How to Exploit Microfluidics for Rapid Prototyping Needs
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Microfluidics Background and Development Goals
Microfluidics emerged in the early 1990s as a revolutionary field combining principles of fluid mechanics, physics, chemistry, and engineering at the microscale. This technology manipulates fluids in channels with dimensions of tens to hundreds of micrometers, enabling precise control over small volumes of liquids. The evolution of microfluidics has been marked by significant advancements in fabrication techniques, from initial silicon-based approaches to more accessible polymer-based methods, dramatically reducing both cost and complexity.
The field has experienced exponential growth over the past two decades, with applications expanding from initial analytical chemistry uses to diverse areas including point-of-care diagnostics, drug discovery, and organ-on-chip platforms. This growth trajectory has been fueled by increasing demand for miniaturized, efficient, and cost-effective solutions across multiple industries, particularly in healthcare and biotechnology sectors.
Current microfluidic technologies face limitations in rapid prototyping scenarios, where traditional fabrication methods like photolithography and soft lithography remain time-consuming and require specialized equipment. This creates a significant barrier for researchers and developers seeking to quickly iterate designs and test concepts, highlighting the need for more agile development approaches.
The primary development goal for microfluidics in rapid prototyping contexts is to establish methodologies that dramatically reduce the design-to-testing cycle time while maintaining precision and reproducibility. This includes developing accessible fabrication techniques that can be implemented with minimal specialized equipment, creating standardized components that can be easily integrated into custom designs, and establishing open platforms that facilitate knowledge sharing across the research community.
Another critical objective is to bridge the gap between academic research and commercial applications by developing scalable manufacturing processes that maintain the advantages of microfluidic systems while enabling mass production. This transition requires addressing challenges related to materials compatibility, channel geometry consistency, and integration with existing laboratory or industrial workflows.
Looking forward, the field aims to develop "plug-and-play" microfluidic systems that allow non-specialists to rapidly prototype and implement custom solutions for specific applications. This democratization of microfluidic technology would significantly expand its impact across various sectors, from personalized medicine to environmental monitoring and beyond, by enabling more researchers and developers to leverage its capabilities without extensive specialized training.
The field has experienced exponential growth over the past two decades, with applications expanding from initial analytical chemistry uses to diverse areas including point-of-care diagnostics, drug discovery, and organ-on-chip platforms. This growth trajectory has been fueled by increasing demand for miniaturized, efficient, and cost-effective solutions across multiple industries, particularly in healthcare and biotechnology sectors.
Current microfluidic technologies face limitations in rapid prototyping scenarios, where traditional fabrication methods like photolithography and soft lithography remain time-consuming and require specialized equipment. This creates a significant barrier for researchers and developers seeking to quickly iterate designs and test concepts, highlighting the need for more agile development approaches.
The primary development goal for microfluidics in rapid prototyping contexts is to establish methodologies that dramatically reduce the design-to-testing cycle time while maintaining precision and reproducibility. This includes developing accessible fabrication techniques that can be implemented with minimal specialized equipment, creating standardized components that can be easily integrated into custom designs, and establishing open platforms that facilitate knowledge sharing across the research community.
Another critical objective is to bridge the gap between academic research and commercial applications by developing scalable manufacturing processes that maintain the advantages of microfluidic systems while enabling mass production. This transition requires addressing challenges related to materials compatibility, channel geometry consistency, and integration with existing laboratory or industrial workflows.
Looking forward, the field aims to develop "plug-and-play" microfluidic systems that allow non-specialists to rapidly prototype and implement custom solutions for specific applications. This democratization of microfluidic technology would significantly expand its impact across various sectors, from personalized medicine to environmental monitoring and beyond, by enabling more researchers and developers to leverage its capabilities without extensive specialized training.
Market Analysis for Rapid Prototyping Microfluidic Solutions
The global microfluidics market for rapid prototyping applications has been experiencing significant growth, driven by increasing demand for point-of-care diagnostics, drug discovery, and personalized medicine. The market was valued at approximately $13.5 billion in 2021 and is projected to reach $37.6 billion by 2028, growing at a CAGR of 15.7% during the forecast period.
Rapid prototyping in microfluidics addresses critical market needs across multiple sectors. In healthcare, there is substantial demand for quick development of diagnostic devices, particularly following the COVID-19 pandemic which highlighted the necessity for rapid test development capabilities. The pharmaceutical industry represents another major market segment, where microfluidic technologies enable faster drug screening and reduced development costs.
Academic and research institutions constitute a significant customer base, requiring flexible prototyping solutions for various experimental setups. This sector values cost-effective systems that can be easily modified for different research applications. The industrial sector, particularly in chemical processing and environmental monitoring, shows growing interest in customizable microfluidic solutions for process optimization and quality control.
Regional analysis reveals North America currently dominates the market with approximately 40% share, attributed to strong research infrastructure and substantial investment in biotechnology. Europe follows with 30% market share, demonstrating particular strength in academic research applications. The Asia-Pacific region represents the fastest-growing market segment with 18% annual growth, driven by expanding healthcare infrastructure and increasing R&D investments in China, Japan, and South Korea.
