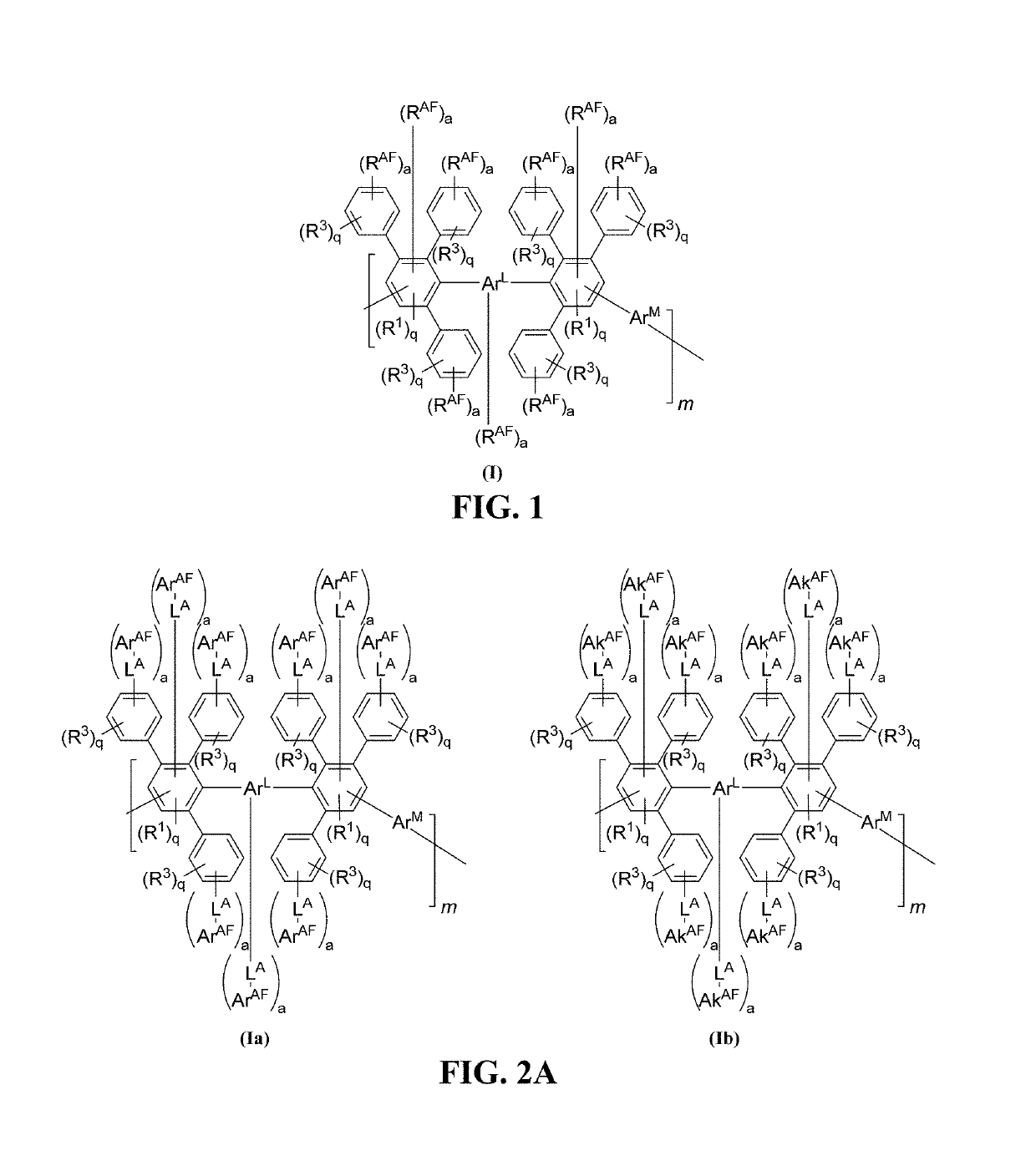
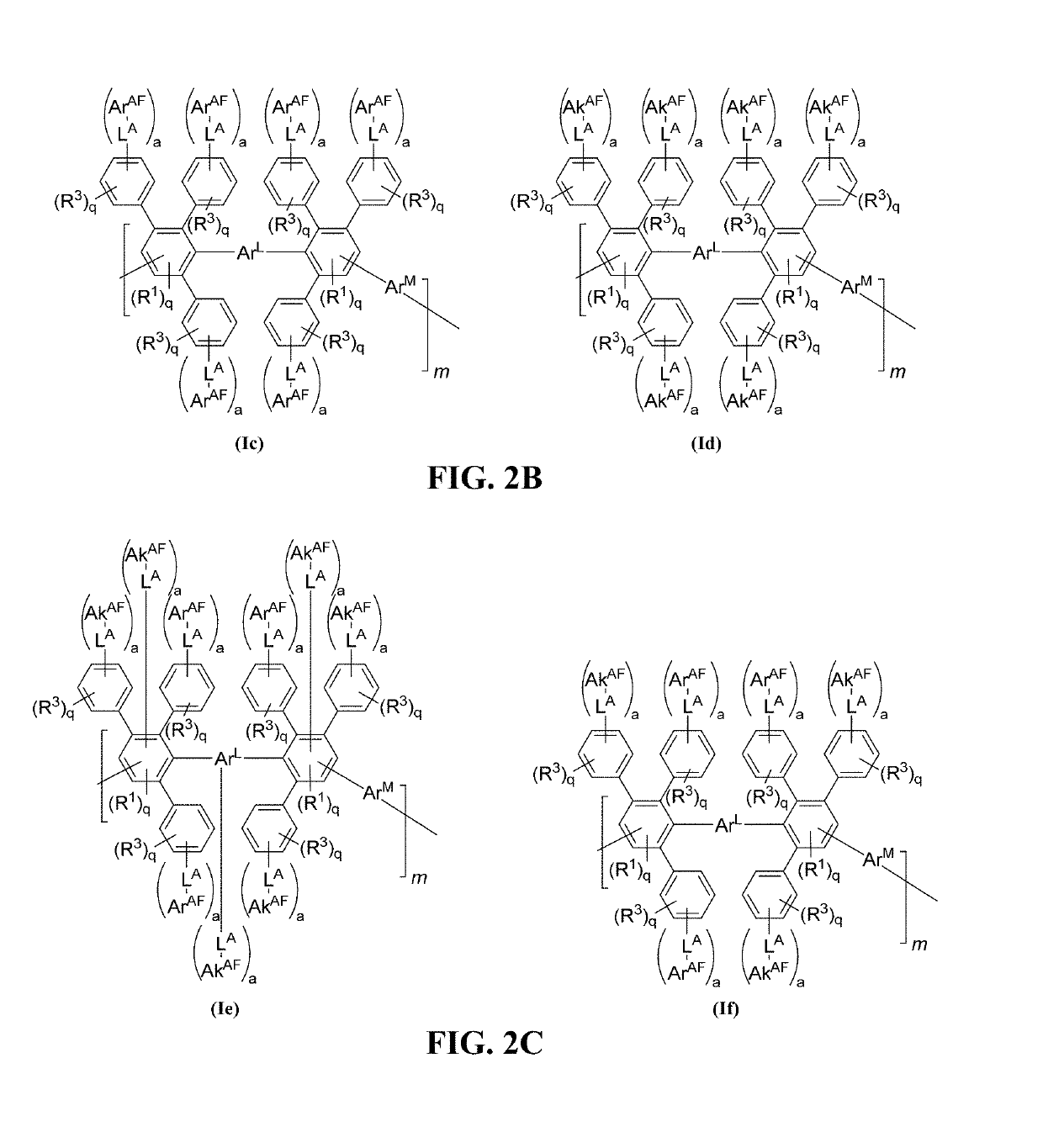
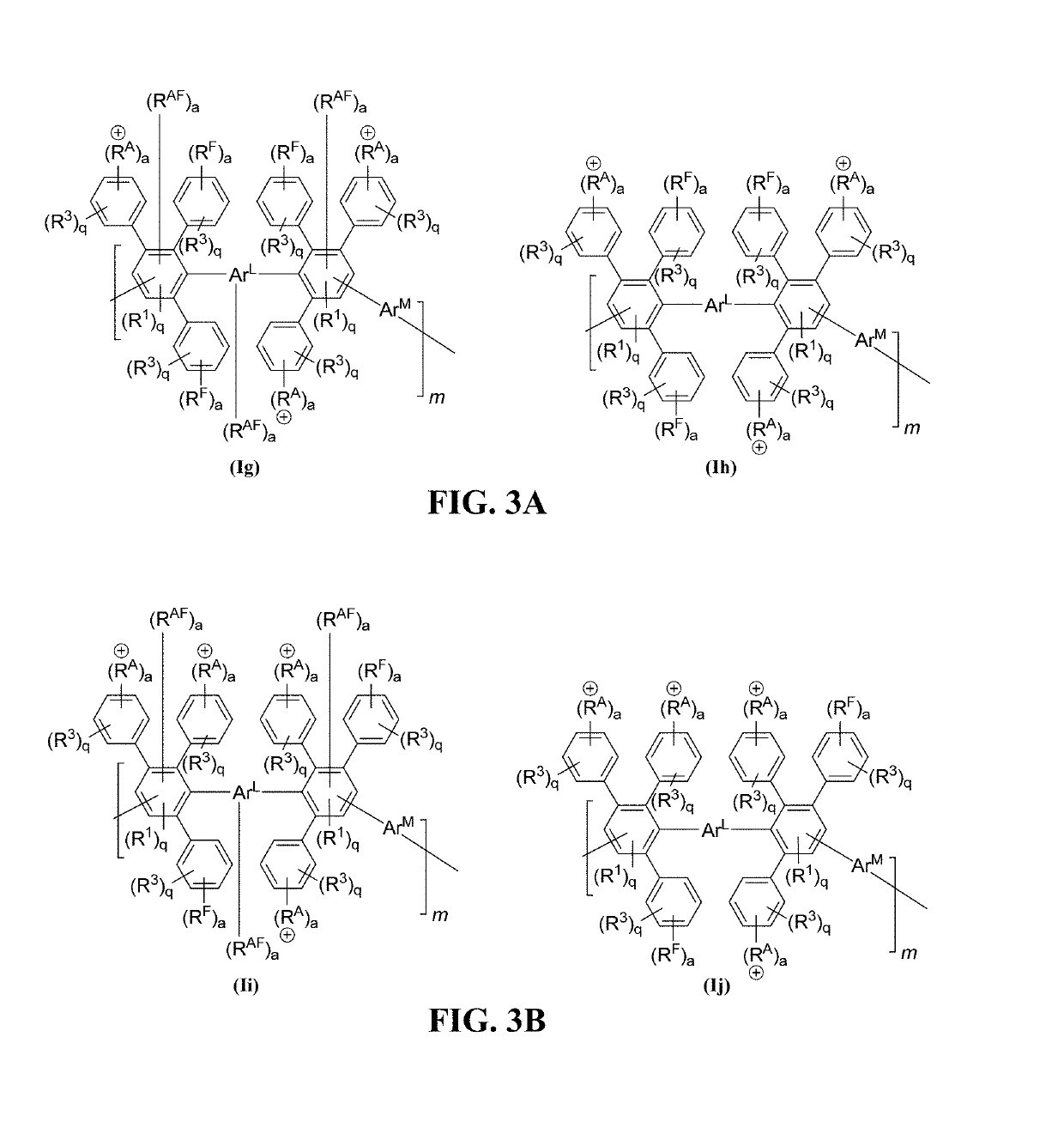
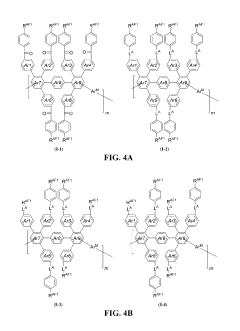
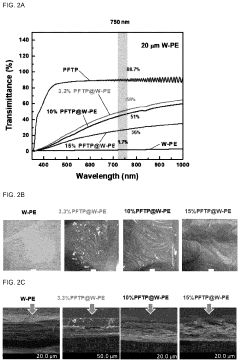
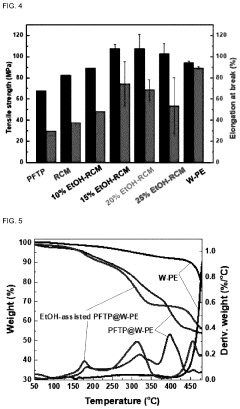
Advances in polymer design for anion exchange membranes
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Polymer AEM Technology Background and Objectives
Anion exchange membranes (AEMs) have emerged as a critical component in various electrochemical energy conversion and storage technologies over the past several decades. The development of these membranes represents a significant shift from traditional proton exchange membrane systems, offering advantages in terms of cost, efficiency, and environmental impact. The evolution of polymer-based AEMs can be traced back to the 1960s, with significant acceleration in research and development occurring in the early 2000s as the limitations of acidic systems became increasingly apparent.
The technological trajectory of AEMs has been characterized by persistent challenges in achieving the optimal balance between ionic conductivity, chemical stability, and mechanical durability. Early polymer designs focused primarily on quaternary ammonium functionalized polymers, which, while demonstrating promising anion conductivity, suffered from poor alkaline stability. This limitation prompted exploration of alternative polymer backbones and cationic functional groups to enhance membrane performance under operating conditions.
