Research on Capillary-Driven Flow in Microfluidic ELISA
OCT 13, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Microfluidic ELISA Technology Background and Objectives
Microfluidic Enzyme-Linked Immunosorbent Assay (ELISA) represents a significant advancement in diagnostic technology, evolving from traditional ELISA methods first developed in the 1970s. The integration of microfluidics with ELISA has transformed this biochemical technique into a more efficient, portable, and cost-effective diagnostic tool. This technological convergence began gaining momentum in the early 2000s, with substantial research acceleration occurring over the past decade.
Capillary-driven flow, a fundamental physical phenomenon in microfluidic systems, has emerged as a critical component in the development of next-generation ELISA platforms. This passive transport mechanism eliminates the need for external pumping systems, significantly reducing complexity, power requirements, and overall system footprint. The natural capillary action within microchannels enables precise fluid control at the microscale level, which is essential for accurate and reproducible immunoassay results.
The evolution of microfluidic ELISA technology has been characterized by progressive miniaturization, integration of multiple analytical steps, and enhancement of detection sensitivity. Recent advances in materials science, microfabrication techniques, and surface chemistry have further accelerated development in this field. Particularly noteworthy is the transition from polydimethylsiloxane (PDMS) to paper-based and other alternative substrate materials, which has expanded accessibility and application scenarios.
The primary technical objectives in capillary-driven microfluidic ELISA research include achieving consistent and predictable flow rates, optimizing surface chemistry for both capillary action and biomolecule immobilization, and developing robust manufacturing processes suitable for mass production. Additionally, researchers aim to enhance detection limits while maintaining the simplicity and user-friendliness that make these systems attractive for point-of-care applications.
Current research trends indicate a growing focus on mathematical modeling and simulation of capillary flow dynamics in complex microstructures, which enables more precise design of microfluidic channels. There is also significant interest in developing multi-functional surfaces that can simultaneously facilitate capillary action while providing optimal conditions for antibody-antigen interactions.
Looking forward, the field is moving toward fully integrated sample-to-answer systems that leverage capillary-driven flow for all stages of the ELISA process. The ultimate goal is to develop platforms that maintain the analytical performance of laboratory-based ELISA while offering the advantages of portability, rapid analysis, minimal sample volume requirements, and reduced dependence on trained personnel. This convergence of capabilities positions microfluidic ELISA as a transformative technology for healthcare delivery, particularly in resource-limited settings and for applications requiring rapid on-site testing.
Capillary-driven flow, a fundamental physical phenomenon in microfluidic systems, has emerged as a critical component in the development of next-generation ELISA platforms. This passive transport mechanism eliminates the need for external pumping systems, significantly reducing complexity, power requirements, and overall system footprint. The natural capillary action within microchannels enables precise fluid control at the microscale level, which is essential for accurate and reproducible immunoassay results.
The evolution of microfluidic ELISA technology has been characterized by progressive miniaturization, integration of multiple analytical steps, and enhancement of detection sensitivity. Recent advances in materials science, microfabrication techniques, and surface chemistry have further accelerated development in this field. Particularly noteworthy is the transition from polydimethylsiloxane (PDMS) to paper-based and other alternative substrate materials, which has expanded accessibility and application scenarios.
The primary technical objectives in capillary-driven microfluidic ELISA research include achieving consistent and predictable flow rates, optimizing surface chemistry for both capillary action and biomolecule immobilization, and developing robust manufacturing processes suitable for mass production. Additionally, researchers aim to enhance detection limits while maintaining the simplicity and user-friendliness that make these systems attractive for point-of-care applications.
Current research trends indicate a growing focus on mathematical modeling and simulation of capillary flow dynamics in complex microstructures, which enables more precise design of microfluidic channels. There is also significant interest in developing multi-functional surfaces that can simultaneously facilitate capillary action while providing optimal conditions for antibody-antigen interactions.
Looking forward, the field is moving toward fully integrated sample-to-answer systems that leverage capillary-driven flow for all stages of the ELISA process. The ultimate goal is to develop platforms that maintain the analytical performance of laboratory-based ELISA while offering the advantages of portability, rapid analysis, minimal sample volume requirements, and reduced dependence on trained personnel. This convergence of capabilities positions microfluidic ELISA as a transformative technology for healthcare delivery, particularly in resource-limited settings and for applications requiring rapid on-site testing.
Market Analysis for Capillary-Driven Microfluidic Diagnostics
The global market for capillary-driven microfluidic diagnostics is experiencing robust growth, driven by increasing demand for point-of-care testing solutions and decentralized healthcare services. Current market valuations indicate that the microfluidic-based diagnostics sector reached approximately 15 billion USD in 2022, with capillary-driven systems representing a significant and rapidly expanding segment within this market.