Customer segmentation indicates three primary user groups: large enterprises (35% of market), small-to-medium enterprises (40%), and academic/research institutions (25%). Large enterprises typically seek integrated solutions compatible with existing workflows, while SMEs prioritize cost-effectiveness and versatility. Academic users value open platforms that allow extensive customization.
Key market drivers include increasing pressure to reduce time-to-market for new products, growing adoption of lab-on-a-chip technologies, and rising demand for point-of-care diagnostics. The shift toward personalized medicine creates substantial opportunities for microfluidic rapid prototyping solutions that can facilitate customized diagnostic and treatment approaches.
Market barriers include high initial investment costs, technical complexity requiring specialized expertise, and challenges in scaling from prototype to mass production. Additionally, regulatory hurdles particularly affect medical applications, where extensive validation is required before commercialization.
Rapid prototyping in microfluidics addresses critical market needs across multiple sectors. In healthcare, there is substantial demand for quick development of diagnostic devices, particularly following the COVID-19 pandemic which highlighted the necessity for rapid test development capabilities. The pharmaceutical industry represents another major market segment, where microfluidic technologies enable faster drug screening and reduced development costs.
Academic and research institutions constitute a significant customer base, requiring flexible prototyping solutions for various experimental setups. This sector values cost-effective systems that can be easily modified for different research applications. The industrial sector, particularly in chemical processing and environmental monitoring, shows growing interest in customizable microfluidic solutions for process optimization and quality control.
Regional analysis reveals North America currently dominates the market with approximately 40% share, attributed to strong research infrastructure and substantial investment in biotechnology. Europe follows with 30% market share, demonstrating particular strength in academic research applications. The Asia-Pacific region represents the fastest-growing market segment with 18% annual growth, driven by expanding healthcare infrastructure and increasing R&D investments in China, Japan, and South Korea.
Customer segmentation indicates three primary user groups: large enterprises (35% of market), small-to-medium enterprises (40%), and academic/research institutions (25%). Large enterprises typically seek integrated solutions compatible with existing workflows, while SMEs prioritize cost-effectiveness and versatility. Academic users value open platforms that allow extensive customization.
Key market drivers include increasing pressure to reduce time-to-market for new products, growing adoption of lab-on-a-chip technologies, and rising demand for point-of-care diagnostics. The shift toward personalized medicine creates substantial opportunities for microfluidic rapid prototyping solutions that can facilitate customized diagnostic and treatment approaches.
Market barriers include high initial investment costs, technical complexity requiring specialized expertise, and challenges in scaling from prototype to mass production. Additionally, regulatory hurdles particularly affect medical applications, where extensive validation is required before commercialization.
Current Microfluidic Technologies and Fabrication Challenges
Microfluidics technology has evolved significantly over the past two decades, transitioning from academic research to commercial applications across various industries. Current microfluidic technologies can be broadly categorized into several platforms: pressure-driven systems, electrokinetic systems, digital microfluidics, centrifugal microfluidics, and acoustic-driven microfluidics. Each platform offers distinct advantages for specific applications, with pressure-driven systems dominating commercial applications due to their reliability and scalability.
Despite technological advancements, fabrication of microfluidic devices remains challenging, particularly for rapid prototyping purposes. Traditional fabrication methods like photolithography and soft lithography require specialized equipment, clean room facilities, and technical expertise. These methods typically involve multiple steps including master mold fabrication, polymer casting, and bonding processes, resulting in lengthy production cycles ranging from days to weeks. Such timeframes are incompatible with rapid prototyping needs where iterative design improvements require quick turnaround times.
Material limitations present another significant challenge. Polydimethylsiloxane (PDMS) remains the most widely used material for prototyping due to its optical transparency, gas permeability, and ease of molding. However, PDMS has drawbacks including solvent absorption, hydrophobicity recovery after surface treatment, and limited compatibility with certain chemicals. Alternative materials like thermoplastics offer better chemical resistance but present more complex fabrication requirements.
Integration challenges further complicate microfluidic device development. Connecting microfluidic chips to macro-scale equipment for fluid handling, detection, and control often requires custom interfaces that are difficult to standardize. This "world-to-chip" interface problem remains a persistent obstacle in the field, particularly for rapid prototyping where quick assembly and testing are essential.
Reproducibility issues also plague current fabrication methods. Manual processes introduce variability between batches, affecting channel dimensions, surface properties, and ultimately device performance. This variability becomes particularly problematic during scale-up from prototype to production, often necessitating significant redesign work.
Emerging fabrication technologies are beginning to address these challenges. 3D printing offers promising solutions for rapid prototyping with direct digital manufacturing capabilities, though resolution limitations and material constraints remain. Laser-based techniques like selective laser ablation provide faster turnaround times but may compromise surface quality. Paper-based microfluidics presents a low-cost alternative for certain applications but lacks the precision and functionality of traditional platforms.
The gap between academic research and industrial implementation remains substantial. While research laboratories continue to develop novel fabrication approaches, industrial adoption is hindered by concerns regarding reproducibility, standardization, and scalability. Bridging this gap requires developing fabrication technologies specifically optimized for rapid prototyping needs.
