Chemical stability challenges in anion exchange membranes
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
AEM Technology Background and Objectives
Anion exchange membranes (AEMs) have emerged as a promising alternative to proton exchange membranes in various electrochemical applications, particularly in fuel cells, electrolyzers, and flow batteries. The development of AEMs can be traced back to the 1960s, but significant advancements have only been achieved in the past two decades. The primary attraction of AEM-based systems lies in their ability to operate under alkaline conditions, which enables the use of non-precious metal catalysts, thereby substantially reducing system costs compared to acidic proton exchange membrane systems.
The evolution of AEM technology has been driven by the need for sustainable energy conversion and storage solutions. Initially, AEMs suffered from poor ionic conductivity and mechanical properties, limiting their practical applications. However, continuous research efforts have led to remarkable improvements in these aspects, with contemporary AEMs exhibiting conductivities approaching those of proton exchange membranes.
Despite these advancements, chemical stability remains the Achilles' heel of AEM technology. Under alkaline operating conditions, AEMs are susceptible to degradation through various mechanisms, including nucleophilic substitution, Hofmann elimination, and direct attack on the polymer backbone. These degradation pathways significantly compromise the long-term performance and durability of AEM-based systems, hindering their commercial viability.
The technical objectives in addressing chemical stability challenges in AEMs are multifaceted. First, there is a critical need to develop fundamental understanding of degradation mechanisms at the molecular level, which requires advanced characterization techniques and computational modeling. Second, innovative molecular designs are needed to enhance the stability of both the polymer backbone and the cationic functional groups against hydroxide attack.
Additionally, researchers aim to establish standardized protocols for evaluating AEM stability, as the current lack of consistency in testing conditions makes it difficult to compare different materials and assess progress. The ultimate goal is to develop AEMs that maintain high ionic conductivity (>100 mS/cm) while demonstrating chemical stability for at least 40,000 hours under operating conditions, which is the benchmark for commercial viability in fuel cell applications.
The technology trend indicates a shift from conventional quaternary ammonium-based AEMs toward more stable cationic groups such as guanidinium, phosphonium, and metal-cation coordinated structures. Simultaneously, there is growing interest in composite and hybrid materials that combine the advantages of different components to achieve enhanced stability without compromising other essential properties.
The evolution of AEM technology has been driven by the need for sustainable energy conversion and storage solutions. Initially, AEMs suffered from poor ionic conductivity and mechanical properties, limiting their practical applications. However, continuous research efforts have led to remarkable improvements in these aspects, with contemporary AEMs exhibiting conductivities approaching those of proton exchange membranes.
Despite these advancements, chemical stability remains the Achilles' heel of AEM technology. Under alkaline operating conditions, AEMs are susceptible to degradation through various mechanisms, including nucleophilic substitution, Hofmann elimination, and direct attack on the polymer backbone. These degradation pathways significantly compromise the long-term performance and durability of AEM-based systems, hindering their commercial viability.
The technical objectives in addressing chemical stability challenges in AEMs are multifaceted. First, there is a critical need to develop fundamental understanding of degradation mechanisms at the molecular level, which requires advanced characterization techniques and computational modeling. Second, innovative molecular designs are needed to enhance the stability of both the polymer backbone and the cationic functional groups against hydroxide attack.
Additionally, researchers aim to establish standardized protocols for evaluating AEM stability, as the current lack of consistency in testing conditions makes it difficult to compare different materials and assess progress. The ultimate goal is to develop AEMs that maintain high ionic conductivity (>100 mS/cm) while demonstrating chemical stability for at least 40,000 hours under operating conditions, which is the benchmark for commercial viability in fuel cell applications.
The technology trend indicates a shift from conventional quaternary ammonium-based AEMs toward more stable cationic groups such as guanidinium, phosphonium, and metal-cation coordinated structures. Simultaneously, there is growing interest in composite and hybrid materials that combine the advantages of different components to achieve enhanced stability without compromising other essential properties.
Market Analysis for AEM Applications
The anion exchange membrane (AEM) market is experiencing significant growth driven by increasing demand for clean energy solutions and sustainable water treatment technologies. The global AEM market was valued at approximately 120 million USD in 2022 and is projected to reach 320 million USD by 2030, representing a compound annual growth rate of 13.1% during the forecast period.
