Purity Assurance: Analytical Methods For Trace Impurities (HPLC, GC, IC)
AUG 22, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Analytical Methods Evolution and Objectives
The evolution of analytical methods for trace impurity detection has undergone significant transformation over the past several decades, driven by increasing regulatory requirements and the growing complexity of pharmaceutical, chemical, and food products. Initially, simple colorimetric and gravimetric methods dominated the field, offering limited sensitivity and specificity. The 1970s marked a turning point with the commercial introduction of High-Performance Liquid Chromatography (HPLC), which revolutionized the ability to detect and quantify trace impurities at parts-per-million levels.
Gas Chromatography (GC) emerged as another powerful technique, particularly suited for volatile compounds, with developments in column technology and detector sensitivity enabling parts-per-billion detection by the 1980s. Ion Chromatography (IC) subsequently evolved to address the specific challenges of ionic impurity detection, filling a critical gap in analytical capabilities.
The 1990s and early 2000s witnessed the integration of mass spectrometry with these chromatographic techniques, dramatically enhancing both identification capabilities and detection limits. Modern HPLC-MS/MS systems can now routinely achieve parts-per-trillion detection, representing a million-fold improvement over early analytical methods.
Recent technological advancements have focused on miniaturization, automation, and high-throughput capabilities. Ultra-High Performance Liquid Chromatography (UHPLC) has reduced analysis times from hours to minutes while improving resolution. Simultaneously, advances in column chemistry, particularly the development of core-shell particles and monolithic columns, have enhanced separation efficiency and selectivity.
The primary objectives of contemporary analytical method development for trace impurities include achieving lower detection limits to meet increasingly stringent regulatory requirements, particularly in pharmaceutical applications where the International Council for Harmonisation (ICH) guidelines mandate identification and quantification of impurities at 0.05% levels or lower.
Enhancing method specificity represents another critical objective, as modern complex matrices often contain numerous potential interferents that can compromise accurate impurity identification. Speed and throughput optimization has become increasingly important in industrial settings, where rapid quality control decisions impact production efficiency.
Method robustness and transferability across different laboratories and instruments remains a significant challenge, particularly for global organizations operating across multiple facilities. The industry is also moving toward green analytical chemistry, with objectives to reduce solvent consumption, minimize waste generation, and develop more environmentally sustainable analytical approaches.
Looking forward, the field aims to develop fully automated, integrated analytical platforms capable of real-time monitoring and adaptive method optimization, potentially leveraging artificial intelligence to interpret complex chromatographic data and identify unknown impurities with minimal human intervention.
Gas Chromatography (GC) emerged as another powerful technique, particularly suited for volatile compounds, with developments in column technology and detector sensitivity enabling parts-per-billion detection by the 1980s. Ion Chromatography (IC) subsequently evolved to address the specific challenges of ionic impurity detection, filling a critical gap in analytical capabilities.
The 1990s and early 2000s witnessed the integration of mass spectrometry with these chromatographic techniques, dramatically enhancing both identification capabilities and detection limits. Modern HPLC-MS/MS systems can now routinely achieve parts-per-trillion detection, representing a million-fold improvement over early analytical methods.
Recent technological advancements have focused on miniaturization, automation, and high-throughput capabilities. Ultra-High Performance Liquid Chromatography (UHPLC) has reduced analysis times from hours to minutes while improving resolution. Simultaneously, advances in column chemistry, particularly the development of core-shell particles and monolithic columns, have enhanced separation efficiency and selectivity.
The primary objectives of contemporary analytical method development for trace impurities include achieving lower detection limits to meet increasingly stringent regulatory requirements, particularly in pharmaceutical applications where the International Council for Harmonisation (ICH) guidelines mandate identification and quantification of impurities at 0.05% levels or lower.
Enhancing method specificity represents another critical objective, as modern complex matrices often contain numerous potential interferents that can compromise accurate impurity identification. Speed and throughput optimization has become increasingly important in industrial settings, where rapid quality control decisions impact production efficiency.
Method robustness and transferability across different laboratories and instruments remains a significant challenge, particularly for global organizations operating across multiple facilities. The industry is also moving toward green analytical chemistry, with objectives to reduce solvent consumption, minimize waste generation, and develop more environmentally sustainable analytical approaches.
Looking forward, the field aims to develop fully automated, integrated analytical platforms capable of real-time monitoring and adaptive method optimization, potentially leveraging artificial intelligence to interpret complex chromatographic data and identify unknown impurities with minimal human intervention.
Market Demand for High-Purity Analysis
The global market for high-purity analytical methods has experienced significant growth in recent years, driven primarily by increasing demands in pharmaceutical, biotechnology, and semiconductor industries. These sectors require stringent quality control measures to ensure product safety, efficacy, and performance. The pharmaceutical industry alone represents approximately 40% of the total market demand for high-purity analysis, with annual growth rates consistently exceeding general market trends.
Healthcare regulations worldwide have become increasingly stringent, particularly regarding impurity detection and quantification in drug substances and products. The FDA, EMA, and other regulatory bodies have established lower acceptable limits for impurities, creating substantial demand for more sensitive and accurate analytical methods. This regulatory pressure has transformed high-purity analysis from a competitive advantage to an essential compliance requirement.
The semiconductor industry presents another major market driver, where even trace impurities can significantly impact device performance. As chip architectures continue to shrink, the tolerance for contaminants has decreased proportionally, necessitating more sophisticated analytical capabilities. Market research indicates that semiconductor manufacturers are willing to invest substantially in advanced analytical technologies that can detect impurities at parts-per-trillion levels.
Environmental monitoring represents an emerging market segment with considerable growth potential. Increasing public awareness and regulatory focus on water quality, air pollution, and soil contamination have expanded the application scope for high-precision analytical methods. Government initiatives worldwide are allocating significant funding for environmental monitoring programs, creating new market opportunities.
Contract research organizations (CROs) and analytical testing laboratories have reported increased client requests for high-sensitivity impurity analysis, with HPLC and GC-MS services showing the highest demand growth. These organizations are investing in equipment upgrades to meet client specifications for lower detection limits and greater analytical precision.