Recent advances in polymer chemistry have enabled more sophisticated approaches to AEM design, including the development of block copolymers, crosslinked networks, and composite materials. These innovations have significantly improved the stability and conductivity profiles of AEMs, pushing the boundaries of what is achievable in alkaline electrochemical systems. The integration of computational modeling and high-throughput screening methodologies has further accelerated the discovery and optimization of novel polymer architectures.
The primary technological objectives in AEM development center on achieving hydroxide conductivity exceeding 100 mS/cm under practical operating conditions while maintaining chemical stability for thousands of hours at elevated temperatures (80-120°C). Additionally, mechanical robustness sufficient to withstand dimensional changes during hydration/dehydration cycles remains a critical target. These performance metrics are essential for enabling the widespread commercial adoption of AEM-based technologies.
Beyond performance considerations, scalability and cost-effectiveness represent important technological goals. Current research increasingly emphasizes polymer designs amenable to large-scale manufacturing processes and utilizing more sustainable, less expensive precursors. This shift reflects the growing recognition that commercial viability requires not only superior performance but also economic competitiveness with established technologies.
The broader technological context for AEM development encompasses applications in fuel cells, electrolyzers, flow batteries, and various separation processes. Each application imposes specific requirements on membrane properties, driving diversification in polymer design approaches. As climate change concerns intensify, the potential of AEM-based technologies to enable more efficient renewable energy utilization and carbon-neutral chemical production has elevated the strategic importance of advances in this field.
The technological trajectory of AEMs has been characterized by persistent challenges in achieving the optimal balance between ionic conductivity, chemical stability, and mechanical durability. Early polymer designs focused primarily on quaternary ammonium functionalized polymers, which, while demonstrating promising anion conductivity, suffered from poor alkaline stability. This limitation prompted exploration of alternative polymer backbones and cationic functional groups to enhance membrane performance under operating conditions.