Fuel cell applications currently dominate the AEM market, accounting for nearly 45% of the total market share. The push for hydrogen-based energy solutions, particularly in transportation and stationary power generation, has accelerated demand for AEM fuel cells. Major automotive manufacturers including Toyota, Hyundai, and Honda have increased investments in AEM fuel cell technology as an alternative to proton exchange membrane (PEM) systems due to potential cost advantages and reduced dependency on platinum catalysts.
Water electrolysis represents the second-largest application segment, comprising approximately 30% of the market. The growing hydrogen economy and renewable energy integration have spurred interest in AEM electrolyzers as efficient hydrogen production systems. Several companies including Enapter, Hydrogenics, and ITM Power have launched commercial AEM electrolyzer products, highlighting the technology's transition from research to commercialization.
Water treatment applications constitute about 15% of the AEM market, with desalination and industrial wastewater treatment being primary drivers. The remaining 10% encompasses emerging applications such as flow batteries, CO2 reduction systems, and specialized separation processes.
Regionally, North America leads the market with 38% share, followed by Europe (32%), Asia-Pacific (25%), and rest of the world (5%). However, the Asia-Pacific region is expected to witness the fastest growth rate of 15.2% annually, driven by China's aggressive clean energy policies and Japan's hydrogen economy initiatives.
A critical market constraint remains the chemical stability challenges of AEMs, which directly impact product lifetime and performance reliability. End-users consistently cite membrane degradation as the primary barrier to wider adoption, with surveys indicating that 78% of potential industrial adopters require membrane lifetimes exceeding 40,000 hours for stationary applications and 5,000 hours for mobile applications.
The competitive landscape features both specialized membrane manufacturers like Fumatech, Tokuyama, and Dioxide Materials, alongside diversified chemical companies including 3M, Solvay, and Asahi Kasei that have entered the space. Recent market consolidation through acquisitions signals growing commercial interest, with four major acquisitions of AEM technology startups occurring in the past three years.
Fuel cell applications currently dominate the AEM market, accounting for nearly 45% of the total market share. The push for hydrogen-based energy solutions, particularly in transportation and stationary power generation, has accelerated demand for AEM fuel cells. Major automotive manufacturers including Toyota, Hyundai, and Honda have increased investments in AEM fuel cell technology as an alternative to proton exchange membrane (PEM) systems due to potential cost advantages and reduced dependency on platinum catalysts.
Water electrolysis represents the second-largest application segment, comprising approximately 30% of the market. The growing hydrogen economy and renewable energy integration have spurred interest in AEM electrolyzers as efficient hydrogen production systems. Several companies including Enapter, Hydrogenics, and ITM Power have launched commercial AEM electrolyzer products, highlighting the technology's transition from research to commercialization.
Water treatment applications constitute about 15% of the AEM market, with desalination and industrial wastewater treatment being primary drivers. The remaining 10% encompasses emerging applications such as flow batteries, CO2 reduction systems, and specialized separation processes.
Regionally, North America leads the market with 38% share, followed by Europe (32%), Asia-Pacific (25%), and rest of the world (5%). However, the Asia-Pacific region is expected to witness the fastest growth rate of 15.2% annually, driven by China's aggressive clean energy policies and Japan's hydrogen economy initiatives.
A critical market constraint remains the chemical stability challenges of AEMs, which directly impact product lifetime and performance reliability. End-users consistently cite membrane degradation as the primary barrier to wider adoption, with surveys indicating that 78% of potential industrial adopters require membrane lifetimes exceeding 40,000 hours for stationary applications and 5,000 hours for mobile applications.
The competitive landscape features both specialized membrane manufacturers like Fumatech, Tokuyama, and Dioxide Materials, alongside diversified chemical companies including 3M, Solvay, and Asahi Kasei that have entered the space. Recent market consolidation through acquisitions signals growing commercial interest, with four major acquisitions of AEM technology startups occurring in the past three years.
Current Challenges in AEM Chemical Stability
Anion exchange membranes (AEMs) face significant chemical stability challenges that currently limit their widespread application in fuel cells, electrolyzers, and other electrochemical devices. The primary degradation mechanism involves nucleophilic attack by hydroxide ions on the cationic functional groups, particularly under the alkaline operating conditions necessary for these applications. This degradation leads to decreased ion conductivity, mechanical integrity loss, and ultimately, device failure.