Despite technological advancements, fabrication of microfluidic devices remains challenging, particularly for rapid prototyping purposes. Traditional fabrication methods like photolithography and soft lithography require specialized equipment, clean room facilities, and technical expertise. These methods typically involve multiple steps including master mold fabrication, polymer casting, and bonding processes, resulting in lengthy production cycles ranging from days to weeks. Such timeframes are incompatible with rapid prototyping needs where iterative design improvements require quick turnaround times.
Material limitations present another significant challenge. Polydimethylsiloxane (PDMS) remains the most widely used material for prototyping due to its optical transparency, gas permeability, and ease of molding. However, PDMS has drawbacks including solvent absorption, hydrophobicity recovery after surface treatment, and limited compatibility with certain chemicals. Alternative materials like thermoplastics offer better chemical resistance but present more complex fabrication requirements.
Integration challenges further complicate microfluidic device development. Connecting microfluidic chips to macro-scale equipment for fluid handling, detection, and control often requires custom interfaces that are difficult to standardize. This "world-to-chip" interface problem remains a persistent obstacle in the field, particularly for rapid prototyping where quick assembly and testing are essential.
Reproducibility issues also plague current fabrication methods. Manual processes introduce variability between batches, affecting channel dimensions, surface properties, and ultimately device performance. This variability becomes particularly problematic during scale-up from prototype to production, often necessitating significant redesign work.
Emerging fabrication technologies are beginning to address these challenges. 3D printing offers promising solutions for rapid prototyping with direct digital manufacturing capabilities, though resolution limitations and material constraints remain. Laser-based techniques like selective laser ablation provide faster turnaround times but may compromise surface quality. Paper-based microfluidics presents a low-cost alternative for certain applications but lacks the precision and functionality of traditional platforms.
The gap between academic research and industrial implementation remains substantial. While research laboratories continue to develop novel fabrication approaches, industrial adoption is hindered by concerns regarding reproducibility, standardization, and scalability. Bridging this gap requires developing fabrication technologies specifically optimized for rapid prototyping needs.
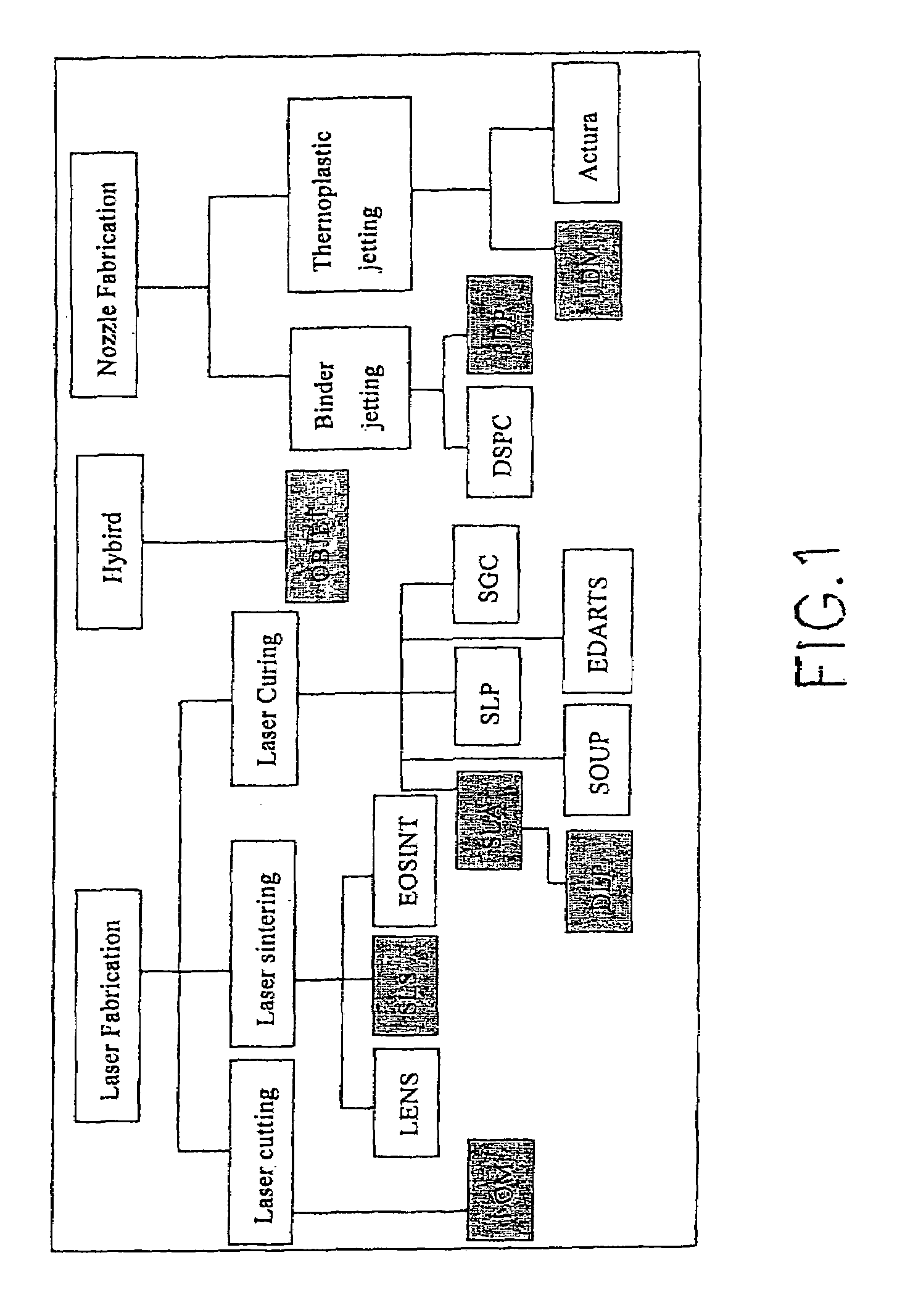
Current Approaches to Microfluidic Rapid Prototyping
01 Soft lithography techniques for microfluidic device fabrication
