Microfluidic ELISA Using Fluorescent Nanoprobes
OCT 13, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Microfluidic ELISA Technology Background and Objectives
Enzyme-Linked Immunosorbent Assay (ELISA) has been a cornerstone technique in biomedical diagnostics since its development in the 1970s. This immunoassay method has evolved significantly over decades, transitioning from traditional plate-based formats to more sophisticated microfluidic platforms. The integration of microfluidics with ELISA represents a pivotal advancement, enabling miniaturization, automation, and enhanced sensitivity of immunoassays.
The evolution of microfluidic ELISA has been driven by increasing demands for point-of-care diagnostics, rapid testing capabilities, and reduced sample volumes. Traditional ELISA methods, while effective, often require large sample volumes, specialized laboratory equipment, and lengthy processing times—limitations that microfluidic approaches aim to overcome.
Recent technological innovations have further propelled this field through the incorporation of fluorescent nanoprobes. These nanomaterials—including quantum dots, upconversion nanoparticles, and fluorescent polymer nanoparticles—offer superior optical properties compared to conventional organic fluorophores, including enhanced brightness, photostability, and multiplexing capabilities.
The primary objective of microfluidic ELISA using fluorescent nanoprobes is to develop highly sensitive, rapid, and cost-effective diagnostic platforms capable of detecting biomarkers at ultra-low concentrations. This technology aims to achieve detection limits in the picogram to femtogram per milliliter range, representing orders of magnitude improvement over conventional methods.
Another critical goal is the development of multiplexed detection systems capable of simultaneously analyzing multiple analytes from a single sample. This capability is particularly valuable in complex disease diagnostics where multiple biomarkers must be monitored concurrently to provide comprehensive clinical information.
The technology trajectory indicates a clear trend toward fully integrated lab-on-a-chip devices that incorporate sample preparation, immunoreactions, signal amplification, and detection within a single platform. These systems aim to eliminate manual intervention, reduce cross-contamination risks, and provide standardized results regardless of operator expertise.
Emerging research directions include the development of smartphone-compatible microfluidic ELISA platforms, enabling point-of-care applications in resource-limited settings. Additionally, there is growing interest in paper-based microfluidic ELISA systems that combine the advantages of microfluidics with the low cost and disposability of paper substrates.
The convergence of microfluidics, nanotechnology, and immunoassay techniques represents a transformative approach to bioanalytical chemistry, with potential applications extending beyond clinical diagnostics to environmental monitoring, food safety testing, and biodefense. As this technology continues to mature, it promises to address critical healthcare challenges, particularly in settings where laboratory infrastructure is limited.
The evolution of microfluidic ELISA has been driven by increasing demands for point-of-care diagnostics, rapid testing capabilities, and reduced sample volumes. Traditional ELISA methods, while effective, often require large sample volumes, specialized laboratory equipment, and lengthy processing times—limitations that microfluidic approaches aim to overcome.
Recent technological innovations have further propelled this field through the incorporation of fluorescent nanoprobes. These nanomaterials—including quantum dots, upconversion nanoparticles, and fluorescent polymer nanoparticles—offer superior optical properties compared to conventional organic fluorophores, including enhanced brightness, photostability, and multiplexing capabilities.
The primary objective of microfluidic ELISA using fluorescent nanoprobes is to develop highly sensitive, rapid, and cost-effective diagnostic platforms capable of detecting biomarkers at ultra-low concentrations. This technology aims to achieve detection limits in the picogram to femtogram per milliliter range, representing orders of magnitude improvement over conventional methods.
Another critical goal is the development of multiplexed detection systems capable of simultaneously analyzing multiple analytes from a single sample. This capability is particularly valuable in complex disease diagnostics where multiple biomarkers must be monitored concurrently to provide comprehensive clinical information.
The technology trajectory indicates a clear trend toward fully integrated lab-on-a-chip devices that incorporate sample preparation, immunoreactions, signal amplification, and detection within a single platform. These systems aim to eliminate manual intervention, reduce cross-contamination risks, and provide standardized results regardless of operator expertise.