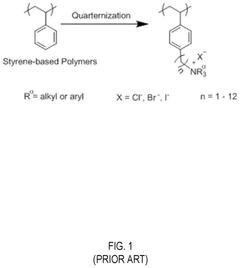
The quaternary ammonium groups, commonly used as ion exchange sites in AEMs, are particularly susceptible to degradation through Hofmann elimination, nucleophilic substitution, and ylide formation pathways. These reactions occur when hydroxide ions attack the β-hydrogen atoms or directly substitute at the α-carbon positions, resulting in the detachment of functional groups and membrane performance deterioration.
Temperature exacerbates these stability issues significantly. Most current AEMs show acceptable stability at ambient temperatures but degrade rapidly at the elevated temperatures (60-80°C) required for efficient electrochemical device operation. This temperature-dependent degradation creates a challenging performance-stability tradeoff that has yet to be satisfactorily resolved.
Water management presents another critical challenge for AEM stability. Dehydration can accelerate degradation processes by increasing the local concentration of hydroxide ions, while excessive hydration leads to dimensional instability and mechanical failures. This delicate balance between hydration levels necessary for ion transport and those that maintain chemical stability remains difficult to achieve consistently.
The presence of carbon dioxide further complicates AEM operation by converting hydroxide ions to carbonate and bicarbonate ions, reducing ionic conductivity and creating additional chemical stress on the membrane structure. This "carbonation" effect is particularly problematic in fuel cell applications where atmospheric CO₂ exposure is unavoidable.
Current testing protocols for AEM stability lack standardization, making direct comparisons between different membrane technologies challenging. Accelerated stress tests often fail to accurately predict real-world performance, while long-term stability studies are time-consuming and expensive. This testing inconsistency hampers rapid development and validation of more stable membrane materials.
Manufacturing scalability of chemically stable AEMs represents another significant hurdle. Laboratory-scale synthesis methods that produce highly stable membranes often involve complex, multi-step processes that are difficult to scale industrially. The trade-off between chemical stability, mechanical properties, and cost-effective production remains a central challenge in AEM development.
The quaternary ammonium groups, commonly used as ion exchange sites in AEMs, are particularly susceptible to degradation through Hofmann elimination, nucleophilic substitution, and ylide formation pathways. These reactions occur when hydroxide ions attack the β-hydrogen atoms or directly substitute at the α-carbon positions, resulting in the detachment of functional groups and membrane performance deterioration.
Temperature exacerbates these stability issues significantly. Most current AEMs show acceptable stability at ambient temperatures but degrade rapidly at the elevated temperatures (60-80°C) required for efficient electrochemical device operation. This temperature-dependent degradation creates a challenging performance-stability tradeoff that has yet to be satisfactorily resolved.
Water management presents another critical challenge for AEM stability. Dehydration can accelerate degradation processes by increasing the local concentration of hydroxide ions, while excessive hydration leads to dimensional instability and mechanical failures. This delicate balance between hydration levels necessary for ion transport and those that maintain chemical stability remains difficult to achieve consistently.
The presence of carbon dioxide further complicates AEM operation by converting hydroxide ions to carbonate and bicarbonate ions, reducing ionic conductivity and creating additional chemical stress on the membrane structure. This "carbonation" effect is particularly problematic in fuel cell applications where atmospheric CO₂ exposure is unavoidable.
Current testing protocols for AEM stability lack standardization, making direct comparisons between different membrane technologies challenging. Accelerated stress tests often fail to accurately predict real-world performance, while long-term stability studies are time-consuming and expensive. This testing inconsistency hampers rapid development and validation of more stable membrane materials.
Manufacturing scalability of chemically stable AEMs represents another significant hurdle. Laboratory-scale synthesis methods that produce highly stable membranes often involve complex, multi-step processes that are difficult to scale industrially. The trade-off between chemical stability, mechanical properties, and cost-effective production remains a central challenge in AEM development.