Healthcare providers worldwide are increasingly adopting microfluidic ELISA technologies due to their ability to deliver laboratory-quality results in resource-limited settings. The primary market drivers include rising incidence of infectious diseases, growing geriatric population requiring frequent monitoring, and increasing preference for home-based healthcare solutions that reduce hospital visits and associated costs.
Regionally, North America currently dominates the market share, accounting for approximately 40% of global revenue, followed by Europe at 30% and Asia-Pacific at 20%. However, the highest growth rates are being observed in emerging economies across Asia-Pacific and Latin America, where healthcare infrastructure development and increasing healthcare expenditure are creating new market opportunities.
The COVID-19 pandemic has significantly accelerated market growth, with a compound annual growth rate exceeding 12% projected through 2028. This acceleration stems from heightened awareness of rapid diagnostic capabilities and substantial investments in developing portable diagnostic platforms that can be deployed in various settings.
Consumer demand patterns reveal a strong preference for diagnostic solutions that offer rapid results (under 30 minutes), minimal sample volume requirements, and user-friendly interfaces. Healthcare facilities are particularly interested in systems that can be integrated with existing electronic health record systems and provide data analytics capabilities.
Market segmentation analysis shows that clinical diagnostics applications currently dominate the capillary-driven microfluidic market, followed by research applications and food safety testing. Within clinical diagnostics, infectious disease testing represents the largest application segment, followed by cardiac markers and metabolic disorder testing.
Key customer groups include hospitals and clinical laboratories (45% of market share), research institutions (25%), pharmaceutical companies (15%), and individual consumers through home testing kits (10%). The remaining market share is distributed among food safety organizations and environmental monitoring agencies.
Pricing trends indicate a gradual decrease in per-test costs as manufacturing processes become more efficient and competition intensifies. However, premium pricing remains viable for solutions offering superior sensitivity, specificity, or multiplex testing capabilities. The average price point for capillary-driven microfluidic ELISA tests ranges from 5-25 USD per test, depending on complexity and application.
Healthcare providers worldwide are increasingly adopting microfluidic ELISA technologies due to their ability to deliver laboratory-quality results in resource-limited settings. The primary market drivers include rising incidence of infectious diseases, growing geriatric population requiring frequent monitoring, and increasing preference for home-based healthcare solutions that reduce hospital visits and associated costs.
Regionally, North America currently dominates the market share, accounting for approximately 40% of global revenue, followed by Europe at 30% and Asia-Pacific at 20%. However, the highest growth rates are being observed in emerging economies across Asia-Pacific and Latin America, where healthcare infrastructure development and increasing healthcare expenditure are creating new market opportunities.
The COVID-19 pandemic has significantly accelerated market growth, with a compound annual growth rate exceeding 12% projected through 2028. This acceleration stems from heightened awareness of rapid diagnostic capabilities and substantial investments in developing portable diagnostic platforms that can be deployed in various settings.
Consumer demand patterns reveal a strong preference for diagnostic solutions that offer rapid results (under 30 minutes), minimal sample volume requirements, and user-friendly interfaces. Healthcare facilities are particularly interested in systems that can be integrated with existing electronic health record systems and provide data analytics capabilities.
Market segmentation analysis shows that clinical diagnostics applications currently dominate the capillary-driven microfluidic market, followed by research applications and food safety testing. Within clinical diagnostics, infectious disease testing represents the largest application segment, followed by cardiac markers and metabolic disorder testing.
Key customer groups include hospitals and clinical laboratories (45% of market share), research institutions (25%), pharmaceutical companies (15%), and individual consumers through home testing kits (10%). The remaining market share is distributed among food safety organizations and environmental monitoring agencies.
Pricing trends indicate a gradual decrease in per-test costs as manufacturing processes become more efficient and competition intensifies. However, premium pricing remains viable for solutions offering superior sensitivity, specificity, or multiplex testing capabilities. The average price point for capillary-driven microfluidic ELISA tests ranges from 5-25 USD per test, depending on complexity and application.
Current Challenges in Capillary-Driven Flow Systems
Despite significant advancements in capillary-driven flow systems for microfluidic ELISA applications, several critical challenges continue to impede their widespread adoption and optimal performance. One of the most persistent issues is flow control inconsistency, where variations in surface properties, channel dimensions, and environmental conditions lead to unpredictable flow behaviors. Even minor manufacturing inconsistencies at the microscale can dramatically alter capillary action, resulting in significant test-to-test variability that undermines diagnostic reliability.