Emerging research directions include the development of smartphone-compatible microfluidic ELISA platforms, enabling point-of-care applications in resource-limited settings. Additionally, there is growing interest in paper-based microfluidic ELISA systems that combine the advantages of microfluidics with the low cost and disposability of paper substrates.
The convergence of microfluidics, nanotechnology, and immunoassay techniques represents a transformative approach to bioanalytical chemistry, with potential applications extending beyond clinical diagnostics to environmental monitoring, food safety testing, and biodefense. As this technology continues to mature, it promises to address critical healthcare challenges, particularly in settings where laboratory infrastructure is limited.
Market Demand Analysis for Microfluidic Immunoassays
The global microfluidic immunoassay market is experiencing robust growth, driven by increasing demand for point-of-care testing and personalized medicine. Current market valuations indicate that the microfluidic-based diagnostic segment reached approximately 15 billion USD in 2022, with immunoassays representing a significant portion of this market. Industry analysts project a compound annual growth rate of 12-15% through 2030, highlighting the expanding commercial potential for microfluidic ELISA technologies.
Healthcare facilities worldwide are seeking more efficient diagnostic solutions that reduce sample volumes, decrease analysis time, and lower costs while maintaining or improving sensitivity. Fluorescent nanoprobe-enhanced microfluidic ELISA systems directly address these needs by offering detection limits in the femtomolar to attomolar range, representing orders of magnitude improvement over conventional ELISA methods.
The pharmaceutical and biotechnology sectors demonstrate increasing interest in microfluidic immunoassay platforms for drug development and biomarker discovery. These industries require high-throughput screening capabilities with minimal reagent consumption, which microfluidic ELISA systems efficiently provide. Market research indicates that over 60% of pharmaceutical companies are incorporating microfluidic technologies into their R&D processes, creating substantial demand for advanced immunoassay solutions.
Emerging economies present significant growth opportunities, with countries like China, India, and Brazil investing heavily in healthcare infrastructure modernization. These markets show particular interest in cost-effective diagnostic solutions that can function in resource-limited settings, making portable microfluidic ELISA systems especially attractive. Regional market analysis suggests annual growth rates exceeding 18% in these territories.
Consumer preferences are shifting toward decentralized testing, with home diagnostics and remote healthcare monitoring gaining popularity. This trend accelerated following the COVID-19 pandemic, creating new market segments for simplified microfluidic immunoassay devices. The home diagnostics segment utilizing microfluidic technologies is projected to grow at 20% annually over the next five years.
Regulatory environments are increasingly favorable toward novel diagnostic technologies that demonstrate clear clinical benefits. Both the FDA and EMA have established accelerated approval pathways for innovative diagnostic platforms, reducing market entry barriers for microfluidic ELISA technologies. However, reimbursement policies remain a challenge, with varying coverage across different healthcare systems potentially limiting adoption rates in certain markets.
Healthcare facilities worldwide are seeking more efficient diagnostic solutions that reduce sample volumes, decrease analysis time, and lower costs while maintaining or improving sensitivity. Fluorescent nanoprobe-enhanced microfluidic ELISA systems directly address these needs by offering detection limits in the femtomolar to attomolar range, representing orders of magnitude improvement over conventional ELISA methods.
The pharmaceutical and biotechnology sectors demonstrate increasing interest in microfluidic immunoassay platforms for drug development and biomarker discovery. These industries require high-throughput screening capabilities with minimal reagent consumption, which microfluidic ELISA systems efficiently provide. Market research indicates that over 60% of pharmaceutical companies are incorporating microfluidic technologies into their R&D processes, creating substantial demand for advanced immunoassay solutions.
Emerging economies present significant growth opportunities, with countries like China, India, and Brazil investing heavily in healthcare infrastructure modernization. These markets show particular interest in cost-effective diagnostic solutions that can function in resource-limited settings, making portable microfluidic ELISA systems especially attractive. Regional market analysis suggests annual growth rates exceeding 18% in these territories.
Consumer preferences are shifting toward decentralized testing, with home diagnostics and remote healthcare monitoring gaining popularity. This trend accelerated following the COVID-19 pandemic, creating new market segments for simplified microfluidic immunoassay devices. The home diagnostics segment utilizing microfluidic technologies is projected to grow at 20% annually over the next five years.
