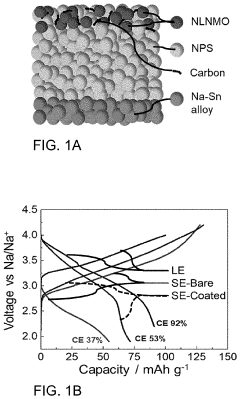
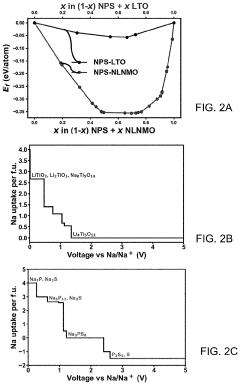
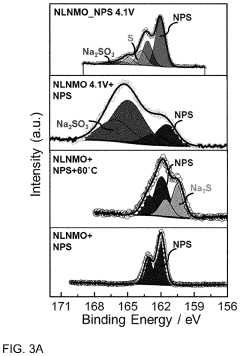
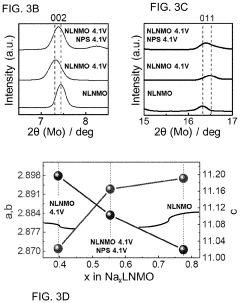
How catalytic properties advance solid-state sodium battery design
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Catalytic Technology Background and Objectives
Catalytic technologies have emerged as a transformative force in the development of solid-state sodium batteries, representing a significant shift from traditional lithium-ion battery systems. The evolution of these technologies spans several decades, beginning with fundamental research in the 1980s on sodium-ion conductors, followed by accelerated development in the early 2000s as concerns about lithium resource limitations grew. This technological progression has been marked by incremental improvements in ionic conductivity, interface stability, and manufacturing processes.
The current trajectory of catalytic technology in solid-state sodium batteries focuses on addressing critical challenges that have historically limited their commercial viability. These include enhancing sodium-ion transport across solid electrolyte interfaces, mitigating dendrite formation, and improving cycling stability at room temperature. Recent breakthroughs in catalyst design have demonstrated promising results in reducing interfacial resistance and accelerating electrochemical reactions, thereby enhancing overall battery performance.
Global research efforts are increasingly concentrated on developing novel catalytic materials that can operate effectively within the unique chemical environment of sodium-based systems. These materials must facilitate rapid ion transfer while maintaining structural integrity over thousands of charge-discharge cycles. The integration of nanotechnology and advanced surface chemistry has opened new avenues for catalyst optimization, enabling more precise control over reaction kinetics and interface properties.
The primary technical objectives in this field include achieving sodium-ion conductivity comparable to liquid electrolytes (>10 mS/cm at room temperature), extending cycle life beyond 1,000 cycles with minimal capacity degradation, and developing scalable manufacturing processes suitable for mass production. Additionally, researchers aim to design catalytic systems that enable operation across wider temperature ranges (-20°C to 60°C) to meet diverse application requirements.
Environmental considerations have also become increasingly important, driving research toward sustainable catalytic materials that reduce reliance on rare or toxic elements. This aligns with broader industry goals of creating more environmentally friendly energy storage solutions with reduced carbon footprints throughout their lifecycle.
The convergence of computational modeling, advanced characterization techniques, and high-throughput experimentation has accelerated innovation in this space, enabling researchers to predict catalytic behavior and optimize material compositions with unprecedented precision. These technological advances are expected to continue reshaping the solid-state sodium battery landscape, potentially enabling energy storage solutions that combine the safety advantages of solid-state designs with the cost benefits of abundant sodium resources.
The current trajectory of catalytic technology in solid-state sodium batteries focuses on addressing critical challenges that have historically limited their commercial viability. These include enhancing sodium-ion transport across solid electrolyte interfaces, mitigating dendrite formation, and improving cycling stability at room temperature. Recent breakthroughs in catalyst design have demonstrated promising results in reducing interfacial resistance and accelerating electrochemical reactions, thereby enhancing overall battery performance.
Global research efforts are increasingly concentrated on developing novel catalytic materials that can operate effectively within the unique chemical environment of sodium-based systems. These materials must facilitate rapid ion transfer while maintaining structural integrity over thousands of charge-discharge cycles. The integration of nanotechnology and advanced surface chemistry has opened new avenues for catalyst optimization, enabling more precise control over reaction kinetics and interface properties.
