Integration of Microfluidic ELISA with Microarray Detection Systems
OCT 13, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Microfluidic ELISA-Microarray Integration Background and Objectives
Microfluidic enzyme-linked immunosorbent assay (ELISA) technology has evolved significantly over the past two decades, transitioning from conventional laboratory-based immunoassays to miniaturized platforms that offer enhanced sensitivity, reduced sample volumes, and accelerated reaction kinetics. The integration of microfluidic ELISA with microarray detection systems represents a convergence of two powerful analytical technologies that promises to revolutionize diagnostic capabilities across multiple sectors including healthcare, environmental monitoring, and food safety.
The historical trajectory of this integration began in the early 2000s with rudimentary attempts to combine microfluidic channels with simple antibody arrays. By 2010, researchers had developed more sophisticated systems incorporating multiple detection modalities, and recent advancements have focused on achieving higher throughput, automation, and system integration. This technological evolution has been driven by increasing demands for point-of-care diagnostics, personalized medicine applications, and rapid screening capabilities in resource-limited settings.
Current technological trends indicate a shift toward fully integrated lab-on-a-chip devices that combine sample preparation, microfluidic ELISA reactions, and high-density microarray detection within a single platform. These systems increasingly incorporate advanced materials such as stimuli-responsive polymers, nanostructured surfaces, and novel biorecognition elements to enhance analytical performance and expand application versatility.
The primary objective of integrating microfluidic ELISA with microarray detection is to develop comprehensive analytical platforms that combine the quantitative precision and sensitivity of ELISA with the high-throughput, multiplexed capabilities of microarrays. This integration aims to overcome limitations of traditional ELISA methods, including lengthy processing times, large reagent consumption, and limited multiplexing capacity.
Specific technical goals include: reducing sample and reagent volumes to nanoliter scales; decreasing assay times from hours to minutes; achieving multiplexed detection of dozens to hundreds of analytes simultaneously; maintaining or improving detection sensitivity compared to conventional methods; and developing user-friendly interfaces that minimize the need for specialized training. Additionally, there is significant focus on creating systems compatible with portable or handheld instrumentation to enable point-of-need applications.
The ultimate vision for this technology integration extends beyond mere analytical improvements to enabling paradigm shifts in how diagnostic testing is performed across multiple sectors. In healthcare, these integrated systems could facilitate rapid, comprehensive patient profiling at the bedside; in environmental monitoring, they could enable real-time detection of multiple contaminants in field settings; and in food safety, they could provide comprehensive screening capabilities throughout the supply chain.
The historical trajectory of this integration began in the early 2000s with rudimentary attempts to combine microfluidic channels with simple antibody arrays. By 2010, researchers had developed more sophisticated systems incorporating multiple detection modalities, and recent advancements have focused on achieving higher throughput, automation, and system integration. This technological evolution has been driven by increasing demands for point-of-care diagnostics, personalized medicine applications, and rapid screening capabilities in resource-limited settings.
Current technological trends indicate a shift toward fully integrated lab-on-a-chip devices that combine sample preparation, microfluidic ELISA reactions, and high-density microarray detection within a single platform. These systems increasingly incorporate advanced materials such as stimuli-responsive polymers, nanostructured surfaces, and novel biorecognition elements to enhance analytical performance and expand application versatility.
The primary objective of integrating microfluidic ELISA with microarray detection is to develop comprehensive analytical platforms that combine the quantitative precision and sensitivity of ELISA with the high-throughput, multiplexed capabilities of microarrays. This integration aims to overcome limitations of traditional ELISA methods, including lengthy processing times, large reagent consumption, and limited multiplexing capacity.
Specific technical goals include: reducing sample and reagent volumes to nanoliter scales; decreasing assay times from hours to minutes; achieving multiplexed detection of dozens to hundreds of analytes simultaneously; maintaining or improving detection sensitivity compared to conventional methods; and developing user-friendly interfaces that minimize the need for specialized training. Additionally, there is significant focus on creating systems compatible with portable or handheld instrumentation to enable point-of-need applications.
The ultimate vision for this technology integration extends beyond mere analytical improvements to enabling paradigm shifts in how diagnostic testing is performed across multiple sectors. In healthcare, these integrated systems could facilitate rapid, comprehensive patient profiling at the bedside; in environmental monitoring, they could enable real-time detection of multiple contaminants in field settings; and in food safety, they could provide comprehensive screening capabilities throughout the supply chain.
Market Analysis for Integrated Diagnostic Platforms
The global market for integrated diagnostic platforms combining microfluidic ELISA with microarray detection systems is experiencing robust growth, driven by increasing demand for rapid, accurate, and cost-effective diagnostic solutions. This market segment is projected to reach $7.8 billion by 2027, growing at a CAGR of 8.3% from 2022 to 2027, significantly outpacing the broader in-vitro diagnostics market.
Healthcare facilities represent the largest end-user segment, accounting for approximately 45% of the market share. This dominance stems from the critical need for high-throughput diagnostic capabilities in hospitals and reference laboratories. Research institutions constitute the second-largest segment at 30%, followed by pharmaceutical companies at 15%, and other end-users at 10%.
Geographically, North America leads the market with a 38% share, attributed to advanced healthcare infrastructure, substantial R&D investments, and favorable reimbursement policies. Europe follows at 30%, with Asia-Pacific emerging as the fastest-growing region at a CAGR of 10.5%, driven by improving healthcare access, increasing chronic disease prevalence, and rising healthcare expenditure in countries like China and India.