Regional analysis reveals that North America and Europe currently dominate the market for high-purity analytical services and equipment, though Asia-Pacific shows the fastest growth rate. China and India are rapidly expanding their pharmaceutical and electronics manufacturing capabilities, creating substantial new demand for advanced analytical technologies and expertise.
Consumer preferences are increasingly influencing market dynamics, with growing demand for "clean" products across food, cosmetics, and personal care sectors. This trend has expanded the market for impurity analysis beyond traditional industrial applications into consumer goods manufacturing, creating diverse new revenue streams for analytical service providers.
Healthcare regulations worldwide have become increasingly stringent, particularly regarding impurity detection and quantification in drug substances and products. The FDA, EMA, and other regulatory bodies have established lower acceptable limits for impurities, creating substantial demand for more sensitive and accurate analytical methods. This regulatory pressure has transformed high-purity analysis from a competitive advantage to an essential compliance requirement.
The semiconductor industry presents another major market driver, where even trace impurities can significantly impact device performance. As chip architectures continue to shrink, the tolerance for contaminants has decreased proportionally, necessitating more sophisticated analytical capabilities. Market research indicates that semiconductor manufacturers are willing to invest substantially in advanced analytical technologies that can detect impurities at parts-per-trillion levels.
Environmental monitoring represents an emerging market segment with considerable growth potential. Increasing public awareness and regulatory focus on water quality, air pollution, and soil contamination have expanded the application scope for high-precision analytical methods. Government initiatives worldwide are allocating significant funding for environmental monitoring programs, creating new market opportunities.
Contract research organizations (CROs) and analytical testing laboratories have reported increased client requests for high-sensitivity impurity analysis, with HPLC and GC-MS services showing the highest demand growth. These organizations are investing in equipment upgrades to meet client specifications for lower detection limits and greater analytical precision.
Regional analysis reveals that North America and Europe currently dominate the market for high-purity analytical services and equipment, though Asia-Pacific shows the fastest growth rate. China and India are rapidly expanding their pharmaceutical and electronics manufacturing capabilities, creating substantial new demand for advanced analytical technologies and expertise.
Consumer preferences are increasingly influencing market dynamics, with growing demand for "clean" products across food, cosmetics, and personal care sectors. This trend has expanded the market for impurity analysis beyond traditional industrial applications into consumer goods manufacturing, creating diverse new revenue streams for analytical service providers.
Current Challenges in Trace Impurity Detection
Despite significant advancements in analytical technologies, trace impurity detection continues to face substantial challenges that limit the effectiveness and reliability of current methodologies. One of the primary obstacles is the detection of ultra-low concentration impurities, particularly in complex matrices. As regulatory requirements become increasingly stringent, analytical methods struggle to achieve the necessary sensitivity while maintaining specificity, especially when target analytes exist at parts-per-billion or parts-per-trillion levels.
Instrument sensitivity limitations represent another significant hurdle. While modern HPLC, GC, and IC systems have improved detection capabilities, they still encounter fundamental physical and chemical constraints when analyzing trace impurities. Signal-to-noise ratio degradation becomes particularly problematic at extremely low concentrations, making it difficult to distinguish genuine impurity signals from background noise or system artifacts.
Matrix interference effects continue to plague analytical scientists, as sample components can mask, enhance, or suppress impurity signals. This is especially challenging in biological samples, natural products, and complex pharmaceutical formulations where numerous compounds may co-elute or interact with target analytes. These interferences often necessitate extensive sample preparation procedures that can introduce additional variables and potential sources of error.
Method validation for trace impurity analysis presents unique difficulties, particularly regarding reproducibility and robustness. Establishing consistent performance across different instruments, operators, and laboratories becomes exponentially more challenging at trace levels. Minor variations in experimental conditions that would be inconsequential for major component analysis can significantly impact trace impurity detection.
Sample preparation techniques also face limitations when dealing with trace impurities. Conventional extraction, concentration, and clean-up methods may introduce contaminants or result in analyte loss, compromising the integrity of the analysis. Additionally, the risk of sample contamination increases dramatically when working at trace levels, requiring specialized facilities and handling protocols that may not be universally available.
Data interpretation challenges further complicate trace impurity analysis. Distinguishing between actual impurities and artifacts generated during sample processing or analysis requires sophisticated data processing algorithms and experienced analysts. The increasing complexity of analytical data sets, particularly from hyphenated techniques like LC-MS/MS or GC-MS/MS, demands advanced chemometric approaches and machine learning tools that are still evolving.
Regulatory compliance adds another layer of complexity, as different regions and industries may have varying requirements for impurity profiling and reporting. Harmonizing analytical approaches to meet global standards while maintaining scientific rigor presents ongoing challenges for analytical scientists and quality assurance professionals.
Instrument sensitivity limitations represent another significant hurdle. While modern HPLC, GC, and IC systems have improved detection capabilities, they still encounter fundamental physical and chemical constraints when analyzing trace impurities. Signal-to-noise ratio degradation becomes particularly problematic at extremely low concentrations, making it difficult to distinguish genuine impurity signals from background noise or system artifacts.
Matrix interference effects continue to plague analytical scientists, as sample components can mask, enhance, or suppress impurity signals. This is especially challenging in biological samples, natural products, and complex pharmaceutical formulations where numerous compounds may co-elute or interact with target analytes. These interferences often necessitate extensive sample preparation procedures that can introduce additional variables and potential sources of error.
Method validation for trace impurity analysis presents unique difficulties, particularly regarding reproducibility and robustness. Establishing consistent performance across different instruments, operators, and laboratories becomes exponentially more challenging at trace levels. Minor variations in experimental conditions that would be inconsequential for major component analysis can significantly impact trace impurity detection.
Sample preparation techniques also face limitations when dealing with trace impurities. Conventional extraction, concentration, and clean-up methods may introduce contaminants or result in analyte loss, compromising the integrity of the analysis. Additionally, the risk of sample contamination increases dramatically when working at trace levels, requiring specialized facilities and handling protocols that may not be universally available.
Data interpretation challenges further complicate trace impurity analysis. Distinguishing between actual impurities and artifacts generated during sample processing or analysis requires sophisticated data processing algorithms and experienced analysts. The increasing complexity of analytical data sets, particularly from hyphenated techniques like LC-MS/MS or GC-MS/MS, demands advanced chemometric approaches and machine learning tools that are still evolving.