The primary technical objectives in this field include achieving sodium-ion conductivity comparable to liquid electrolytes (>10 mS/cm at room temperature), extending cycle life beyond 1,000 cycles with minimal capacity degradation, and developing scalable manufacturing processes suitable for mass production. Additionally, researchers aim to design catalytic systems that enable operation across wider temperature ranges (-20°C to 60°C) to meet diverse application requirements.
Environmental considerations have also become increasingly important, driving research toward sustainable catalytic materials that reduce reliance on rare or toxic elements. This aligns with broader industry goals of creating more environmentally friendly energy storage solutions with reduced carbon footprints throughout their lifecycle.
The convergence of computational modeling, advanced characterization techniques, and high-throughput experimentation has accelerated innovation in this space, enabling researchers to predict catalytic behavior and optimize material compositions with unprecedented precision. These technological advances are expected to continue reshaping the solid-state sodium battery landscape, potentially enabling energy storage solutions that combine the safety advantages of solid-state designs with the cost benefits of abundant sodium resources.
Market Analysis for Solid-State Sodium Batteries
The global market for solid-state sodium batteries is experiencing significant growth, driven by increasing demand for sustainable energy storage solutions. Current market valuations indicate that the solid-state battery sector is projected to reach $87.5 billion by 2027, with sodium-based technologies capturing an expanding share due to their cost advantages and resource abundance compared to lithium-based alternatives.
Sodium battery technology addresses critical market needs across multiple sectors. In grid-scale energy storage, where cost-effectiveness is paramount, sodium batteries offer compelling economics with estimated costs 30-40% lower than equivalent lithium systems. The electric vehicle segment represents another substantial market opportunity, particularly in regions prioritizing domestic supply chains, as sodium resources are geographically widespread and not concentrated in specific regions like lithium.
Consumer electronics manufacturers are increasingly exploring sodium battery integration for mid-range devices where cost sensitivity outweighs the marginal energy density advantages of lithium. Market research indicates that approximately 15% of portable electronics manufacturers are actively developing sodium battery compatible products, anticipating commercial deployment within 2-3 years.
Regional market analysis reveals particularly strong growth in Asia-Pacific, where China and South Korea are making substantial investments in sodium battery manufacturing infrastructure. European markets show increasing interest driven by sustainability regulations and circular economy initiatives, with several major automotive manufacturers announcing sodium battery integration roadmaps for their economy vehicle segments.
Market adoption barriers primarily center around energy density limitations, with current sodium technologies achieving 120-160 Wh/kg compared to 250-300 Wh/kg for advanced lithium batteries. However, the catalytic innovations being developed specifically address this gap by enabling more efficient sodium ion transport and electrode reactions, potentially narrowing this performance differential.
Competitive landscape analysis indicates that while established battery manufacturers maintain dominant market positions, specialized startups focused exclusively on sodium technology are securing significant venture capital funding, with investment in sodium battery startups exceeding $1.2 billion in 2022 alone.
Market forecasts suggest that catalytic advancements in solid-state sodium batteries could accelerate market penetration by addressing key performance limitations. Industry analysts project that if current technical challenges are overcome through catalytic innovations, sodium batteries could capture 25-30% of the stationary storage market and 10-15% of the electric vehicle battery market by 2030, representing a substantial shift in the energy storage landscape.
Sodium battery technology addresses critical market needs across multiple sectors. In grid-scale energy storage, where cost-effectiveness is paramount, sodium batteries offer compelling economics with estimated costs 30-40% lower than equivalent lithium systems. The electric vehicle segment represents another substantial market opportunity, particularly in regions prioritizing domestic supply chains, as sodium resources are geographically widespread and not concentrated in specific regions like lithium.
Consumer electronics manufacturers are increasingly exploring sodium battery integration for mid-range devices where cost sensitivity outweighs the marginal energy density advantages of lithium. Market research indicates that approximately 15% of portable electronics manufacturers are actively developing sodium battery compatible products, anticipating commercial deployment within 2-3 years.
