Research on Digital Microfluidic ELISA Automation
OCT 13, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Digital Microfluidic ELISA Background and Objectives
Digital Microfluidic (DMF) technology has emerged as a revolutionary approach in the field of laboratory diagnostics over the past two decades. This technology manipulates discrete droplets of fluid on a hydrophobic surface using electrowetting principles, offering unprecedented control over small-volume biochemical reactions. The evolution of DMF has been marked by significant milestones, from initial proof-of-concept demonstrations in the early 2000s to recent integrated systems capable of performing complex multi-step assays.
The integration of DMF with Enzyme-Linked Immunosorbent Assay (ELISA), one of the most widely used diagnostic techniques in clinical and research settings, represents a particularly promising direction. Traditional ELISA procedures are labor-intensive, time-consuming, and require specialized laboratory equipment and trained personnel. The automation of ELISA through DMF technology addresses these limitations by miniaturizing the process, reducing reagent consumption, and eliminating manual intervention steps.
Recent technological advances in electrode design, surface chemistry, and control systems have accelerated the development of DMF-ELISA platforms. The transition from academic prototypes to commercially viable systems has been facilitated by improvements in manufacturing processes, particularly in the areas of electrode patterning and dielectric layer deposition. Parallel developments in biosensor integration and detection methodologies have further enhanced the sensitivity and specificity of these systems.
The primary objective of this research is to comprehensively evaluate the current state of Digital Microfluidic ELISA automation and identify pathways for technological advancement. Specifically, we aim to assess the technical feasibility of developing a fully integrated DMF-ELISA platform capable of performing multiple assays simultaneously with minimal user intervention. This includes examining critical parameters such as droplet manipulation reliability, cross-contamination prevention, detection sensitivity, and system robustness.
Additionally, this research seeks to establish a technological roadmap for overcoming existing limitations in DMF-ELISA systems. These include challenges related to protein adsorption on hydrophobic surfaces, biofouling during extended operations, integration of sample preparation steps, and scaling of electrode arrays for higher throughput. The development of standardized protocols and quality control measures for DMF-ELISA represents another key objective, essential for clinical adoption and regulatory approval.
Furthermore, this investigation aims to explore emerging trends in the field, including the integration of artificial intelligence for automated assay optimization, the development of reconfigurable DMF platforms for diverse assay types, and the potential for point-of-care applications in resource-limited settings. By identifying these technological trajectories, this research will provide valuable insights for strategic R&D investments and product development initiatives.
The integration of DMF with Enzyme-Linked Immunosorbent Assay (ELISA), one of the most widely used diagnostic techniques in clinical and research settings, represents a particularly promising direction. Traditional ELISA procedures are labor-intensive, time-consuming, and require specialized laboratory equipment and trained personnel. The automation of ELISA through DMF technology addresses these limitations by miniaturizing the process, reducing reagent consumption, and eliminating manual intervention steps.
Recent technological advances in electrode design, surface chemistry, and control systems have accelerated the development of DMF-ELISA platforms. The transition from academic prototypes to commercially viable systems has been facilitated by improvements in manufacturing processes, particularly in the areas of electrode patterning and dielectric layer deposition. Parallel developments in biosensor integration and detection methodologies have further enhanced the sensitivity and specificity of these systems.
The primary objective of this research is to comprehensively evaluate the current state of Digital Microfluidic ELISA automation and identify pathways for technological advancement. Specifically, we aim to assess the technical feasibility of developing a fully integrated DMF-ELISA platform capable of performing multiple assays simultaneously with minimal user intervention. This includes examining critical parameters such as droplet manipulation reliability, cross-contamination prevention, detection sensitivity, and system robustness.
Additionally, this research seeks to establish a technological roadmap for overcoming existing limitations in DMF-ELISA systems. These include challenges related to protein adsorption on hydrophobic surfaces, biofouling during extended operations, integration of sample preparation steps, and scaling of electrode arrays for higher throughput. The development of standardized protocols and quality control measures for DMF-ELISA represents another key objective, essential for clinical adoption and regulatory approval.
Furthermore, this investigation aims to explore emerging trends in the field, including the integration of artificial intelligence for automated assay optimization, the development of reconfigurable DMF platforms for diverse assay types, and the potential for point-of-care applications in resource-limited settings. By identifying these technological trajectories, this research will provide valuable insights for strategic R&D investments and product development initiatives.
Market Analysis for Automated ELISA Systems
The global market for automated ELISA systems has been experiencing robust growth, driven by increasing demand for high-throughput diagnostic solutions across healthcare, pharmaceutical research, and life sciences sectors. The market was valued at approximately $1.2 billion in 2022 and is projected to reach $1.8 billion by 2027, representing a compound annual growth rate (CAGR) of 8.5%. This growth trajectory is particularly significant in regions with advanced healthcare infrastructure such as North America and Europe, which collectively account for over 60% of the global market share.
Digital microfluidic (DMF) ELISA automation represents a disruptive innovation within this market landscape, offering significant advantages over conventional automated ELISA systems. Traditional automated systems, while efficient, still require relatively large sample volumes and considerable laboratory space. In contrast, DMF technology enables miniaturization, reduced reagent consumption (up to 90% less than conventional methods), and enhanced portability, addressing critical market needs for point-of-care testing and resource-limited settings.