Current Solutions for AEM Chemical Stability Enhancement
01 Polymer modification for enhanced chemical stability
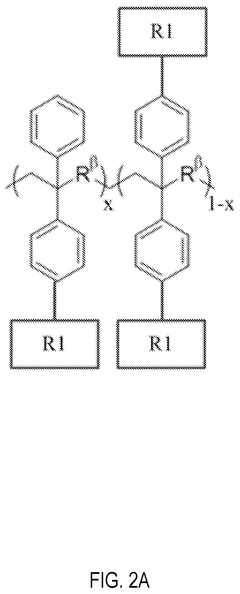
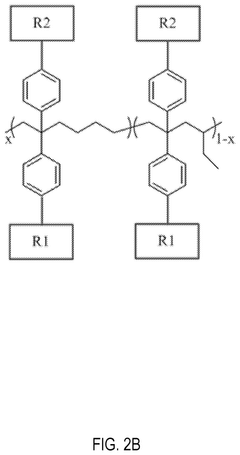
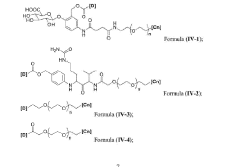
Chemical stability of anion exchange membranes can be improved through polymer modification techniques. These include crosslinking of polymer chains, incorporation of stable functional groups, and development of composite structures. Modified polymers show increased resistance to degradation in alkaline environments, which is crucial for applications in fuel cells and electrolysis. These modifications help maintain ion conductivity while preventing membrane deterioration during long-term operation.- Polymer backbone modifications for enhanced stability: Modifying the polymer backbone structure of anion exchange membranes can significantly improve their chemical stability. Approaches include incorporating fluorinated segments, aromatic rings, or crosslinking structures that resist degradation in alkaline environments. These modifications help prevent backbone scission and maintain membrane integrity during long-term operation in harsh conditions, resulting in membranes with extended service life and consistent performance.
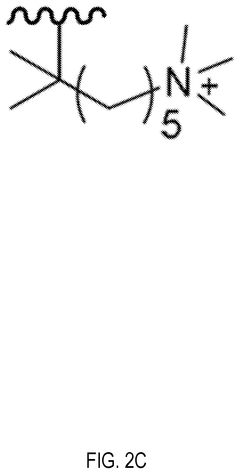
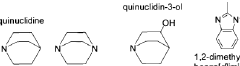
- Cation group design for alkaline resistance: The design of cation functional groups plays a crucial role in the chemical stability of anion exchange membranes. Novel cation structures such as sterically hindered quaternary ammonium groups, imidazolium derivatives, and phosphonium-based functional groups can resist nucleophilic attack and hydroxide degradation. These advanced cation designs minimize the Hofmann elimination reaction and other degradation pathways that typically limit membrane longevity in alkaline environments.
- Composite and hybrid membrane structures: Composite and hybrid membrane structures combine different materials to enhance chemical stability while maintaining ion conductivity. These membranes often incorporate inorganic components such as metal oxides, layered double hydroxides, or nanoparticles within a polymer matrix. The inorganic components provide additional stability against chemical degradation, while the polymer matrix ensures flexibility and processability. This synergistic approach results in membranes with superior alkaline resistance and mechanical properties.
- Protective coatings and surface treatments: Applying protective coatings or surface treatments to anion exchange membranes can significantly improve their chemical stability. These treatments create a barrier against aggressive chemical species while maintaining ion transport capabilities. Methods include plasma treatment, layer-by-layer deposition of protective polymers, and grafting of chemically resistant functional groups onto the membrane surface. These approaches effectively extend membrane lifetime by protecting vulnerable components from direct exposure to harsh alkaline environments.
- Stabilizing additives and reinforcement materials: Incorporating stabilizing additives and reinforcement materials into anion exchange membranes can enhance their chemical durability. Additives such as antioxidants, radical scavengers, and alkaline-resistant fillers help neutralize degradative species and maintain membrane integrity. Reinforcement materials like nanofibers, carbon-based materials, or high-strength polymers provide mechanical support that prevents physical deterioration during chemical attack. These components work together to create membranes with improved dimensional stability and resistance to chemical degradation.