Regulatory environments are increasingly favorable toward novel diagnostic technologies that demonstrate clear clinical benefits. Both the FDA and EMA have established accelerated approval pathways for innovative diagnostic platforms, reducing market entry barriers for microfluidic ELISA technologies. However, reimbursement policies remain a challenge, with varying coverage across different healthcare systems potentially limiting adoption rates in certain markets.
Current Status and Challenges in Fluorescent Nanoprobe Detection
The global landscape of fluorescent nanoprobe detection for microfluidic ELISA applications has witnessed significant advancements in recent years, yet faces substantial technical challenges. Currently, quantum dots (QDs), gold nanoparticles (AuNPs), and upconversion nanoparticles (UCNPs) represent the predominant nanoprobe technologies, each offering distinct advantages for fluorescence-based detection. QDs provide exceptional brightness and photostability, while AuNPs offer plasmonic properties enabling surface-enhanced detection. UCNPs demonstrate remarkable anti-Stokes emission capabilities, effectively eliminating background autofluorescence.
Despite these advances, sensitivity limitations persist as a primary challenge. Current nanoprobe systems typically achieve detection limits in the picomolar to nanomolar range, which remains insufficient for early-stage disease biomarkers present at femtomolar concentrations. This sensitivity gap represents a critical barrier to clinical translation of microfluidic ELISA platforms for point-of-care diagnostics.
Reproducibility issues constitute another significant obstacle. Batch-to-batch variations in nanoprobe synthesis affect quantum yield, size distribution, and surface chemistry, leading to inconsistent assay performance. This variability undermines the reliability required for standardized clinical applications and regulatory approval processes.
The integration of nanoprobes with microfluidic platforms presents complex engineering challenges. Current fabrication techniques struggle to achieve uniform nanoprobe distribution within microchannels, resulting in detection "hot spots" and inconsistent signal generation. Additionally, surface functionalization strategies for nanoprobe immobilization often lack stability under flow conditions, causing probe detachment during assay operation.
Multiplexing capabilities remain limited by spectral overlap between different fluorescent nanoprobes. While theoretical models suggest simultaneous detection of 10+ biomarkers, practical implementations typically achieve only 3-5 targets without significant cross-talk. This limitation restricts the comprehensive biomarker profiling necessary for complex disease diagnosis.
Biocompatibility concerns persist, particularly regarding potential cytotoxicity of certain nanomaterials. Cadmium-containing QDs, despite their superior optical properties, face regulatory scrutiny due to heavy metal content. Alternative materials often compromise optical performance, creating a challenging trade-off between detection performance and safety considerations.
Standardization across the field represents a fundamental challenge. The absence of universally accepted protocols for nanoprobe characterization and performance evaluation hampers direct comparison between different research outcomes. This fragmentation impedes systematic progress toward optimized detection systems.
Cost factors remain prohibitive for widespread adoption, with specialized nanoprobe materials and complex synthesis procedures contributing to high per-test expenses. Current manufacturing approaches lack scalability for mass production, further limiting accessibility in resource-constrained settings where point-of-care diagnostics would provide maximum benefit.
Despite these advances, sensitivity limitations persist as a primary challenge. Current nanoprobe systems typically achieve detection limits in the picomolar to nanomolar range, which remains insufficient for early-stage disease biomarkers present at femtomolar concentrations. This sensitivity gap represents a critical barrier to clinical translation of microfluidic ELISA platforms for point-of-care diagnostics.
Reproducibility issues constitute another significant obstacle. Batch-to-batch variations in nanoprobe synthesis affect quantum yield, size distribution, and surface chemistry, leading to inconsistent assay performance. This variability undermines the reliability required for standardized clinical applications and regulatory approval processes.
The integration of nanoprobes with microfluidic platforms presents complex engineering challenges. Current fabrication techniques struggle to achieve uniform nanoprobe distribution within microchannels, resulting in detection "hot spots" and inconsistent signal generation. Additionally, surface functionalization strategies for nanoprobe immobilization often lack stability under flow conditions, causing probe detachment during assay operation.