Regulatory compliance adds another layer of complexity, as different regions and industries may have varying requirements for impurity profiling and reporting. Harmonizing analytical approaches to meet global standards while maintaining scientific rigor presents ongoing challenges for analytical scientists and quality assurance professionals.
State-of-the-Art HPLC, GC, and IC Techniques
01 HPLC methods for trace impurity detection
High-Performance Liquid Chromatography (HPLC) is widely used for detecting and quantifying trace impurities in various samples. This analytical technique offers high sensitivity and selectivity, making it suitable for identifying low-level contaminants. HPLC methods can be optimized with different detectors such as UV-Vis, fluorescence, or mass spectrometry to enhance detection capabilities for specific trace impurities. These methods are particularly valuable in pharmaceutical analysis, environmental monitoring, and food safety testing.- HPLC methods for trace impurity analysis: High-Performance Liquid Chromatography (HPLC) is widely used for detecting and quantifying trace impurities in various samples. This technique offers high sensitivity and selectivity, making it suitable for analyzing complex matrices. Advanced HPLC methods incorporate specialized columns, optimized mobile phases, and detection systems such as UV-Vis, fluorescence, or mass spectrometry to achieve lower detection limits for trace impurities. These methods are particularly valuable in pharmaceutical, environmental, and food safety applications where impurity profiling is critical.
- Gas chromatography techniques for volatile impurities: Gas Chromatography (GC) is an essential analytical method for detecting volatile and semi-volatile trace impurities. This technique separates compounds based on their volatility and interaction with the stationary phase. GC methods often employ specialized detectors such as flame ionization detector (FID), electron capture detector (ECD), or mass spectrometry (MS) to achieve high sensitivity for trace analysis. These techniques are particularly valuable for analyzing residual solvents, volatile organic compounds, and other trace contaminants in various matrices including pharmaceuticals, environmental samples, and industrial products.
- Ion chromatography for ionic impurity detection: Ion Chromatography (IC) is specifically designed for the analysis of ionic species and trace ionic impurities in various samples. This technique separates anions and cations based on their interactions with ion exchange resins. IC methods typically employ conductivity detection, which can be enhanced with suppression techniques to improve sensitivity. This analytical approach is particularly valuable for detecting inorganic anions, organic acids, amines, and metal ions at trace levels in pharmaceuticals, water samples, and food products where ionic impurities can significantly impact quality and safety.
- Sample preparation techniques for trace analysis: Effective sample preparation is crucial for successful trace impurity analysis. Various techniques such as solid-phase extraction (SPE), liquid-liquid extraction (LLE), solid-phase microextraction (SPME), and QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) are employed to isolate and concentrate trace impurities from complex matrices. These preparation methods help eliminate matrix interferences, enhance sensitivity, and improve the reliability of analytical results. Advanced sample preparation approaches often combine multiple techniques to achieve optimal extraction efficiency and selectivity for specific trace impurities.
- Hyphenated techniques for enhanced trace impurity detection: Hyphenated analytical techniques combine two or more analytical methods to enhance the detection and characterization of trace impurities. Common combinations include LC-MS/MS, GC-MS, LC-NMR, and IC-MS, which provide both separation and identification capabilities. These integrated approaches offer improved selectivity, sensitivity, and structural information for trace impurities. Hyphenated techniques are particularly valuable for complex samples where conventional single methods may not provide sufficient resolution or identification power, enabling comprehensive impurity profiling at trace levels in pharmaceuticals, environmental samples, and food products.
02 Gas Chromatography techniques for volatile impurities
Gas Chromatography (GC) is an effective analytical method for separating and analyzing volatile and semi-volatile trace impurities. This technique is particularly useful for organic compounds and can be coupled with various detectors such as flame ionization detector (FID), electron capture detector (ECD), or mass spectrometry (MS) to enhance sensitivity and specificity. GC methods are commonly applied in environmental analysis, petrochemical industry, and forensic science to detect trace levels of contaminants that can be vaporized without decomposition.Expand Specific Solutions03 Ion Chromatography for ionic impurity analysis
Ion Chromatography (IC) is specialized for the detection and quantification of ionic trace impurities in various matrices. This technique is particularly effective for analyzing anions, cations, and polar molecules at trace levels. IC offers high selectivity and sensitivity for ionic species that might be difficult to detect using other chromatographic methods. Applications include water quality testing, pharmaceutical analysis, and food safety, where ionic contaminants need to be monitored at very low concentrations.Expand Specific Solutions04 Sample preparation techniques for trace analysis
Effective sample preparation is crucial for accurate trace impurity analysis. Techniques such as solid-phase extraction (SPE), liquid-liquid extraction, and microextraction methods help concentrate analytes and remove matrix interferences before chromatographic analysis. Advanced sample preparation approaches like QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) have been developed specifically for trace analysis applications. These preparation methods significantly improve detection limits and analytical precision when working with complex matrices containing trace impurities.Expand Specific Solutions05 Combined analytical approaches for comprehensive impurity profiling
Combining multiple analytical techniques provides comprehensive characterization of trace impurities in complex samples. Hyphenated techniques such as LC-MS, GC-MS, and IC-MS offer enhanced selectivity and sensitivity by leveraging the strengths of each method. Multi-dimensional chromatography approaches like 2D-LC and GC×GC provide improved separation power for complex mixtures containing trace impurities. These combined analytical strategies are particularly valuable in regulatory compliance testing, quality control, and research applications where complete impurity profiling is required.Expand Specific Solutions
Leading Manufacturers and Research Institutions
The analytical methods for trace impurities market is currently in a growth phase, characterized by increasing regulatory demands for pharmaceutical purity and quality control. The global market for analytical instruments in pharmaceutical testing is estimated at $5-6 billion, with HPLC dominating approximately 45% of this segment. Leading players include established pharmaceutical companies like Sanofi-Aventis and Teva Pharmaceutical, who have developed proprietary analytical methods, alongside specialized analytical equipment manufacturers such as Hitachi and Carl Zeiss Microscopy. The technology landscape shows varying maturity levels: HPLC and GC are well-established with continuous refinement, while newer IC applications are still evolving. Companies like DuPont and BASF are advancing method development for complex matrices, while pharmaceutical manufacturers including Zydus Lifesciences and Alembic are implementing these technologies to meet stringent regulatory requirements.
