Comprehensive Lifecycle Analysis of Carbon Tetrachloride Products
JUL 31, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
CCl4 Background and Objectives
Carbon tetrachloride (CCl4) has been a significant industrial chemical since its discovery in the mid-19th century. Initially used as a solvent and cleaning agent, its applications expanded to include use as a refrigerant, fire extinguishing agent, and precursor in various chemical processes. However, the recognition of its ozone-depleting properties and potential health hazards has led to a dramatic shift in its global production and use over the past few decades.
The evolution of CCl4 technology has been marked by several key milestones. Its synthesis was first reported in 1839 by Henri Victor Regnault, who produced it by chlorinating chloroform. Industrial production began in the early 20th century, with the compound finding widespread use due to its non-flammability and excellent solvent properties. The mid-20th century saw a peak in CCl4 production, driven by its use in refrigeration and as a feedstock for chlorofluorocarbon (CFC) production.
However, the 1987 Montreal Protocol marked a turning point in CCl4 history. As a potent ozone-depleting substance, CCl4 was phased out for most uses in developed countries by 1996 and in developing countries by 2010. This shift has dramatically altered the technological landscape surrounding CCl4, with research and development efforts now focused on alternatives, remediation, and monitoring technologies.
The primary objective of a comprehensive lifecycle analysis of CCl4 products is to understand the full environmental and health impacts associated with its production, use, and disposal. This analysis aims to quantify the carbon footprint, ozone depletion potential, and other environmental indicators across the entire lifecycle of CCl4-based products. Additionally, it seeks to identify potential areas for improvement in current practices and explore sustainable alternatives.
Key technological goals in this field include developing more accurate methods for detecting and measuring CCl4 in the environment, improving remediation techniques for contaminated sites, and creating environmentally friendly substitutes for remaining CCl4 applications. There is also a growing focus on understanding and mitigating the long-term effects of historical CCl4 use, particularly its persistence in the atmosphere and potential impact on global climate systems.
As global efforts to combat climate change and protect the ozone layer intensify, the importance of comprehensive lifecycle analyses for compounds like CCl4 continues to grow. These studies provide crucial data for policymakers, industry leaders, and researchers, guiding decisions on chemical management, environmental protection, and sustainable development strategies. The ongoing research in this area reflects a broader trend towards more holistic approaches to understanding and mitigating the environmental impacts of industrial chemicals.
The evolution of CCl4 technology has been marked by several key milestones. Its synthesis was first reported in 1839 by Henri Victor Regnault, who produced it by chlorinating chloroform. Industrial production began in the early 20th century, with the compound finding widespread use due to its non-flammability and excellent solvent properties. The mid-20th century saw a peak in CCl4 production, driven by its use in refrigeration and as a feedstock for chlorofluorocarbon (CFC) production.
However, the 1987 Montreal Protocol marked a turning point in CCl4 history. As a potent ozone-depleting substance, CCl4 was phased out for most uses in developed countries by 1996 and in developing countries by 2010. This shift has dramatically altered the technological landscape surrounding CCl4, with research and development efforts now focused on alternatives, remediation, and monitoring technologies.
The primary objective of a comprehensive lifecycle analysis of CCl4 products is to understand the full environmental and health impacts associated with its production, use, and disposal. This analysis aims to quantify the carbon footprint, ozone depletion potential, and other environmental indicators across the entire lifecycle of CCl4-based products. Additionally, it seeks to identify potential areas for improvement in current practices and explore sustainable alternatives.
Key technological goals in this field include developing more accurate methods for detecting and measuring CCl4 in the environment, improving remediation techniques for contaminated sites, and creating environmentally friendly substitutes for remaining CCl4 applications. There is also a growing focus on understanding and mitigating the long-term effects of historical CCl4 use, particularly its persistence in the atmosphere and potential impact on global climate systems.
As global efforts to combat climate change and protect the ozone layer intensify, the importance of comprehensive lifecycle analyses for compounds like CCl4 continues to grow. These studies provide crucial data for policymakers, industry leaders, and researchers, guiding decisions on chemical management, environmental protection, and sustainable development strategies. The ongoing research in this area reflects a broader trend towards more holistic approaches to understanding and mitigating the environmental impacts of industrial chemicals.