The point-of-care testing segment within this market is witnessing particularly accelerated growth, expected to expand at a CAGR of 11.2% through 2027. This surge reflects the global shift toward decentralized healthcare delivery models and the growing emphasis on preventive healthcare approaches.
Key market drivers include the rising incidence of chronic and infectious diseases, growing demand for personalized medicine, technological advancements in microfluidics and biosensors, and increasing healthcare expenditure worldwide. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of rapid diagnostic capabilities.
However, market challenges persist, including high development costs, complex regulatory approval processes, and technical challenges in system integration. The average development timeline for bringing an integrated diagnostic platform to market ranges from 3 to 5 years, with regulatory approval accounting for approximately 30% of this timeframe.
Customer adoption trends indicate growing preference for multiplex testing capabilities, reduced sample volume requirements, and seamless data integration with healthcare information systems. Healthcare providers increasingly value diagnostic platforms that offer comprehensive testing menus while maintaining ease of use and minimal training requirements.
Healthcare facilities represent the largest end-user segment, accounting for approximately 45% of the market share. This dominance stems from the critical need for high-throughput diagnostic capabilities in hospitals and reference laboratories. Research institutions constitute the second-largest segment at 30%, followed by pharmaceutical companies at 15%, and other end-users at 10%.
Geographically, North America leads the market with a 38% share, attributed to advanced healthcare infrastructure, substantial R&D investments, and favorable reimbursement policies. Europe follows at 30%, with Asia-Pacific emerging as the fastest-growing region at a CAGR of 10.5%, driven by improving healthcare access, increasing chronic disease prevalence, and rising healthcare expenditure in countries like China and India.
The point-of-care testing segment within this market is witnessing particularly accelerated growth, expected to expand at a CAGR of 11.2% through 2027. This surge reflects the global shift toward decentralized healthcare delivery models and the growing emphasis on preventive healthcare approaches.
Key market drivers include the rising incidence of chronic and infectious diseases, growing demand for personalized medicine, technological advancements in microfluidics and biosensors, and increasing healthcare expenditure worldwide. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of rapid diagnostic capabilities.
However, market challenges persist, including high development costs, complex regulatory approval processes, and technical challenges in system integration. The average development timeline for bringing an integrated diagnostic platform to market ranges from 3 to 5 years, with regulatory approval accounting for approximately 30% of this timeframe.
Customer adoption trends indicate growing preference for multiplex testing capabilities, reduced sample volume requirements, and seamless data integration with healthcare information systems. Healthcare providers increasingly value diagnostic platforms that offer comprehensive testing menus while maintaining ease of use and minimal training requirements.
Current Technological Landscape and Barriers
The global microfluidic ELISA and microarray detection systems market has witnessed significant growth in recent years, driven by increasing demand for point-of-care diagnostics and personalized medicine. Currently, the technological landscape is characterized by a diverse range of approaches, with varying degrees of integration between microfluidic ELISA platforms and microarray detection systems. Leading commercial solutions include lab-on-a-chip devices that incorporate both immunoassay capabilities and multiplexed detection mechanisms, though complete integration remains challenging.
Major technological advancements have emerged from research institutions and biotech companies, particularly in North America, Europe, and East Asia. These innovations focus on miniaturization, automation, and enhanced sensitivity. Recent developments include polymer-based microfluidic chips with integrated optical detection systems, digital microfluidic platforms utilizing electrowetting principles, and paper-based microfluidic devices offering cost-effective alternatives for resource-limited settings.
Despite these advances, significant barriers impede the widespread adoption and commercialization of integrated microfluidic ELISA-microarray systems. Technical challenges include achieving consistent fluid handling across multiple detection zones, maintaining uniform binding kinetics throughout the microarray, and ensuring reproducible signal amplification. The miniaturization of optical detection components while maintaining sensitivity presents another substantial hurdle, particularly for fluorescence-based detection methods requiring sophisticated optics.
Manufacturing scalability represents a critical bottleneck, with many current fabrication techniques being labor-intensive and difficult to translate to mass production. The integration of different materials—polymers, glass, metals, and biological components—creates compatibility issues affecting both manufacturing and performance. Surface chemistry optimization remains particularly challenging, as consistent antibody immobilization across microarray spots is essential for quantitative analysis.
Regulatory barriers further complicate commercialization efforts. The novel nature of these integrated systems often places them in uncertain regulatory categories, requiring extensive validation studies. Clinical adoption faces resistance due to concerns about analytical performance compared to established laboratory methods, particularly regarding sensitivity, specificity, and reproducibility across different sample matrices.
Cost considerations present additional obstacles, as the integration of sophisticated detection systems with disposable microfluidic components creates tension between performance requirements and economic viability. Current manufacturing approaches often result in devices that are either too expensive for widespread adoption or compromise on analytical performance to achieve cost targets.
Major technological advancements have emerged from research institutions and biotech companies, particularly in North America, Europe, and East Asia. These innovations focus on miniaturization, automation, and enhanced sensitivity. Recent developments include polymer-based microfluidic chips with integrated optical detection systems, digital microfluidic platforms utilizing electrowetting principles, and paper-based microfluidic devices offering cost-effective alternatives for resource-limited settings.