The demand for DMF ELISA automation is being fueled by several key market drivers. First, the growing prevalence of chronic diseases necessitates more accessible and efficient diagnostic tools. Second, the increasing focus on personalized medicine requires more sensitive and specific testing methodologies. Third, the global push toward decentralized healthcare delivery models is creating opportunities for portable diagnostic platforms. Fourth, cost pressures within healthcare systems worldwide are driving demand for more economical testing solutions that reduce reagent waste and labor costs.
Market segmentation reveals particularly strong growth potential in clinical diagnostics (estimated at 45% of the market), pharmaceutical research and development (30%), and academic research (15%). Within clinical diagnostics, infectious disease testing represents the largest application segment, followed by autoimmune disease diagnostics and cancer biomarker detection. The pharmaceutical sector's demand is primarily driven by drug discovery applications and clinical trials, where high-throughput screening capabilities are essential.
Geographically, emerging markets in Asia-Pacific, particularly China and India, are expected to witness the highest growth rates (12-14% annually) due to expanding healthcare infrastructure, increasing R&D investments, and growing awareness about advanced diagnostic technologies. However, market penetration in these regions faces challenges related to high initial investment costs and technical expertise requirements.
Customer needs analysis indicates that end-users prioritize automation reliability, ease of integration with existing laboratory information systems, reduced hands-on time, and improved reproducibility of results. Additionally, there is growing demand for systems that can handle smaller sample volumes, particularly in pediatric applications and precious sample testing scenarios.
Digital microfluidic (DMF) ELISA automation represents a disruptive innovation within this market landscape, offering significant advantages over conventional automated ELISA systems. Traditional automated systems, while efficient, still require relatively large sample volumes and considerable laboratory space. In contrast, DMF technology enables miniaturization, reduced reagent consumption (up to 90% less than conventional methods), and enhanced portability, addressing critical market needs for point-of-care testing and resource-limited settings.
The demand for DMF ELISA automation is being fueled by several key market drivers. First, the growing prevalence of chronic diseases necessitates more accessible and efficient diagnostic tools. Second, the increasing focus on personalized medicine requires more sensitive and specific testing methodologies. Third, the global push toward decentralized healthcare delivery models is creating opportunities for portable diagnostic platforms. Fourth, cost pressures within healthcare systems worldwide are driving demand for more economical testing solutions that reduce reagent waste and labor costs.
Market segmentation reveals particularly strong growth potential in clinical diagnostics (estimated at 45% of the market), pharmaceutical research and development (30%), and academic research (15%). Within clinical diagnostics, infectious disease testing represents the largest application segment, followed by autoimmune disease diagnostics and cancer biomarker detection. The pharmaceutical sector's demand is primarily driven by drug discovery applications and clinical trials, where high-throughput screening capabilities are essential.
Geographically, emerging markets in Asia-Pacific, particularly China and India, are expected to witness the highest growth rates (12-14% annually) due to expanding healthcare infrastructure, increasing R&D investments, and growing awareness about advanced diagnostic technologies. However, market penetration in these regions faces challenges related to high initial investment costs and technical expertise requirements.
Customer needs analysis indicates that end-users prioritize automation reliability, ease of integration with existing laboratory information systems, reduced hands-on time, and improved reproducibility of results. Additionally, there is growing demand for systems that can handle smaller sample volumes, particularly in pediatric applications and precious sample testing scenarios.
Current Challenges in Digital Microfluidic ELISA
Despite significant advancements in digital microfluidic (DMF) ELISA technology, several critical challenges continue to impede its widespread adoption and full automation. The integration of multiple biochemical processes on a single DMF platform remains problematic, particularly when considering the diverse reaction conditions required for different ELISA steps. Temperature control, incubation timing, and reagent compatibility must be precisely managed across the entire workflow, creating significant engineering hurdles.
Droplet manipulation reliability presents another major obstacle. Current DMF systems struggle with consistent droplet movement, especially when handling complex biological samples like serum or whole blood. These samples often contain proteins and other biomolecules that can adsorb onto the device surface, causing biofouling and compromising the electrowetting mechanism. This phenomenon, known as electrode fouling, reduces operational efficiency and leads to unpredictable droplet behavior during extended automated protocols.
Cross-contamination between sequential assay steps represents a persistent challenge in DMF ELISA systems. The shared pathways for different reagents and the potential for residue accumulation on electrode surfaces can compromise assay specificity and sensitivity. Current solutions involving washing steps or surface treatments add complexity to the automation process and may not completely eliminate contamination risks.
Scalability issues further complicate DMF ELISA automation. As the number of simultaneous assays increases, the complexity of electrode arrays, control systems, and fluidic routing grows exponentially. This scaling challenge limits throughput capabilities and makes multiplexed testing difficult to implement effectively on current platforms.
Detection sensitivity remains suboptimal compared to conventional laboratory-based ELISA methods. The miniaturized reaction volumes in DMF systems, while advantageous for reagent conservation, can limit detection capabilities for low-abundance biomarkers. Integration of highly sensitive detection technologies without compromising the compact nature of DMF platforms presents significant engineering challenges.