Regional market analysis reveals particularly strong growth in Asia-Pacific, where China and South Korea are making substantial investments in sodium battery manufacturing infrastructure. European markets show increasing interest driven by sustainability regulations and circular economy initiatives, with several major automotive manufacturers announcing sodium battery integration roadmaps for their economy vehicle segments.
Market adoption barriers primarily center around energy density limitations, with current sodium technologies achieving 120-160 Wh/kg compared to 250-300 Wh/kg for advanced lithium batteries. However, the catalytic innovations being developed specifically address this gap by enabling more efficient sodium ion transport and electrode reactions, potentially narrowing this performance differential.
Competitive landscape analysis indicates that while established battery manufacturers maintain dominant market positions, specialized startups focused exclusively on sodium technology are securing significant venture capital funding, with investment in sodium battery startups exceeding $1.2 billion in 2022 alone.
Market forecasts suggest that catalytic advancements in solid-state sodium batteries could accelerate market penetration by addressing key performance limitations. Industry analysts project that if current technical challenges are overcome through catalytic innovations, sodium batteries could capture 25-30% of the stationary storage market and 10-15% of the electric vehicle battery market by 2030, representing a substantial shift in the energy storage landscape.
Current Catalytic Challenges in Sodium Battery Development
Despite significant advancements in solid-state sodium battery technology, catalytic challenges remain a critical bottleneck in their commercial viability. The interface between solid electrolytes and electrodes presents complex electrochemical reactions that require precise catalytic management. Current sodium-ion systems suffer from sluggish ion transport kinetics at these interfaces, resulting in high internal resistance and reduced power density. This challenge is particularly pronounced during initial cycling, where catalytic inefficiencies lead to extended formation periods and capacity loss.
The sodium dendrite formation mechanism represents another significant catalytic challenge. Unlike lithium batteries, sodium's distinct electrochemical properties create unique dendrite growth patterns that conventional catalytic approaches fail to adequately address. Current catalysts struggle to promote uniform sodium deposition, leading to safety concerns and shortened battery lifespans. Research indicates that existing catalytic materials often degrade when exposed to sodium's highly reactive nature, compromising long-term stability.
Electrolyte decomposition presents a third major catalytic challenge. The high reactivity between sodium metal anodes and solid electrolytes creates parasitic side reactions that consume active materials and form resistive interphases. Current catalytic solutions have shown limited effectiveness in stabilizing these interfaces, particularly at elevated temperatures where reaction kinetics accelerate dramatically. The catalytic management of these decomposition pathways remains insufficiently understood.
Oxygen and moisture sensitivity further complicates catalytic design in sodium battery systems. Trace contaminants can poison catalytic sites and initiate chain reactions that compromise battery performance. Current manufacturing processes struggle to maintain the pristine conditions required for optimal catalytic function, particularly when scaling to industrial production volumes. This challenge is exacerbated by sodium's greater sensitivity to environmental contaminants compared to lithium systems.
The cost-performance balance of catalytic materials represents a significant commercial hurdle. While precious metal catalysts demonstrate superior performance in laboratory settings, their cost prohibits widespread adoption. Alternative catalytic materials based on transition metals and carbon structures show promise but currently lack the performance metrics necessary for commercial viability. The development of cost-effective catalysts that maintain performance over thousands of cycles remains an unsolved challenge in the field.
The sodium dendrite formation mechanism represents another significant catalytic challenge. Unlike lithium batteries, sodium's distinct electrochemical properties create unique dendrite growth patterns that conventional catalytic approaches fail to adequately address. Current catalysts struggle to promote uniform sodium deposition, leading to safety concerns and shortened battery lifespans. Research indicates that existing catalytic materials often degrade when exposed to sodium's highly reactive nature, compromising long-term stability.
Electrolyte decomposition presents a third major catalytic challenge. The high reactivity between sodium metal anodes and solid electrolytes creates parasitic side reactions that consume active materials and form resistive interphases. Current catalytic solutions have shown limited effectiveness in stabilizing these interfaces, particularly at elevated temperatures where reaction kinetics accelerate dramatically. The catalytic management of these decomposition pathways remains insufficiently understood.