Soft lithography is a key technique for rapid prototyping of microfluidic devices, involving the use of elastomeric materials like PDMS (polydimethylsiloxane) to create microchannels and structures. This approach allows for quick iteration of designs without requiring expensive cleanroom facilities. The process typically involves creating a master mold, casting the elastomer, curing, and bonding to create sealed microfluidic channels. This technique enables researchers and engineers to rapidly test and refine microfluidic designs for various applications.- Soft lithography techniques for microfluidic device fabrication: Soft lithography is a key technique for rapid prototyping of microfluidic devices, involving the use of elastomeric materials like PDMS (polydimethylsiloxane) to create microchannels and structures. This approach allows for quick iteration of designs without requiring expensive cleanroom facilities. The process typically involves creating a master mold, casting the elastomer, curing, and bonding to create sealed microfluidic channels. This technique enables researchers and engineers to rapidly test and refine microfluidic designs for various applications.
- 3D printing for microfluidic device prototyping: 3D printing technologies have emerged as valuable tools for rapid prototyping of microfluidic devices. These additive manufacturing approaches allow for the direct fabrication of complex three-dimensional microfluidic structures without the need for multiple fabrication steps. Various 3D printing methods including stereolithography, digital light processing, and fused deposition modeling can be employed to create microfluidic chips with intricate channel geometries. This approach significantly reduces development time and enables rapid iteration of designs for applications in biomedical research, chemical analysis, and point-of-care diagnostics.
- Automated design and fabrication systems for microfluidics: Automated systems that integrate design software with fabrication equipment enable streamlined rapid prototyping of microfluidic devices. These systems allow users to design microfluidic layouts through computer-aided design interfaces and directly translate them to physical prototypes through automated manufacturing processes. Such integration reduces human error, increases reproducibility, and accelerates the development cycle. Advanced systems may incorporate simulation capabilities to predict fluid behavior before physical fabrication, further optimizing the design process.
- Novel materials for microfluidic rapid prototyping: The development of novel materials has expanded the capabilities of microfluidic rapid prototyping. Beyond traditional PDMS, materials such as thermoplastics, photocurable resins, hydrogels, and paper-based substrates offer unique properties for specific applications. These materials can provide benefits such as improved chemical resistance, optical transparency, biocompatibility, or reduced cost. Selection of appropriate materials based on application requirements is crucial for successful microfluidic device development, with considerations including surface properties, mechanical strength, and compatibility with biological samples.
- Modular and reconfigurable microfluidic systems: Modular approaches to microfluidic device fabrication allow for rapid assembly and reconfiguration of components without complete redesign and fabrication. These systems typically consist of standardized microfluidic building blocks that can be connected in various configurations to create customized fluid handling networks. This approach enables quick prototyping through the assembly of pre-fabricated components, facilitating iterative testing and optimization. Modular systems are particularly valuable for educational settings, proof-of-concept demonstrations, and applications requiring frequent reconfiguration of fluid handling architectures.
