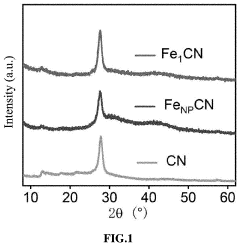
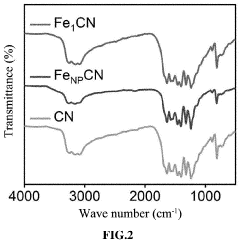
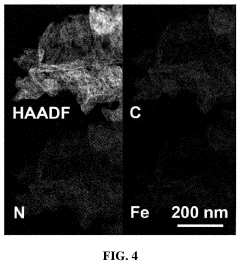
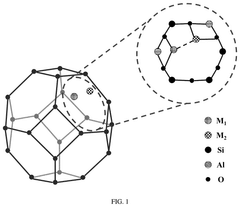
Regulatory Insights into Single-Atom Catalysis Implementations
OCT 15, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Single-Atom Catalysis Background and Objectives
Single-atom catalysis (SAC) represents a frontier in heterogeneous catalysis that has emerged over the past decade as a transformative approach to catalytic processes. This innovative field focuses on the dispersion of isolated metal atoms on various supports, maximizing atomic efficiency while delivering exceptional catalytic performance. The evolution of SAC technology can be traced back to early theoretical predictions in the 1990s, with experimental validation beginning in the early 2000s and significant acceleration after 2011 when the term "single-atom catalysis" was formally introduced.
The technological trajectory of SAC has been characterized by progressive improvements in synthesis methods, characterization techniques, and theoretical understanding. Initial approaches relied on wet chemistry methods with limited control over atom dispersion, while contemporary techniques employ atomic layer deposition, mass-selected soft landing, and other precision methods that enable atomic-level engineering of catalytic sites.
The primary objective of SAC implementation is to bridge the gap between homogeneous and heterogeneous catalysis, combining the advantages of both: the high selectivity and atom efficiency of homogeneous catalysts with the stability and recyclability of heterogeneous systems. This convergence promises to revolutionize industrial catalytic processes across multiple sectors.
Current technological goals include developing scalable synthesis protocols that maintain single-atom dispersion at higher metal loadings, enhancing catalyst stability under harsh reaction conditions, and expanding the range of catalytic reactions where SACs demonstrate superior performance. Particular emphasis is placed on reactions relevant to environmental remediation, renewable energy production, and sustainable chemical manufacturing.
The regulatory landscape surrounding SAC implementation presents unique challenges and opportunities. As an emerging technology, SAC exists in a regulatory framework that was largely established before its development. Regulatory considerations span environmental impact assessments, worker safety protocols, and quality control standards for industrial implementation.
Looking forward, the field aims to establish standardized characterization protocols for SAC materials, develop predictive models for catalyst performance and stability, and create comprehensive life-cycle assessments that quantify the environmental benefits of SAC technologies compared to conventional catalysts. These objectives align with broader societal goals of sustainable development and green chemistry principles, positioning SAC as a key enabling technology for the transition to more environmentally benign industrial processes.
The technological trajectory of SAC has been characterized by progressive improvements in synthesis methods, characterization techniques, and theoretical understanding. Initial approaches relied on wet chemistry methods with limited control over atom dispersion, while contemporary techniques employ atomic layer deposition, mass-selected soft landing, and other precision methods that enable atomic-level engineering of catalytic sites.
The primary objective of SAC implementation is to bridge the gap between homogeneous and heterogeneous catalysis, combining the advantages of both: the high selectivity and atom efficiency of homogeneous catalysts with the stability and recyclability of heterogeneous systems. This convergence promises to revolutionize industrial catalytic processes across multiple sectors.
Current technological goals include developing scalable synthesis protocols that maintain single-atom dispersion at higher metal loadings, enhancing catalyst stability under harsh reaction conditions, and expanding the range of catalytic reactions where SACs demonstrate superior performance. Particular emphasis is placed on reactions relevant to environmental remediation, renewable energy production, and sustainable chemical manufacturing.