Standardization across different DMF ELISA platforms is notably lacking. The absence of universal protocols, reagent specifications, and quality control measures hinders reproducibility between different systems and laboratories. This standardization gap impedes clinical validation and regulatory approval processes necessary for widespread adoption in diagnostic settings.
Manufacturing complexity and associated costs present substantial barriers to commercialization. Current fabrication methods for DMF devices often involve sophisticated cleanroom processes and expensive materials, resulting in high production costs that limit accessibility and commercial viability. Developing cost-effective, scalable manufacturing approaches while maintaining device performance remains a significant industry challenge.
Droplet manipulation reliability presents another major obstacle. Current DMF systems struggle with consistent droplet movement, especially when handling complex biological samples like serum or whole blood. These samples often contain proteins and other biomolecules that can adsorb onto the device surface, causing biofouling and compromising the electrowetting mechanism. This phenomenon, known as electrode fouling, reduces operational efficiency and leads to unpredictable droplet behavior during extended automated protocols.
Cross-contamination between sequential assay steps represents a persistent challenge in DMF ELISA systems. The shared pathways for different reagents and the potential for residue accumulation on electrode surfaces can compromise assay specificity and sensitivity. Current solutions involving washing steps or surface treatments add complexity to the automation process and may not completely eliminate contamination risks.
Scalability issues further complicate DMF ELISA automation. As the number of simultaneous assays increases, the complexity of electrode arrays, control systems, and fluidic routing grows exponentially. This scaling challenge limits throughput capabilities and makes multiplexed testing difficult to implement effectively on current platforms.
Detection sensitivity remains suboptimal compared to conventional laboratory-based ELISA methods. The miniaturized reaction volumes in DMF systems, while advantageous for reagent conservation, can limit detection capabilities for low-abundance biomarkers. Integration of highly sensitive detection technologies without compromising the compact nature of DMF platforms presents significant engineering challenges.
Standardization across different DMF ELISA platforms is notably lacking. The absence of universal protocols, reagent specifications, and quality control measures hinders reproducibility between different systems and laboratories. This standardization gap impedes clinical validation and regulatory approval processes necessary for widespread adoption in diagnostic settings.
Manufacturing complexity and associated costs present substantial barriers to commercialization. Current fabrication methods for DMF devices often involve sophisticated cleanroom processes and expensive materials, resulting in high production costs that limit accessibility and commercial viability. Developing cost-effective, scalable manufacturing approaches while maintaining device performance remains a significant industry challenge.
Current Digital Microfluidic ELISA Solutions
01 Digital microfluidic platforms for ELISA automation
Digital microfluidic platforms enable the automation of ELISA procedures by manipulating small droplets of reagents on a surface using electrowetting principles. These platforms allow for precise control of droplet movement, mixing, and detection, eliminating the need for manual pipetting and reducing sample volumes. The automation of ELISA on digital microfluidic platforms increases throughput, improves reproducibility, and reduces human error in diagnostic testing.- Digital microfluidic platforms for ELISA automation: Digital microfluidic platforms enable the automation of ELISA procedures by manipulating small droplets of reagents on a surface using electrowetting principles. These platforms allow for precise control of droplet movement, mixing, and detection, reducing sample and reagent volumes while increasing throughput. The technology integrates electrodes, control systems, and detection mechanisms to perform all ELISA steps in a miniaturized format, offering advantages in speed, sensitivity, and reproducibility compared to conventional methods.
- Droplet manipulation techniques for immunoassays: Advanced droplet manipulation techniques are essential for performing immunoassays on digital microfluidic platforms. These techniques include electrowetting-based movement, magnetic bead-based separation, and acoustic manipulation of droplets containing antibodies, antigens, and detection reagents. The precise control of droplet operations such as dispensing, merging, mixing, and splitting enables complex multi-step ELISA protocols to be performed with minimal human intervention, enhancing assay performance and reliability.
- Integrated detection systems for microfluidic ELISA: Integrated detection systems are crucial components of digital microfluidic ELISA platforms, enabling real-time monitoring and quantification of immunoassay results. These systems incorporate optical, electrochemical, or fluorescence-based detection methods directly into the microfluidic chip. Advanced imaging technologies, photodetectors, and signal processing algorithms work together to provide sensitive and accurate measurement of analyte concentrations, while minimizing background noise and interference from the microfluidic environment.
- Microfluidic chip design for ELISA applications: Specialized microfluidic chip designs optimize ELISA performance in digital platforms. These designs feature strategically arranged electrode arrays, hydrophobic and hydrophilic surface treatments, and reaction chambers optimized for antibody-antigen interactions. The chip architecture incorporates reagent reservoirs, waste collection areas, and detection zones while maintaining biocompatibility with biological samples and reagents. Advanced fabrication techniques ensure precise channel dimensions and surface properties that enhance assay sensitivity and reproducibility.
- Automation software and control systems for digital microfluidic ELISA: Sophisticated automation software and control systems are essential for orchestrating the complex operations required in digital microfluidic ELISA. These systems provide programmable protocols for droplet manipulation sequences, timing of reagent additions, incubation periods, and washing steps. Real-time feedback mechanisms monitor assay progress and make adjustments as needed. User-friendly interfaces allow operators to design custom assay protocols, while data management components handle result storage, analysis, and interpretation, enabling seamless integration with laboratory information systems.