Surface modification stability presents another major hurdle, as the hydrophilic or hydrophobic properties crucial for directing capillary flow often degrade over time or during storage. This degradation can significantly alter flow characteristics, reducing shelf life and compromising test reproducibility, particularly problematic for point-of-care applications in resource-limited settings.
Temperature and humidity sensitivity further complicates capillary-driven systems, with environmental fluctuations substantially affecting flow rates and potentially leading to incomplete reactions or false results. This environmental dependency limits the geographical deployability of such diagnostic platforms without additional control mechanisms.
The integration of multiple assay steps remains technically challenging, as sequential fluid handling operations (sample preparation, reagent delivery, washing, and detection) require precise timing and flow control that passive capillary systems struggle to maintain autonomously. Current designs often necessitate user intervention, increasing complexity and error potential.
Scaling limitations also constrain the technology, with difficulties in maintaining consistent capillary action across larger microfluidic networks or when processing higher sample volumes. This restricts throughput capabilities and application scope compared to pressure-driven alternatives.
Biomolecule adsorption presents a significant biochemical challenge, as proteins and other biomolecules can non-specifically adsorb to channel surfaces, altering both flow characteristics and assay sensitivity. This phenomenon can lead to channel clogging, increased background signals, and reduced detection limits.
Manufacturing complexity and cost considerations further limit widespread implementation, as fabrication of precise microchannels with controlled surface properties requires sophisticated techniques that challenge mass production economics. The balance between manufacturing precision and cost-effectiveness remains elusive for many applications.
Bubble formation during fluid flow represents another persistent technical issue, disrupting flow continuity and potentially blocking channels or reaction chambers. These air pockets can prevent complete reagent mixing or washing, significantly impacting assay performance and reliability.
Surface modification stability presents another major hurdle, as the hydrophilic or hydrophobic properties crucial for directing capillary flow often degrade over time or during storage. This degradation can significantly alter flow characteristics, reducing shelf life and compromising test reproducibility, particularly problematic for point-of-care applications in resource-limited settings.
Temperature and humidity sensitivity further complicates capillary-driven systems, with environmental fluctuations substantially affecting flow rates and potentially leading to incomplete reactions or false results. This environmental dependency limits the geographical deployability of such diagnostic platforms without additional control mechanisms.
The integration of multiple assay steps remains technically challenging, as sequential fluid handling operations (sample preparation, reagent delivery, washing, and detection) require precise timing and flow control that passive capillary systems struggle to maintain autonomously. Current designs often necessitate user intervention, increasing complexity and error potential.
Scaling limitations also constrain the technology, with difficulties in maintaining consistent capillary action across larger microfluidic networks or when processing higher sample volumes. This restricts throughput capabilities and application scope compared to pressure-driven alternatives.
Biomolecule adsorption presents a significant biochemical challenge, as proteins and other biomolecules can non-specifically adsorb to channel surfaces, altering both flow characteristics and assay sensitivity. This phenomenon can lead to channel clogging, increased background signals, and reduced detection limits.
Manufacturing complexity and cost considerations further limit widespread implementation, as fabrication of precise microchannels with controlled surface properties requires sophisticated techniques that challenge mass production economics. The balance between manufacturing precision and cost-effectiveness remains elusive for many applications.
Bubble formation during fluid flow represents another persistent technical issue, disrupting flow continuity and potentially blocking channels or reaction chambers. These air pockets can prevent complete reagent mixing or washing, significantly impacting assay performance and reliability.
Current Capillary-Driven Flow Implementation Strategies
01 Microfluidic devices for capillary-driven flow
Microfluidic devices utilize capillary forces to drive fluid flow through small channels without external pumps. These devices incorporate specific channel geometries, surface treatments, and materials to control flow dynamics. The capillary action is influenced by surface tension, contact angle, and channel dimensions, allowing for precise fluid manipulation in diagnostic, analytical, and lab-on-a-chip applications.- Microfluidic devices utilizing capillary-driven flow: Microfluidic devices leverage capillary-driven flow for fluid transport without external pumps. These systems utilize surface tension and capillary forces to move liquids through microchannels, enabling applications in diagnostics, lab-on-a-chip technologies, and analytical chemistry. The geometry, surface properties, and channel dimensions are optimized to control flow rates and fluid behavior in these miniaturized systems.
- Capillary flow in porous media and membranes: Capillary-driven flow in porous materials and membranes is governed by the interaction between fluid properties and the material's structure. The pore size distribution, tortuosity, and surface chemistry influence the wicking behavior and flow dynamics. These principles are applied in separation technologies, filtration systems, and absorbent materials where controlled fluid transport through porous structures is essential.