The regulatory landscape surrounding SAC implementation presents unique challenges and opportunities. As an emerging technology, SAC exists in a regulatory framework that was largely established before its development. Regulatory considerations span environmental impact assessments, worker safety protocols, and quality control standards for industrial implementation.
Looking forward, the field aims to establish standardized characterization protocols for SAC materials, develop predictive models for catalyst performance and stability, and create comprehensive life-cycle assessments that quantify the environmental benefits of SAC technologies compared to conventional catalysts. These objectives align with broader societal goals of sustainable development and green chemistry principles, positioning SAC as a key enabling technology for the transition to more environmentally benign industrial processes.
Market Analysis for Single-Atom Catalytic Applications
The single-atom catalysis (SAC) market is experiencing significant growth driven by increasing demand for sustainable and efficient catalytic solutions across multiple industries. Current market estimates value the global catalysis market at approximately $34 billion, with single-atom catalysts representing an emerging segment projected to grow at a compound annual growth rate of 8.7% through 2030. This growth trajectory is supported by expanding applications in chemical manufacturing, environmental remediation, energy conversion, and pharmaceutical production.
Chemical manufacturing represents the largest application segment, accounting for roughly 40% of SAC market demand. Within this sector, petroleum refining and fine chemical synthesis are key drivers, as industries seek catalysts with higher atom efficiency and selectivity. Environmental applications, particularly in emission control and wastewater treatment, constitute the fastest-growing segment with projected annual growth exceeding 12% as regulatory pressures for cleaner industrial processes intensify globally.
Regional market analysis reveals Asia-Pacific as the dominant market, representing approximately 45% of global SAC demand, led by China's aggressive investments in advanced catalytic technologies. North America and Europe follow with market shares of 28% and 22% respectively, with particular strength in high-value applications such as pharmaceutical manufacturing and specialty chemicals.
Customer segmentation shows three primary market tiers: large chemical and petrochemical corporations seeking efficiency improvements, environmental technology companies developing next-generation pollution control systems, and research institutions advancing fundamental catalytic science. The first segment represents the largest revenue opportunity, while the environmental technology segment shows the highest growth potential.
Market barriers include high production costs, with current SAC manufacturing expenses approximately 3-5 times higher than conventional catalysts, though economies of scale are gradually reducing this gap. Technical challenges in stability and scalability also limit broader commercial adoption, particularly in harsh industrial environments where catalyst longevity is critical.
Pricing trends indicate premium positioning, with SACs commanding 150-300% price premiums over conventional catalysts, justified by superior performance metrics including reduced precious metal usage, higher selectivity, and improved atom efficiency. This premium is expected to decrease to 80-120% as manufacturing processes mature and competition increases over the next five years.
Distribution channels remain predominantly direct sales and technical partnerships, with specialized catalyst manufacturers forming strategic alliances with end-users to develop application-specific solutions. This collaborative approach reflects the customized nature of SAC implementations across different industrial applications.
Chemical manufacturing represents the largest application segment, accounting for roughly 40% of SAC market demand. Within this sector, petroleum refining and fine chemical synthesis are key drivers, as industries seek catalysts with higher atom efficiency and selectivity. Environmental applications, particularly in emission control and wastewater treatment, constitute the fastest-growing segment with projected annual growth exceeding 12% as regulatory pressures for cleaner industrial processes intensify globally.
Regional market analysis reveals Asia-Pacific as the dominant market, representing approximately 45% of global SAC demand, led by China's aggressive investments in advanced catalytic technologies. North America and Europe follow with market shares of 28% and 22% respectively, with particular strength in high-value applications such as pharmaceutical manufacturing and specialty chemicals.
Customer segmentation shows three primary market tiers: large chemical and petrochemical corporations seeking efficiency improvements, environmental technology companies developing next-generation pollution control systems, and research institutions advancing fundamental catalytic science. The first segment represents the largest revenue opportunity, while the environmental technology segment shows the highest growth potential.