Recent advances in polymer chemistry have enabled more sophisticated approaches to AEM design, including the development of block copolymers, crosslinked networks, and composite materials. These innovations have significantly improved the stability and conductivity profiles of AEMs, pushing the boundaries of what is achievable in alkaline electrochemical systems. The integration of computational modeling and high-throughput screening methodologies has further accelerated the discovery and optimization of novel polymer architectures.
The primary technological objectives in AEM development center on achieving hydroxide conductivity exceeding 100 mS/cm under practical operating conditions while maintaining chemical stability for thousands of hours at elevated temperatures (80-120°C). Additionally, mechanical robustness sufficient to withstand dimensional changes during hydration/dehydration cycles remains a critical target. These performance metrics are essential for enabling the widespread commercial adoption of AEM-based technologies.
Beyond performance considerations, scalability and cost-effectiveness represent important technological goals. Current research increasingly emphasizes polymer designs amenable to large-scale manufacturing processes and utilizing more sustainable, less expensive precursors. This shift reflects the growing recognition that commercial viability requires not only superior performance but also economic competitiveness with established technologies.
The broader technological context for AEM development encompasses applications in fuel cells, electrolyzers, flow batteries, and various separation processes. Each application imposes specific requirements on membrane properties, driving diversification in polymer design approaches. As climate change concerns intensify, the potential of AEM-based technologies to enable more efficient renewable energy utilization and carbon-neutral chemical production has elevated the strategic importance of advances in this field.
Market Analysis for AEM Applications
The anion exchange membrane (AEM) market is experiencing significant growth, driven by increasing demand for clean energy solutions and sustainable water treatment technologies. The global AEM market was valued at approximately 4.2 billion USD in 2022 and is projected to reach 7.8 billion USD by 2028, representing a compound annual growth rate of 10.7%. This growth trajectory is primarily fueled by the expanding applications of AEMs in fuel cells, electrolyzers, flow batteries, and water purification systems.
In the energy sector, AEM fuel cells (AEMFCs) are gaining traction as viable alternatives to proton exchange membrane fuel cells (PEMFCs) due to their potential cost advantages and comparable performance metrics. The elimination of expensive platinum catalysts in AEMFCs represents a significant market driver, particularly as global efforts to reduce dependency on precious metals intensify. The hydrogen economy's expansion, with projected investments exceeding 300 billion USD globally by 2030, provides substantial market opportunities for AEM technologies.
Water treatment applications constitute another major market segment for AEMs, with electrodialysis and desalination processes increasingly adopting these membranes. The global water treatment market, valued at 265 billion USD in 2022, is expected to grow at 7.5% annually, with AEM technologies capturing an expanding share due to their energy efficiency and operational advantages over conventional technologies.
Regionally, North America and Europe currently dominate the AEM market, accounting for approximately 60% of global demand. However, Asia-Pacific represents the fastest-growing region, with China, Japan, and South Korea making substantial investments in hydrogen technologies and advanced water treatment solutions. The Chinese government's commitment to achieving carbon neutrality by 2060 has catalyzed significant funding for fuel cell technologies, creating favorable market conditions for AEM adoption.
Industry segmentation reveals that transportation applications, particularly in the automotive sector, represent the largest end-use market for AEMs, followed by stationary power generation and industrial processes. The commercial vehicle segment, including buses and trucks, shows particularly strong growth potential for AEM fuel cells due to their extended range capabilities and faster refueling compared to battery electric alternatives.
Market challenges include competition from established technologies such as proton exchange membranes and the need for further performance improvements to achieve widespread commercial adoption. Price sensitivity remains a significant factor, with end-users requiring demonstrable cost advantages over incumbent technologies to justify transition investments.
In the energy sector, AEM fuel cells (AEMFCs) are gaining traction as viable alternatives to proton exchange membrane fuel cells (PEMFCs) due to their potential cost advantages and comparable performance metrics. The elimination of expensive platinum catalysts in AEMFCs represents a significant market driver, particularly as global efforts to reduce dependency on precious metals intensify. The hydrogen economy's expansion, with projected investments exceeding 300 billion USD globally by 2030, provides substantial market opportunities for AEM technologies.
Water treatment applications constitute another major market segment for AEMs, with electrodialysis and desalination processes increasingly adopting these membranes. The global water treatment market, valued at 265 billion USD in 2022, is expected to grow at 7.5% annually, with AEM technologies capturing an expanding share due to their energy efficiency and operational advantages over conventional technologies.
Regionally, North America and Europe currently dominate the AEM market, accounting for approximately 60% of global demand. However, Asia-Pacific represents the fastest-growing region, with China, Japan, and South Korea making substantial investments in hydrogen technologies and advanced water treatment solutions. The Chinese government's commitment to achieving carbon neutrality by 2060 has catalyzed significant funding for fuel cell technologies, creating favorable market conditions for AEM adoption.
Industry segmentation reveals that transportation applications, particularly in the automotive sector, represent the largest end-use market for AEMs, followed by stationary power generation and industrial processes. The commercial vehicle segment, including buses and trucks, shows particularly strong growth potential for AEM fuel cells due to their extended range capabilities and faster refueling compared to battery electric alternatives.