02 Droplet manipulation techniques for immunoassays
Various techniques for manipulating droplets in digital microfluidic systems enable efficient ELISA automation. These include electrowetting-on-dielectric (EWOD), dielectrophoresis, and magnetic actuation methods. These techniques allow for controlled movement, splitting, merging, and mixing of droplets containing antibodies, antigens, and detection reagents. Advanced droplet manipulation enhances the precision and speed of immunoassay protocols while minimizing reagent consumption.Expand Specific Solutions03 Integrated detection systems for microfluidic ELISA
Integrated detection systems in digital microfluidic ELISA platforms enable real-time monitoring and quantification of immunoassay results. These systems incorporate optical, electrochemical, or fluorescence-based detection methods directly into the microfluidic chip. The integration of detection components eliminates the need for external readers, reduces analysis time, and enables point-of-care applications. These systems can achieve sensitivity comparable to conventional ELISA while offering greater portability and ease of use.Expand Specific Solutions04 Microfluidic chip designs for automated immunoassays
Specialized microfluidic chip designs facilitate the automation of ELISA procedures. These designs incorporate electrode arrays, hydrophobic and hydrophilic regions, and reaction chambers optimized for immunoassay protocols. Some designs feature multiple layers for separating different assay components or include integrated heating elements for temperature-controlled reactions. Advanced chip architectures enable parallel processing of multiple samples, increasing throughput and efficiency in clinical diagnostics.Expand Specific Solutions05 Software control systems for digital microfluidic ELISA
Software control systems are essential for orchestrating the complex sequence of operations in digital microfluidic ELISA automation. These systems coordinate droplet movements, timing of reagent additions, incubation periods, and detection steps. Advanced algorithms optimize assay protocols, monitor system performance, and process analytical data. User-friendly interfaces allow operators to program custom assay protocols and visualize results, making automated ELISA accessible to users without specialized training in microfluidics.Expand Specific Solutions
Leading Companies in Digital Microfluidics
The digital microfluidic ELISA automation market is currently in its growth phase, characterized by increasing adoption of lab-on-a-chip technologies for diagnostic applications. The global market size is estimated to reach approximately $1.2 billion by 2027, with a CAGR of 8-10%. From a technological maturity perspective, companies like Advanced Liquid Logic (acquired by Illumina) and Baebies have established foundational digital microfluidic platforms, while BOE Technology Group and Samsung Electronics are leveraging their display expertise to develop integrated solutions. Academic institutions including University of Toronto and Nanyang Technological University are driving innovation through research partnerships. Miroculus and Digital Biosystems represent specialized players focusing on microfluidic ELISA applications, while established diagnostic companies like Abbott Laboratories and Tecan Trading AG are incorporating this technology into their product portfolios to enhance automation capabilities and improve assay performance.
The Governing Council of the University of Toronto
Technical Solution: The University of Toronto's Wheeler Microfluidics Laboratory has pioneered significant advancements in digital microfluidic (DMF) ELISA automation. Their platform employs electrowetting-on-dielectric (EWOD) principles with a two-plate configuration—a bottom plate containing an array of individually addressable electrodes and a top plate serving as a ground electrode. This design enables precise manipulation of nanoliter-scale droplets containing samples and reagents. The Toronto team has developed specialized surface modifications using heterogeneous immunoassay formats where capture antibodies are immobilized on the DMF device surface. Their innovation includes magnetic particle-based protocols that enhance washing efficiency and reduce non-specific binding. The system integrates real-time fluorescence detection capabilities and has demonstrated detection limits in the pg/mL range for various biomarkers. Their automated DMF-ELISA platform reduces total assay time from 4+ hours to under 90 minutes while consuming less than 1% of the reagent volumes required by conventional methods.
Strengths: Exceptional reagent efficiency with nanoliter-scale operations; versatile platform supporting multiple assay formats (direct, sandwich, competitive); robust magnetic particle-based protocols improving sensitivity; comprehensive automation reducing operator variability. Weaknesses: Complex fabrication process for DMF devices limiting mass production; challenges with surface fouling during repeated use; current designs primarily optimized for research applications rather than high-throughput clinical settings; requires specialized expertise for protocol development.
Miroculus, Inc.
Technical Solution: Miroculus has developed the Miro Canvas platform, a digital microfluidic system specifically optimized for automated ELISA workflows. Their technology utilizes an array of individually addressable electrodes to manipulate droplets through electrowetting principles. The platform features a cartridge-based design with integrated reagent storage and reconstitution capabilities, allowing complete sample-to-answer ELISA processing. Their proprietary surface chemistry modifications reduce protein adsorption and non-specific binding, addressing a key challenge in microfluidic immunoassays. The system incorporates real-time fluorescence detection with multiple excitation/emission channels for multiplexed analysis. Miroculus' software suite provides automated protocol execution, quality control monitoring, and data analysis. The platform achieves detection limits comparable to conventional ELISA (picogram/mL range) while using approximately 10-fold less reagent volume and reducing hands-on time by over 70%.