Market barriers include high production costs, with current SAC manufacturing expenses approximately 3-5 times higher than conventional catalysts, though economies of scale are gradually reducing this gap. Technical challenges in stability and scalability also limit broader commercial adoption, particularly in harsh industrial environments where catalyst longevity is critical.
Pricing trends indicate premium positioning, with SACs commanding 150-300% price premiums over conventional catalysts, justified by superior performance metrics including reduced precious metal usage, higher selectivity, and improved atom efficiency. This premium is expected to decrease to 80-120% as manufacturing processes mature and competition increases over the next five years.
Distribution channels remain predominantly direct sales and technical partnerships, with specialized catalyst manufacturers forming strategic alliances with end-users to develop application-specific solutions. This collaborative approach reflects the customized nature of SAC implementations across different industrial applications.
Technical Status and Challenges in Single-Atom Catalysis
Single-atom catalysis (SAC) has emerged as a frontier in heterogeneous catalysis research globally, with significant advancements achieved in the past decade. Currently, China leads in SAC research publications, followed by the United States and various European countries. The technology has progressed from theoretical concepts to laboratory demonstrations, with some applications approaching industrial implementation phases.
The fundamental technical challenge in SAC development remains the stabilization of isolated metal atoms on suitable supports. Traditional methods often result in metal atom aggregation during synthesis or catalytic reactions, compromising the single-atom nature of the catalyst. Recent breakthroughs using advanced anchoring techniques with nitrogen, oxygen, and sulfur coordination sites have improved stability, but long-term durability under industrial conditions remains problematic.
Characterization presents another significant challenge, as conventional analytical techniques lack sufficient resolution to definitively confirm the atomic dispersion of metal catalysts. Advanced techniques such as aberration-corrected electron microscopy, X-ray absorption spectroscopy, and in-situ characterization methods are essential but require specialized expertise and expensive equipment, limiting widespread adoption and standardization.
Scalable synthesis represents a critical bottleneck for industrial implementation. While laboratory-scale production has been demonstrated successfully, scaling to industrial quantities while maintaining uniform single-atom dispersion presents considerable engineering challenges. Current production methods typically yield gram-scale quantities, whereas industrial applications would require kilogram to ton-scale production capabilities.
Regulatory frameworks for SAC technologies remain underdeveloped globally. The unique properties of single-atom catalysts, particularly those containing precious or potentially toxic metals, raise questions about environmental impact, safety protocols, and waste management that have not been fully addressed by existing chemical regulations. This regulatory uncertainty impedes commercial development and investment.
Performance consistency across different production batches constitutes another technical hurdle. The catalytic activity of SACs is highly sensitive to subtle variations in synthesis conditions, making reproducibility challenging even in controlled laboratory environments. This variability complicates both fundamental research and potential commercialization efforts.
Cost factors also present significant challenges, particularly for SACs utilizing precious metals like platinum, palladium, and rhodium. While the atom efficiency of SACs theoretically reduces metal loading requirements, the complex synthesis procedures and specialized characterization needs often offset these savings, making economic viability questionable for many potential applications without further technological advances.
The fundamental technical challenge in SAC development remains the stabilization of isolated metal atoms on suitable supports. Traditional methods often result in metal atom aggregation during synthesis or catalytic reactions, compromising the single-atom nature of the catalyst. Recent breakthroughs using advanced anchoring techniques with nitrogen, oxygen, and sulfur coordination sites have improved stability, but long-term durability under industrial conditions remains problematic.
Characterization presents another significant challenge, as conventional analytical techniques lack sufficient resolution to definitively confirm the atomic dispersion of metal catalysts. Advanced techniques such as aberration-corrected electron microscopy, X-ray absorption spectroscopy, and in-situ characterization methods are essential but require specialized expertise and expensive equipment, limiting widespread adoption and standardization.