- Mathematical modeling and simulation of capillary flow dynamics: Mathematical models and computational simulations are developed to predict and analyze capillary-driven flow behavior. These models incorporate factors such as surface tension, contact angle, viscosity, and geometric parameters to describe fluid movement in various systems. Advanced simulation techniques help optimize designs for applications requiring precise control of capillary flow and enable better understanding of complex flow phenomena at micro and nano scales.
- Capillary flow control mechanisms and structures: Specialized structures and mechanisms are designed to control capillary-driven flow in various applications. These include capillary valves, flow restrictors, capillary pumps, and geometric features that can start, stop, or regulate flow rates. By manipulating channel geometries, surface properties, or incorporating smart materials, these control mechanisms enable precise fluid handling in passive microfluidic systems without external power sources.
- Applications of capillary-driven flow in electronic and energy systems: Capillary-driven flow principles are applied in electronic cooling systems, heat pipes, and energy conversion devices. These applications utilize the passive fluid transport capabilities of capillary structures to manage thermal energy, enhance heat transfer, or facilitate electrochemical processes. The integration of capillary flow dynamics in these systems improves efficiency, reduces power consumption, and enables novel device architectures for electronics and energy technologies.
02 Capillary flow in porous media and membranes
Capillary-driven flow in porous materials and membranes is governed by the interaction between fluid properties and the material's structure. The flow dynamics depend on pore size distribution, tortuosity, and surface chemistry. These principles are applied in filtration systems, chromatography, and lateral flow assays where controlled fluid transport through porous matrices is essential for proper functionality.Expand Specific Solutions03 Mathematical modeling and simulation of capillary flow
Advanced mathematical models and computational simulations are used to predict and analyze capillary-driven flow dynamics. These models incorporate factors such as surface tension, viscosity, contact angle hysteresis, and geometric constraints to accurately represent fluid behavior. Simulation techniques help optimize designs for microfluidic systems and understand complex flow phenomena at microscale and nanoscale levels.Expand Specific Solutions04 Capillary flow control mechanisms
Various mechanisms are employed to control capillary-driven flow, including geometric features, surface modifications, and active elements. These control mechanisms can regulate flow rate, direction, and timing through techniques such as capillary valves, flow restrictors, and capillary pumps. By manipulating channel dimensions, surface energy gradients, and introducing specific structures, precise fluid handling can be achieved in microfluidic systems.Expand Specific Solutions05 Applications of capillary-driven flow in sensing and diagnostics
Capillary-driven flow is extensively utilized in sensing and diagnostic platforms, particularly in point-of-care testing devices. These applications leverage spontaneous fluid transport to enable sample processing, reagent mixing, and detection without external power sources. The controlled flow dynamics facilitate precise timing of reactions, separation of analytes, and sequential delivery of reagents, making them ideal for resource-limited settings.Expand Specific Solutions
Key Industry Players in Microfluidic Diagnostics
Capillary-driven microfluidic ELISA technology is currently in the growth phase, with an estimated market size of $3-5 billion and expanding at 8-10% annually. The competitive landscape features a diverse mix of academic institutions (Colorado State University, Zhejiang University, University of Washington) conducting fundamental research alongside established corporations (LG Chem, IBM, Agilent Technologies) developing commercial applications. Technical maturity varies significantly across players, with companies like miDIAGNOSTICS and Biosurfit leading with near-market solutions, while research institutions like Georgia Tech Research Corp and Katholieke Universiteit Leuven focus on advancing fundamental capillary flow mechanics. The field is characterized by increasing cross-sector collaboration between academic and industrial partners to overcome remaining technical challenges in sample preparation, flow control, and detection sensitivity.
Zhejiang University
Technical Solution: Zhejiang University has developed innovative paper-based capillary-driven microfluidic ELISA platforms that combine the advantages of traditional paper diagnostics with advanced microfluidic principles. Their technology utilizes specially treated cellulose substrates with patterned hydrophobic barriers created through wax printing or photolithography techniques, forming defined capillary flow paths with channel widths ranging from 200μm to 2mm[1]. The university's research team has engineered multi-layer paper structures that enable vertical fluid transport through capillary action, creating three-dimensional assay architectures that increase functional density. Their platform incorporates strategically positioned dry reagent zones that dissolve sequentially as sample fluid advances through capillary action, enabling multi-step ELISA protocols without external equipment. Zhejiang University has demonstrated detection of various biomarkers including cancer antigens, infectious disease markers, and environmental contaminants with detection limits in the ng/mL range using colorimetric detection visible to the naked eye or with smartphone cameras[3]. Their technology achieves complete ELISA procedures in 10-30 minutes using sample volumes as low as 5-10μL, with estimated manufacturing costs below $1 per device.