02 3D printing for microfluidic device prototyping
3D printing technologies have emerged as valuable tools for rapid prototyping of microfluidic devices. These additive manufacturing approaches allow for the direct fabrication of complex three-dimensional microfluidic structures without the need for multiple fabrication steps. Various 3D printing methods including stereolithography, digital light processing, and fused deposition modeling can be employed to create microfluidic devices with intricate channel geometries. This approach significantly reduces the time from design to testing and enables customization for specific applications.Expand Specific Solutions03 Computer-aided design and simulation for microfluidic prototyping
Computer-aided design (CAD) and simulation tools play a crucial role in microfluidic rapid prototyping by enabling the optimization of designs before physical fabrication. These software tools allow engineers to model fluid flow, predict device performance, and identify potential issues prior to manufacturing. Simulation capabilities can include computational fluid dynamics, particle tracing, and chemical reaction modeling. This approach reduces the number of physical prototypes needed, saving time and materials in the development process.Expand Specific Solutions04 Rapid bonding and assembly techniques for microfluidic devices
Various bonding and assembly techniques have been developed to facilitate rapid prototyping of microfluidic devices. These include plasma treatment for PDMS-glass bonding, adhesive bonding, thermal bonding, and solvent bonding methods. Quick assembly approaches using mechanical clamping or magnetic fixtures have also been implemented. These techniques enable fast iteration through multiple design versions by reducing the time required for device assembly and allowing for modular approaches to microfluidic system development.Expand Specific Solutions05 Materials innovation for microfluidic rapid prototyping
Novel materials have been developed specifically for microfluidic rapid prototyping applications. These include photocurable resins with optimized optical and mechanical properties, thermoplastics with improved processing characteristics, and hybrid materials that combine advantages of multiple material types. Some materials feature biocompatibility for cell-based applications, while others offer chemical resistance or optical transparency. These material innovations enable faster prototyping cycles and expand the application range of microfluidic devices.Expand Specific Solutions
Leading Companies and Research Institutions in Microfluidics
Microfluidics for rapid prototyping is currently in a growth phase, with the market expected to reach significant expansion due to increasing applications in healthcare, pharmaceuticals, and biotechnology. The technology enables miniaturization of laboratory processes, offering advantages in speed, cost, and sample conservation. Academic institutions like University of California, Shenzhen University, and Technical University of Munich are driving fundamental research, while commercial players including BOE Technology, IBM, 10X Genomics, and Netri SAS are advancing practical applications. The field is approaching maturity in certain segments such as lab-on-a-chip devices, but emerging applications in organ-on-chip technology and personalized medicine represent new frontiers where companies like Roche Diagnostics and AMSilk are making significant investments to capitalize on future growth opportunities.
The Johns Hopkins University
Technical Solution: Johns Hopkins University has developed innovative approaches to microfluidic rapid prototyping through their biomedical engineering and materials science departments. Their technology focuses on accessible microfluidic fabrication methods that enable rapid iteration and testing. Johns Hopkins researchers have pioneered paper-based microfluidics that utilize wax printing and laser cutting techniques to create low-cost, rapidly prototyped devices for point-of-care diagnostics. Their approach includes modular microfluidic building blocks that can be assembled in various configurations to quickly test different designs without complete refabrication. The university has developed specialized hydrogel-based microfluidic systems that can be rapidly photopatterned to create complex 3D microenvironments for tissue engineering and drug testing applications. Their technology incorporates computational fluid dynamics modeling integrated with rapid fabrication workflows to optimize designs before physical implementation. Johns Hopkins has also created open-source design tools and fabrication protocols specifically tailored for rapid microfluidic prototyping in resource-limited settings, enabling broader access to microfluidic technology development.
Strengths: Strong interdisciplinary collaboration between engineering, medicine, and materials science; focus on accessible and translatable technologies; innovative approaches to low-cost fabrication methods. Weaknesses: As an academic institution, may face challenges in scaling technologies to commercial production; research priorities may shift with funding opportunities; intellectual property considerations may complicate technology transfer.
Netri SAS
Technical Solution: Netri specializes in organ-on-chip technology using advanced microfluidic systems for neuroscience applications. Their NeuroFluidic platform enables the creation of controlled microenvironments that mimic physiological conditions for neuronal cultures. For rapid prototyping, Netri has developed a unique approach combining microfluidic engineering with neurobiology expertise. Their technology utilizes standardized microfluidic building blocks that can be rapidly assembled into customized configurations, allowing researchers to quickly prototype different neural circuit designs. Netri employs direct laser writing and soft lithography techniques optimized for rapid iteration, reducing fabrication time from weeks to days. Their microfluidic chips feature precisely controlled microchannels that guide axonal growth between compartmentalized neuronal populations, enabling the study of neural network formation and function. The company has also developed automated perfusion systems that maintain stable microenvironments while allowing for precise spatiotemporal control of biochemical signals, critical for neurological disease modeling and drug testing applications.
Strengths: Highly specialized expertise in neuroscience applications; innovative compartmentalized designs allow for complex neural circuit modeling; rapid customization capabilities for specific research questions. Weaknesses: Narrow focus on neuroscience applications may limit broader utility; complex biological integration requires specialized knowledge; relatively new technology with evolving standardization.