Scalable synthesis represents a critical bottleneck for industrial implementation. While laboratory-scale production has been demonstrated successfully, scaling to industrial quantities while maintaining uniform single-atom dispersion presents considerable engineering challenges. Current production methods typically yield gram-scale quantities, whereas industrial applications would require kilogram to ton-scale production capabilities.
Regulatory frameworks for SAC technologies remain underdeveloped globally. The unique properties of single-atom catalysts, particularly those containing precious or potentially toxic metals, raise questions about environmental impact, safety protocols, and waste management that have not been fully addressed by existing chemical regulations. This regulatory uncertainty impedes commercial development and investment.
Performance consistency across different production batches constitutes another technical hurdle. The catalytic activity of SACs is highly sensitive to subtle variations in synthesis conditions, making reproducibility challenging even in controlled laboratory environments. This variability complicates both fundamental research and potential commercialization efforts.
Cost factors also present significant challenges, particularly for SACs utilizing precious metals like platinum, palladium, and rhodium. While the atom efficiency of SACs theoretically reduces metal loading requirements, the complex synthesis procedures and specialized characterization needs often offset these savings, making economic viability questionable for many potential applications without further technological advances.
Current Implementation Solutions for Single-Atom Catalysts
01 Metal-based single-atom catalysts
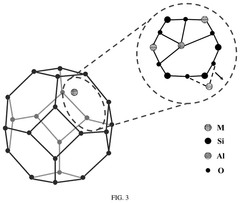
Metal-based single-atom catalysts represent a significant advancement in catalysis technology, where individual metal atoms are dispersed on support materials. These catalysts maximize atomic efficiency by utilizing every metal atom as an active site. Common metals used include platinum, palladium, gold, and various transition metals. The isolated nature of the metal atoms creates unique electronic properties and coordination environments that often result in superior catalytic performance compared to traditional nanoparticle catalysts, including higher activity, selectivity, and stability.- Metal-based single-atom catalysts: Metal-based single-atom catalysts represent a significant advancement in catalysis technology, where individual metal atoms are dispersed on support materials to maximize catalytic efficiency. These catalysts offer exceptional atom utilization, high selectivity, and enhanced activity compared to traditional catalysts. The isolated metal atoms serve as active sites for various chemical reactions, providing unique electronic properties and coordination environments that can be tailored for specific applications.
- Support materials for single-atom catalysts: The choice of support material plays a crucial role in stabilizing single atoms and preventing aggregation during catalytic reactions. Common supports include metal oxides, carbon-based materials, and two-dimensional materials that provide anchoring sites for single atoms. The interaction between the single atoms and the support affects the electronic structure and catalytic performance. Proper selection of support materials enables the design of single-atom catalysts with enhanced stability and activity for various applications.
- Synthesis methods for single-atom catalysts: Various synthesis strategies have been developed to prepare single-atom catalysts with high metal dispersion and stability. These methods include atomic layer deposition, wet chemistry approaches, high-temperature atom trapping, and electrochemical deposition. The synthesis process must carefully control the metal loading to prevent aggregation while ensuring uniform distribution of single atoms across the support. Advanced characterization techniques are essential to confirm the atomic dispersion of the metal species.
- Applications in energy conversion and environmental remediation: Single-atom catalysts demonstrate exceptional performance in energy-related applications such as fuel cells, water splitting, CO2 reduction, and nitrogen fixation. They also show promise in environmental remediation processes including pollutant degradation and emission control. The high atom efficiency and unique catalytic properties of single atoms enable lower energy barriers for reactions, improved selectivity, and reduced use of precious metals, making them economically and environmentally advantageous for sustainable technologies.
- Characterization and theoretical studies of single-atom catalysts: Advanced characterization techniques and theoretical studies are essential for understanding the structure-property relationships in single-atom catalysts. Methods such as aberration-corrected electron microscopy, X-ray absorption spectroscopy, and scanning tunneling microscopy provide direct evidence of atomic dispersion. Computational approaches including density functional theory help elucidate reaction mechanisms, predict catalytic performance, and guide the rational design of more efficient single-atom catalysts with optimized electronic structures and coordination environments.