Despite these advances, significant barriers impede the widespread adoption and commercialization of integrated microfluidic ELISA-microarray systems. Technical challenges include achieving consistent fluid handling across multiple detection zones, maintaining uniform binding kinetics throughout the microarray, and ensuring reproducible signal amplification. The miniaturization of optical detection components while maintaining sensitivity presents another substantial hurdle, particularly for fluorescence-based detection methods requiring sophisticated optics.
Manufacturing scalability represents a critical bottleneck, with many current fabrication techniques being labor-intensive and difficult to translate to mass production. The integration of different materials—polymers, glass, metals, and biological components—creates compatibility issues affecting both manufacturing and performance. Surface chemistry optimization remains particularly challenging, as consistent antibody immobilization across microarray spots is essential for quantitative analysis.
Regulatory barriers further complicate commercialization efforts. The novel nature of these integrated systems often places them in uncertain regulatory categories, requiring extensive validation studies. Clinical adoption faces resistance due to concerns about analytical performance compared to established laboratory methods, particularly regarding sensitivity, specificity, and reproducibility across different sample matrices.
Cost considerations present additional obstacles, as the integration of sophisticated detection systems with disposable microfluidic components creates tension between performance requirements and economic viability. Current manufacturing approaches often result in devices that are either too expensive for widespread adoption or compromise on analytical performance to achieve cost targets.
Existing Integration Approaches and Architectures
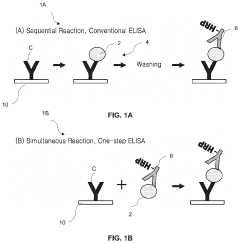
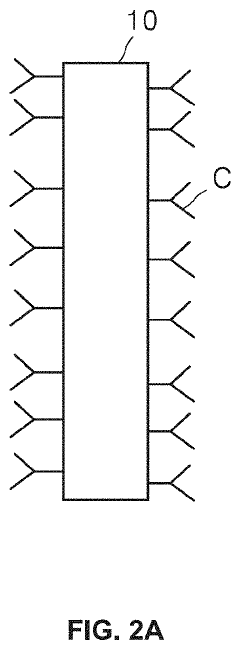
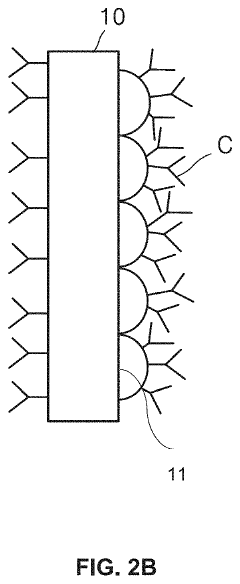
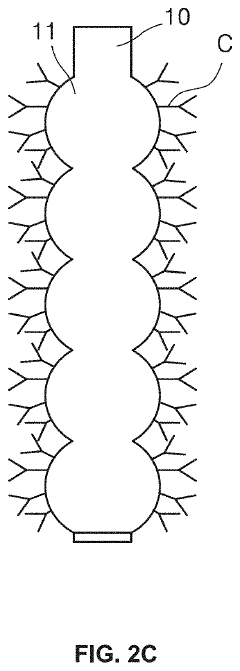
01 Microfluidic ELISA platforms with integrated detection systems
Microfluidic platforms that integrate ELISA methodology with detection systems allow for miniaturized immunoassays with enhanced sensitivity and reduced sample volumes. These systems incorporate microchannels for fluid handling, reaction chambers, and integrated sensors that enable automated sample processing and analysis. The integration of detection components directly within the microfluidic chip improves assay performance while reducing analysis time and reagent consumption compared to conventional ELISA methods.- Microfluidic ELISA platforms with integrated detection systems: Microfluidic platforms that integrate ELISA with detection systems offer advantages such as reduced sample volume, faster analysis times, and improved sensitivity. These systems incorporate microchannels and chambers for sample processing, reagent mixing, and detection zones where antibody-antigen interactions occur. The integration of detection components directly within the microfluidic chip enables real-time monitoring of immunoassay results, making these platforms suitable for point-of-care diagnostics.
- Microarray detection technologies for multiplexed analysis: Microarray detection systems integrated with microfluidic ELISA enable multiplexed analysis of multiple biomarkers simultaneously. These systems utilize arrays of capture molecules (antibodies, aptamers, or other binding agents) patterned on surfaces within microfluidic channels. Detection methods include fluorescence, chemiluminescence, or electrochemical sensing to quantify binding events across the array. This approach significantly increases throughput while maintaining the sensitivity and specificity of traditional ELISA methods.
- Novel fabrication methods for integrated microfluidic ELISA systems: Advanced fabrication techniques enable the creation of sophisticated microfluidic ELISA platforms with integrated detection capabilities. These methods include soft lithography, 3D printing, laser ablation, and injection molding to create precise microstructures. Surface modification techniques are employed to control protein adsorption and enhance antibody immobilization. These fabrication approaches allow for mass production of disposable devices while maintaining consistent performance across manufactured units.
- Automated sample processing and detection in microfluidic ELISA systems: Automation of sample processing and detection in microfluidic ELISA systems improves reproducibility and ease of use. These systems incorporate components for automated sample introduction, reagent delivery, washing steps, and signal detection. Integrated pumps, valves, and flow control mechanisms enable precise fluid handling within the microchannels. Software-controlled operation allows for standardized protocols and minimal user intervention, making these platforms accessible to non-specialized personnel in clinical or field settings.