Market Demand Analysis
The market demand for carbon tetrachloride (CCl4) products has undergone significant changes in recent decades due to environmental concerns and regulatory restrictions. Historically, CCl4 was widely used as a solvent, cleaning agent, and feedstock for various chemical processes. However, its ozone-depleting properties led to a sharp decline in demand following the implementation of the Montreal Protocol in 1987.
Despite the overall reduction in demand, certain niche markets continue to drive the need for CCl4 products. The pharmaceutical industry remains a key consumer, utilizing CCl4 in the production of specific medications and as a reagent in analytical processes. Additionally, the agrochemical sector employs CCl4 in the synthesis of certain pesticides and herbicides, albeit in reduced quantities compared to historical usage.
The electronics industry has emerged as a potential growth area for CCl4 demand. High-purity CCl4 is used in the production of semiconductor materials and in plasma etching processes for manufacturing integrated circuits. As the global demand for electronic devices continues to rise, this sector may contribute to a modest increase in CCl4 consumption.
Environmental remediation efforts have also created a unique market for CCl4 products. Ironically, the compound is used in some cleanup technologies designed to address soil and groundwater contamination caused by historical CCl4 use. This application, while limited in scale, demonstrates the complex nature of the market demand for this chemical.
The global market for CCl4 products is characterized by regional variations in demand patterns. Developed countries have largely phased out CCl4 use in most applications, while some developing nations still permit limited use in certain sectors. This geographical disparity in demand reflects differences in regulatory frameworks and industrial development stages across regions.
Looking ahead, the market demand for CCl4 products is expected to remain constrained due to ongoing environmental concerns and the availability of alternative substances. However, research into new applications, particularly in advanced materials and nanotechnology, may open up novel niche markets. The development of more environmentally friendly production methods and closed-loop recycling systems could also influence future demand by addressing some of the sustainability challenges associated with CCl4 use.
In conclusion, the market demand analysis for CCl4 products reveals a complex landscape shaped by regulatory pressures, technological advancements, and evolving industrial needs. While traditional large-scale applications have largely disappeared, specialized uses in pharmaceuticals, electronics, and environmental remediation continue to sustain a reduced but persistent demand for this controversial chemical compound.
Despite the overall reduction in demand, certain niche markets continue to drive the need for CCl4 products. The pharmaceutical industry remains a key consumer, utilizing CCl4 in the production of specific medications and as a reagent in analytical processes. Additionally, the agrochemical sector employs CCl4 in the synthesis of certain pesticides and herbicides, albeit in reduced quantities compared to historical usage.
The electronics industry has emerged as a potential growth area for CCl4 demand. High-purity CCl4 is used in the production of semiconductor materials and in plasma etching processes for manufacturing integrated circuits. As the global demand for electronic devices continues to rise, this sector may contribute to a modest increase in CCl4 consumption.
Environmental remediation efforts have also created a unique market for CCl4 products. Ironically, the compound is used in some cleanup technologies designed to address soil and groundwater contamination caused by historical CCl4 use. This application, while limited in scale, demonstrates the complex nature of the market demand for this chemical.
The global market for CCl4 products is characterized by regional variations in demand patterns. Developed countries have largely phased out CCl4 use in most applications, while some developing nations still permit limited use in certain sectors. This geographical disparity in demand reflects differences in regulatory frameworks and industrial development stages across regions.
Looking ahead, the market demand for CCl4 products is expected to remain constrained due to ongoing environmental concerns and the availability of alternative substances. However, research into new applications, particularly in advanced materials and nanotechnology, may open up novel niche markets. The development of more environmentally friendly production methods and closed-loop recycling systems could also influence future demand by addressing some of the sustainability challenges associated with CCl4 use.
In conclusion, the market demand analysis for CCl4 products reveals a complex landscape shaped by regulatory pressures, technological advancements, and evolving industrial needs. While traditional large-scale applications have largely disappeared, specialized uses in pharmaceuticals, electronics, and environmental remediation continue to sustain a reduced but persistent demand for this controversial chemical compound.