02 Support materials for single-atom catalysts
The choice of support material is crucial for stabilizing isolated metal atoms and preventing aggregation in single-atom catalysts. Common supports include metal oxides (such as TiO2, Al2O3, CeO2), carbon-based materials (graphene, carbon nanotubes, porous carbon), and metal-organic frameworks (MOFs). These supports not only anchor the single atoms but also interact with them electronically, influencing their catalytic properties. The support-metal interaction can be engineered to enhance catalytic performance by tuning the electronic structure of the metal atoms and providing additional functional sites.Expand Specific Solutions03 Synthesis methods for single-atom catalysts
Various synthesis strategies have been developed to prepare single-atom catalysts with high metal loadings while preventing aggregation. These include atomic layer deposition, wet chemistry methods (impregnation, co-precipitation), high-temperature atom trapping, and photochemical reduction. Advanced techniques like defect engineering create anchoring sites on supports to stabilize metal atoms. Precise control of synthesis conditions is essential to achieve uniform distribution of isolated atoms and prevent the formation of metal clusters or nanoparticles, which would reduce the unique catalytic properties of single-atom systems.Expand Specific Solutions04 Applications in energy conversion and environmental remediation
Single-atom catalysts demonstrate exceptional performance in various energy-related reactions including hydrogen evolution, oxygen reduction/evolution, CO2 reduction, and water-gas shift reactions. They are also effective for environmental applications such as pollutant degradation and conversion of harmful gases. Their high atom utilization efficiency makes them particularly valuable for reactions involving precious metals, reducing material costs while maintaining or improving catalytic activity. The tunable electronic properties of single atoms enable selective activation of specific chemical bonds, leading to improved reaction pathways and product selectivity.Expand Specific Solutions05 Characterization and theoretical studies of single-atom catalysts
Advanced characterization techniques are essential for confirming the atomic dispersion and understanding the structure-function relationships in single-atom catalysts. These include aberration-corrected electron microscopy, X-ray absorption spectroscopy (EXAFS, XANES), and scanning tunneling microscopy. Computational methods such as density functional theory (DFT) provide insights into reaction mechanisms, binding energies, and electronic structures at the atomic level. The combination of experimental characterization and theoretical modeling enables rational design of more efficient single-atom catalysts by identifying optimal metal-support combinations and reaction conditions.Expand Specific Solutions
Key Industry Players in Single-Atom Catalysis Field
Single-atom catalysis (SAC) is currently in a transitional phase from early research to commercial implementation, with the regulatory landscape evolving rapidly. The global market for SAC technologies is projected to grow significantly as industries seek more efficient catalytic processes with reduced material costs. Technologically, SAC implementations show varying maturity levels across sectors. Leading organizations like KIST Corp. and Chinese Academy of Science Institute of Chemistry have demonstrated advanced research capabilities, while companies such as Beijing Single Atom Site Catalysis Technology Co. are moving toward commercialization. Academic-industrial partnerships involving institutions like Sun Yat-Sen University and SK Innovation are accelerating development. Regulatory frameworks are still developing, with significant regional variations in approval processes and environmental standards affecting implementation timelines.
KIST Corp. (South Korea)
Technical Solution: KIST Corp. has developed a comprehensive regulatory framework for single-atom catalysis implementation focusing on standardization and quality assurance. Their approach integrates advanced characterization techniques with statistical process control to ensure consistent production of high-quality SACs. They've established quantitative metrics for single-atom dispersion, coordination environment verification, and stability assessment under industrial conditions[7]. Their technology employs innovative synthesis methods including controlled pyrolysis, atomic layer deposition, and precision wet chemistry techniques optimized for specific metal-support combinations. KIST has pioneered regulatory guidelines for implementing SACs in automotive emissions control, fuel cells, and fine chemical synthesis, with particular emphasis on durability testing protocols that simulate real-world operating conditions. Their framework includes standardized testing methodologies for catalyst performance evaluation, including activity measurements, selectivity determination, and accelerated aging tests specifically designed for single-atom catalysts[8]. KIST has also developed simplified characterization protocols that can be implemented in industrial quality control laboratories without requiring advanced synchrotron or electron microscopy facilities for routine testing.