Hitachi Ltd.
Technical Solution: Hitachi has developed advanced analytical instrumentation specifically designed for pharmaceutical impurity analysis. Their HPLC systems feature ultra-low dispersion flow paths and specialized detector technologies including their proprietary VWD (Variable Wavelength Detector) that provides exceptional sensitivity for trace impurity detection. Hitachi's GC solutions incorporate innovative sample introduction systems and highly inert flow paths to minimize analyte degradation and adsorption, critical for accurate impurity quantification. Their ion chromatography platforms feature high-pressure capabilities (up to 5000 psi) enabling the use of smaller particle size columns for enhanced resolution of closely related impurities. Hitachi has integrated machine learning algorithms into their chromatography data systems to improve peak detection, integration, and impurity identification in complex samples. Their systems feature automated method scouting capabilities that systematically evaluate multiple column chemistries and mobile phase compositions to optimize separation of critical impurity pairs.
Strengths: Exceptional instrument reliability and precision for routine quality control applications; innovative detector technologies provide superior sensitivity; comprehensive software solutions streamline method development and validation. Weaknesses: Higher initial capital investment compared to some competitors; proprietary technologies may limit method transfer to other platforms; requires specialized training for optimal utilization.
Teva Pharmaceutical Industries Ltd.
Technical Solution: Teva has developed a systematic approach to impurity profiling using a combination of orthogonal analytical techniques. Their platform centers on stability-indicating HPLC methods using modern column technologies (core-shell, monolithic) coupled with diode array detection (DAD) and evaporative light scattering detection (ELSD) for comprehensive impurity detection. For genotoxic impurities, Teva employs highly sensitive GC-MS/MS methods with specialized derivatization techniques to enhance volatility and detection of polar compounds. Their ion chromatography platform features capillary IC with suppressed conductivity detection for trace level analysis of inorganic impurities in API and drug products. Teva has implemented automated sample preparation workflows using robotic liquid handling systems to minimize variability and contamination risks during impurity analysis. Their methods incorporate internal standardization and isotope dilution techniques to improve quantitative accuracy across diverse sample matrices.
Strengths: Robust methods suitable for generic drug development and manufacturing; efficient workflows support high-volume testing requirements; cost-effective approaches balance regulatory compliance with operational efficiency. Weaknesses: Less cutting-edge than some competitors in terms of novel detection technologies; method development may require more iterations to achieve optimal performance; limited application to complex biologics.
Breakthrough Patents in Separation Science
Analytical methods for analyzing and determining impurities in dianhydrogalactitol
PatentActiveUS9029164B2
Innovation
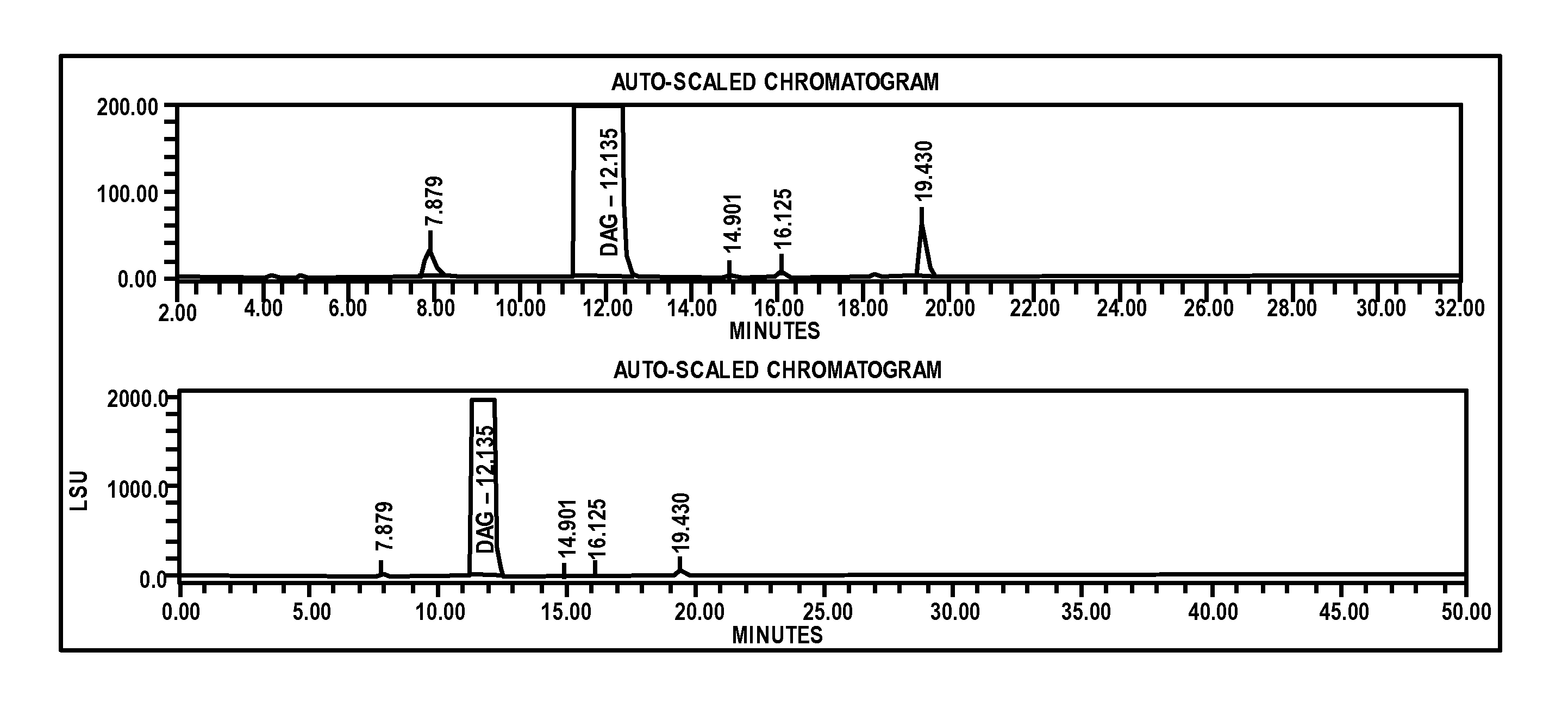
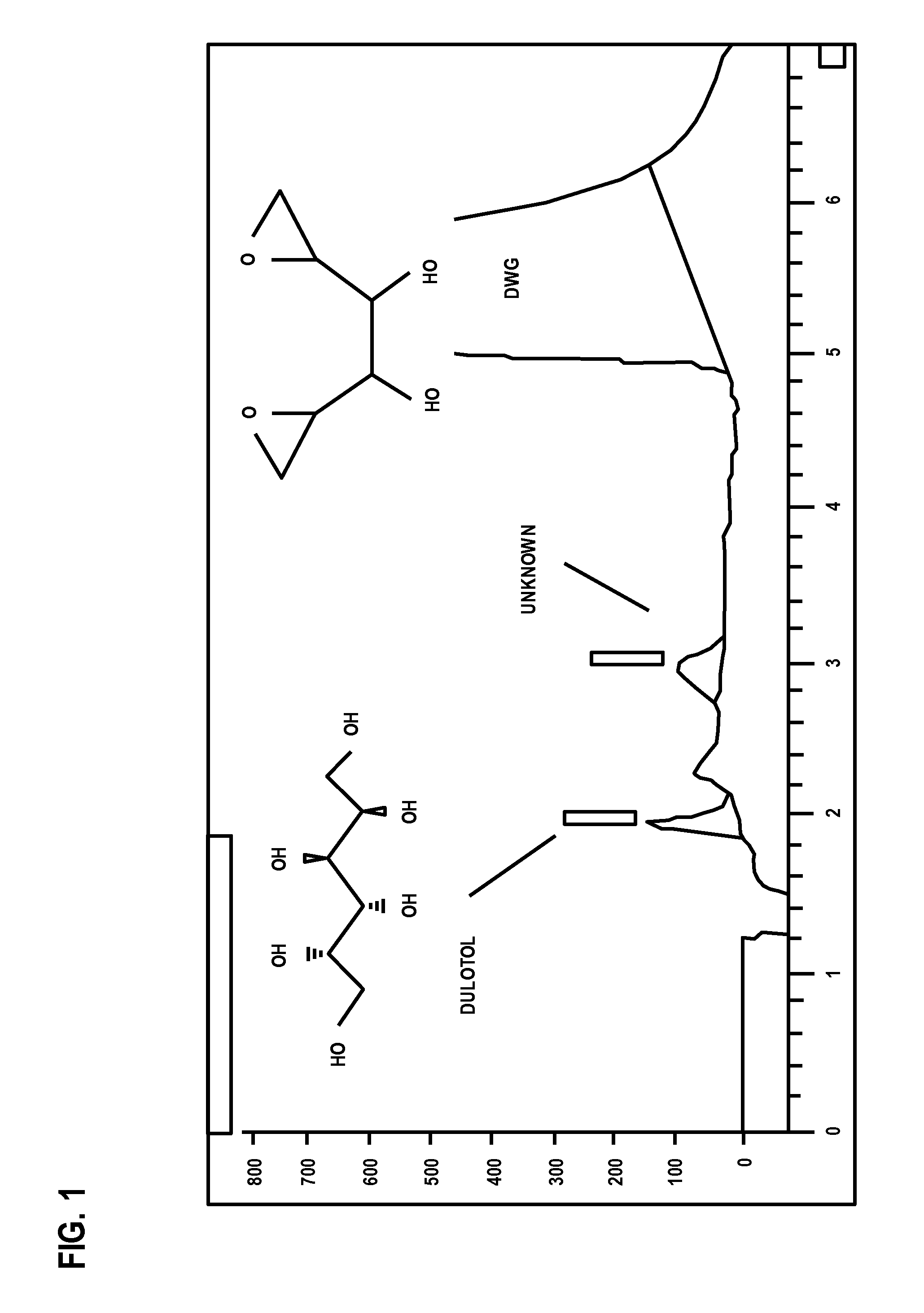
- An improved analytical method utilizing high-performance liquid chromatography (HPLC) with refractive index (RI) detection and evaporative light scattering detection (ELSD), combined with electrospray tandem mass spectrometry, to separate and quantify dianhydrogalactitol from impurities like dulcitol and degradation products using gradient or isocratic elution with sodium hydroxide or ammonium hydroxide-based mobile phases.