- Enhanced sensitivity and specificity in microfluidic ELISA microarray systems: Various strategies are employed to enhance the sensitivity and specificity of microfluidic ELISA integrated with microarray detection. These include signal amplification methods such as enzymatic amplification cascades, nanoparticle-based enhancement, and digital detection approaches. Advanced surface chemistry and blocking strategies reduce non-specific binding and background signals. Additionally, optimized microfluidic designs improve reaction kinetics and washing efficiency, leading to lower detection limits and broader dynamic ranges compared to conventional plate-based ELISA methods.
02 Microarray detection technologies for multiplexed analysis
Microarray detection systems enable simultaneous analysis of multiple analytes within a single microfluidic ELISA platform. These technologies utilize patterned arrays of capture molecules (antibodies, antigens, or other binding proteins) arranged on a substrate surface. When combined with microfluidic ELISA, these systems allow for high-throughput screening with spatial resolution, enabling the detection of multiple biomarkers from a single sample. Advanced imaging and signal processing techniques enhance the sensitivity and specificity of these multiplexed assays.Expand Specific Solutions03 Novel detection methods for microfluidic immunoassays
Various novel detection methods have been developed to enhance the performance of microfluidic ELISA integrated with microarray systems. These include fluorescence-based detection, chemiluminescence, electrochemical detection, and surface plasmon resonance. These detection methods improve sensitivity, lower detection limits, and expand the dynamic range of microfluidic immunoassays. Some systems incorporate label-free detection approaches that eliminate the need for secondary antibodies or enzyme labels, simplifying the assay workflow while maintaining high sensitivity.Expand Specific Solutions04 Automated sample processing and analysis systems
Automated systems for microfluidic ELISA with integrated microarray detection incorporate features for sample preparation, reagent handling, incubation, washing, and detection in a single platform. These systems utilize programmable fluid handling components, temperature control elements, and integrated detection modules to perform complete immunoassay workflows with minimal user intervention. Automation reduces human error, improves reproducibility, and enables high-throughput analysis for clinical or research applications.Expand Specific Solutions05 Point-of-care and portable microfluidic ELISA devices
Portable and point-of-care microfluidic ELISA devices with integrated microarray detection systems enable rapid diagnostics in resource-limited settings. These compact systems incorporate miniaturized components, simplified user interfaces, and often smartphone-based or portable readers for signal detection and analysis. The integration of sample preparation, assay execution, and result interpretation in a single portable device facilitates on-site testing with sensitivity comparable to laboratory-based methods while requiring minimal infrastructure and technical expertise.Expand Specific Solutions
Leading Organizations in Microfluidic Diagnostic Systems
The integration of Microfluidic ELISA with Microarray Detection Systems is currently in a growth phase, with the market expanding as healthcare facilities increasingly adopt these advanced diagnostic technologies. The global market size is projected to reach significant value due to rising demand for point-of-care testing and personalized medicine applications. Technologically, this field shows varying maturity levels across companies. Industry leaders like Samsung Electronics, Beckman Coulter, and Siemens AG have developed commercial platforms with advanced capabilities, while academic institutions including The Regents of the University of California, Tsinghua University, and Johns Hopkins University are driving innovation through fundamental research. Research-focused companies like Abbott Point of Care and FUJIFILM are bridging the gap between academic discoveries and commercial applications, accelerating the technology's transition toward widespread clinical adoption.
Beckman Coulter, Inc.
Technical Solution: Beckman Coulter has developed an integrated microfluidic ELISA platform that combines their proprietary microfluidic handling technology with high-sensitivity microarray detection. Their system utilizes a centrifugal force-driven microfluidic disc architecture that enables simultaneous processing of multiple samples. The platform incorporates automated sample preparation, reagent delivery, and waste removal within a single disposable cartridge. Their technology employs a patented microarray spotting technique that achieves high spot density and uniformity, enhancing detection sensitivity. The system integrates fluorescence-based detection with their advanced image processing algorithms to achieve detection limits in the picogram/mL range. Beckman Coulter's platform also features multiplexing capabilities, allowing for the simultaneous detection of up to 100 different biomarkers from a single sample volume as small as 50 μL.
Strengths: Industry-leading automation capabilities reduce operator intervention and error; high multiplexing capacity enables comprehensive biomarker profiling; established global distribution network ensures widespread availability. Weaknesses: Higher initial equipment cost compared to traditional ELISA systems; proprietary reagent requirements may increase operational costs; system complexity requires specialized maintenance.
Abbott Point of Care, Inc.
Technical Solution: Abbott Point of Care has developed the i-STAT Microfluidic ELISA Array system, which integrates their established point-of-care testing expertise with microarray detection capabilities. Their technology utilizes a silicon microfluidic chip with embedded immunoassay reagents and electrochemical detection elements. The system employs a unique approach where sample preparation and analysis occur within a self-contained cartridge that requires minimal user intervention. Abbott's platform incorporates their proprietary electrochemical detection technology that measures electrical signals generated by enzymatic reactions on the microarray surface, eliminating the need for optical detection systems. The technology achieves rapid results (typically under 15 minutes) while maintaining sensitivity comparable to laboratory-based ELISA methods. Their system features built-in quality control mechanisms and wireless connectivity for immediate result transmission to electronic medical record systems.