02 Quaternary ammonium functional groups optimization
Quaternary ammonium groups are commonly used in anion exchange membranes but are susceptible to degradation. Optimization strategies include using sterically hindered quaternary ammonium structures, incorporating bulky substituents to protect the cationic sites, and developing new quaternary ammonium derivatives with enhanced stability. These approaches minimize the Hofmann elimination reaction and nucleophilic substitution that typically cause degradation in alkaline environments.Expand Specific Solutions03 Reinforcement with inorganic materials
Incorporating inorganic materials into anion exchange membranes significantly improves their chemical stability. Materials such as metal oxides, silica, and layered double hydroxides create composite membranes with enhanced resistance to chemical degradation. These inorganic components act as stabilizers and can reduce water uptake while maintaining adequate ion conductivity. The resulting hybrid membranes demonstrate superior durability in harsh alkaline conditions compared to conventional polymer-only membranes.Expand Specific Solutions04 Novel polymer backbones for stability
Development of novel polymer backbones represents a fundamental approach to improving the chemical stability of anion exchange membranes. Fluorinated polymers, aromatic polymers, and polybenzimidazole-based materials demonstrate exceptional resistance to chemical degradation. These advanced polymer structures maintain their mechanical integrity and functional properties even after prolonged exposure to alkaline environments, making them suitable for demanding electrochemical applications.Expand Specific Solutions05 Protective coatings and multilayer structures
Applying protective coatings or developing multilayer structures can significantly enhance the chemical stability of anion exchange membranes. These approaches involve creating barrier layers that shield the functional ion-exchange groups from aggressive chemical environments. Thin-film composite designs, surface modification techniques, and laminated structures help extend membrane lifetime by preventing direct contact between vulnerable membrane components and degrading species while maintaining essential ion transport properties.Expand Specific Solutions
Leading Companies and Research Institutions in AEM Development
The chemical stability challenges in anion exchange membranes represent a critical technological hurdle in the emerging clean energy sector. This field is currently in an early growth phase, with market projections indicating significant expansion as fuel cell and electrolysis technologies mature. Leading research institutions including Wuhan University of Technology, Dalian Institute of Chemical Physics, and Georgia Tech Research Corp. are advancing fundamental solutions, while commercial players like Tokuyama Corp., Nitto Denko, and BASF are developing practical applications. The competitive landscape features a mix of established chemical companies and specialized startups, with academic-industrial partnerships becoming increasingly important. Technical maturity remains moderate, with companies like UOP LLC and Toray Advanced Materials focusing on improving membrane durability and alkaline stability for commercial viability in energy storage and conversion applications.
Nitto Denko Corp.
Technical Solution: Nitto Denko has developed proprietary hydrocarbon-based anion exchange membranes with enhanced chemical stability through strategic molecular design. Their approach focuses on quaternary ammonium functional groups attached to aromatic polymer backbones with sterically hindered structures that protect the cationic sites from hydroxide attack. The company employs cross-linking techniques to create three-dimensional networks that restrict chain mobility and reduce degradation pathways. Their membranes incorporate fluorinated segments in the polymer backbone to enhance hydrophobicity while maintaining adequate ion conductivity. Nitto's latest generation membranes demonstrate stability in alkaline conditions (1M KOH) at 80°C for over 1000 hours with less than 10% conductivity loss, representing significant improvement over previous generations. The company has also developed specialized coating technologies that create protective layers on membrane surfaces to further enhance durability in real-world applications.
Strengths: Superior chemical stability in highly alkaline environments compared to many competitors; excellent mechanical properties allowing for thinner membranes with lower resistance; established manufacturing infrastructure enabling scale-up. Weaknesses: Higher production costs compared to hydrocarbon alternatives; potential environmental concerns with fluorinated components; trade-off between conductivity and stability still presents challenges for high-temperature applications.
Dalian Institute of Chemical Physics of CAS
Technical Solution: Dalian Institute of Chemical Physics (DICP) has developed advanced anion exchange membranes utilizing side-chain-type quaternized poly(arylene ether sulfone) polymers with strategically positioned functional groups. Their proprietary synthesis approach incorporates bulky hydrophobic segments that create a microstructure with distinct hydrophilic and hydrophobic domains, enhancing both ion conductivity and chemical stability. DICP researchers have implemented a multi-stage crosslinking strategy that forms covalent networks resistant to dimensional changes while maintaining adequate water uptake for ion transport. Their membranes feature specially designed quaternary ammonium groups with steric hindrance that significantly reduces susceptibility to hydroxide attack through Hofmann elimination. Recent advancements include the incorporation of graphene oxide nanosheets as reinforcing fillers, which create tortuous pathways that limit hydroxide access to vulnerable cationic sites while enhancing mechanical properties. Testing has demonstrated these membranes maintain over 85% of their initial conductivity after 1000 hours in alkaline conditions (1M KOH) at 60°C, with minimal changes in mechanical properties and ion exchange capacity.
Strengths: Excellent balance of alkaline stability and hydroxide conductivity; innovative microstructure design that enhances mechanical properties; cost-effective synthesis methods suitable for scale-up. Weaknesses: Performance degradation accelerates at temperatures above 80°C; water management remains challenging in practical devices; long-term stability under variable operating conditions needs further improvement.