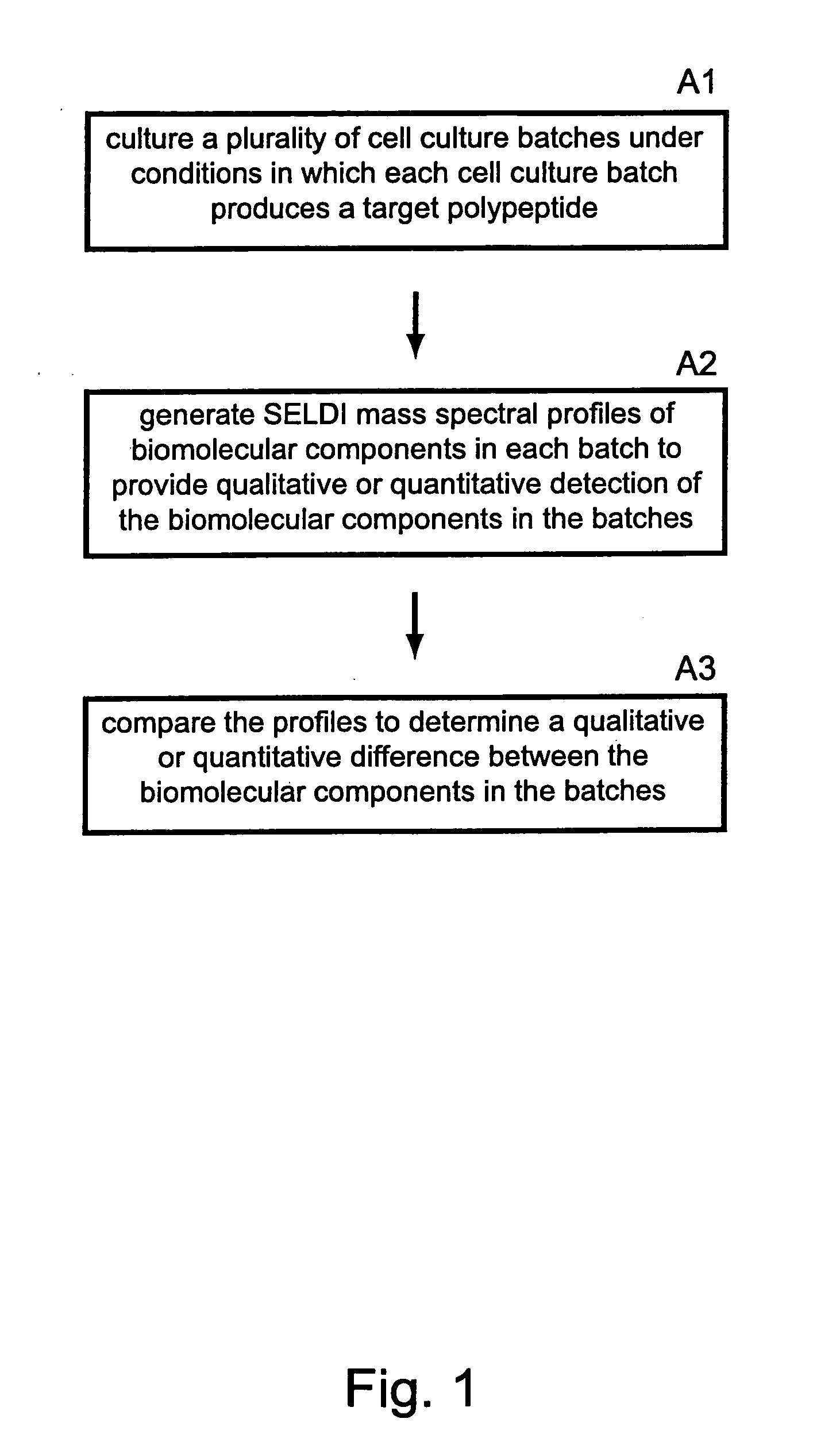
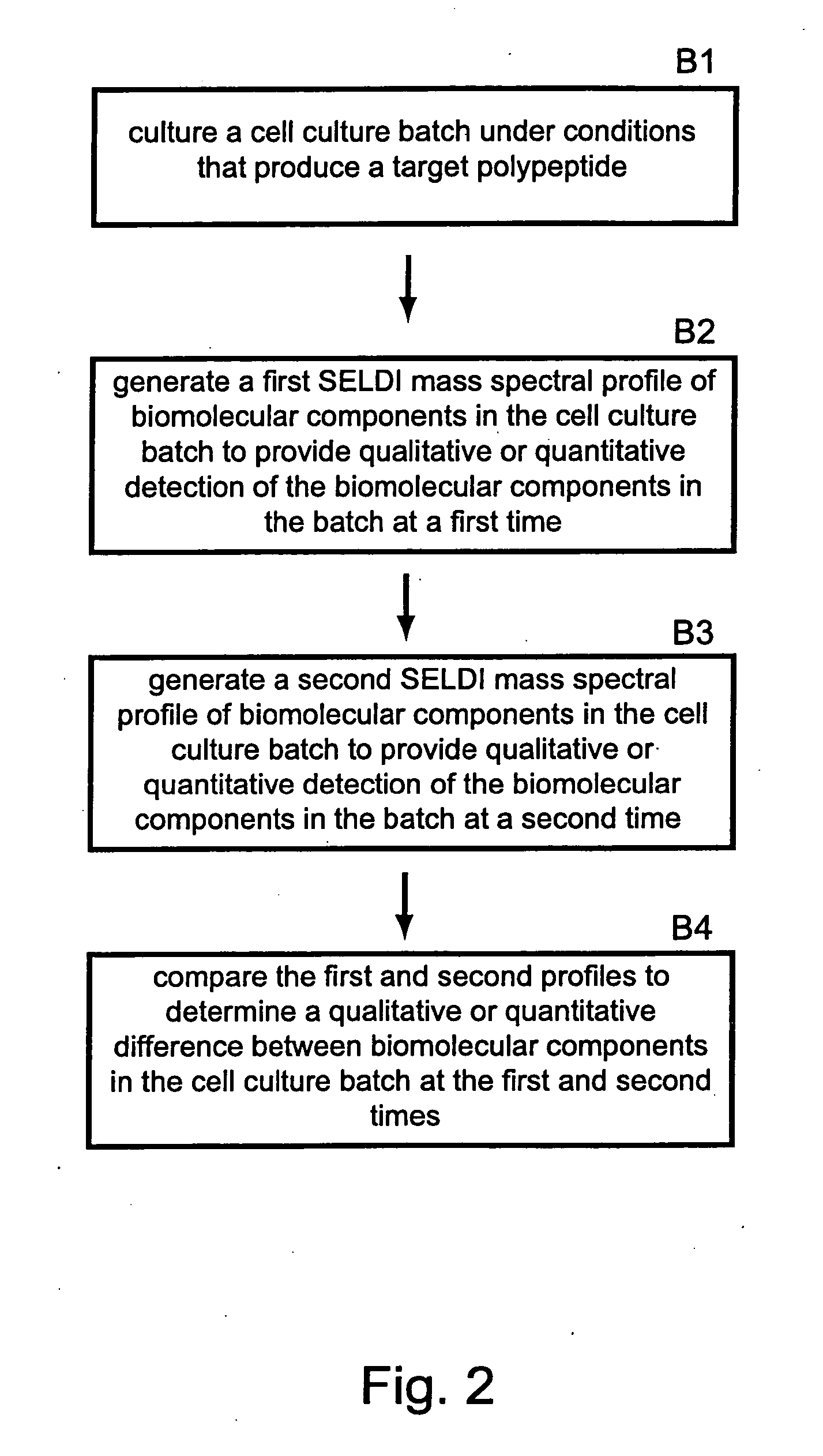
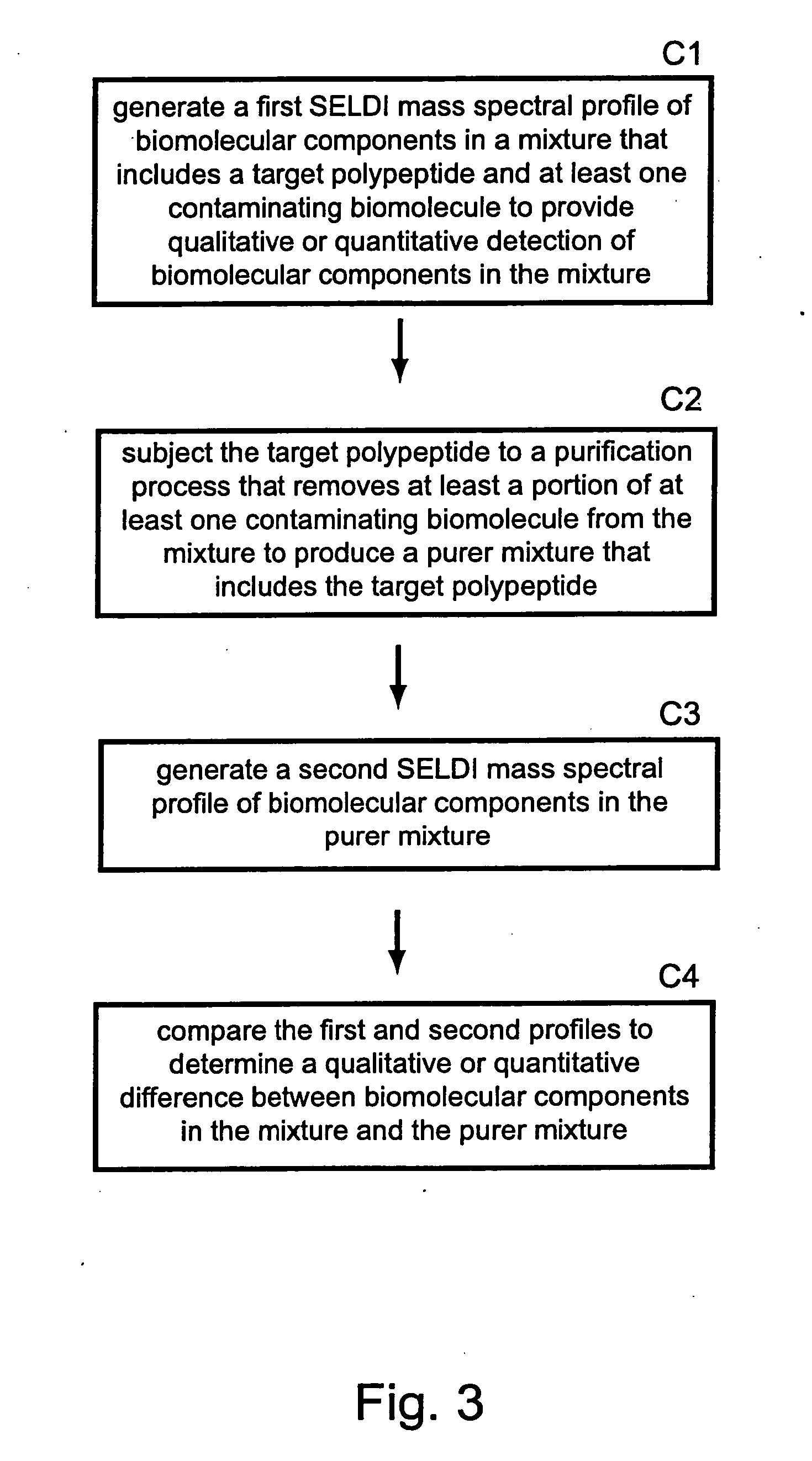
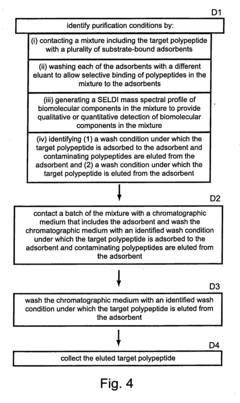
Methods for monitoring polypeptide production and purification using surface enhanced laser desorption/ionization mass spectrometry
PatentInactiveUS20070059776A1
Innovation
- The use of surface-enhanced laser desorption/ionization (SELDI) mass spectrometry to generate mass spectral profiles of biomolecular components in cell culture media, allowing for the identification and quantification of target polypeptides and differentiation from impurities with improved sensitivity, and optimization of purification processes.
Regulatory Compliance and Quality Standards
The regulatory landscape for analytical methods in purity assurance is increasingly complex and stringent, with global regulatory bodies establishing comprehensive frameworks to ensure product safety and efficacy. The FDA's guidance on Analytical Procedures and Methods Validation for Drugs and Biologics provides detailed requirements for HPLC, GC, and IC methodologies, emphasizing method validation parameters including specificity, linearity, accuracy, precision, and robustness. These parameters are critical for ensuring reliable detection and quantification of trace impurities.
The European Medicines Agency (EMA) has established parallel guidelines through ICH Q2(R1) for analytical method validation, with specific focus on chromatographic techniques. These guidelines mandate thorough documentation of system suitability tests and method transfer protocols when implementing analytical methods across different laboratories or manufacturing sites.