Strengths: Rapid time-to-result enables immediate clinical decision-making; portable design allows for true point-of-care implementation; established regulatory approval pathway based on previous i-STAT successes. Weaknesses: Limited multiplexing capability compared to optical microarray systems; electrochemical detection may be susceptible to certain types of sample interference; higher per-test cost than centralized laboratory testing.
Critical Patents and Innovations in Microfluidic ELISA-Microarray Systems
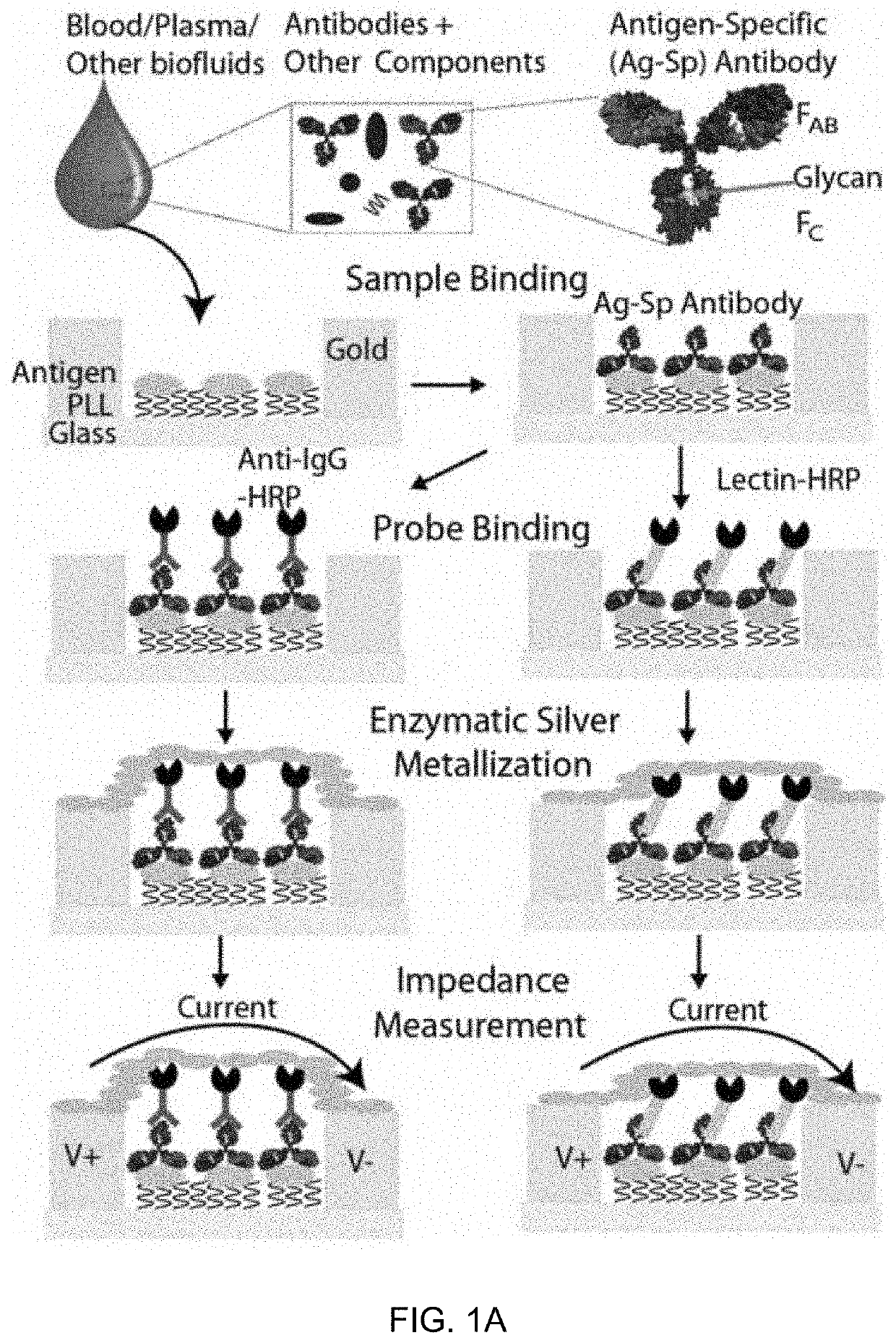
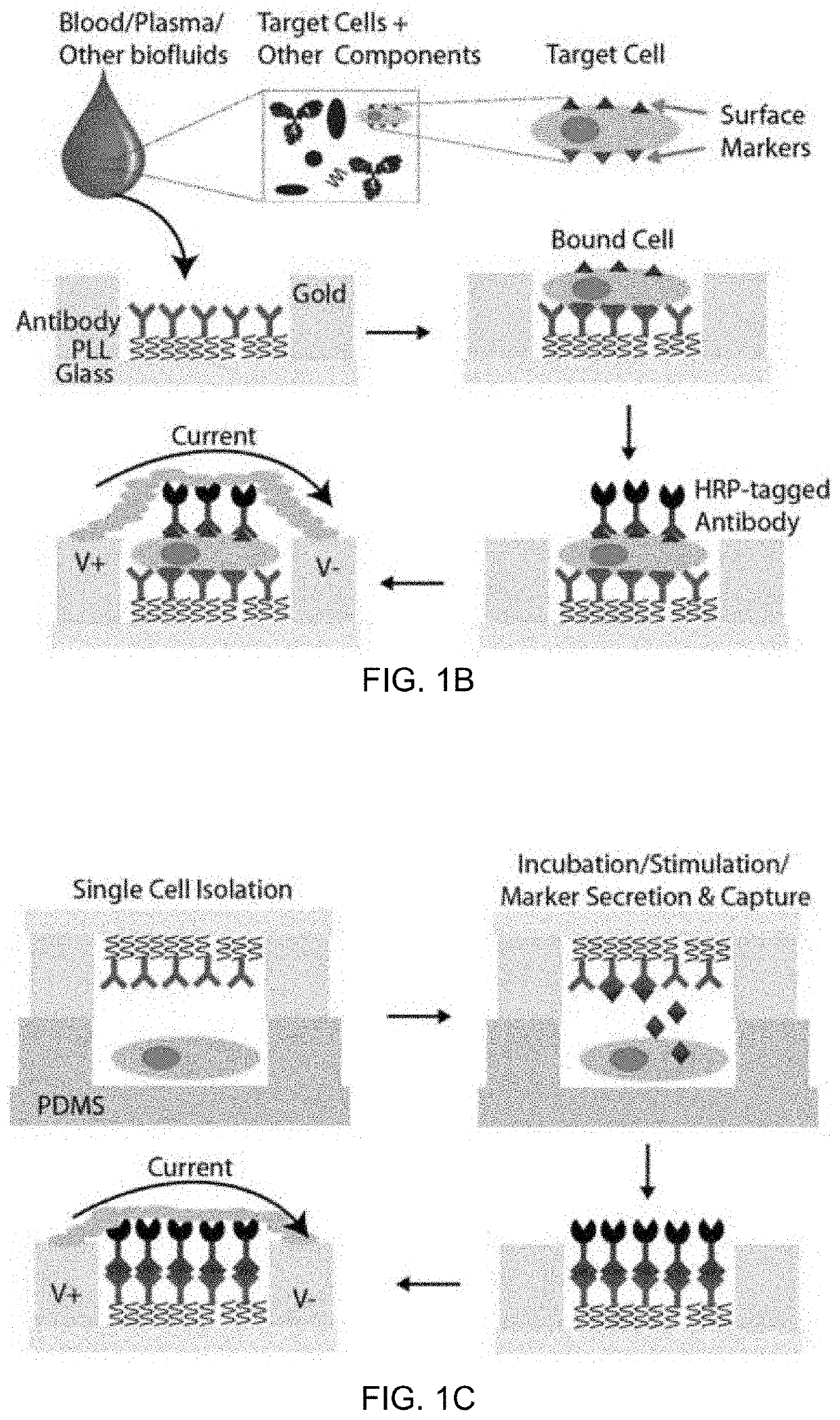
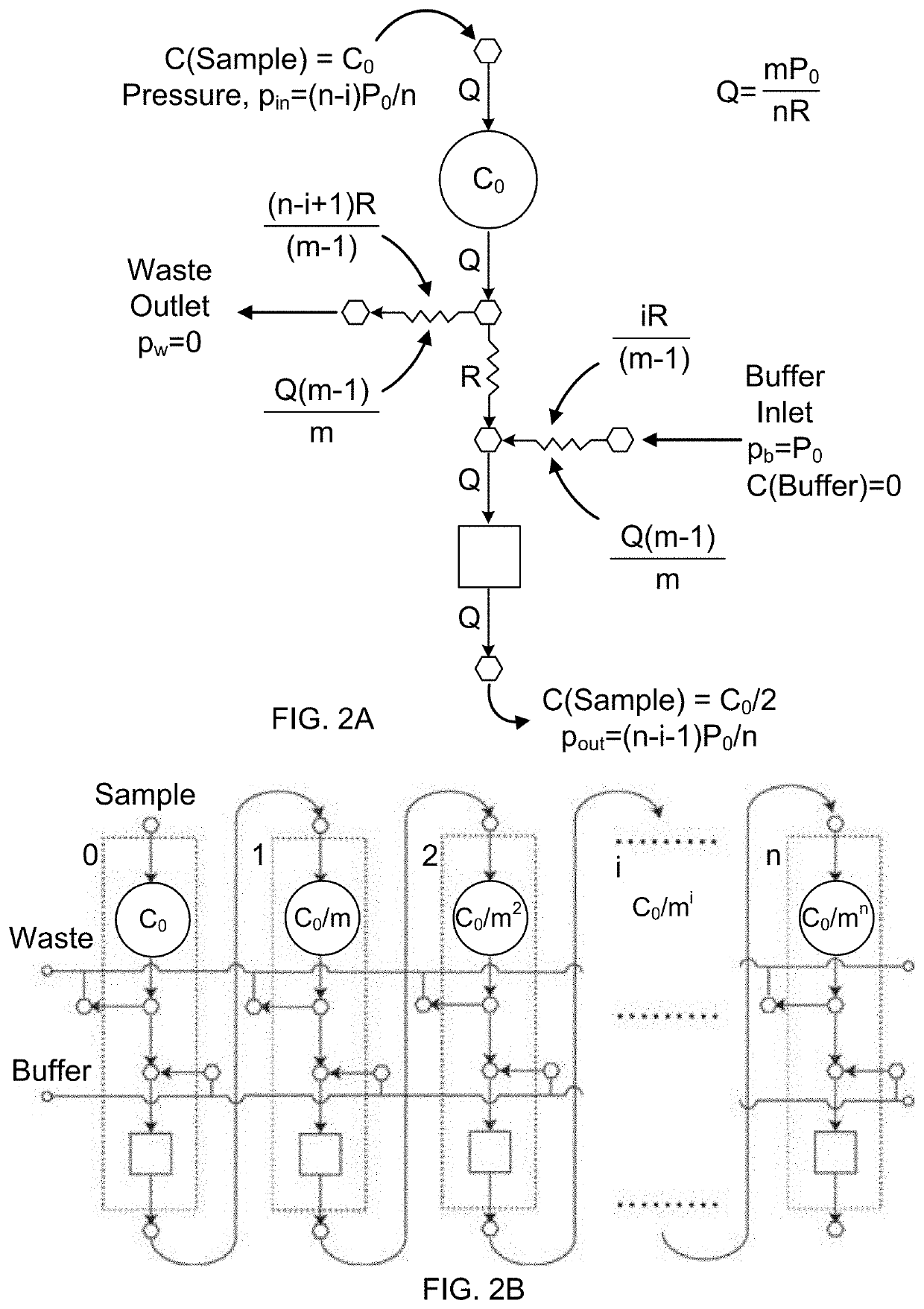
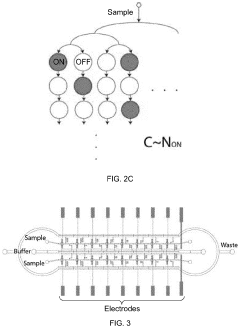
An integrated microfluidic electrode array system for enzyme-linked immuno-sorbent assay (easy-elisa) for point-of-care detection of molecular and cellular biomarkers
PatentInactiveUS20210373014A1
Innovation
- A microfluidic Electrode Array System for Enzyme-Linked Immuno-Sorbent Assay (EASy-ELISA) that uses direct electrical impedance-based detection and quantitation, enabling sensitive and quantitative detection of molecular and cellular biomarkers without intermediate optics or optical detectors, integrated with a microfluidic serial dilution network for sample handling and distribution.
Surface and diffusion enhanced biosensor
PatentInactiveUS20190391142A1
Innovation
- The development of biosensor assemblies with enhanced surface areas, featuring microstructures or nanostructures on detection structures, which increase the concentration of immobilized biomolecules, thereby improving sensitivity and reducing analysis time by optimizing the surface-to-volume ratio and minimizing the hook effect.
Regulatory Framework for Integrated Diagnostic Platforms