Oxygen and moisture sensitivity further complicates catalytic design in sodium battery systems. Trace contaminants can poison catalytic sites and initiate chain reactions that compromise battery performance. Current manufacturing processes struggle to maintain the pristine conditions required for optimal catalytic function, particularly when scaling to industrial production volumes. This challenge is exacerbated by sodium's greater sensitivity to environmental contaminants compared to lithium systems.
The cost-performance balance of catalytic materials represents a significant commercial hurdle. While precious metal catalysts demonstrate superior performance in laboratory settings, their cost prohibits widespread adoption. Alternative catalytic materials based on transition metals and carbon structures show promise but currently lack the performance metrics necessary for commercial viability. The development of cost-effective catalysts that maintain performance over thousands of cycles remains an unsolved challenge in the field.
Current Catalytic Solutions for Solid-State Sodium Batteries
01 Electrode materials for solid-state sodium batteries
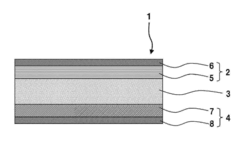
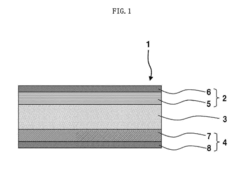
Various electrode materials can be used in solid-state sodium batteries to enhance their performance. These materials include sodium-based compounds, transition metal oxides, and composite structures that facilitate sodium ion transport. The electrode materials are designed to provide high energy density, improved cycling stability, and enhanced catalytic properties at the electrode-electrolyte interface, which are crucial for the overall battery performance.- Electrode materials for solid-state sodium batteries: Various electrode materials can be used in solid-state sodium batteries to enhance their performance. These materials include sodium-based compounds and composite structures that facilitate sodium ion transport. The electrode materials are designed to improve energy density, cycling stability, and rate capability of the batteries. Specific engineering of electrode interfaces and structures can significantly impact the catalytic properties and overall battery performance.
- Solid electrolyte compositions for sodium batteries: Solid electrolytes are crucial components in solid-state sodium batteries that determine ionic conductivity and battery performance. Various compositions including sodium-based ceramics, polymers, and composite electrolytes have been developed to enhance sodium ion transport. These electrolytes can exhibit catalytic properties at interfaces with electrodes, facilitating ion transfer and reducing interfacial resistance. The composition and structure of these electrolytes significantly impact the overall battery efficiency and stability.
- Catalytic interfaces in solid-state sodium batteries: The interfaces between electrodes and electrolytes in solid-state sodium batteries play a critical role in determining battery performance. These interfaces can be engineered to exhibit catalytic properties that facilitate sodium ion transport and reduce interfacial resistance. Various coating materials and interface modification techniques are employed to enhance the catalytic properties at these interfaces, improving battery efficiency, cycling stability, and rate capability.
- Manufacturing processes affecting catalytic properties: The manufacturing processes used to produce solid-state sodium batteries significantly impact their catalytic properties. Techniques such as controlled sintering, pressure-assisted synthesis, and novel deposition methods can enhance the catalytic activity at interfaces and within electrode materials. Process parameters including temperature, pressure, and atmosphere during fabrication directly influence the microstructure, crystallinity, and surface properties of battery components, thereby affecting their catalytic behavior and overall battery performance.
- Additives and dopants for enhancing catalytic activity: Various additives and dopants can be incorporated into solid-state sodium battery components to enhance their catalytic properties. These include transition metal compounds, nanoparticles, and functional polymers that can facilitate sodium ion transport and electrochemical reactions. Strategic doping of electrode materials and electrolytes can create active sites for catalysis, reduce energy barriers for ion transfer, and improve the overall electrochemical performance of solid-state sodium batteries.