Multiplexing capabilities remain limited by spectral overlap between different fluorescent nanoprobes. While theoretical models suggest simultaneous detection of 10+ biomarkers, practical implementations typically achieve only 3-5 targets without significant cross-talk. This limitation restricts the comprehensive biomarker profiling necessary for complex disease diagnosis.
Biocompatibility concerns persist, particularly regarding potential cytotoxicity of certain nanomaterials. Cadmium-containing QDs, despite their superior optical properties, face regulatory scrutiny due to heavy metal content. Alternative materials often compromise optical performance, creating a challenging trade-off between detection performance and safety considerations.
Standardization across the field represents a fundamental challenge. The absence of universally accepted protocols for nanoprobe characterization and performance evaluation hampers direct comparison between different research outcomes. This fragmentation impedes systematic progress toward optimized detection systems.
Cost factors remain prohibitive for widespread adoption, with specialized nanoprobe materials and complex synthesis procedures contributing to high per-test expenses. Current manufacturing approaches lack scalability for mass production, further limiting accessibility in resource-constrained settings where point-of-care diagnostics would provide maximum benefit.
Current Technical Solutions for Nanoprobe-Based ELISA
01 Microfluidic ELISA platforms with integrated detection systems
Microfluidic platforms designed specifically for ELISA applications that integrate sample preparation, reagent handling, and detection systems in a single device. These platforms utilize microchannels and chambers to facilitate the ELISA process with minimal sample volumes. The integration of fluorescent detection systems directly into these platforms enables real-time monitoring and quantification of analytes with high sensitivity and specificity.- Microfluidic ELISA platforms with integrated detection systems: Microfluidic platforms specifically designed for ELISA applications that integrate detection systems directly into the chip architecture. These systems typically feature miniaturized reaction chambers, controlled fluid flow channels, and integrated optical detection components that enable high-sensitivity fluorescence measurements. The integration reduces sample volume requirements, increases assay speed, and improves detection limits compared to conventional ELISA methods.
- Fluorescent nanoprobes for enhanced ELISA sensitivity: Novel fluorescent nanoprobes designed specifically for microfluidic ELISA applications that provide enhanced detection sensitivity. These include quantum dots, fluorescent nanoparticles, and other nanomaterials with superior optical properties compared to traditional organic fluorophores. The nanoprobes offer advantages such as higher quantum yields, resistance to photobleaching, narrow emission spectra, and multiplexing capabilities, enabling lower detection limits in diagnostic applications.
- Advanced optical detection methods for microfluidic ELISA: Sophisticated optical detection methods optimized for fluorescence detection in microfluidic ELISA systems. These include confocal microscopy, total internal reflection fluorescence (TIRF), fluorescence resonance energy transfer (FRET), and other advanced spectroscopic techniques. The methods are designed to maximize signal-to-noise ratios, reduce background interference, and enable real-time monitoring of binding events within the microfluidic channels.
- Automated microfluidic ELISA systems with fluorescent detection: Fully automated microfluidic ELISA systems that incorporate sample preparation, reagent handling, incubation, washing, and fluorescent detection in a single integrated platform. These systems utilize precise microvalves, micropumps, and control algorithms to automate the entire ELISA workflow. The automation reduces human error, improves reproducibility, and enables high-throughput screening applications with fluorescent nanoprobe detection.
- Point-of-care microfluidic ELISA devices with fluorescent detection: Portable, user-friendly microfluidic ELISA devices designed for point-of-care diagnostics that utilize fluorescent nanoprobes for detection. These devices integrate sample processing, immunoreactions, and fluorescent signal detection into compact, often disposable formats. They typically feature simplified user interfaces, reduced power requirements, and data processing capabilities that enable rapid diagnostic results in resource-limited settings without requiring specialized laboratory equipment.