Technical Challenges
The comprehensive lifecycle analysis of carbon tetrachloride products faces several significant technical challenges. One of the primary obstacles is the complexity of tracking and quantifying emissions throughout the entire product lifecycle. Carbon tetrachloride, being a volatile organic compound, can easily escape into the atmosphere during production, transportation, use, and disposal phases. Developing accurate measurement techniques and monitoring systems to capture these emissions at various stages presents a formidable challenge.
Another technical hurdle lies in the assessment of environmental impacts across different ecosystems. Carbon tetrachloride, once released, can affect air quality, water systems, and soil composition. The interaction of this compound with various environmental matrices and its potential for bioaccumulation in food chains necessitates sophisticated modeling and analytical techniques. Current methodologies may struggle to fully capture the long-term effects and potential synergistic interactions with other pollutants.
The variability in production processes and end-use applications of carbon tetrachloride products further complicates the lifecycle analysis. Different manufacturing techniques, purification methods, and industrial applications can lead to diverse emission profiles and environmental footprints. Standardizing the assessment methodology across this spectrum of scenarios while maintaining accuracy and relevance is a significant technical challenge.
Addressing the end-of-life phase in the lifecycle analysis poses unique difficulties. The persistence of carbon tetrachloride in the environment and its potential for long-range transport make it challenging to accurately assess the full environmental impact of disposal or recycling processes. Developing effective remediation technologies and quantifying their efficacy in mitigating long-term environmental damage remains an area of ongoing research and technical development.
Furthermore, the global nature of carbon tetrachloride production and use introduces complexities in data collection and analysis. Variations in regulatory frameworks, reporting standards, and environmental monitoring capabilities across different countries can lead to inconsistencies in the quality and comparability of lifecycle data. Harmonizing these diverse data sources and methodologies to produce a comprehensive, globally relevant lifecycle analysis is a significant technical challenge that requires innovative approaches in data integration and validation.
Lastly, the evolving understanding of climate change impacts adds another layer of complexity to the lifecycle analysis of carbon tetrachloride products. As a potent ozone-depleting substance and greenhouse gas, accurately modeling its long-term climate effects requires continuous updates to climate models and impact assessment methodologies. This dynamic nature of climate science necessitates frequent revisions and adaptations in lifecycle analysis techniques, posing an ongoing technical challenge for researchers and analysts in this field.
Another technical hurdle lies in the assessment of environmental impacts across different ecosystems. Carbon tetrachloride, once released, can affect air quality, water systems, and soil composition. The interaction of this compound with various environmental matrices and its potential for bioaccumulation in food chains necessitates sophisticated modeling and analytical techniques. Current methodologies may struggle to fully capture the long-term effects and potential synergistic interactions with other pollutants.
The variability in production processes and end-use applications of carbon tetrachloride products further complicates the lifecycle analysis. Different manufacturing techniques, purification methods, and industrial applications can lead to diverse emission profiles and environmental footprints. Standardizing the assessment methodology across this spectrum of scenarios while maintaining accuracy and relevance is a significant technical challenge.
Addressing the end-of-life phase in the lifecycle analysis poses unique difficulties. The persistence of carbon tetrachloride in the environment and its potential for long-range transport make it challenging to accurately assess the full environmental impact of disposal or recycling processes. Developing effective remediation technologies and quantifying their efficacy in mitigating long-term environmental damage remains an area of ongoing research and technical development.
Furthermore, the global nature of carbon tetrachloride production and use introduces complexities in data collection and analysis. Variations in regulatory frameworks, reporting standards, and environmental monitoring capabilities across different countries can lead to inconsistencies in the quality and comparability of lifecycle data. Harmonizing these diverse data sources and methodologies to produce a comprehensive, globally relevant lifecycle analysis is a significant technical challenge that requires innovative approaches in data integration and validation.
Lastly, the evolving understanding of climate change impacts adds another layer of complexity to the lifecycle analysis of carbon tetrachloride products. As a potent ozone-depleting substance and greenhouse gas, accurately modeling its long-term climate effects requires continuous updates to climate models and impact assessment methodologies. This dynamic nature of climate science necessitates frequent revisions and adaptations in lifecycle analysis techniques, posing an ongoing technical challenge for researchers and analysts in this field.