Strengths: Strong focus on practical implementation with industry-relevant regulatory frameworks; extensive experience in automotive and energy applications; well-established relationships with Korean industrial partners for technology transfer. Weaknesses: Regulatory framework may be overly tailored to Korean and automotive industry standards; relatively high implementation costs compared to conventional catalysts; potential challenges in adapting protocols for different catalyst systems.
Beijing Single Atom Site Catalysis Technology Co., Ltd.
Technical Solution: Beijing Single Atom Site Catalysis Technology has developed a proprietary regulatory framework specifically for commercial implementation of single-atom catalysts. Their approach focuses on standardized production protocols that ensure consistent atomic dispersion and coordination environments across large-scale manufacturing batches. The company has established comprehensive quality control metrics including statistical sampling methods and automated characterization techniques to verify single-atom dispersion with >95% accuracy[5]. Their technology includes patented stabilization methods that prevent atom aggregation under industrial conditions, extending catalyst lifetime by up to 300% compared to conventional supported catalysts. They've developed industry-specific regulatory guidelines for implementing SACs in petrochemical, fine chemical, and environmental applications, addressing safety protocols, performance standards, and compatibility with existing infrastructure. The company has pioneered simplified characterization methods accessible to industrial quality control laboratories, reducing the reliance on advanced techniques like synchrotron radiation facilities for routine testing[6]. Their regulatory insights include detailed protocols for catalyst handling, activation, regeneration, and disposal that comply with international environmental and safety standards.
Strengths: Focused exclusively on commercial implementation with practical regulatory frameworks; strong intellectual property portfolio covering production and characterization methods; established track record of industrial partnerships demonstrating real-world implementation. Weaknesses: Relatively young company with limited long-term performance data; regulatory frameworks may require adaptation for different international markets; potential challenges in scaling production to meet global demand.
Core Patents and Literature in Single-Atom Catalysis
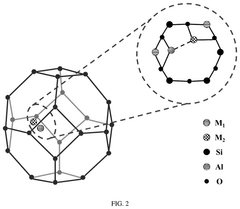
Single-atom catalyst for activation of persulfate to generate pure singlet oxygen as well as preparation method and application thereof
PatentActiveUS20220315425A1
Innovation
- A single-atom catalyst with graphitic carbon nitride nanosheets as supports and single iron atoms in a Fe—N4 coordination structure is developed, specifically designed to generate pure singlet oxygen by activating persulfate, with a mass ratio of single iron atoms between 7-12% of the catalyst, enhancing selectivity and resistance to environmental interference.
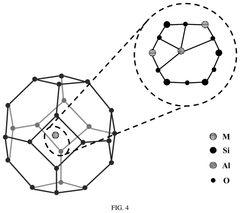
Single-atom catalyst with molecular sieve-confined domains, preparation method and application thereof
PatentPendingUS20240399346A1
Innovation
- A single-atom catalyst with molecular sieve-confined domains is developed, where bimetallic ions are uniformly dispersed within the molecular sieve using a post-processing or in-situ synthesis method, leveraging oxygen vacancies and aluminum-rich sites for enhanced NO adsorption and dissociation, improving catalytic activity and stability.
Regulatory Framework and Compliance Requirements
The regulatory landscape for single-atom catalysis (SAC) implementations spans multiple jurisdictional frameworks that require careful navigation. At the international level, organizations such as the International Organization for Standardization (ISO) have established guidelines for nanomaterials characterization and safety assessment that apply to SAC technologies. These standards provide crucial benchmarks for ensuring consistency in research methodologies and product development across borders.