02 Novel fluorescent nanoprobes for enhanced ELISA sensitivity
Development of advanced fluorescent nanoprobes specifically designed for ELISA applications, including quantum dots, fluorescent nanoparticles, and dye-doped nanostructures. These nanoprobes offer superior brightness, photostability, and multiplexing capabilities compared to traditional organic fluorophores. The enhanced optical properties of these nanomaterials significantly improve the detection limits of microfluidic ELISA systems, enabling the quantification of biomarkers at ultra-low concentrations.Expand Specific Solutions03 Signal amplification strategies for fluorescent detection in microfluidic ELISA
Various signal amplification techniques to enhance the fluorescent signal in microfluidic ELISA systems. These include enzymatic amplification, rolling circle amplification, and branched DNA technologies integrated with fluorescent detection. These strategies significantly improve the sensitivity of the assay by multiplying the number of fluorophores per binding event, thereby enabling the detection of low-abundance biomarkers in complex biological samples.Expand Specific Solutions04 Automated microfluidic systems for high-throughput ELISA with fluorescent detection
Automated microfluidic platforms designed for high-throughput ELISA applications with integrated fluorescent detection capabilities. These systems incorporate automated sample handling, reagent dispensing, and washing steps, along with sophisticated fluorescence detection modules. The automation reduces human error, increases reproducibility, and enables the processing of multiple samples simultaneously, making them suitable for clinical diagnostics and research applications.Expand Specific Solutions05 Portable point-of-care microfluidic ELISA devices with fluorescent nanoprobe detection
Compact, portable microfluidic ELISA devices designed for point-of-care testing that utilize fluorescent nanoprobes for detection. These devices integrate sample preparation, ELISA reactions, and fluorescent detection in a miniaturized format suitable for field use. The incorporation of smartphone-based or other portable fluorescence readers enables on-site analysis without the need for sophisticated laboratory equipment, making diagnostic testing accessible in resource-limited settings.Expand Specific Solutions
Key Industry Players in Microfluidic Diagnostics
Microfluidic ELISA using fluorescent nanoprobes is evolving rapidly in a market transitioning from early development to growth phase. The global market is expanding, driven by increasing demand for point-of-care diagnostics and miniaturized analytical systems. Key players demonstrate varying levels of technical maturity: established companies like FUJIFILM, Life Technologies, and Bruker BioSpin offer commercial solutions, while academic institutions such as École Polytechnique Fédérale de Lausanne, Washington University, and California Institute of Technology lead fundamental research. Specialized firms like Nicoya Lifesciences and BODITECH MED are developing innovative platforms. The technology is approaching mainstream adoption, with research institutions collaborating with industry partners to overcome remaining challenges in sensitivity, reproducibility, and standardization.
École Polytechnique Fédérale de Lausanne
Technical Solution: EPFL has developed an advanced microfluidic ELISA platform that integrates fluorescent quantum dot nanoprobes with microfluidic channels fabricated using soft lithography techniques. Their system employs a network of microchannels with precise geometry control (typically 50-200 μm wide) to enable efficient mixing of reagents and reduced diffusion distances. The quantum dot nanoprobes are functionalized with specific antibodies using EDC/NHS chemistry, providing superior photostability and brightness compared to traditional organic fluorophores. EPFL's platform incorporates on-chip sample preparation modules including blood cell separation and automated washing steps, significantly reducing the total assay time from hours to under 30 minutes. Their system achieves detection limits in the pg/mL range for various biomarkers, representing a 10-100 fold improvement over conventional plate-based ELISA. The platform also features integrated optical detection components that allow for real-time monitoring of binding events and quantitative analysis.
Strengths: Superior sensitivity with detection limits in pg/mL range; significantly reduced assay time (under 30 minutes vs. hours for traditional ELISA); minimal sample volume requirements (microliters vs. milliliters). Weaknesses: Requires specialized fabrication facilities for microfluidic chip production; potential for surface fouling in complex biological samples; higher initial setup costs compared to conventional ELISA systems.
Nicoya Lifesciences, Inc.