Key Innovations in Microfluidic Chip Fabrication
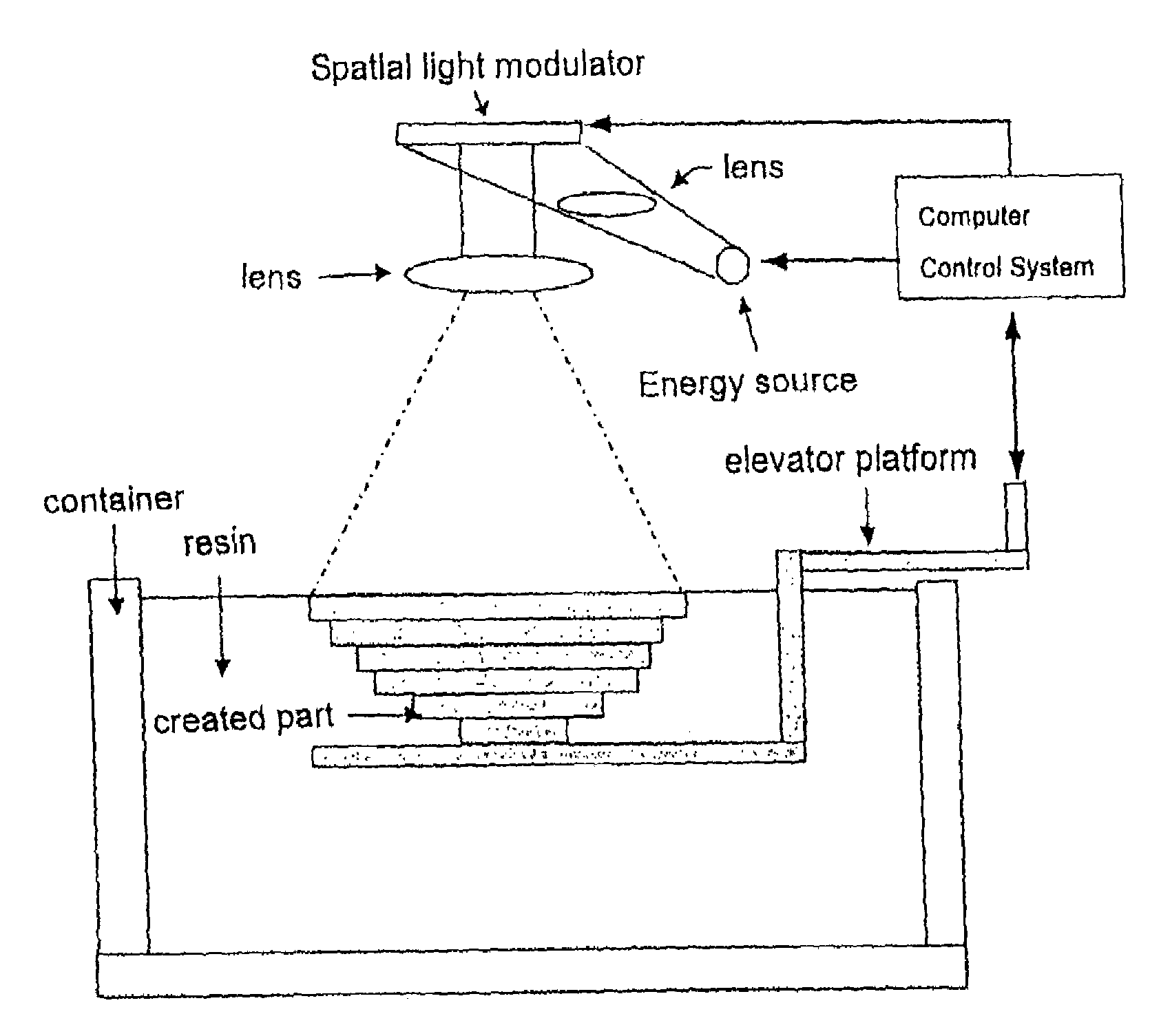
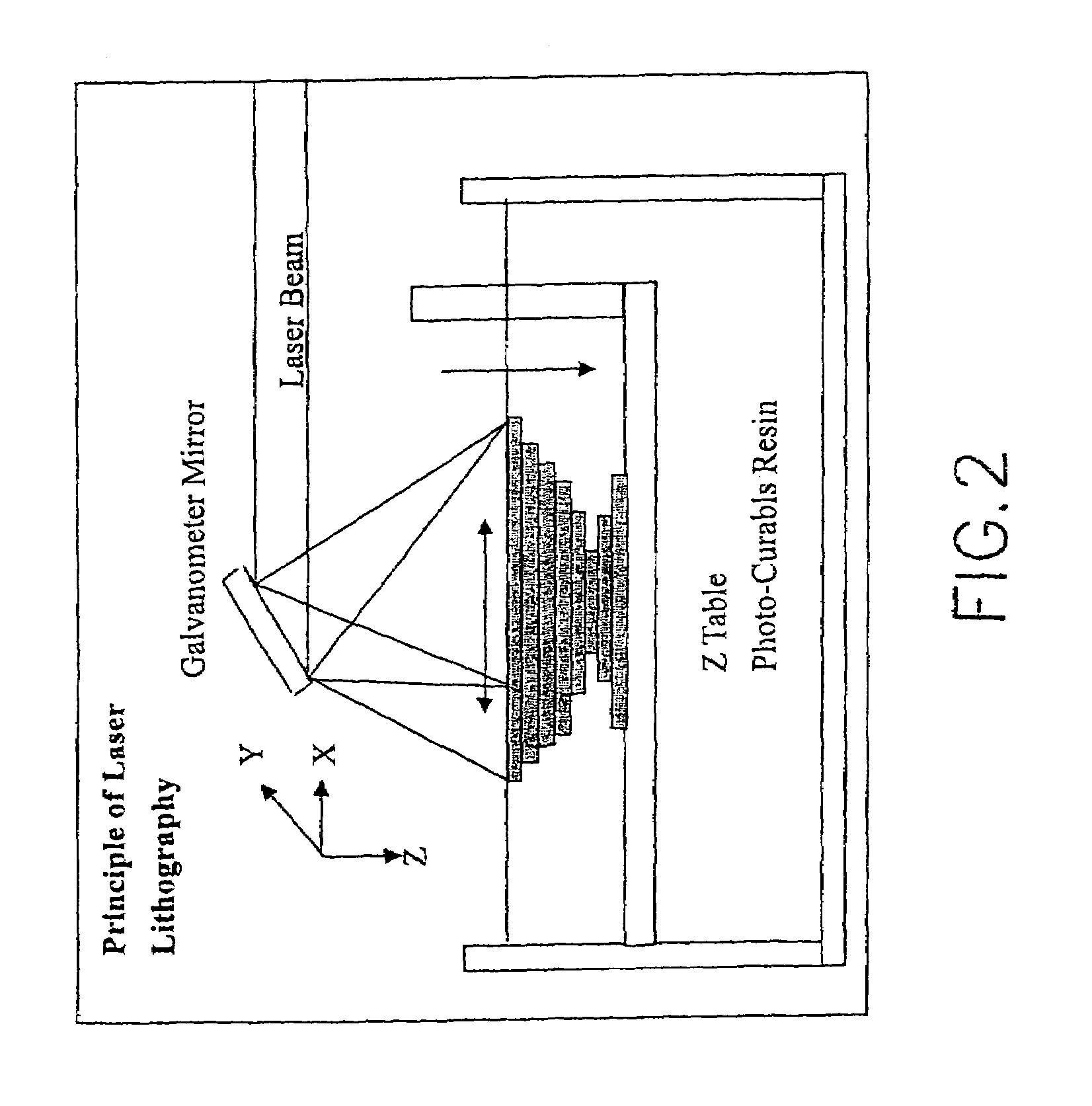
Method for rapid prototyping by using linear light as sources
PatentInactiveUS7158849B2
Innovation
- A method utilizing linear light sources and a Digital Micromirror Device (DMD) to cause multiple physical or chemical changes in raw materials, forming a 3-D image by overlapping 2-D layers, with the support of a portable DMD or Liquid Crystal Display (LCD) for precise scanning and material processing.
Materials Science Advancements for Microfluidic Applications
Recent advancements in materials science have significantly expanded the capabilities of microfluidic systems for rapid prototyping applications. Traditional materials like polydimethylsiloxane (PDMS) continue to dominate the field due to their optical transparency, biocompatibility, and ease of fabrication. However, new composite materials incorporating nanoparticles and specialized polymers have emerged, offering enhanced mechanical properties and chemical resistance while maintaining the flexibility needed for complex microfluidic designs.
Thermoplastic materials such as cyclic olefin copolymer (COC) and polymethyl methacrylate (PMMA) have gained prominence for their superior chemical resistance and mass production potential. These materials enable more robust prototyping cycles and can withstand harsh chemical environments often encountered in biological and chemical analysis applications. The development of UV-curable resins compatible with stereolithography (SLA) 3D printing has further revolutionized the rapid fabrication of microfluidic devices with intricate channel geometries.
Surface modification techniques have evolved to address the inherent hydrophobicity of many polymeric materials. Plasma treatment, chemical vapor deposition, and layer-by-layer assembly methods now allow precise control over surface properties, enabling selective functionalization of microchannels for specific applications. These advancements facilitate improved fluid handling, reduced non-specific binding, and enhanced biocompatibility for cell-based assays.