Market challenges include competition from established technologies such as proton exchange membranes and the need for further performance improvements to achieve widespread commercial adoption. Price sensitivity remains a significant factor, with end-users requiring demonstrable cost advantages over incumbent technologies to justify transition investments.
Current Challenges in Polymer AEM Development
Despite significant advancements in anion exchange membrane (AEM) technology, several critical challenges persist in polymer design that hinder widespread commercial adoption. The most prominent issue remains chemical stability, particularly in alkaline environments. Current polymer backbones and cationic functional groups undergo degradation through multiple mechanisms, including nucleophilic substitution, Hofmann elimination, and oxidative attacks, severely limiting operational lifetimes under practical conditions.
Mechanical stability presents another significant hurdle, as many high-performance polymers exhibit excessive water uptake leading to dimensional instability. This swelling behavior compromises the structural integrity of membranes during operation cycles, resulting in performance deterioration and potential system failures. The inherent trade-off between ion conductivity and mechanical properties continues to challenge material scientists seeking balanced performance metrics.
Ion conductivity limitations represent a persistent bottleneck in AEM development. While hydroxide conductivity has improved substantially in recent years, it still lags behind proton conductivity in comparable proton exchange membranes by approximately an order of magnitude. This conductivity gap stems from the intrinsically lower mobility of hydroxide ions and insufficient understanding of ion transport mechanisms within polymer matrices.
Scalable synthesis routes pose considerable manufacturing challenges. Many promising laboratory-scale polymers utilize complex, multi-step synthesis procedures with expensive reagents and environmentally problematic solvents. These approaches often yield inconsistent products with batch-to-batch variations, making quality control difficult and increasing production costs substantially.
Interface compatibility issues emerge when integrating AEMs with electrodes and other fuel cell components. Poor interfacial contact leads to increased resistance and accelerated degradation at critical junctions. Current polymer designs frequently fail to address these interfacial phenomena, focusing primarily on bulk membrane properties rather than system-level integration.
Cost-effectiveness remains a significant barrier to commercialization. The specialized monomers, complex synthesis procedures, and limited economies of scale contribute to prohibitively high production costs compared to established technologies. Additionally, the performance-durability-cost triangle presents difficult optimization challenges that have not been adequately resolved with current polymer designs.
Environmental considerations are increasingly important, with many current synthesis approaches utilizing hazardous chemicals and generating substantial waste streams. Sustainable alternatives that maintain performance while reducing environmental impact represent an emerging challenge in the field that requires innovative approaches to green chemistry and manufacturing.
Mechanical stability presents another significant hurdle, as many high-performance polymers exhibit excessive water uptake leading to dimensional instability. This swelling behavior compromises the structural integrity of membranes during operation cycles, resulting in performance deterioration and potential system failures. The inherent trade-off between ion conductivity and mechanical properties continues to challenge material scientists seeking balanced performance metrics.
Ion conductivity limitations represent a persistent bottleneck in AEM development. While hydroxide conductivity has improved substantially in recent years, it still lags behind proton conductivity in comparable proton exchange membranes by approximately an order of magnitude. This conductivity gap stems from the intrinsically lower mobility of hydroxide ions and insufficient understanding of ion transport mechanisms within polymer matrices.
Scalable synthesis routes pose considerable manufacturing challenges. Many promising laboratory-scale polymers utilize complex, multi-step synthesis procedures with expensive reagents and environmentally problematic solvents. These approaches often yield inconsistent products with batch-to-batch variations, making quality control difficult and increasing production costs substantially.
Interface compatibility issues emerge when integrating AEMs with electrodes and other fuel cell components. Poor interfacial contact leads to increased resistance and accelerated degradation at critical junctions. Current polymer designs frequently fail to address these interfacial phenomena, focusing primarily on bulk membrane properties rather than system-level integration.
Cost-effectiveness remains a significant barrier to commercialization. The specialized monomers, complex synthesis procedures, and limited economies of scale contribute to prohibitively high production costs compared to established technologies. Additionally, the performance-durability-cost triangle presents difficult optimization challenges that have not been adequately resolved with current polymer designs.
Environmental considerations are increasingly important, with many current synthesis approaches utilizing hazardous chemicals and generating substantial waste streams. Sustainable alternatives that maintain performance while reducing environmental impact represent an emerging challenge in the field that requires innovative approaches to green chemistry and manufacturing.
State-of-the-Art Polymer AEM Solutions
01 Quaternary ammonium functionalized polymers
Quaternary ammonium functional groups are commonly incorporated into polymer backbones to create anion exchange membranes with high ionic conductivity. These functional groups provide positively charged sites that facilitate the transport of anions across the membrane. Various polymer architectures including block copolymers and crosslinked networks can be functionalized with quaternary ammonium groups to achieve desired mechanical stability and ion exchange capacity.- Quaternary ammonium functionalized polymers: Quaternary ammonium functional groups are commonly incorporated into polymer backbones to create anion exchange membranes with high ionic conductivity. These functional groups provide positively charged sites that facilitate the transport of anions across the membrane. Various polymer architectures including block copolymers and crosslinked networks can be functionalized with quaternary ammonium groups to enhance membrane performance and stability in alkaline environments.