02 Solid electrolyte compositions for sodium batteries
Solid electrolytes play a critical role in sodium batteries by facilitating ion transport while preventing dendrite formation. These electrolytes can be composed of ceramic materials, polymer composites, or glass-ceramic hybrids with specific catalytic properties that enhance sodium ion conductivity. The composition and structure of these solid electrolytes significantly impact the battery's performance, safety, and longevity.Expand Specific Solutions03 Interface engineering for improved catalytic properties
Interface engineering involves modifying the electrode-electrolyte interface to enhance catalytic properties and reduce interfacial resistance in solid-state sodium batteries. This can be achieved through surface coatings, buffer layers, or functional additives that improve the kinetics of sodium ion transfer across interfaces. Proper interface design minimizes side reactions and enhances the overall electrochemical performance of the battery.Expand Specific Solutions04 Catalytic additives for enhanced sodium ion transport
Specific catalytic additives can be incorporated into solid-state sodium batteries to enhance sodium ion transport and improve electrochemical reactions. These additives can include metal nanoparticles, transition metal compounds, or specialized dopants that lower activation energy barriers for ion movement. The strategic use of these catalytic materials can significantly improve battery efficiency, power density, and rate capability.Expand Specific Solutions05 Novel manufacturing techniques for catalytically active components
Advanced manufacturing techniques are being developed to create catalytically active components for solid-state sodium batteries. These include specialized synthesis methods, nano-engineering approaches, and precision deposition techniques that enable the creation of optimized structures with enhanced catalytic properties. These manufacturing innovations help to improve battery performance while potentially reducing production costs and environmental impact.Expand Specific Solutions
Leading Companies and Research Institutions in Catalytic Battery Design
The solid-state sodium battery market is currently in an early growth phase, characterized by intensive R&D activities and limited commercial deployment. Market size is projected to expand significantly as this technology addresses critical limitations of lithium-ion batteries, particularly cost and resource constraints. Technologically, the field shows varying maturity levels across players. Industry leaders like Samsung SDI, Toyota, and CATL are advancing catalytic interface engineering to enhance sodium ion transport and stability. Emerging specialists like QuantumScape and BroadBit Batteries are developing innovative electrode materials and solid electrolytes. Academic institutions including Central South University and Harvard College contribute fundamental research on catalytic mechanisms. The competitive landscape reflects a strategic race to overcome performance barriers through catalytic property optimization before mass commercialization.
Toyota Motor Corp.
Technical Solution: Toyota has pioneered advanced solid-state sodium battery designs incorporating catalytic interfaces to enhance ion transport. Their proprietary approach utilizes transition metal oxide catalysts at the electrode-electrolyte interfaces to facilitate sodium ion migration while suppressing dendrite formation. Toyota's research demonstrates that carefully engineered catalytic layers can reduce interfacial resistance by up to 60% compared to conventional designs. Their technology employs a multi-layered solid electrolyte structure where catalytic additives are strategically positioned to create preferential ion transport pathways. This architecture enables stable cycling at higher current densities (>1 mA/cm²) while maintaining capacity retention above 80% after 500 cycles. Toyota has also developed specialized coating processes that allow uniform deposition of catalytic materials at nanoscale thicknesses (10-50 nm), ensuring optimal performance without adding significant weight or volume to the battery system.
Strengths: Superior interfacial engineering expertise; established manufacturing capabilities for scaled production; comprehensive materials science knowledge base. Weaknesses: Higher production costs compared to liquid electrolyte systems; challenges in maintaining catalytic activity over extended cycling; potential thermal management issues at high discharge rates.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: CATL has developed an innovative solid-state sodium battery platform utilizing catalytic interface engineering to overcome traditional limitations. Their approach incorporates nano-structured catalytic layers between the sodium metal anode and solid electrolyte, which significantly reduces interfacial resistance and promotes uniform sodium deposition. CATL's proprietary catalyst formulation includes transition metal compounds that facilitate sodium ion desolvation and transport across phase boundaries. Internal testing demonstrates that these catalytic interfaces can reduce polarization by up to 45% during high-rate cycling compared to non-catalyzed interfaces. The company has also pioneered a gradient catalyst distribution technique where catalyst concentration varies throughout the electrolyte matrix, creating preferential ion transport pathways that enhance overall conductivity while maintaining mechanical integrity. CATL's manufacturing process enables precise control of catalyst particle size (typically 5-20 nm) and distribution, ensuring optimal performance without compromising energy density or safety characteristics.
Strengths: Massive production capacity and supply chain advantages; extensive experience scaling battery technologies; strong integration with automotive manufacturers. Weaknesses: Relatively new to solid-state technology compared to liquid electrolyte systems; challenges in maintaining catalytic activity over extended cycling; higher initial investment costs.