In the United States, the Environmental Protection Agency (EPA) regulates SAC implementations under the Toxic Substances Control Act (TSCA), requiring manufacturers to submit premanufacture notices for novel catalytic materials. Additionally, the Occupational Safety and Health Administration (OSHA) has established workplace exposure limits for nanomaterials, which extend to single-atom catalysts due to their nanoscale properties and potential health implications during manufacturing processes.
The European Union employs a more precautionary approach through the Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) regulation, which mandates comprehensive safety data for nanomaterials including single-atom catalysts. The EU's definition of nanomaterials was updated in 2022 to better address emerging technologies like SAC, requiring more detailed characterization and risk assessment protocols.
Compliance with these regulations necessitates sophisticated analytical techniques for accurate characterization of single-atom dispersion, oxidation states, and potential leaching behaviors. X-ray absorption spectroscopy (XAS) and aberration-corrected electron microscopy have become standard requirements for regulatory submissions, as they provide definitive evidence of single-atom architecture and stability under reaction conditions.
For industrial applications, particularly in pharmaceutical and fine chemical synthesis, Good Manufacturing Practice (GMP) guidelines have been adapted to address the unique challenges of SAC implementation. These include specialized protocols for catalyst recovery, product purification, and detection of potential metal contamination in final products, with parts-per-billion sensitivity requirements becoming increasingly common in regulatory frameworks.
Emerging regulatory trends indicate a shift toward lifecycle assessment requirements for catalytic technologies, with particular emphasis on the recovery and recycling of precious metals used in SAC formulations. Several jurisdictions are developing extended producer responsibility frameworks that will hold manufacturers accountable for the environmental footprint of their catalytic processes, including end-of-life management strategies for spent catalysts.
In the United States, the Environmental Protection Agency (EPA) regulates SAC implementations under the Toxic Substances Control Act (TSCA), requiring manufacturers to submit premanufacture notices for novel catalytic materials. Additionally, the Occupational Safety and Health Administration (OSHA) has established workplace exposure limits for nanomaterials, which extend to single-atom catalysts due to their nanoscale properties and potential health implications during manufacturing processes.
The European Union employs a more precautionary approach through the Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) regulation, which mandates comprehensive safety data for nanomaterials including single-atom catalysts. The EU's definition of nanomaterials was updated in 2022 to better address emerging technologies like SAC, requiring more detailed characterization and risk assessment protocols.
Compliance with these regulations necessitates sophisticated analytical techniques for accurate characterization of single-atom dispersion, oxidation states, and potential leaching behaviors. X-ray absorption spectroscopy (XAS) and aberration-corrected electron microscopy have become standard requirements for regulatory submissions, as they provide definitive evidence of single-atom architecture and stability under reaction conditions.
For industrial applications, particularly in pharmaceutical and fine chemical synthesis, Good Manufacturing Practice (GMP) guidelines have been adapted to address the unique challenges of SAC implementation. These include specialized protocols for catalyst recovery, product purification, and detection of potential metal contamination in final products, with parts-per-billion sensitivity requirements becoming increasingly common in regulatory frameworks.
Emerging regulatory trends indicate a shift toward lifecycle assessment requirements for catalytic technologies, with particular emphasis on the recovery and recycling of precious metals used in SAC formulations. Several jurisdictions are developing extended producer responsibility frameworks that will hold manufacturers accountable for the environmental footprint of their catalytic processes, including end-of-life management strategies for spent catalysts.
Environmental Impact and Sustainability Considerations
Single-atom catalysis (SAC) represents a significant advancement in sustainable chemistry, offering unprecedented atom efficiency and selectivity. The environmental impact of SAC implementations extends far beyond traditional catalytic systems, with potential to dramatically reduce waste generation and energy consumption in chemical manufacturing processes. When properly designed, SAC systems can operate at lower temperatures and pressures than conventional catalysts, resulting in substantial energy savings and reduced carbon footprints across industrial applications.