- Crosslinked polymer networks for enhanced stability: Crosslinking strategies are employed in anion exchange membrane design to improve mechanical strength, dimensional stability, and chemical resistance. These networks can be formed through various mechanisms including thermal crosslinking, radiation-induced crosslinking, or chemical crosslinking agents. The crosslinked structure helps maintain membrane integrity under operating conditions while balancing ion conductivity requirements for efficient anion transport.
- Block copolymer architectures for phase separation: Block copolymer designs utilize microphase separation to create distinct hydrophilic ion-conducting channels and hydrophobic mechanical support domains. This architecture allows for optimized ion transport pathways while maintaining structural integrity. Various block configurations including diblock, triblock, and multiblock structures can be tailored to achieve the desired balance between conductivity and mechanical properties in anion exchange membranes.
- Alkaline-stable polymer backbones: Development of polymer backbones with enhanced resistance to alkaline degradation is crucial for long-term membrane stability. Polymers containing aromatic rings, fluorinated segments, or sterically hindered structures demonstrate improved chemical stability under high pH conditions. These alkaline-stable backbones help prevent membrane degradation and maintain performance during extended operation in fuel cells and electrolysis applications.
- Composite and hybrid membrane structures: Composite and hybrid approaches combine different materials to leverage their complementary properties in anion exchange membranes. These designs may incorporate inorganic fillers, reinforcement fibers, or multiple polymer layers to enhance mechanical strength, conductivity, and selectivity. Hybrid structures can also include interpenetrating polymer networks or pore-filling strategies to optimize the balance between ion transport and structural integrity.
02 Crosslinked polymer networks for enhanced stability
Crosslinking strategies are employed in anion exchange membrane design to improve chemical and mechanical stability under alkaline conditions. These networks help maintain dimensional stability during hydration/dehydration cycles and resist degradation in high pH environments. Various crosslinking agents and techniques can be used to create robust polymer networks while maintaining sufficient ion conductivity for practical applications.Expand Specific Solutions03 Block copolymer architectures for phase separation
Block copolymer designs utilize microphase separation to create distinct hydrophilic ion-conducting channels and hydrophobic mechanical support domains. This architecture allows for simultaneous optimization of ion conductivity and mechanical properties. By controlling the block length ratios and chemistry, researchers can tune the morphology of the membrane to create efficient ion transport pathways while maintaining structural integrity.Expand Specific Solutions04 Alkaline-stable polymer backbones
Development of polymer backbones with enhanced resistance to alkaline degradation is crucial for long-term membrane performance. Fluorinated polymers, aromatic polymers, and certain aliphatic structures demonstrate superior stability in high pH environments. These alkaline-stable backbones prevent degradation mechanisms such as Hofmann elimination and nucleophilic substitution that typically limit the lifetime of anion exchange membranes in fuel cell and electrolyzer applications.Expand Specific Solutions05 Novel cation groups beyond quaternary ammonium
Alternative cationic functional groups are being explored to overcome the stability limitations of traditional quaternary ammonium groups. These include phosphonium, imidazolium, guanidinium, and metal-organic frameworks that can provide improved alkaline stability while maintaining high anion conductivity. These novel cation groups offer different degradation resistance profiles and can be tailored to specific operating conditions and applications.Expand Specific Solutions
Leading Organizations in AEM Research and Production
The anion exchange membrane (AEM) technology landscape is currently in a growth phase, with increasing market adoption driven by applications in fuel cells, electrolyzers, and energy storage systems. The global market is expanding rapidly, projected to reach significant scale as clean energy technologies gain traction. Technologically, the field shows moderate maturity with ongoing innovation addressing key challenges of chemical stability, conductivity, and durability. Leading players include established chemical companies like BASF Corp. and AGC Inc., who leverage their materials expertise, alongside specialized innovators such as Bettergy Corp. and POCell Tech focusing on novel polymer designs. Academic-industrial partnerships are prominent, with institutions like Rensselaer Polytechnic Institute and Georgia Tech collaborating with industry. Asian companies, particularly from South Korea (SK Innovation, Hanwha Solutions) and China (Huaneng Clean Energy Research Institute), are increasingly active in this space, indicating the global competitive nature of AEM development.
Promerus LLC
Technical Solution: Promerus has pioneered the development of polynorbornene-based anion exchange membranes with unique structural advantages. Their approach leverages the rigid bicyclic structure of norbornene derivatives to create polymers with exceptional dimensional stability and mechanical strength. The company's proprietary polymerization technology allows precise control over the polymer architecture, enabling the strategic placement of quaternary ammonium functional groups along the polymer backbone. This results in well-defined ion transport pathways while minimizing susceptibility to alkaline degradation. Promerus has developed a series of membranes with varying ion exchange capacities (1.5-2.5 meq/g) to address different application requirements. Their latest innovation involves the incorporation of sterically hindered quaternary ammonium groups that demonstrate significantly improved alkaline stability, with less than 5% degradation after 1000 hours in 1M KOH at 80°C.