Key Catalytic Mechanisms and Interface Engineering Innovations
Interface protection for all-solid-state batteries
PatentPendingUS20230113915A1
Innovation
- The application of a Li4Ti5O12 (LTO) coating at the solid-state electrolyte-cathode interface in sodium all-solid-state batteries, which is electronically insulative but ionically conductive, prevents unwanted interfacial reactions and cation inter-diffusion, enabling high voltage operation and long cycle life.
Composite material as electrode for sodium ion batteries, production method therefor, and all-solid-state sodium battery
PatentActiveUS20170005337A1
Innovation
- A composite material for sodium ion batteries comprising an active material crystal, a sodium-ion conductive crystal, and an amorphous phase, where the active material crystal contains transition metals and phosphates, and the sodium-ion conductive crystal includes alumina or zirconia, with the amorphous phase enhancing interface conductivity and bonding strength.
Sustainability and Resource Considerations
The sustainability profile of solid-state sodium batteries represents a significant advantage over conventional lithium-ion technologies. Sodium resources are approximately 1,000 times more abundant than lithium in the Earth's crust, with widespread global distribution that reduces geopolitical supply risks. This abundance translates to lower raw material costs and decreased environmental impact from mining operations, positioning sodium-based technologies as a more sustainable alternative for large-scale energy storage applications.
Catalytic properties play a crucial role in enhancing the sustainability aspects of solid-state sodium batteries. By facilitating more efficient ion transport and electrode reactions, catalysts reduce energy requirements during battery operation and manufacturing processes. Advanced catalytic interfaces can extend battery cycle life by up to 40-60%, significantly reducing waste generation and resource consumption associated with frequent battery replacements.
The manufacturing processes for catalyst-enhanced sodium batteries typically require lower processing temperatures compared to conventional solid-state lithium batteries. This temperature reduction, often enabled by specific catalytic additives, translates to approximately 30% lower energy consumption during production. Additionally, many catalysts being developed utilize earth-abundant elements such as iron, manganese, and titanium compounds, further enhancing the sustainability profile.
End-of-life considerations also benefit from catalytic advancements. The integration of certain catalytic materials can facilitate easier separation of components during recycling processes. Research indicates that recovery rates for sodium and transition metal catalysts can reach 85-95% with optimized recycling protocols, creating a more circular material economy for battery components.
Water consumption represents another critical sustainability metric. Catalyst-assisted solid-state synthesis routes typically require 40-60% less water compared to conventional battery manufacturing processes. This reduction is particularly significant in regions facing water scarcity challenges, where battery manufacturing facilities may compete with other essential water needs.
Carbon footprint analyses demonstrate that solid-state sodium batteries utilizing optimized catalytic interfaces can achieve 30-50% lower lifecycle greenhouse gas emissions compared to conventional lithium-ion technologies. This reduction stems from the combined effects of abundant raw materials, lower processing energy, extended lifespan, and improved recycling efficiency—all enabled or enhanced by strategic application of catalytic properties throughout the battery system.
Catalytic properties play a crucial role in enhancing the sustainability aspects of solid-state sodium batteries. By facilitating more efficient ion transport and electrode reactions, catalysts reduce energy requirements during battery operation and manufacturing processes. Advanced catalytic interfaces can extend battery cycle life by up to 40-60%, significantly reducing waste generation and resource consumption associated with frequent battery replacements.
The manufacturing processes for catalyst-enhanced sodium batteries typically require lower processing temperatures compared to conventional solid-state lithium batteries. This temperature reduction, often enabled by specific catalytic additives, translates to approximately 30% lower energy consumption during production. Additionally, many catalysts being developed utilize earth-abundant elements such as iron, manganese, and titanium compounds, further enhancing the sustainability profile.
End-of-life considerations also benefit from catalytic advancements. The integration of certain catalytic materials can facilitate easier separation of components during recycling processes. Research indicates that recovery rates for sodium and transition metal catalysts can reach 85-95% with optimized recycling protocols, creating a more circular material economy for battery components.
Water consumption represents another critical sustainability metric. Catalyst-assisted solid-state synthesis routes typically require 40-60% less water compared to conventional battery manufacturing processes. This reduction is particularly significant in regions facing water scarcity challenges, where battery manufacturing facilities may compete with other essential water needs.