Paper-based microfluidics represents another significant materials innovation, offering extremely low-cost, disposable platforms for point-of-care diagnostics. The integration of functional materials such as conductive inks, hydrogels, and stimuli-responsive polymers has expanded the analytical capabilities of these simple devices, making them particularly valuable for resource-limited settings.
Hybrid materials combining the advantages of different material classes have emerged as powerful solutions for complex microfluidic applications. Glass-polymer hybrids offer the chemical resistance of glass with the fabrication ease of polymers, while polymer-metal composites provide electrical conductivity for integrated sensing capabilities. These hybrid approaches enable multifunctional microfluidic platforms that can perform multiple analytical steps simultaneously.
Biodegradable and environmentally friendly materials are gaining attention as sustainability concerns grow. Materials derived from natural sources such as cellulose, chitosan, and alginate offer biocompatibility and reduced environmental impact while maintaining the functional requirements for microfluidic applications. These materials are particularly promising for disposable diagnostic devices and environmental monitoring systems.
Thermoplastic materials such as cyclic olefin copolymer (COC) and polymethyl methacrylate (PMMA) have gained prominence for their superior chemical resistance and mass production potential. These materials enable more robust prototyping cycles and can withstand harsh chemical environments often encountered in biological and chemical analysis applications. The development of UV-curable resins compatible with stereolithography (SLA) 3D printing has further revolutionized the rapid fabrication of microfluidic devices with intricate channel geometries.