Strengths: User-friendly interface with pre-programmed ELISA protocols; robust surface chemistry minimizing cross-contamination; flexible cartridge design accommodating different assay formats; integrated detection system eliminating manual transfers. Weaknesses: Limited menu of validated assay protocols compared to established platforms; higher initial capital investment; dependence on proprietary consumables; potential challenges with highly complex sample matrices requiring extensive preparation.
Key Patents and Research in DMF-ELISA
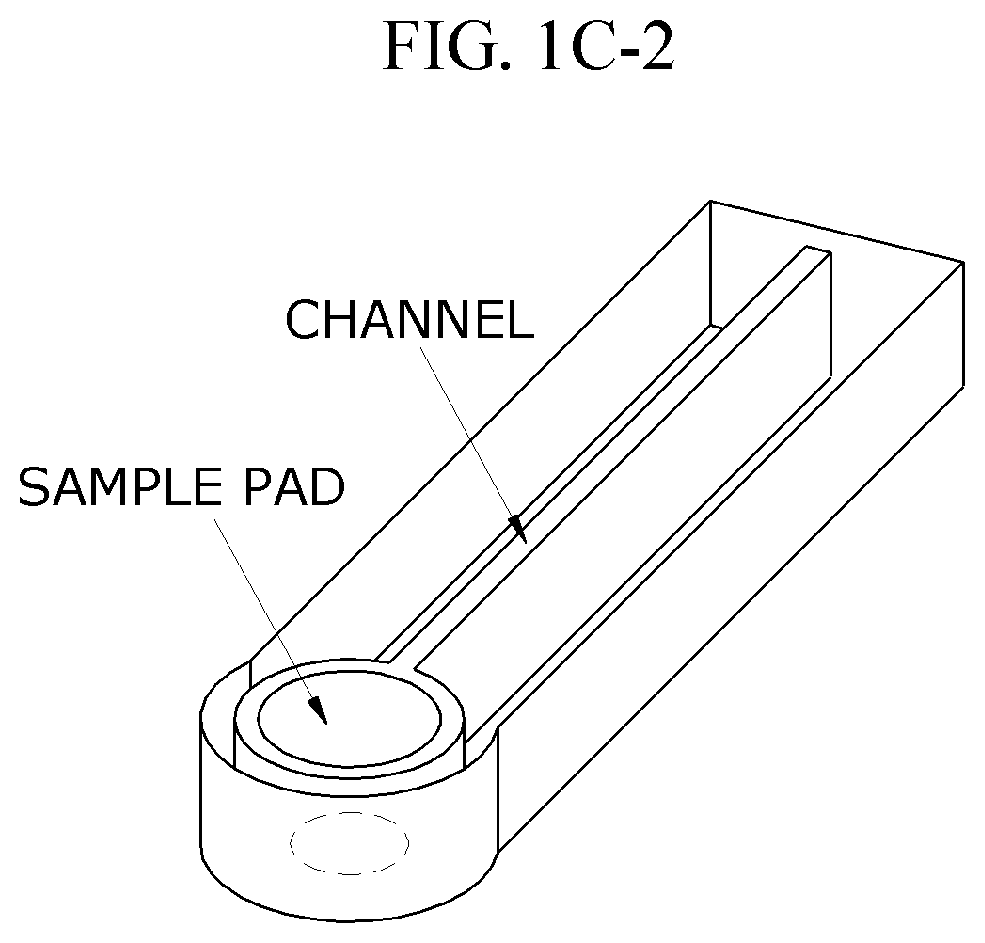
Droplet generating method
PatentActiveUS20210207191A1
Innovation
- A droplet generating method using a micro-pipe with a vibrating equipment to create relative periodic vibrations, allowing the micro-pipe's outlet end to touch a second liquid, thereby overcoming surface tension and adhesion forces to produce uniform droplets with controlled sizes and volumes.