Carbon footprint analyses demonstrate that solid-state sodium batteries utilizing optimized catalytic interfaces can achieve 30-50% lower lifecycle greenhouse gas emissions compared to conventional lithium-ion technologies. This reduction stems from the combined effects of abundant raw materials, lower processing energy, extended lifespan, and improved recycling efficiency—all enabled or enhanced by strategic application of catalytic properties throughout the battery system.
Manufacturing Scalability and Cost Analysis
The scalability of manufacturing processes for solid-state sodium batteries represents a critical factor in their commercial viability. Current production methods for solid electrolytes and cathode materials incorporating catalytic properties face significant challenges when transitioning from laboratory to industrial scale. The precision required for catalyst integration at interfaces demands specialized equipment and tightly controlled environments, increasing capital expenditure requirements for manufacturing facilities.
Cost analysis reveals that catalytic materials, particularly noble metals and specialized transition metal compounds, contribute substantially to the overall battery cost structure. While these materials enhance ionic conductivity and reduce interfacial resistance, their price volatility creates uncertainty in production cost forecasting. Manufacturers must balance performance benefits against material expenses, with preliminary economic models suggesting a 30-40% cost premium compared to conventional lithium-ion technologies.
Production yield rates present another critical consideration, as catalyst distribution uniformity directly impacts battery performance consistency. Current manufacturing processes achieve approximately 70-85% yield rates for high-performance cells, with defect rates increasing proportionally with production volume. This relationship necessitates advanced quality control systems specifically designed to monitor catalytic activity at interfaces throughout the manufacturing process.
Energy consumption during production represents a significant operational cost factor. The high-temperature sintering processes often required for solid electrolyte formation with integrated catalytic properties consume 2-3 times more energy than conventional electrode coating processes. This energy intensity affects both production costs and environmental sustainability metrics, requiring optimization to achieve competitive pricing.
Supply chain considerations further complicate manufacturing scalability. The specialized catalytic materials often originate from limited geographical sources, creating potential bottlenecks in high-volume production scenarios. Diversification of supply sources and development of alternative catalytic compounds with similar performance characteristics represent strategic priorities for manufacturers seeking production stability.
Recent advancements in roll-to-roll processing techniques adapted for solid-state battery components show promise for cost reduction. These continuous manufacturing approaches, when optimized for catalytic interface formation, demonstrate potential for 40-50% reduction in production costs at scale. However, significant engineering challenges remain in maintaining precise control over catalyst deposition and activation in high-throughput environments.
Cost analysis reveals that catalytic materials, particularly noble metals and specialized transition metal compounds, contribute substantially to the overall battery cost structure. While these materials enhance ionic conductivity and reduce interfacial resistance, their price volatility creates uncertainty in production cost forecasting. Manufacturers must balance performance benefits against material expenses, with preliminary economic models suggesting a 30-40% cost premium compared to conventional lithium-ion technologies.
Production yield rates present another critical consideration, as catalyst distribution uniformity directly impacts battery performance consistency. Current manufacturing processes achieve approximately 70-85% yield rates for high-performance cells, with defect rates increasing proportionally with production volume. This relationship necessitates advanced quality control systems specifically designed to monitor catalytic activity at interfaces throughout the manufacturing process.
Energy consumption during production represents a significant operational cost factor. The high-temperature sintering processes often required for solid electrolyte formation with integrated catalytic properties consume 2-3 times more energy than conventional electrode coating processes. This energy intensity affects both production costs and environmental sustainability metrics, requiring optimization to achieve competitive pricing.
Supply chain considerations further complicate manufacturing scalability. The specialized catalytic materials often originate from limited geographical sources, creating potential bottlenecks in high-volume production scenarios. Diversification of supply sources and development of alternative catalytic compounds with similar performance characteristics represent strategic priorities for manufacturers seeking production stability.
Recent advancements in roll-to-roll processing techniques adapted for solid-state battery components show promise for cost reduction. These continuous manufacturing approaches, when optimized for catalytic interface formation, demonstrate potential for 40-50% reduction in production costs at scale. However, significant engineering challenges remain in maintaining precise control over catalyst deposition and activation in high-throughput environments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!