Surface modification techniques have evolved to address the inherent hydrophobicity of many polymeric materials. Plasma treatment, chemical vapor deposition, and layer-by-layer assembly methods now allow precise control over surface properties, enabling selective functionalization of microchannels for specific applications. These advancements facilitate improved fluid handling, reduced non-specific binding, and enhanced biocompatibility for cell-based assays.
Paper-based microfluidics represents another significant materials innovation, offering extremely low-cost, disposable platforms for point-of-care diagnostics. The integration of functional materials such as conductive inks, hydrogels, and stimuli-responsive polymers has expanded the analytical capabilities of these simple devices, making them particularly valuable for resource-limited settings.
Hybrid materials combining the advantages of different material classes have emerged as powerful solutions for complex microfluidic applications. Glass-polymer hybrids offer the chemical resistance of glass with the fabrication ease of polymers, while polymer-metal composites provide electrical conductivity for integrated sensing capabilities. These hybrid approaches enable multifunctional microfluidic platforms that can perform multiple analytical steps simultaneously.
Biodegradable and environmentally friendly materials are gaining attention as sustainability concerns grow. Materials derived from natural sources such as cellulose, chitosan, and alginate offer biocompatibility and reduced environmental impact while maintaining the functional requirements for microfluidic applications. These materials are particularly promising for disposable diagnostic devices and environmental monitoring systems.
Standardization and Scalability Considerations
The standardization of microfluidic technologies represents a critical challenge for their widespread adoption in rapid prototyping applications. Currently, the field suffers from a lack of universal design standards, protocols, and components, which significantly impedes knowledge transfer and reproducibility across different laboratories and industries. Establishing standardized chip designs, connection interfaces, and control systems would enable more efficient collaboration and accelerate innovation cycles in microfluidic rapid prototyping.
Material standardization presents another crucial consideration. The diversity of materials used in microfluidics—ranging from PDMS, glass, and thermoplastics to paper and hydrogels—creates challenges for establishing uniform fabrication protocols. A systematic classification of materials based on their properties, compatibility with biological samples, and suitability for specific applications would streamline the selection process for rapid prototyping needs.
Scalability pathways must be clearly defined to bridge the gap between prototype development and mass production. Microfluidic technologies often face the "valley of death" when transitioning from laboratory-scale demonstrations to commercial manufacturing. Developing scalable fabrication techniques that maintain precision while increasing throughput is essential. Injection molding, roll-to-roll processing, and advanced 3D printing technologies offer promising approaches for high-volume production while preserving the intricate features required for microfluidic functionality.
Modular design principles can significantly enhance both standardization and scalability. By developing interchangeable microfluidic components with standardized interfaces, researchers and engineers can rapidly assemble complex systems from pre-validated building blocks. This approach reduces development time, increases reliability, and facilitates iterative design improvements—all critical factors for effective rapid prototyping.
Quality control metrics and validation protocols represent another dimension requiring standardization. Establishing industry-wide benchmarks for channel dimensions, flow rates, mixing efficiencies, and detection sensitivities would enable meaningful comparisons between different microfluidic platforms. Automated testing systems that can rapidly characterize microfluidic device performance would accelerate the prototyping cycle and ensure consistent quality across production batches.
The integration of digital design tools and simulation capabilities with standardized microfluidic components would create a powerful ecosystem for rapid prototyping. Cloud-based design libraries, coupled with accurate computational fluid dynamics models, could enable researchers to virtually test designs before physical fabrication, significantly reducing development iterations and material waste.
Material standardization presents another crucial consideration. The diversity of materials used in microfluidics—ranging from PDMS, glass, and thermoplastics to paper and hydrogels—creates challenges for establishing uniform fabrication protocols. A systematic classification of materials based on their properties, compatibility with biological samples, and suitability for specific applications would streamline the selection process for rapid prototyping needs.
Scalability pathways must be clearly defined to bridge the gap between prototype development and mass production. Microfluidic technologies often face the "valley of death" when transitioning from laboratory-scale demonstrations to commercial manufacturing. Developing scalable fabrication techniques that maintain precision while increasing throughput is essential. Injection molding, roll-to-roll processing, and advanced 3D printing technologies offer promising approaches for high-volume production while preserving the intricate features required for microfluidic functionality.
Modular design principles can significantly enhance both standardization and scalability. By developing interchangeable microfluidic components with standardized interfaces, researchers and engineers can rapidly assemble complex systems from pre-validated building blocks. This approach reduces development time, increases reliability, and facilitates iterative design improvements—all critical factors for effective rapid prototyping.
Quality control metrics and validation protocols represent another dimension requiring standardization. Establishing industry-wide benchmarks for channel dimensions, flow rates, mixing efficiencies, and detection sensitivities would enable meaningful comparisons between different microfluidic platforms. Automated testing systems that can rapidly characterize microfluidic device performance would accelerate the prototyping cycle and ensure consistent quality across production batches.
The integration of digital design tools and simulation capabilities with standardized microfluidic components would create a powerful ecosystem for rapid prototyping. Cloud-based design libraries, coupled with accurate computational fluid dynamics models, could enable researchers to virtually test designs before physical fabrication, significantly reducing development iterations and material waste.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!