The regulatory landscape for integrated diagnostic platforms combining microfluidic ELISA with microarray detection systems presents significant complexity due to their hybrid nature. These platforms fall under multiple regulatory frameworks globally, with the FDA in the United States classifying them primarily as in vitro diagnostic devices (IVDs) under the Medical Device Regulations. The European Union regulates them through the In Vitro Diagnostic Regulation (IVDR 2017/746), which replaced the previous directive in May 2022, introducing more stringent requirements for clinical evidence, post-market surveillance, and risk classification.
The path to regulatory approval for these integrated systems typically requires demonstration of analytical validity, clinical validity, and clinical utility. Manufacturers must navigate premarket approval (PMA) or 510(k) clearance pathways in the US, depending on the risk classification and novelty of the technology. The integrated nature of these platforms often complicates the regulatory process, as authorities must evaluate both the microfluidic components and the detection systems as a unified diagnostic tool.
Quality management systems compliant with ISO 13485 standards are mandatory for manufacturers of these integrated diagnostic platforms. Additionally, compliance with IEC 61010 for laboratory equipment safety and IEC 62304 for medical device software is essential when these platforms incorporate automated analysis software, which is increasingly common in microarray detection systems.
Regulatory bodies are increasingly adopting a risk-based approach to these technologies, with classification determined by intended use, technological characteristics, and potential impact on patient outcomes. High-risk applications, such as cancer diagnostics or infectious disease detection, face more rigorous scrutiny and validation requirements than lower-risk applications.
Global harmonization efforts, including the Medical Device Single Audit Program (MDSAP) and International Medical Device Regulators Forum (IMDRF) initiatives, are gradually reducing regulatory divergence across major markets. However, significant regional differences persist, creating challenges for global deployment of these integrated diagnostic platforms.
Emerging regulatory considerations specific to these integrated systems include data privacy concerns when patient diagnostic information is digitized and potentially shared across healthcare networks, cybersecurity requirements for networked devices, and validation protocols for artificial intelligence components increasingly incorporated into analysis software for microarray detection.
The regulatory framework continues to evolve as these technologies advance, with regulatory agencies developing new guidance documents specifically addressing the unique challenges of integrated diagnostic platforms. Manufacturers are advised to engage early and frequently with regulatory authorities through pre-submission consultations to navigate this complex landscape effectively.
The path to regulatory approval for these integrated systems typically requires demonstration of analytical validity, clinical validity, and clinical utility. Manufacturers must navigate premarket approval (PMA) or 510(k) clearance pathways in the US, depending on the risk classification and novelty of the technology. The integrated nature of these platforms often complicates the regulatory process, as authorities must evaluate both the microfluidic components and the detection systems as a unified diagnostic tool.