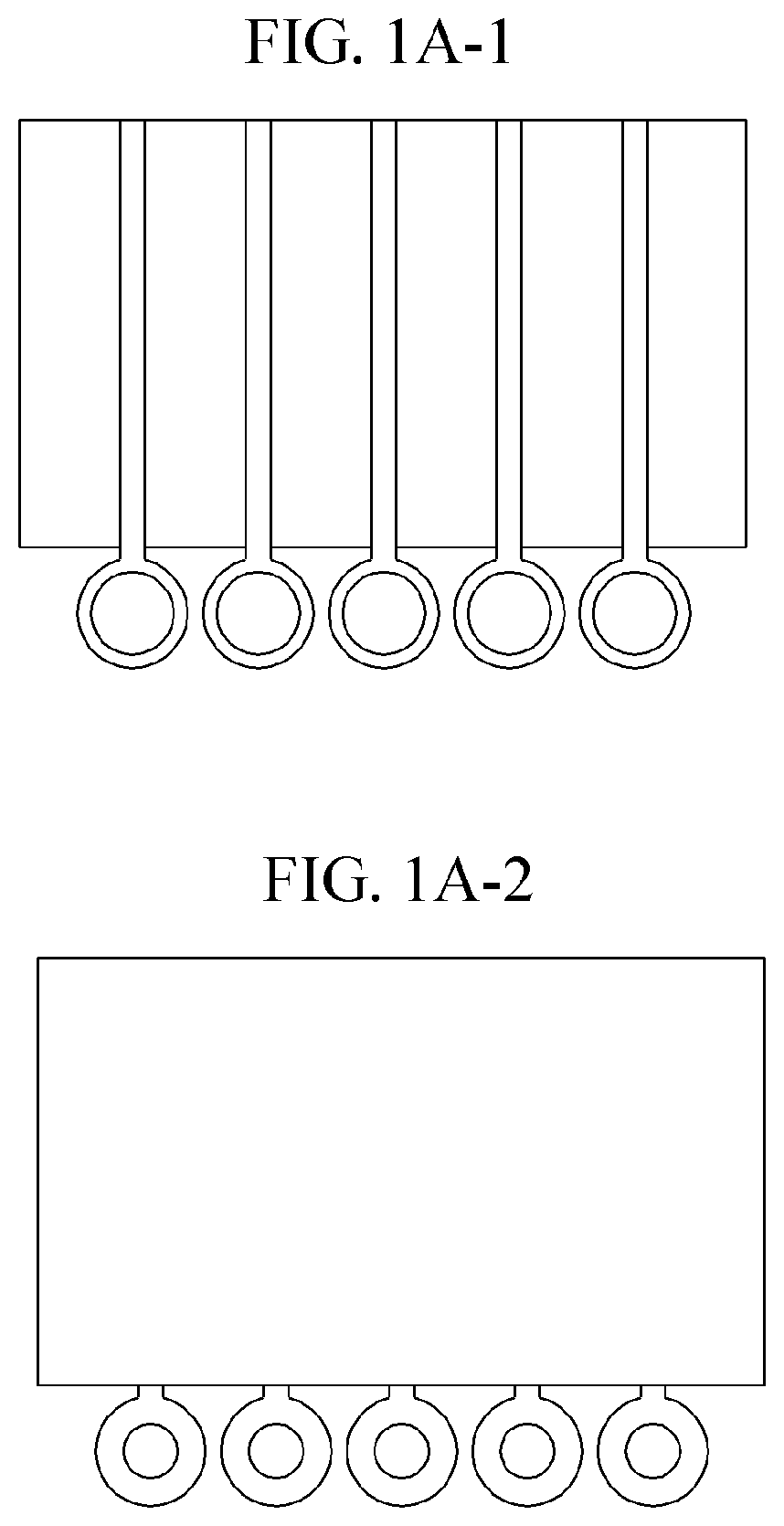
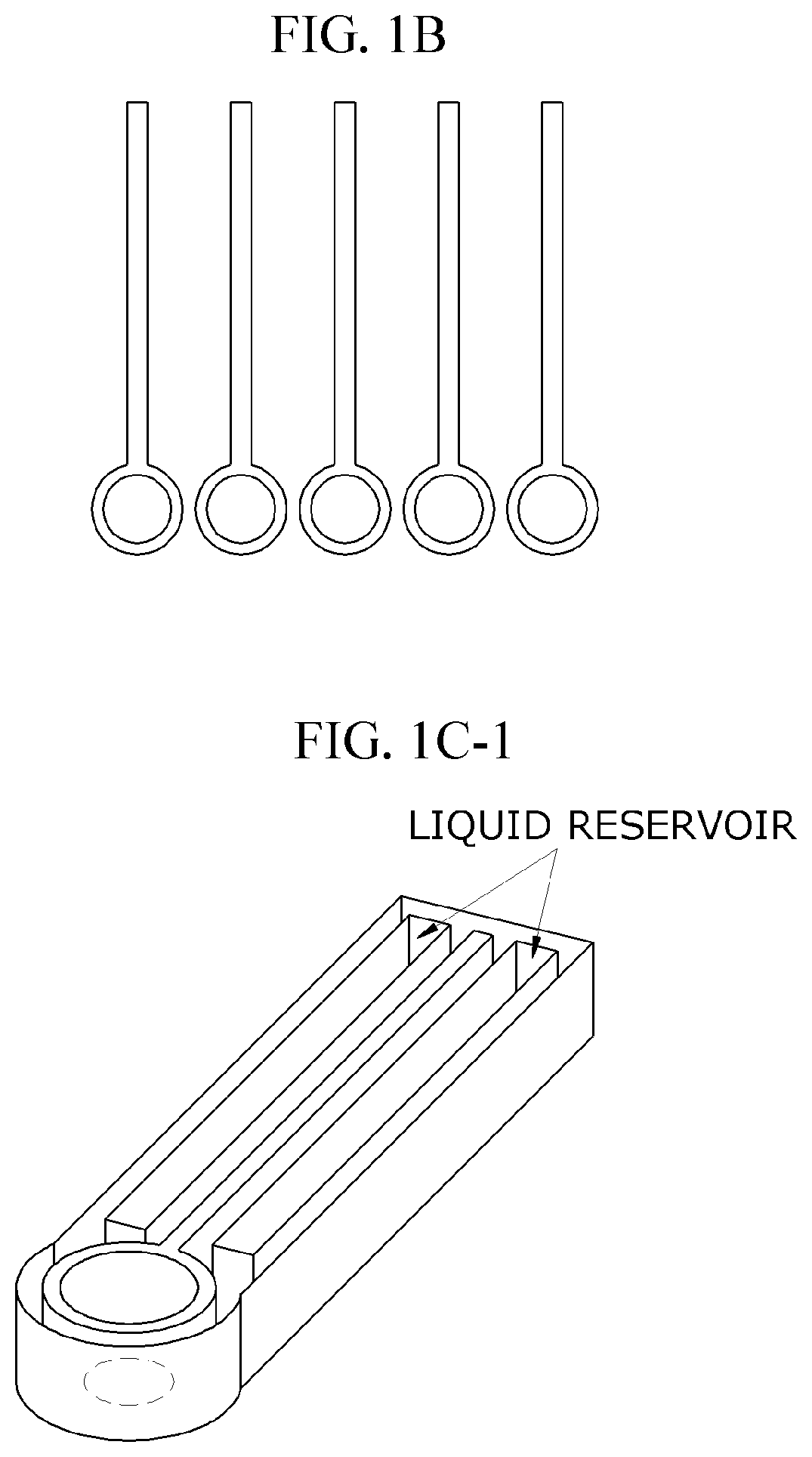
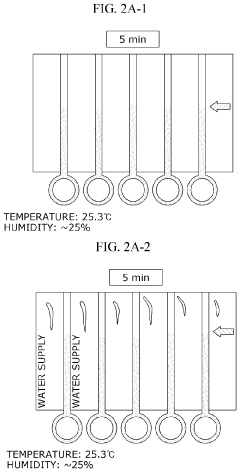
Lateral-flow microfluidic chip and flow velocity control method thereof
PatentInactiveUS20190374940A1
Innovation
- Increasing vapor pressure around specific channels in the microfluidic chip by using a separate vapor supply device or a liquid reservoir adjacent to the channel, which suppresses fluid evaporation and accelerates flow velocity, allowing for efficient sequential reactions without additional processes or equipment.
Regulatory Considerations for Clinical Applications
The implementation of Digital Microfluidic ELISA Automation in clinical settings necessitates thorough compliance with various regulatory frameworks. In the United States, the FDA classifies such systems under medical devices, requiring premarket approval (PMA) or 510(k) clearance depending on risk classification. Digital microfluidic platforms typically fall under Class II medical devices, demanding demonstration of substantial equivalence to predicate devices already on the market.
European regulatory bodies enforce the In Vitro Diagnostic Regulation (IVDR), which replaced the IVD Directive in 2022, imposing stricter requirements for clinical evidence, risk management, and post-market surveillance. Digital microfluidic ELISA systems must meet these enhanced standards, with particular attention to performance evaluation and technical documentation.
Quality management systems compliant with ISO 13485 are essential for manufacturers developing automated ELISA platforms. This standard ensures consistent design, development, production, and service processes that meet both regulatory requirements and customer needs. Additionally, IEC 61010 standards for laboratory equipment safety and IEC 62304 for medical device software must be addressed during development.
Clinical validation presents significant regulatory challenges, requiring demonstration of analytical performance (sensitivity, specificity, precision) and clinical performance (diagnostic accuracy, clinical utility). Manufacturers must conduct appropriate clinical studies and collect sufficient data to support intended use claims, particularly for novel biomarkers or diagnostic applications.