Strengths: Extremely low production costs through paper-based manufacturing make their technology highly accessible for resource-limited settings. The simplicity of operation requires minimal user training and no external equipment. Weaknesses: Paper-based platforms typically offer lower sensitivity compared to polymer or glass-based microfluidic systems, and may have more limited shelf life due to potential degradation of paper substrates in challenging environmental conditions.
miDIAGNOSTICS NV
Technical Solution: miDIAGNOSTICS has pioneered a proprietary capillary-driven microfluidic ELISA technology called "nanoFluidic Processor" that leverages silicon chip manufacturing techniques for high-precision microfluidic channels. Their platform utilizes precisely engineered microstructures with dimensions ranging from 200nm to 20μm that generate strong and consistent capillary forces for fluid transport[2]. The company's technology incorporates specialized surface treatments that maintain stable hydrophilic properties over extended storage periods (>12 months), addressing a common challenge in capillary-driven systems. Their innovation includes integrated capillary pumps with sequential activation based on differential capillary pressure, enabling automated multi-step ELISA protocols without external equipment. The platform features embedded dry reagents that reconstitute upon sample introduction, eliminating reagent handling steps. miDIAGNOSTICS has demonstrated clinical applications for their technology in infectious disease testing, achieving detection sensitivities in the pg/mL range with coefficient of variation <10% across tests[4]. Their devices incorporate smartphone-compatible optical detection systems for point-of-care applications.
Strengths: Exceptional manufacturing precision through silicon fabrication techniques enables highly reproducible capillary flow characteristics. Their integrated dry reagent technology simplifies user workflow and extends shelf life. Weaknesses: The silicon-based manufacturing approach, while precise, may result in higher production costs compared to polymer-based alternatives, potentially limiting accessibility in resource-limited settings.
Critical Patents and Research in Microfluidic ELISA
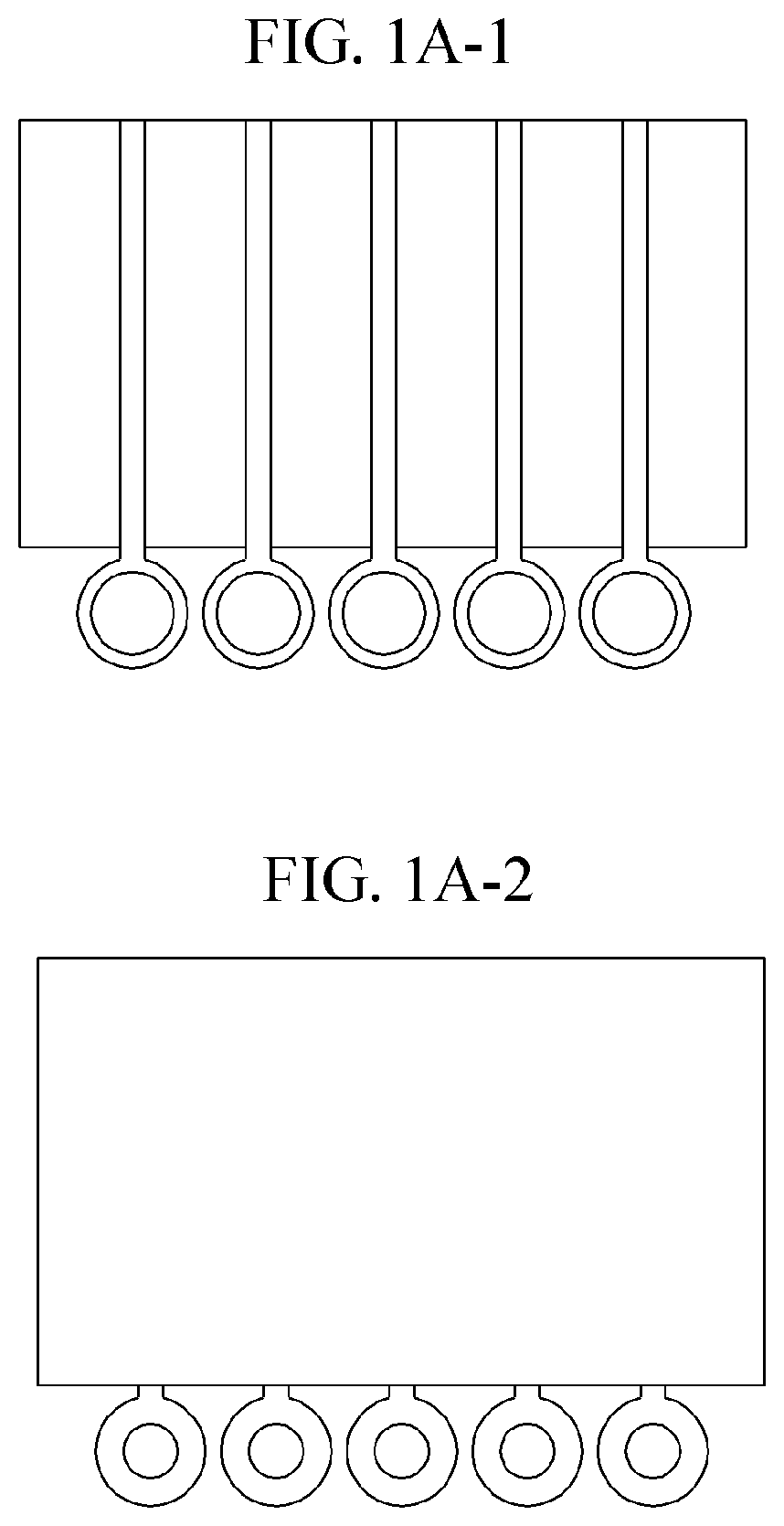
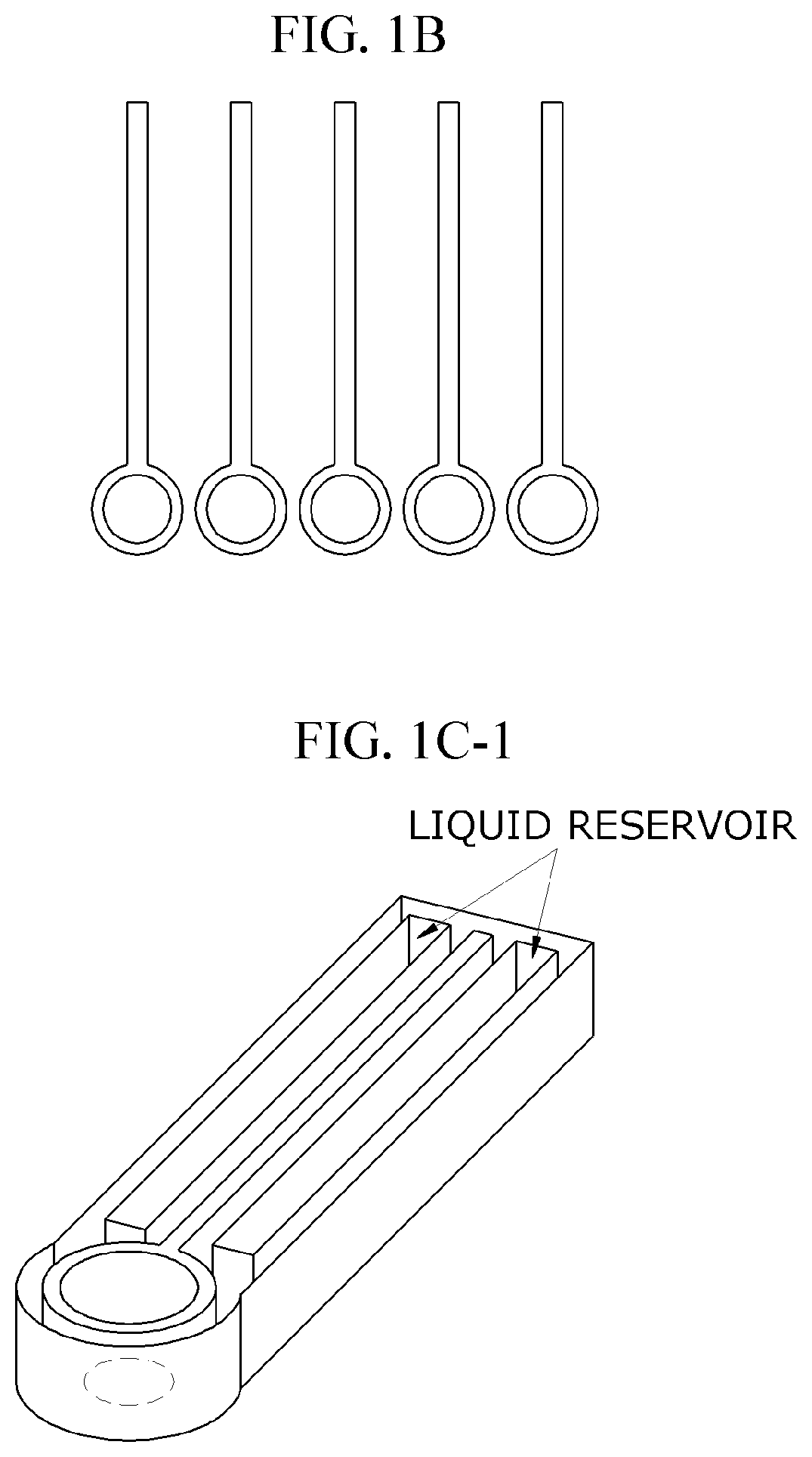
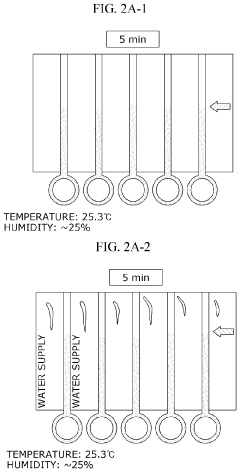
Lateral-flow microfluidic chip and flow velocity control method thereof
PatentInactiveUS20190374940A1
Innovation
- Increasing vapor pressure around specific channels in the microfluidic chip by using a separate vapor supply device or a liquid reservoir adjacent to the channel, which suppresses fluid evaporation and accelerates flow velocity, allowing for efficient sequential reactions without additional processes or equipment.