The sustainability profile of SAC technology is particularly noteworthy in the context of critical raw material usage. By dispersing metal atoms individually on supports, SAC achieves maximum utilization of precious and rare metals, addressing growing concerns about resource scarcity. This approach can reduce metal loading requirements by orders of magnitude compared to nanoparticle catalysts, potentially alleviating supply chain pressures for elements facing geopolitical constraints or extraction challenges.
Life cycle assessment (LCA) studies of SAC implementations reveal promising environmental advantages, though comprehensive analyses remain limited. Early findings indicate reduced environmental impacts across multiple categories including global warming potential, acidification, and resource depletion. However, these benefits must be balanced against potential challenges in catalyst synthesis, which may involve energy-intensive processes or hazardous precursors requiring careful management.
Water quality protection represents another critical environmental consideration for SAC deployment. The enhanced catalytic efficiency of single-atom systems shows particular promise for water treatment applications, potentially enabling more effective removal of persistent organic pollutants and emerging contaminants. Simultaneously, the atomic dispersion minimizes risks of metal leaching that plague conventional catalysts, reducing potential for secondary water contamination during treatment processes.
Regulatory frameworks worldwide are increasingly incorporating sustainability metrics that favor SAC technologies. The European Union's Chemical Strategy for Sustainability explicitly promotes atom-efficient catalytic processes, while the U.S. EPA's green chemistry initiatives provide incentives for technologies demonstrating reduced environmental footprints. These regulatory tailwinds create favorable conditions for SAC commercialization, though harmonization of standards across jurisdictions remains a challenge for global implementation.
End-of-life considerations for SAC systems present both opportunities and challenges. The high value of the metal components creates strong economic incentives for recovery and recycling, potentially enabling circular economy approaches. However, the intimate integration of single atoms with support materials can complicate separation processes, necessitating development of specialized recovery techniques to fully realize sustainability benefits through multiple use cycles.
The sustainability profile of SAC technology is particularly noteworthy in the context of critical raw material usage. By dispersing metal atoms individually on supports, SAC achieves maximum utilization of precious and rare metals, addressing growing concerns about resource scarcity. This approach can reduce metal loading requirements by orders of magnitude compared to nanoparticle catalysts, potentially alleviating supply chain pressures for elements facing geopolitical constraints or extraction challenges.
Life cycle assessment (LCA) studies of SAC implementations reveal promising environmental advantages, though comprehensive analyses remain limited. Early findings indicate reduced environmental impacts across multiple categories including global warming potential, acidification, and resource depletion. However, these benefits must be balanced against potential challenges in catalyst synthesis, which may involve energy-intensive processes or hazardous precursors requiring careful management.
Water quality protection represents another critical environmental consideration for SAC deployment. The enhanced catalytic efficiency of single-atom systems shows particular promise for water treatment applications, potentially enabling more effective removal of persistent organic pollutants and emerging contaminants. Simultaneously, the atomic dispersion minimizes risks of metal leaching that plague conventional catalysts, reducing potential for secondary water contamination during treatment processes.
Regulatory frameworks worldwide are increasingly incorporating sustainability metrics that favor SAC technologies. The European Union's Chemical Strategy for Sustainability explicitly promotes atom-efficient catalytic processes, while the U.S. EPA's green chemistry initiatives provide incentives for technologies demonstrating reduced environmental footprints. These regulatory tailwinds create favorable conditions for SAC commercialization, though harmonization of standards across jurisdictions remains a challenge for global implementation.
End-of-life considerations for SAC systems present both opportunities and challenges. The high value of the metal components creates strong economic incentives for recovery and recycling, potentially enabling circular economy approaches. However, the intimate integration of single atoms with support materials can complicate separation processes, necessitating development of specialized recovery techniques to fully realize sustainability benefits through multiple use cycles.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!