Key Patents and Innovations in AEM Stability
Alkaline anion exchange membranes derived from diphenylethylene and co-monomer feedstock
PatentActiveUS12303880B2
Innovation
- The development of ion exchange membrane materials using copolymers derived from diphenylalkylene and alkadiene, where the polymer backbone is functionalized with haloalkylated precursor substrates and subsequently modified with ionic groups, such as ammonium groups, to create a stable and conductive membrane structure.
Cross-linked high stable anion exchange blend membranes with polyethyleneglycols as hydrophilic membrane phase
PatentWO2017220065A1
Innovation
- The development of crosslinked highly stable anion exchange blend membranes using halomethylated polymers quaternized with sterically hindered tertiary nitrogen compounds, combined with a hydrophilic polyethylene glycol phase for enhanced conductivity and stability, and optionally incorporating sulfonated or sulfinated polymers for covalent crosslinking, to improve mechanical and chemical stability.
Environmental Impact and Sustainability of AEM Technologies
The development of anion exchange membrane (AEM) technologies represents a significant advancement in clean energy systems, particularly for fuel cells and electrolyzers. However, the environmental implications of these technologies must be thoroughly assessed to ensure their sustainability credentials align with their clean energy objectives.
AEM technologies offer several environmental advantages compared to traditional proton exchange membrane (PEM) systems. Most notably, AEMs eliminate the need for platinum group metals (PGMs) as catalysts, instead utilizing non-precious metal alternatives. This shift significantly reduces the environmental footprint associated with mining and processing these rare and environmentally damaging resources. The extraction of PGMs typically involves energy-intensive processes and generates substantial waste streams, including acid mine drainage and heavy metal contamination.
The manufacturing processes for AEMs generally require less energy and produce fewer harmful byproducts than their PEM counterparts. This advantage stems from the simpler synthesis routes and less stringent processing conditions needed for AEM production. Life cycle assessments indicate that AEM fuel cells can achieve up to 30% lower carbon footprints compared to traditional systems when considering the entire production chain.
Despite these benefits, the chemical stability challenges in AEMs present environmental concerns. The degradation of membrane materials over time necessitates more frequent replacement, potentially increasing waste generation. The alkaline degradation products from AEMs may include various organic compounds and quaternary ammonium fragments, which require proper disposal protocols to prevent environmental contamination.
Water management in AEM systems presents another sustainability consideration. While AEMs typically require less water than PEM technologies, the quality requirements differ. The alkaline operating environment can lead to precipitation of carbonates when exposed to atmospheric CO2, potentially requiring water treatment systems that consume additional energy and resources.
End-of-life management for AEM technologies remains underdeveloped compared to more established energy technologies. Current recycling methods for membrane materials are limited, though research into recovery processes for the non-precious metal catalysts shows promise. Developing closed-loop systems for AEM components represents a critical area for future sustainability improvements.
The overall environmental impact of addressing chemical stability challenges in AEMs must be evaluated holistically. While more stable membranes may require more complex chemistry and potentially more environmentally intensive manufacturing processes, these impacts must be balanced against the extended operational lifetimes and reduced replacement frequency they enable.
AEM technologies offer several environmental advantages compared to traditional proton exchange membrane (PEM) systems. Most notably, AEMs eliminate the need for platinum group metals (PGMs) as catalysts, instead utilizing non-precious metal alternatives. This shift significantly reduces the environmental footprint associated with mining and processing these rare and environmentally damaging resources. The extraction of PGMs typically involves energy-intensive processes and generates substantial waste streams, including acid mine drainage and heavy metal contamination.
The manufacturing processes for AEMs generally require less energy and produce fewer harmful byproducts than their PEM counterparts. This advantage stems from the simpler synthesis routes and less stringent processing conditions needed for AEM production. Life cycle assessments indicate that AEM fuel cells can achieve up to 30% lower carbon footprints compared to traditional systems when considering the entire production chain.
Despite these benefits, the chemical stability challenges in AEMs present environmental concerns. The degradation of membrane materials over time necessitates more frequent replacement, potentially increasing waste generation. The alkaline degradation products from AEMs may include various organic compounds and quaternary ammonium fragments, which require proper disposal protocols to prevent environmental contamination.
Water management in AEM systems presents another sustainability consideration. While AEMs typically require less water than PEM technologies, the quality requirements differ. The alkaline operating environment can lead to precipitation of carbonates when exposed to atmospheric CO2, potentially requiring water treatment systems that consume additional energy and resources.