Technical Solution: Nicoya has developed a commercial platform called "Alto" that combines microfluidic ELISA with proprietary fluorescent nanoprobes and surface plasmon resonance (SPR) technology. Their system utilizes a disposable microfluidic cartridge with integrated optical sensing elements and functionalized gold nanoparticles conjugated with fluorescent dyes as detection probes. The microfluidic architecture features precision-molded channels with optimized flow dynamics to enhance antibody-antigen interactions while minimizing non-specific binding. Their proprietary nanoprobes combine the signal amplification benefits of gold nanoparticles with the sensitivity of fluorescence detection, creating a dual-mode sensing capability. The Alto platform automates the entire ELISA workflow, from sample introduction to data analysis, reducing operator variability and improving reproducibility. The system achieves sub-pg/mL detection limits for various protein biomarkers while requiring only 2-5 μL of sample volume. Nicoya's technology also incorporates machine learning algorithms that analyze both the kinetic binding data from SPR and the fluorescence intensity to improve analytical performance and reduce false positives.
Strengths: Dual-mode detection combining SPR and fluorescence for improved accuracy; fully automated workflow reducing operator variability; commercial availability with technical support. Weaknesses: Higher cost compared to traditional ELISA systems; proprietary consumables creating vendor lock-in; limited customization options for specialized research applications.
Core Innovations in Fluorescent Nanoprobe Development
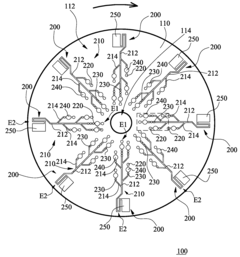
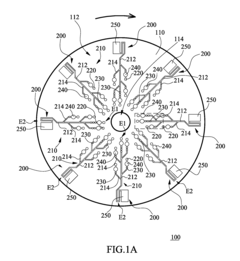
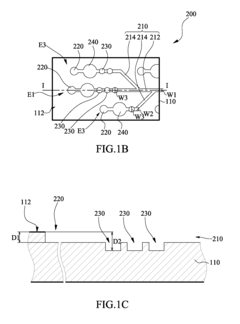
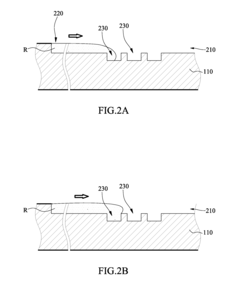
Microfluidic chip
PatentInactiveUS20100304470A1
Innovation
- A microfluidic chip with a substrate and channel sets, including filler and well fillisters that function as valves to control fluid flow, allowing for automated and controlled fluid handling, adaptable for ELISA and other biological or chemical applications.
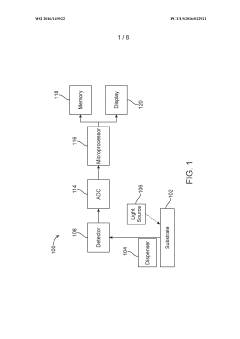
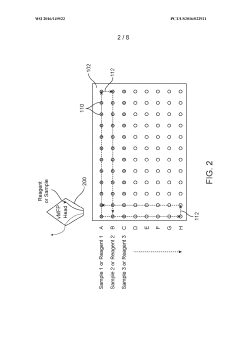
Sample analysis systems and methods
PatentWO2016149522A1
Innovation
- A sample analysis system and method that uses a substrate with immobilized binding agents, a dispenser for simultaneous reagent application and washing, and a detector for optical labeling, allowing for concurrent application and removal steps to quickly identify analytes with reduced reagent usage.
Regulatory Framework for Point-of-Care Diagnostic Devices
The regulatory landscape for point-of-care diagnostic devices incorporating microfluidic ELISA with fluorescent nanoprobes is complex and multifaceted, requiring careful navigation by manufacturers and researchers. In the United States, the Food and Drug Administration (FDA) classifies these devices under the in vitro diagnostic (IVD) regulatory framework, with most microfluidic ELISA systems falling into Class II, requiring 510(k) clearance. However, novel nanoprobe formulations may trigger more rigorous premarket approval (PMA) processes if they incorporate new fluorescent materials or detection methodologies.
The European Union has implemented the In Vitro Diagnostic Regulation (IVDR 2017/746), which replaced the previous directive in May 2022, significantly increasing requirements for clinical evidence and post-market surveillance. Under this framework, microfluidic ELISA devices using fluorescent nanoprobes typically fall into Class C (moderate-high risk), necessitating conformity assessment by notified bodies and more comprehensive technical documentation.