Strengths: Exceptional dimensional stability due to rigid polynorbornene backbone; tunable ion exchange capacity for application-specific optimization; proprietary chemistry for enhanced alkaline stability. Weaknesses: Higher material costs compared to conventional polymers; limited commercial-scale production experience; potential challenges in processing due to polymer rigidity.
Toray Advanced Materials Co., Ltd
Technical Solution: Toray has developed a novel approach to anion exchange membranes based on fluorinated aromatic polymers with pendant quaternary ammonium groups. Their technology utilizes a multi-block copolymer architecture where hydrophobic fluorinated segments provide mechanical strength and chemical stability, while hydrophilic segments containing quaternary ammonium groups facilitate ion transport. Toray's proprietary synthesis method involves controlled radical polymerization to achieve precise molecular weight distribution and block length, resulting in optimized microphase separation. Their latest generation AEMs demonstrate hydroxide conductivity of 120-150 mS/cm at 80°C with alkaline stability maintained for over 2000 hours at pH 14. Toray has also developed composite membranes incorporating nanoparticles that enhance water retention and ion transport properties, particularly beneficial for operation under low humidity conditions.
Strengths: Exceptional hydroxide conductivity; superior alkaline stability due to fluorinated backbone; excellent mechanical properties allowing for thinner membranes. Weaknesses: Complex synthesis process increases manufacturing costs; fluorinated materials raise environmental concerns regarding end-of-life disposal; potential brittleness issues at low humidity conditions.
Key Innovations in AEM Polymer Chemistry
Halo-containing anion exchange membranes and methods thereof
PatentActiveUS10294325B2
Innovation
- Development of polymer compounds with a poly(phenylene) structure incorporating a cationic moiety and/or a halo group to enhance hydrophobicity and anion binding, thereby reducing water affinity and improving membrane performance.
Polyfluorene-based anion exchange composite membrane and method for preparing same
PatentPendingUS20240100490A1
Innovation
- A polyfluorene-based anion exchange composite membrane is developed, featuring a porous polymer support with a cross-linked polyfluorene-based copolymer or copolymer ionomer, where the support is fluorinated or hydrophilized, and a cosolvent is used to improve impregnation, enhancing mechanical and dimensional stability.
Sustainability Aspects of Advanced AEM Polymers
The sustainability implications of advanced anion exchange membrane (AEM) polymers represent a critical dimension in evaluating their long-term viability and environmental impact. Current polymer designs for AEMs have predominantly focused on performance metrics such as ionic conductivity and chemical stability, often overlooking sustainability considerations that are increasingly important in the clean energy transition.
Advanced AEM polymers offer significant sustainability advantages compared to traditional proton exchange membrane technologies, particularly through the elimination of precious metal catalysts like platinum. This reduction in critical raw material dependency aligns with circular economy principles and reduces supply chain vulnerabilities associated with geopolitically sensitive resources.
Life cycle assessment (LCA) studies of emerging AEM polymers reveal promising environmental profiles, with potential reductions in carbon footprint ranging from 30-45% compared to conventional alternatives. However, these benefits are partially offset by the complex synthesis routes of some advanced polymers, which may involve toxic reagents or energy-intensive processing steps that compromise their overall environmental credentials.
Biodegradability remains a significant challenge for most high-performance AEM polymers. The very chemical stability that makes these materials attractive for long-term electrochemical applications also renders them persistent in the environment. Recent innovations incorporating bio-based building blocks and degradable linkages show promise but typically come with performance trade-offs that limit commercial viability.
Manufacturing scalability presents another sustainability dimension, with many laboratory-scale polymer designs requiring synthesis pathways that are difficult to scale without substantial solvent usage or specialized equipment. Advances in green chemistry approaches, including aqueous processing methods and catalyst-free polymerization, are beginning to address these concerns.
End-of-life management strategies for AEM devices remain underdeveloped, with limited recycling infrastructure capable of handling these specialized materials. Research into design-for-disassembly approaches and recovery of valuable ionic groups could significantly improve the circular economy potential of these technologies.
Regulatory frameworks are increasingly influencing polymer design choices, with restrictions on certain fluorinated compounds and halogenated materials driving innovation toward more environmentally benign alternatives. Forward-thinking polymer chemists are now incorporating regulatory compliance into early-stage material design rather than as an afterthought.
The economic sustainability of advanced AEM polymers ultimately depends on achieving cost parity with incumbent technologies while delivering superior environmental performance. Current cost analyses suggest that while material costs remain higher, the system-level economics increasingly favor AEM approaches when considering full lifecycle impacts and operational benefits.
Advanced AEM polymers offer significant sustainability advantages compared to traditional proton exchange membrane technologies, particularly through the elimination of precious metal catalysts like platinum. This reduction in critical raw material dependency aligns with circular economy principles and reduces supply chain vulnerabilities associated with geopolitically sensitive resources.
Life cycle assessment (LCA) studies of emerging AEM polymers reveal promising environmental profiles, with potential reductions in carbon footprint ranging from 30-45% compared to conventional alternatives. However, these benefits are partially offset by the complex synthesis routes of some advanced polymers, which may involve toxic reagents or energy-intensive processing steps that compromise their overall environmental credentials.