End-of-life management for AEM technologies remains underdeveloped compared to more established energy technologies. Current recycling methods for membrane materials are limited, though research into recovery processes for the non-precious metal catalysts shows promise. Developing closed-loop systems for AEM components represents a critical area for future sustainability improvements.
The overall environmental impact of addressing chemical stability challenges in AEMs must be evaluated holistically. While more stable membranes may require more complex chemistry and potentially more environmentally intensive manufacturing processes, these impacts must be balanced against the extended operational lifetimes and reduced replacement frequency they enable.
Standardization and Testing Protocols for AEM Durability
The development of standardized testing protocols for anion exchange membrane (AEM) durability represents a critical need in advancing AEM technology toward commercial viability. Currently, the field suffers from significant inconsistencies in how researchers evaluate and report chemical stability, making cross-study comparisons nearly impossible and hindering technological progress.
A comprehensive standardization framework must address multiple degradation mechanisms simultaneously. This includes protocols for accelerated stress tests that can reliably predict membrane lifetime under various operating conditions while maintaining relevance to real-world applications. The ex-situ testing methods should incorporate controlled exposure to hydroxide environments at various temperatures, with standardized metrics for measuring degradation such as ion conductivity loss, mechanical property changes, and chemical structure alterations.
In-situ testing protocols are equally important, requiring standardized cell configurations, operating parameters, and performance metrics. These should include defined cycling procedures, load profiles, and environmental conditions that reflect realistic operational scenarios. The development of reference materials with well-characterized degradation behaviors would provide valuable benchmarks against which new membranes could be evaluated.
Several international organizations have begun addressing this standardization gap. The International Electrotechnical Commission (IEC) has established working groups focused on AEM fuel cell testing, while the American Society for Testing and Materials (ASTM) is developing specific protocols for polymer electrolyte evaluation. These efforts aim to create universally accepted methodologies that enable meaningful comparisons between different research groups and commercial entities.
The implementation of round-robin testing across multiple laboratories represents another crucial step toward standardization. Such collaborative efforts help identify procedural variables that may affect test results and establish reproducibility limits for various protocols. Recent initiatives by research consortia in Europe, North America, and Asia have begun coordinating these cross-laboratory validation studies.
Data reporting standards must also be established, specifying minimum required information regarding test conditions, membrane properties, and degradation metrics. This includes detailed documentation of membrane composition, preparation methods, pretreatment procedures, and comprehensive characterization before and after stability testing. The development of shared databases for AEM durability data would further accelerate progress by enabling meta-analyses and facilitating the identification of structure-property-durability relationships.
A comprehensive standardization framework must address multiple degradation mechanisms simultaneously. This includes protocols for accelerated stress tests that can reliably predict membrane lifetime under various operating conditions while maintaining relevance to real-world applications. The ex-situ testing methods should incorporate controlled exposure to hydroxide environments at various temperatures, with standardized metrics for measuring degradation such as ion conductivity loss, mechanical property changes, and chemical structure alterations.
In-situ testing protocols are equally important, requiring standardized cell configurations, operating parameters, and performance metrics. These should include defined cycling procedures, load profiles, and environmental conditions that reflect realistic operational scenarios. The development of reference materials with well-characterized degradation behaviors would provide valuable benchmarks against which new membranes could be evaluated.
Several international organizations have begun addressing this standardization gap. The International Electrotechnical Commission (IEC) has established working groups focused on AEM fuel cell testing, while the American Society for Testing and Materials (ASTM) is developing specific protocols for polymer electrolyte evaluation. These efforts aim to create universally accepted methodologies that enable meaningful comparisons between different research groups and commercial entities.
The implementation of round-robin testing across multiple laboratories represents another crucial step toward standardization. Such collaborative efforts help identify procedural variables that may affect test results and establish reproducibility limits for various protocols. Recent initiatives by research consortia in Europe, North America, and Asia have begun coordinating these cross-laboratory validation studies.
Data reporting standards must also be established, specifying minimum required information regarding test conditions, membrane properties, and degradation metrics. This includes detailed documentation of membrane composition, preparation methods, pretreatment procedures, and comprehensive characterization before and after stability testing. The development of shared databases for AEM durability data would further accelerate progress by enabling meta-analyses and facilitating the identification of structure-property-durability relationships.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!