Pharmacopoeial standards, including USP <621> (Chromatography) and EP 2.2.29 (Liquid Chromatography), provide standardized methodologies and acceptance criteria for chromatographic analyses. These standards are regularly updated to incorporate technological advancements and emerging best practices in analytical chemistry.
ISO/IEC 17025 certification has become increasingly important for analytical laboratories, establishing requirements for competence, impartiality, and consistent operation. This standard specifically addresses technical requirements for testing and calibration of analytical instruments used in trace impurity detection.
Quality-by-Design (QbD) principles, as outlined in ICH Q8, Q9, and Q10 guidelines, are now being applied to analytical method development. This approach emphasizes understanding critical method attributes and establishing a design space within which changes to method parameters do not significantly impact the quality of results, providing greater flexibility in method optimization.
Continuous monitoring requirements have evolved to include real-time release testing (RTRT) strategies, where analytical methods for impurity detection are integrated into the manufacturing process. This approach aligns with FDA's Process Analytical Technology (PAT) initiative, enabling more efficient quality control and reducing time-to-market.
Environmental regulations, including REACH in Europe and various global initiatives addressing persistent organic pollutants, have expanded the scope of impurity testing to include environmental contaminants. These regulations necessitate increasingly sensitive analytical methods capable of detecting trace impurities at parts-per-billion or even parts-per-trillion levels.