Data security and privacy regulations, including HIPAA in the US and GDPR in Europe, impose additional compliance requirements when patient data is processed or stored by digital microfluidic systems. Secure data handling protocols, encryption methods, and user authentication systems must be implemented to protect sensitive clinical information.
Regulatory pathways for novel technologies like digital microfluidic ELISA automation often involve early engagement with regulatory bodies through pre-submission meetings or consultation procedures. These interactions help clarify specific requirements and potential regulatory hurdles before formal submission, potentially streamlining the approval process.
Post-market surveillance requirements mandate continuous monitoring of device performance and safety after regulatory approval. Manufacturers must establish systems for adverse event reporting, periodic safety updates, and implementation of corrective actions when necessary, ensuring long-term compliance and patient safety.
European regulatory bodies enforce the In Vitro Diagnostic Regulation (IVDR), which replaced the IVD Directive in 2022, imposing stricter requirements for clinical evidence, risk management, and post-market surveillance. Digital microfluidic ELISA systems must meet these enhanced standards, with particular attention to performance evaluation and technical documentation.
Quality management systems compliant with ISO 13485 are essential for manufacturers developing automated ELISA platforms. This standard ensures consistent design, development, production, and service processes that meet both regulatory requirements and customer needs. Additionally, IEC 61010 standards for laboratory equipment safety and IEC 62304 for medical device software must be addressed during development.
Clinical validation presents significant regulatory challenges, requiring demonstration of analytical performance (sensitivity, specificity, precision) and clinical performance (diagnostic accuracy, clinical utility). Manufacturers must conduct appropriate clinical studies and collect sufficient data to support intended use claims, particularly for novel biomarkers or diagnostic applications.