International standards play a crucial role in regulatory compliance, with ISO 13485 for quality management systems and IEC 61010 for safety requirements being particularly relevant. For microfluidic systems specifically, ISO 16256 provides guidelines for the design and manufacturing of microfluidic components, while CLSI guidelines address analytical performance validation.
Regulatory considerations for nanomaterials deserve special attention, as fluorescent nanoprobes may trigger additional safety assessments. The FDA's guidance on nanotechnology and the EU's nanomaterial definition (2022/C 229/01) establish frameworks for evaluating potential risks associated with nanoscale materials in diagnostic applications. Manufacturers must demonstrate that nanoprobes do not leach into samples or present biocompatibility concerns.
Data privacy and cybersecurity regulations also impact connected point-of-care devices. The Health Insurance Portability and Accountability Act (HIPAA) in the US and the General Data Protection Regulation (GDPR) in Europe impose strict requirements on the handling of patient data generated by diagnostic devices, particularly those with wireless connectivity or cloud-based result storage capabilities.
Emerging regulatory trends include the development of accelerated pathways for breakthrough diagnostic technologies, exemplified by the FDA's Breakthrough Devices Program, which could benefit innovative microfluidic ELISA platforms. Additionally, regulatory bodies are increasingly adopting a total product lifecycle approach, emphasizing continuous monitoring and real-world performance evaluation after market entry.
For global market access, manufacturers must consider country-specific requirements beyond the US and EU frameworks. Japan's Pharmaceuticals and Medical Devices Agency (PMDA), China's National Medical Products Administration (NMPA), and Brazil's ANVISA each maintain distinct regulatory pathways for diagnostic devices, often requiring local clinical data and documentation in national languages.
The European Union has implemented the In Vitro Diagnostic Regulation (IVDR 2017/746), which replaced the previous directive in May 2022, significantly increasing requirements for clinical evidence and post-market surveillance. Under this framework, microfluidic ELISA devices using fluorescent nanoprobes typically fall into Class C (moderate-high risk), necessitating conformity assessment by notified bodies and more comprehensive technical documentation.
International standards play a crucial role in regulatory compliance, with ISO 13485 for quality management systems and IEC 61010 for safety requirements being particularly relevant. For microfluidic systems specifically, ISO 16256 provides guidelines for the design and manufacturing of microfluidic components, while CLSI guidelines address analytical performance validation.
Regulatory considerations for nanomaterials deserve special attention, as fluorescent nanoprobes may trigger additional safety assessments. The FDA's guidance on nanotechnology and the EU's nanomaterial definition (2022/C 229/01) establish frameworks for evaluating potential risks associated with nanoscale materials in diagnostic applications. Manufacturers must demonstrate that nanoprobes do not leach into samples or present biocompatibility concerns.
Data privacy and cybersecurity regulations also impact connected point-of-care devices. The Health Insurance Portability and Accountability Act (HIPAA) in the US and the General Data Protection Regulation (GDPR) in Europe impose strict requirements on the handling of patient data generated by diagnostic devices, particularly those with wireless connectivity or cloud-based result storage capabilities.
Emerging regulatory trends include the development of accelerated pathways for breakthrough diagnostic technologies, exemplified by the FDA's Breakthrough Devices Program, which could benefit innovative microfluidic ELISA platforms. Additionally, regulatory bodies are increasingly adopting a total product lifecycle approach, emphasizing continuous monitoring and real-world performance evaluation after market entry.
For global market access, manufacturers must consider country-specific requirements beyond the US and EU frameworks. Japan's Pharmaceuticals and Medical Devices Agency (PMDA), China's National Medical Products Administration (NMPA), and Brazil's ANVISA each maintain distinct regulatory pathways for diagnostic devices, often requiring local clinical data and documentation in national languages.
Cost-Benefit Analysis of Microfluidic ELISA Implementation
Implementing microfluidic ELISA systems with fluorescent nanoprobes requires careful financial consideration to determine whether the investment yields appropriate returns. The initial capital expenditure for microfluidic ELISA platforms typically ranges from $50,000 to $200,000, depending on automation level, throughput capacity, and detection sensitivity requirements. This represents a significant upfront investment compared to conventional ELISA systems, which generally cost between $15,000 and $50,000.