Industry-specific standards, such as those from ASTM International and AOAC International, provide method-specific guidelines for particular applications of HPLC, GC, and IC in different sectors, ensuring consistency and comparability of analytical results across the industry.
The European Medicines Agency (EMA) has established parallel guidelines through ICH Q2(R1) for analytical method validation, with specific focus on chromatographic techniques. These guidelines mandate thorough documentation of system suitability tests and method transfer protocols when implementing analytical methods across different laboratories or manufacturing sites.
Pharmacopoeial standards, including USP <621> (Chromatography) and EP 2.2.29 (Liquid Chromatography), provide standardized methodologies and acceptance criteria for chromatographic analyses. These standards are regularly updated to incorporate technological advancements and emerging best practices in analytical chemistry.
ISO/IEC 17025 certification has become increasingly important for analytical laboratories, establishing requirements for competence, impartiality, and consistent operation. This standard specifically addresses technical requirements for testing and calibration of analytical instruments used in trace impurity detection.
Quality-by-Design (QbD) principles, as outlined in ICH Q8, Q9, and Q10 guidelines, are now being applied to analytical method development. This approach emphasizes understanding critical method attributes and establishing a design space within which changes to method parameters do not significantly impact the quality of results, providing greater flexibility in method optimization.
Continuous monitoring requirements have evolved to include real-time release testing (RTRT) strategies, where analytical methods for impurity detection are integrated into the manufacturing process. This approach aligns with FDA's Process Analytical Technology (PAT) initiative, enabling more efficient quality control and reducing time-to-market.
Environmental regulations, including REACH in Europe and various global initiatives addressing persistent organic pollutants, have expanded the scope of impurity testing to include environmental contaminants. These regulations necessitate increasingly sensitive analytical methods capable of detecting trace impurities at parts-per-billion or even parts-per-trillion levels.
Industry-specific standards, such as those from ASTM International and AOAC International, provide method-specific guidelines for particular applications of HPLC, GC, and IC in different sectors, ensuring consistency and comparability of analytical results across the industry.
Method Validation and Reproducibility Strategies
Method validation is a critical component in analytical testing for trace impurities, ensuring that HPLC, GC, and IC methods deliver reliable and consistent results. The validation process typically encompasses several key parameters: specificity, linearity, accuracy, precision, detection limit, quantitation limit, range, and robustness. Each parameter must be rigorously evaluated according to international guidelines such as ICH Q2(R1), USP <1225>, and FDA Guidance for Industry.
Specificity validation ensures that the analytical method can accurately identify and quantify the target analyte in the presence of other components. This is particularly crucial for complex matrices where interference can compromise results. Techniques such as peak purity assessment using diode array detection in HPLC or mass spectrometric detection can provide additional confirmation of specificity.
Linearity assessment establishes the method's ability to obtain test results proportional to the concentration of analyte. For trace impurity analysis, this relationship must be demonstrated across the expected concentration range, typically requiring a minimum of five concentration levels and correlation coefficients (r²) exceeding 0.99.
Precision evaluation encompasses repeatability (intra-day), intermediate precision (inter-day), and reproducibility (inter-laboratory). For trace impurity analysis, relative standard deviation (RSD) targets are typically set at ≤10% for repeatability and ≤15% for intermediate precision, though these may vary based on concentration levels and regulatory requirements.
Accuracy validation, often conducted through recovery studies, verifies that the method can quantify the true amount of analyte present. For trace impurities, spiking known quantities at different concentration levels (typically 80%, 100%, and 120% of specification limits) provides recovery data that should fall within 80-120% for concentrations below 0.1%.
Robustness testing evaluates the method's reliability during normal usage by deliberately introducing small variations in method parameters. For HPLC methods, these might include changes in mobile phase composition (±2%), pH (±0.2 units), column temperature (±5°C), or flow rate (±0.1 mL/min).
System suitability testing (SST) serves as an ongoing verification that the analytical system performs adequately during routine use. Key SST parameters include resolution between critical pairs, tailing factor, theoretical plates, and signal-to-noise ratio, with acceptance criteria established during method validation.
Reproducibility strategies often incorporate quality control samples, proficiency testing, and inter-laboratory comparisons. The implementation of statistical process control charts helps monitor method performance over time, enabling early detection of analytical drift or systematic errors before they impact product quality decisions.
Specificity validation ensures that the analytical method can accurately identify and quantify the target analyte in the presence of other components. This is particularly crucial for complex matrices where interference can compromise results. Techniques such as peak purity assessment using diode array detection in HPLC or mass spectrometric detection can provide additional confirmation of specificity.
Linearity assessment establishes the method's ability to obtain test results proportional to the concentration of analyte. For trace impurity analysis, this relationship must be demonstrated across the expected concentration range, typically requiring a minimum of five concentration levels and correlation coefficients (r²) exceeding 0.99.
Precision evaluation encompasses repeatability (intra-day), intermediate precision (inter-day), and reproducibility (inter-laboratory). For trace impurity analysis, relative standard deviation (RSD) targets are typically set at ≤10% for repeatability and ≤15% for intermediate precision, though these may vary based on concentration levels and regulatory requirements.
Accuracy validation, often conducted through recovery studies, verifies that the method can quantify the true amount of analyte present. For trace impurities, spiking known quantities at different concentration levels (typically 80%, 100%, and 120% of specification limits) provides recovery data that should fall within 80-120% for concentrations below 0.1%.
Robustness testing evaluates the method's reliability during normal usage by deliberately introducing small variations in method parameters. For HPLC methods, these might include changes in mobile phase composition (±2%), pH (±0.2 units), column temperature (±5°C), or flow rate (±0.1 mL/min).
System suitability testing (SST) serves as an ongoing verification that the analytical system performs adequately during routine use. Key SST parameters include resolution between critical pairs, tailing factor, theoretical plates, and signal-to-noise ratio, with acceptance criteria established during method validation.
Reproducibility strategies often incorporate quality control samples, proficiency testing, and inter-laboratory comparisons. The implementation of statistical process control charts helps monitor method performance over time, enabling early detection of analytical drift or systematic errors before they impact product quality decisions.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!