Quantitative and self-calibrating chemical analysis using paper-based microfluidic systems
PatentActiveEP2449380A1
Innovation
- A paper-based microfluidic system with hydrophilic testing zones that uses standard fluid samples of varying concentrations for internal self-calibration, reacting with an indicator solution to determine analyte concentration through color intensity changes, allowing for accurate results regardless of external factors and enabling use with diverse recording equipment.
Manufacturing Scalability Considerations
The scalability of manufacturing processes represents a critical consideration for the successful commercialization of microfluidic ELISA devices utilizing capillary-driven flow. Current laboratory-scale fabrication methods often employ techniques such as soft lithography with PDMS, which presents significant challenges when transitioning to mass production. These challenges include inconsistent surface properties, material degradation over time, and labor-intensive fabrication steps that are difficult to automate.
For high-volume manufacturing, injection molding and hot embossing emerge as more viable alternatives, offering reproducibility and cost-effectiveness at scale. However, these methods require substantial initial investment in tooling and equipment. The transition from prototype to mass production necessitates careful material selection that balances capillary flow performance with manufacturing constraints. Thermoplastics like cyclic olefin copolymer (COC) and polymethyl methacrylate (PMMA) demonstrate promising characteristics for scaled production while maintaining consistent surface properties essential for capillary action.
Channel dimension consistency represents another manufacturing challenge, as variations in channel geometry significantly impact capillary flow dynamics. Advanced quality control systems incorporating optical inspection and automated dimensional verification become essential when scaling production. Statistical process control methodologies must be implemented to monitor critical parameters affecting capillary flow performance across production batches.
Surface treatment processes, crucial for controlling hydrophilicity and ensuring reliable capillary action, present additional scaling challenges. Plasma treatment, commonly used in laboratory settings, requires adaptation for inline manufacturing processes. Alternative approaches such as chemical vapor deposition or UV-ozone treatment may offer more scalable solutions for surface modification in production environments.
Assembly and bonding processes must also evolve from manual laboratory techniques to automated systems. Techniques such as ultrasonic welding and laser welding show promise for high-throughput production while maintaining channel integrity. The integration of functional components like detection zones and sample introduction ports requires precision automation to ensure consistent performance across manufactured units.
Cost considerations ultimately drive manufacturing decisions, with material selection, process complexity, and production volume determining economic viability. Modular design approaches that separate critical capillary flow components from structural elements may optimize both performance and manufacturing efficiency. As production scales increase, environmental sustainability becomes increasingly important, necessitating consideration of recyclable materials and energy-efficient manufacturing processes that align with regulatory requirements and market expectations.
For high-volume manufacturing, injection molding and hot embossing emerge as more viable alternatives, offering reproducibility and cost-effectiveness at scale. However, these methods require substantial initial investment in tooling and equipment. The transition from prototype to mass production necessitates careful material selection that balances capillary flow performance with manufacturing constraints. Thermoplastics like cyclic olefin copolymer (COC) and polymethyl methacrylate (PMMA) demonstrate promising characteristics for scaled production while maintaining consistent surface properties essential for capillary action.
Channel dimension consistency represents another manufacturing challenge, as variations in channel geometry significantly impact capillary flow dynamics. Advanced quality control systems incorporating optical inspection and automated dimensional verification become essential when scaling production. Statistical process control methodologies must be implemented to monitor critical parameters affecting capillary flow performance across production batches.
Surface treatment processes, crucial for controlling hydrophilicity and ensuring reliable capillary action, present additional scaling challenges. Plasma treatment, commonly used in laboratory settings, requires adaptation for inline manufacturing processes. Alternative approaches such as chemical vapor deposition or UV-ozone treatment may offer more scalable solutions for surface modification in production environments.