Current Lifecycle Solutions
01 Production methods for carbon tetrachloride
Various methods are employed to produce carbon tetrachloride, including chlorination of methane or other hydrocarbons, and the reaction of carbon disulfide with chlorine. These processes often involve specific reaction conditions and catalysts to optimize yield and purity.- Production methods for carbon tetrachloride: Various methods are employed to produce carbon tetrachloride, including chlorination of methane or other hydrocarbons, and the reaction of carbon disulfide with chlorine. These processes often involve specific reaction conditions and catalysts to optimize yield and purity.
- Applications of carbon tetrachloride in industrial processes: Carbon tetrachloride finds use in various industrial applications, such as a solvent for oils and fats, a cleaning agent for machinery, and as a raw material in the production of refrigerants and other chlorinated compounds. Its unique properties make it valuable in specific manufacturing processes.
- Safety measures and environmental considerations: Due to its toxicity and ozone-depleting properties, the use of carbon tetrachloride is heavily regulated. Safety measures for handling and storage are crucial, and alternatives are being developed to replace its use in various applications. Environmental impact assessments and disposal methods are also important considerations.
- Purification and quality control of carbon tetrachloride: Various techniques are employed to purify carbon tetrachloride and ensure its quality for different applications. These may include distillation, chemical treatments, and advanced analytical methods to detect and remove impurities, ensuring the product meets specific industry standards.
- Alternatives and substitutes for carbon tetrachloride: Research and development efforts focus on finding safer and more environmentally friendly alternatives to carbon tetrachloride for various applications. This includes developing new compounds or modifying existing processes to eliminate the need for carbon tetrachloride while maintaining or improving performance.
02 Applications of carbon tetrachloride in industrial processes
Carbon tetrachloride finds use in various industrial applications, such as a solvent for oils and fats, a cleaning agent for metal parts, and as a raw material in the production of refrigerants and other chlorinated compounds. Its unique properties make it valuable in specific manufacturing processes.Expand Specific Solutions03 Safety measures and environmental considerations
Due to its toxicity and ozone-depleting properties, the use of carbon tetrachloride is heavily regulated. Safety measures for handling and storage are crucial, and alternatives are being developed to replace it in various applications. Environmental impact assessments and disposal methods are also important considerations.Expand Specific Solutions04 Analytical and detection methods for carbon tetrachloride
Various analytical techniques are used to detect and quantify carbon tetrachloride in environmental samples and industrial products. These methods may include gas chromatography, mass spectrometry, and spectrophotometric analyses, which are essential for quality control and environmental monitoring.Expand Specific Solutions05 Remediation and treatment of carbon tetrachloride contamination
Techniques for remediating sites contaminated with carbon tetrachloride are developed, including chemical degradation, bioremediation, and physical removal methods. These approaches aim to reduce environmental impact and restore affected areas to safe conditions.Expand Specific Solutions
Key Industry Players
The comprehensive lifecycle analysis of carbon tetrachloride products is in a mature stage of industry development, with a well-established global market. The technology has reached a high level of maturity, as evidenced by the involvement of major players across various sectors. Companies like Occidental Chemical Corp., State Grid Corp. of China, and Evonik Operations GmbH are actively engaged in research and development, while academic institutions such as Tsinghua University and Zhejiang University contribute to advancing the field. The market size is substantial, driven by industrial applications and environmental concerns. The competitive landscape is diverse, including chemical manufacturers, power companies, and research institutions, indicating a broad interest in improving the lifecycle management of carbon tetrachloride products.
Occidental Chemical Corp.
Technical Solution: Occidental Chemical Corp. has developed a comprehensive lifecycle analysis approach for carbon tetrachloride products. Their method involves advanced tracking and monitoring systems throughout the production, use, and disposal phases. The company utilizes real-time data collection and analysis to assess environmental impacts, including greenhouse gas emissions, energy consumption, and waste generation[1]. They have implemented a closed-loop recycling system that recovers and purifies used carbon tetrachloride, significantly reducing the need for new production and minimizing environmental impact[3]. Additionally, Occidental has invested in green chemistry initiatives to explore alternative synthesis routes that reduce the carbon footprint of carbon tetrachloride production[5].