Data security and privacy regulations, including HIPAA in the US and GDPR in Europe, impose additional compliance requirements when patient data is processed or stored by digital microfluidic systems. Secure data handling protocols, encryption methods, and user authentication systems must be implemented to protect sensitive clinical information.
Regulatory pathways for novel technologies like digital microfluidic ELISA automation often involve early engagement with regulatory bodies through pre-submission meetings or consultation procedures. These interactions help clarify specific requirements and potential regulatory hurdles before formal submission, potentially streamlining the approval process.
Post-market surveillance requirements mandate continuous monitoring of device performance and safety after regulatory approval. Manufacturers must establish systems for adverse event reporting, periodic safety updates, and implementation of corrective actions when necessary, ensuring long-term compliance and patient safety.
Cost-Benefit Analysis of DMF-ELISA Implementation
The implementation of Digital Microfluidic ELISA (DMF-ELISA) systems represents a significant investment for laboratories and healthcare facilities. This analysis examines the economic aspects of adopting this technology compared to traditional ELISA methods, providing stakeholders with comprehensive financial insights for decision-making.
Initial capital expenditure for DMF-ELISA systems ranges from $50,000 to $150,000, depending on system complexity, automation level, and detection capabilities. While this represents a substantial upfront investment compared to conventional ELISA equipment ($15,000-$30,000), the long-term operational economics reveal a different picture.
Consumable costs show marked differences between the two approaches. Traditional ELISA requires multiple disposable components including microplates, pipette tips, and reagent reservoirs, averaging $5-15 per test. DMF-ELISA platforms significantly reduce reagent consumption by up to 90%, with typical per-test consumable costs of $1-3, representing substantial savings for high-volume testing environments.
Labor efficiency constitutes another critical economic factor. Conventional ELISA protocols demand 3-5 hours of technician time for sample preparation, incubation, and result interpretation. DMF-ELISA automation reduces hands-on time to approximately 30-45 minutes, allowing laboratory personnel to manage multiple tests simultaneously or perform other tasks. At average laboratory technician compensation rates, this translates to labor savings of $50-100 per testing batch.
Maintenance requirements also differ significantly. Traditional ELISA equipment typically requires calibration and maintenance 2-4 times annually at $1,500-3,000 per service. DMF-ELISA systems, with more sophisticated electronics and microfluidic components, necessitate specialized maintenance at higher per-service costs ($2,500-5,000) but often with reduced frequency (1-2 times annually) due to fewer moving parts and automated self-diagnostic capabilities.
Return on investment (ROI) calculations indicate that laboratories processing more than 10,000 tests annually can expect to recover DMF-ELISA implementation costs within 2-3 years, primarily through reagent savings and labor efficiency. Smaller facilities with lower testing volumes may experience longer payback periods of 4-6 years, suggesting that adoption decisions should be volume-dependent.
Additional economic benefits include reduced waste management costs, as DMF-ELISA generates approximately 70% less biohazardous waste than traditional methods. Furthermore, the higher sensitivity of DMF-ELISA can reduce false negative/positive results, decreasing costly repeat testing and improving diagnostic accuracy, though these benefits are more difficult to quantify precisely in financial terms.
Initial capital expenditure for DMF-ELISA systems ranges from $50,000 to $150,000, depending on system complexity, automation level, and detection capabilities. While this represents a substantial upfront investment compared to conventional ELISA equipment ($15,000-$30,000), the long-term operational economics reveal a different picture.
Consumable costs show marked differences between the two approaches. Traditional ELISA requires multiple disposable components including microplates, pipette tips, and reagent reservoirs, averaging $5-15 per test. DMF-ELISA platforms significantly reduce reagent consumption by up to 90%, with typical per-test consumable costs of $1-3, representing substantial savings for high-volume testing environments.
Labor efficiency constitutes another critical economic factor. Conventional ELISA protocols demand 3-5 hours of technician time for sample preparation, incubation, and result interpretation. DMF-ELISA automation reduces hands-on time to approximately 30-45 minutes, allowing laboratory personnel to manage multiple tests simultaneously or perform other tasks. At average laboratory technician compensation rates, this translates to labor savings of $50-100 per testing batch.
Maintenance requirements also differ significantly. Traditional ELISA equipment typically requires calibration and maintenance 2-4 times annually at $1,500-3,000 per service. DMF-ELISA systems, with more sophisticated electronics and microfluidic components, necessitate specialized maintenance at higher per-service costs ($2,500-5,000) but often with reduced frequency (1-2 times annually) due to fewer moving parts and automated self-diagnostic capabilities.
Return on investment (ROI) calculations indicate that laboratories processing more than 10,000 tests annually can expect to recover DMF-ELISA implementation costs within 2-3 years, primarily through reagent savings and labor efficiency. Smaller facilities with lower testing volumes may experience longer payback periods of 4-6 years, suggesting that adoption decisions should be volume-dependent.
Additional economic benefits include reduced waste management costs, as DMF-ELISA generates approximately 70% less biohazardous waste than traditional methods. Furthermore, the higher sensitivity of DMF-ELISA can reduce false negative/positive results, decreasing costly repeat testing and improving diagnostic accuracy, though these benefits are more difficult to quantify precisely in financial terms.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!