Biodegradability remains a significant challenge for most high-performance AEM polymers. The very chemical stability that makes these materials attractive for long-term electrochemical applications also renders them persistent in the environment. Recent innovations incorporating bio-based building blocks and degradable linkages show promise but typically come with performance trade-offs that limit commercial viability.
Manufacturing scalability presents another sustainability dimension, with many laboratory-scale polymer designs requiring synthesis pathways that are difficult to scale without substantial solvent usage or specialized equipment. Advances in green chemistry approaches, including aqueous processing methods and catalyst-free polymerization, are beginning to address these concerns.
End-of-life management strategies for AEM devices remain underdeveloped, with limited recycling infrastructure capable of handling these specialized materials. Research into design-for-disassembly approaches and recovery of valuable ionic groups could significantly improve the circular economy potential of these technologies.
Regulatory frameworks are increasingly influencing polymer design choices, with restrictions on certain fluorinated compounds and halogenated materials driving innovation toward more environmentally benign alternatives. Forward-thinking polymer chemists are now incorporating regulatory compliance into early-stage material design rather than as an afterthought.
The economic sustainability of advanced AEM polymers ultimately depends on achieving cost parity with incumbent technologies while delivering superior environmental performance. Current cost analyses suggest that while material costs remain higher, the system-level economics increasingly favor AEM approaches when considering full lifecycle impacts and operational benefits.
Standardization and Testing Protocols for AEMs
The standardization and testing protocols for Anion Exchange Membranes (AEMs) represent a critical yet underdeveloped aspect of the field. Despite significant advances in polymer design for AEMs, the lack of universally accepted testing methodologies has hindered meaningful comparison between different membrane materials and slowed overall progress in the field.
Currently, researchers employ diverse testing conditions including varying temperatures (typically 30-80°C), different hydroxide concentrations, and inconsistent humidity levels. This variability makes direct comparison between published results nearly impossible, creating significant barriers to identifying truly superior membrane designs.
The ion conductivity measurement protocols particularly suffer from inconsistency. Some researchers report conductivity in fully hydrated conditions while others use partially hydrated or controlled humidity environments. The counter-ion used during measurement (Cl-, HCO3-, or OH-) also varies significantly across studies, further complicating comparative analysis.
Mechanical stability testing faces similar challenges, with no standardized protocols for measuring dimensional changes during hydration/dehydration cycles or mechanical strength under operating conditions. The absence of accelerated durability tests that correlate with real-world performance represents another significant gap.
Several international organizations have begun addressing these issues. The International Electrotechnical Commission (IEC) has established working groups focused on developing standardized testing protocols specifically for AEMs. Similarly, ASTM International has initiated efforts to create testing standards that address the unique characteristics of anion exchange membranes.
The U.S. Department of Energy has funded initiatives through its Fuel Cell Technologies Office to develop benchmark testing protocols. These efforts aim to establish standardized methodologies for measuring key performance indicators including hydroxide conductivity, chemical stability, mechanical properties, and gas crossover rates.
Industry-academic partnerships have emerged as crucial drivers of standardization efforts. Companies like Dioxide Materials, Ionomr Innovations, and Fumatech have collaborated with academic institutions to develop reference materials and testing protocols that could serve as industry benchmarks.
Moving forward, the establishment of round-robin testing programs involving multiple laboratories will be essential for validating proposed standardized methods. Additionally, the development of reference AEM materials with well-characterized properties would provide valuable benchmarks against which new membranes could be evaluated.
Currently, researchers employ diverse testing conditions including varying temperatures (typically 30-80°C), different hydroxide concentrations, and inconsistent humidity levels. This variability makes direct comparison between published results nearly impossible, creating significant barriers to identifying truly superior membrane designs.
The ion conductivity measurement protocols particularly suffer from inconsistency. Some researchers report conductivity in fully hydrated conditions while others use partially hydrated or controlled humidity environments. The counter-ion used during measurement (Cl-, HCO3-, or OH-) also varies significantly across studies, further complicating comparative analysis.
Mechanical stability testing faces similar challenges, with no standardized protocols for measuring dimensional changes during hydration/dehydration cycles or mechanical strength under operating conditions. The absence of accelerated durability tests that correlate with real-world performance represents another significant gap.
Several international organizations have begun addressing these issues. The International Electrotechnical Commission (IEC) has established working groups focused on developing standardized testing protocols specifically for AEMs. Similarly, ASTM International has initiated efforts to create testing standards that address the unique characteristics of anion exchange membranes.
The U.S. Department of Energy has funded initiatives through its Fuel Cell Technologies Office to develop benchmark testing protocols. These efforts aim to establish standardized methodologies for measuring key performance indicators including hydroxide conductivity, chemical stability, mechanical properties, and gas crossover rates.
Industry-academic partnerships have emerged as crucial drivers of standardization efforts. Companies like Dioxide Materials, Ionomr Innovations, and Fumatech have collaborated with academic institutions to develop reference materials and testing protocols that could serve as industry benchmarks.
Moving forward, the establishment of round-robin testing programs involving multiple laboratories will be essential for validating proposed standardized methods. Additionally, the development of reference AEM materials with well-characterized properties would provide valuable benchmarks against which new membranes could be evaluated.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!