Quality management systems compliant with ISO 13485 standards are mandatory for manufacturers of these integrated diagnostic platforms. Additionally, compliance with IEC 61010 for laboratory equipment safety and IEC 62304 for medical device software is essential when these platforms incorporate automated analysis software, which is increasingly common in microarray detection systems.
Regulatory bodies are increasingly adopting a risk-based approach to these technologies, with classification determined by intended use, technological characteristics, and potential impact on patient outcomes. High-risk applications, such as cancer diagnostics or infectious disease detection, face more rigorous scrutiny and validation requirements than lower-risk applications.
Global harmonization efforts, including the Medical Device Single Audit Program (MDSAP) and International Medical Device Regulators Forum (IMDRF) initiatives, are gradually reducing regulatory divergence across major markets. However, significant regional differences persist, creating challenges for global deployment of these integrated diagnostic platforms.
Emerging regulatory considerations specific to these integrated systems include data privacy concerns when patient diagnostic information is digitized and potentially shared across healthcare networks, cybersecurity requirements for networked devices, and validation protocols for artificial intelligence components increasingly incorporated into analysis software for microarray detection.
The regulatory framework continues to evolve as these technologies advance, with regulatory agencies developing new guidance documents specifically addressing the unique challenges of integrated diagnostic platforms. Manufacturers are advised to engage early and frequently with regulatory authorities through pre-submission consultations to navigate this complex landscape effectively.
Manufacturing Scalability and Cost Optimization Strategies
The scalability of manufacturing processes for integrated microfluidic ELISA-microarray systems presents significant challenges that must be addressed to enable widespread adoption. Current production methods often rely on labor-intensive manual assembly, resulting in high unit costs and limited throughput. Transitioning to automated manufacturing requires substantial investment in specialized equipment and process development, creating barriers for smaller companies and research institutions. The integration of different materials—polymers, glass, and various surface coatings—further complicates mass production efforts due to varying thermal properties and bonding requirements.
Cost optimization strategies must focus on materials selection, as this represents 30-40% of total manufacturing expenses. Replacing traditional glass substrates with engineered polymers like cyclic olefin copolymer (COC) can reduce material costs by up to 60% while maintaining optical clarity and chemical compatibility. Implementing design for manufacturing (DFM) principles early in development can eliminate unnecessary complexity and reduce production steps, potentially decreasing assembly costs by 25-35%.
Standardization of components and interfaces represents another critical optimization pathway. The development of modular microfluidic platforms with standardized connections would enable economies of scale through higher production volumes of common elements. Several industry consortia are working to establish such standards, though fragmentation remains a challenge. Companies like Dolomite Microfluidics and uFluidix have demonstrated success with standardized microfluidic building blocks that can be configured for various applications, reducing custom engineering requirements.
Advanced manufacturing technologies show promise for scaling production while maintaining precision. Injection molding can produce complex microfluidic structures at high volumes once initial tooling investments are recovered. Roll-to-roll manufacturing techniques adapted from the electronics industry offer continuous production capabilities for certain microfluidic components, potentially reducing unit costs by 40-70% at high volumes. 3D printing technologies, particularly micro-stereolithography, are evolving rapidly and may enable cost-effective small-batch production with reduced setup costs.
Supply chain optimization represents an often-overlooked aspect of cost reduction. Vertical integration of critical manufacturing steps can eliminate markup costs from multiple vendors, while strategic outsourcing of standardized components can leverage specialized expertise and equipment. Several contract manufacturing organizations now offer microfluidic-specific services, providing access to advanced production capabilities without requiring capital investment.
Cost optimization strategies must focus on materials selection, as this represents 30-40% of total manufacturing expenses. Replacing traditional glass substrates with engineered polymers like cyclic olefin copolymer (COC) can reduce material costs by up to 60% while maintaining optical clarity and chemical compatibility. Implementing design for manufacturing (DFM) principles early in development can eliminate unnecessary complexity and reduce production steps, potentially decreasing assembly costs by 25-35%.
Standardization of components and interfaces represents another critical optimization pathway. The development of modular microfluidic platforms with standardized connections would enable economies of scale through higher production volumes of common elements. Several industry consortia are working to establish such standards, though fragmentation remains a challenge. Companies like Dolomite Microfluidics and uFluidix have demonstrated success with standardized microfluidic building blocks that can be configured for various applications, reducing custom engineering requirements.
Advanced manufacturing technologies show promise for scaling production while maintaining precision. Injection molding can produce complex microfluidic structures at high volumes once initial tooling investments are recovered. Roll-to-roll manufacturing techniques adapted from the electronics industry offer continuous production capabilities for certain microfluidic components, potentially reducing unit costs by 40-70% at high volumes. 3D printing technologies, particularly micro-stereolithography, are evolving rapidly and may enable cost-effective small-batch production with reduced setup costs.
Supply chain optimization represents an often-overlooked aspect of cost reduction. Vertical integration of critical manufacturing steps can eliminate markup costs from multiple vendors, while strategic outsourcing of standardized components can leverage specialized expertise and equipment. Several contract manufacturing organizations now offer microfluidic-specific services, providing access to advanced production capabilities without requiring capital investment.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!