Assembly and bonding processes must also evolve from manual laboratory techniques to automated systems. Techniques such as ultrasonic welding and laser welding show promise for high-throughput production while maintaining channel integrity. The integration of functional components like detection zones and sample introduction ports requires precision automation to ensure consistent performance across manufactured units.
Cost considerations ultimately drive manufacturing decisions, with material selection, process complexity, and production volume determining economic viability. Modular design approaches that separate critical capillary flow components from structural elements may optimize both performance and manufacturing efficiency. As production scales increase, environmental sustainability becomes increasingly important, necessitating consideration of recyclable materials and energy-efficient manufacturing processes that align with regulatory requirements and market expectations.
Regulatory Pathway for POC Diagnostic Applications
The regulatory landscape for Point-of-Care (POC) diagnostic applications incorporating microfluidic ELISA with capillary-driven flow presents a complex pathway that developers must navigate carefully. In the United States, the Food and Drug Administration (FDA) oversees these devices primarily through the Center for Devices and Radiological Health (CDRH), classifying them based on risk levels. Capillary-driven microfluidic ELISA devices typically fall under Class II (moderate risk) or Class III (high risk) depending on their intended use and diagnostic claims.
For these technologies, developers must generally pursue either the 510(k) premarket notification pathway (demonstrating substantial equivalence to a legally marketed device) or the more rigorous Premarket Approval (PMA) process. The choice depends on the novelty of the capillary flow mechanism and the clinical significance of the diagnostic results. Additionally, the FDA's newer De Novo classification process may be applicable for novel capillary-driven technologies without predicate devices.
In the European Union, the regulatory framework has shifted significantly with the implementation of the In Vitro Diagnostic Regulation (IVDR 2017/746), replacing the previous IVDD directive. This transition introduces a risk-based classification system with stricter requirements for clinical evidence, technical documentation, and post-market surveillance. Capillary-driven microfluidic ELISA devices are typically classified in Class B or C under the IVDR, requiring conformity assessment involving a Notified Body.
The regulatory pathway also includes consideration of international standards relevant to microfluidic technologies, such as ISO 13485 for quality management systems and IEC 62304 for software validation if the device incorporates digital components. For capillary-driven systems specifically, demonstrating reproducibility and reliability of the passive flow mechanisms becomes a critical regulatory consideration.
Emerging markets present additional regulatory challenges, with China's NMPA, Japan's PMDA, and India's CDSCO each maintaining distinct requirements for POC diagnostic approval. These authorities increasingly emphasize local clinical validation studies that account for population-specific factors that might affect capillary flow performance in different environmental conditions.
A strategic approach to regulatory compliance for capillary-driven microfluidic ELISA technologies should include early engagement with regulatory bodies through pre-submission consultations, careful design of validation studies that specifically address the unique aspects of passive capillary flow, and development of comprehensive risk management documentation that addresses potential failure modes specific to this technology.
For these technologies, developers must generally pursue either the 510(k) premarket notification pathway (demonstrating substantial equivalence to a legally marketed device) or the more rigorous Premarket Approval (PMA) process. The choice depends on the novelty of the capillary flow mechanism and the clinical significance of the diagnostic results. Additionally, the FDA's newer De Novo classification process may be applicable for novel capillary-driven technologies without predicate devices.
In the European Union, the regulatory framework has shifted significantly with the implementation of the In Vitro Diagnostic Regulation (IVDR 2017/746), replacing the previous IVDD directive. This transition introduces a risk-based classification system with stricter requirements for clinical evidence, technical documentation, and post-market surveillance. Capillary-driven microfluidic ELISA devices are typically classified in Class B or C under the IVDR, requiring conformity assessment involving a Notified Body.
The regulatory pathway also includes consideration of international standards relevant to microfluidic technologies, such as ISO 13485 for quality management systems and IEC 62304 for software validation if the device incorporates digital components. For capillary-driven systems specifically, demonstrating reproducibility and reliability of the passive flow mechanisms becomes a critical regulatory consideration.
Emerging markets present additional regulatory challenges, with China's NMPA, Japan's PMDA, and India's CDSCO each maintaining distinct requirements for POC diagnostic approval. These authorities increasingly emphasize local clinical validation studies that account for population-specific factors that might affect capillary flow performance in different environmental conditions.
A strategic approach to regulatory compliance for capillary-driven microfluidic ELISA technologies should include early engagement with regulatory bodies through pre-submission consultations, careful design of validation studies that specifically address the unique aspects of passive capillary flow, and development of comprehensive risk management documentation that addresses potential failure modes specific to this technology.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!