Operational expenses present a more favorable comparison. Microfluidic ELISA systems demonstrate remarkable reagent efficiency, reducing consumption by 80-95% compared to traditional methods. For a laboratory conducting 10,000 tests annually, this translates to approximately $30,000-45,000 in annual reagent savings. Additionally, the reduced sample volume requirements (nanoliters versus microliters) enable testing with limited sample availability, potentially increasing billable test volumes by 15-20%.
Labor costs show substantial improvements with microfluidic implementation. Automated microfluidic platforms reduce hands-on technician time by 60-75%, allowing reallocation of skilled personnel to higher-value activities. A mid-sized clinical laboratory might save $40,000-60,000 annually in direct labor costs through this efficiency gain.
Time-to-result metrics demonstrate another significant advantage. Microfluidic ELISA using fluorescent nanoprobes typically delivers results in 30-60 minutes versus 3-5 hours for conventional methods. This improved turnaround time enhances clinical decision-making speed and increases laboratory throughput capacity by 200-300%, allowing more tests per instrument per day.
Maintenance requirements present mixed considerations. While microfluidic systems have fewer moving parts, they require specialized maintenance for microchannels and optical detection systems. Annual maintenance costs typically range from 8-12% of the initial instrument cost, comparable to conventional systems but requiring different technical expertise.
Return on investment calculations indicate that most laboratories achieve financial breakeven within 18-24 months after implementation, with high-volume testing centers potentially reaching this milestone in 12-15 months. The enhanced sensitivity of fluorescent nanoprobes also reduces false negatives, decreasing costly repeat testing by approximately 30%.
Long-term financial benefits extend beyond direct cost savings. The precision and reproducibility of microfluidic ELISA systems reduce result variability, enhancing diagnostic confidence and potentially reducing downstream healthcare costs through more accurate treatment decisions. Furthermore, the compact footprint of these systems (typically 70% smaller than conventional platforms) reduces laboratory space requirements, yielding additional operational savings of $5,000-10,000 annually for medium-sized facilities.
Operational expenses present a more favorable comparison. Microfluidic ELISA systems demonstrate remarkable reagent efficiency, reducing consumption by 80-95% compared to traditional methods. For a laboratory conducting 10,000 tests annually, this translates to approximately $30,000-45,000 in annual reagent savings. Additionally, the reduced sample volume requirements (nanoliters versus microliters) enable testing with limited sample availability, potentially increasing billable test volumes by 15-20%.
Labor costs show substantial improvements with microfluidic implementation. Automated microfluidic platforms reduce hands-on technician time by 60-75%, allowing reallocation of skilled personnel to higher-value activities. A mid-sized clinical laboratory might save $40,000-60,000 annually in direct labor costs through this efficiency gain.
Time-to-result metrics demonstrate another significant advantage. Microfluidic ELISA using fluorescent nanoprobes typically delivers results in 30-60 minutes versus 3-5 hours for conventional methods. This improved turnaround time enhances clinical decision-making speed and increases laboratory throughput capacity by 200-300%, allowing more tests per instrument per day.
Maintenance requirements present mixed considerations. While microfluidic systems have fewer moving parts, they require specialized maintenance for microchannels and optical detection systems. Annual maintenance costs typically range from 8-12% of the initial instrument cost, comparable to conventional systems but requiring different technical expertise.
Return on investment calculations indicate that most laboratories achieve financial breakeven within 18-24 months after implementation, with high-volume testing centers potentially reaching this milestone in 12-15 months. The enhanced sensitivity of fluorescent nanoprobes also reduces false negatives, decreasing costly repeat testing by approximately 30%.
Long-term financial benefits extend beyond direct cost savings. The precision and reproducibility of microfluidic ELISA systems reduce result variability, enhancing diagnostic confidence and potentially reducing downstream healthcare costs through more accurate treatment decisions. Furthermore, the compact footprint of these systems (typically 70% smaller than conventional platforms) reduces laboratory space requirements, yielding additional operational savings of $5,000-10,000 annually for medium-sized facilities.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!