Strengths: Comprehensive tracking system, closed-loop recycling, and green chemistry initiatives. Weaknesses: Potential high implementation costs and complexity in managing the entire lifecycle.
Tsinghua University
Technical Solution: Tsinghua University has developed a comprehensive lifecycle analysis framework for carbon tetrachloride products, focusing on interdisciplinary research and collaboration. Their approach combines environmental chemistry, materials science, and systems engineering to create a holistic understanding of carbon tetrachloride's impacts. The university has developed a high-resolution atmospheric transport model to track the global distribution and fate of carbon tetrachloride emissions[13]. They have also pioneered a novel photocatalytic degradation technique that can effectively break down carbon tetrachloride in contaminated soil and water, offering a promising remediation solution[15]. Additionally, Tsinghua researchers have created a multi-criteria decision analysis tool that helps industries evaluate and optimize their carbon tetrachloride use based on environmental, economic, and social factors[17].
Strengths: Interdisciplinary approach, advanced atmospheric modeling, and innovative remediation techniques. Weaknesses: Potential gaps between academic research and industrial application, and the need for extensive field testing of proposed solutions.
Key Innovations Analysis
A full-life-cycle carbon footprint analysis method, device, system, and storage medium
PatentPendingCN120197808A
Innovation
- The whole-life cycle carbon footprint analysis method is adopted, including obtaining the carbon footprint data of the park product, conducting uncertainty analysis to obtain consistent carbon footprint data, quantifying the carbon footprint information of the whole-life cycle product, and conducting sensitivity analysis to extract key factors influencing carbon emissions.
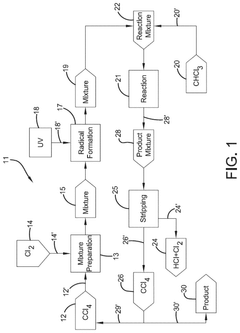
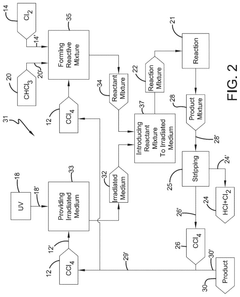
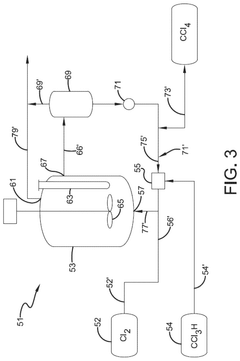
Photochlorination of partially-chlorinated chloromethanes to carbon tetrachloride
PatentPendingUS20250236573A1
Innovation
- A method involving the photochlorination of a chloromethanes stream, including chloroform and methyl chloride/methylene chloride, with stoichiometric chlorine and carbon tetrachloride, using electromagnetic radiation to form carbon tetrachloride with minimal impurities, allowing direct use in subsequent synthetic processes without additional purification.
Environmental Regulations
Environmental regulations play a crucial role in shaping the lifecycle analysis of carbon tetrachloride products. These regulations have evolved significantly over the past few decades, driven by increasing awareness of the environmental and health impacts associated with this chemical compound.
The Montreal Protocol, signed in 1987, marked a turning point in the regulation of carbon tetrachloride. This international treaty aimed to phase out the production of ozone-depleting substances, including carbon tetrachloride. As a result, the production and use of carbon tetrachloride for non-feedstock purposes have been severely restricted in many countries.
In the United States, the Environmental Protection Agency (EPA) has implemented stringent regulations under the Clean Air Act and the Toxic Substances Control Act. These regulations mandate strict controls on the production, use, and disposal of carbon tetrachloride. The EPA has also established reporting requirements for facilities that handle this substance, ensuring better tracking and management throughout its lifecycle.
The European Union has adopted similar measures through the REACH (Registration, Evaluation, Authorization, and Restriction of Chemicals) regulation. REACH requires manufacturers and importers to assess and manage the risks associated with carbon tetrachloride and other chemicals. This comprehensive approach covers the entire lifecycle of the substance, from production to end-of-life disposal.
Developing countries have also begun to implement stricter regulations on carbon tetrachloride. China, for instance, has introduced a national plan to phase out the production and consumption of ozone-depleting substances, including carbon tetrachloride. This shift in policy has significant implications for the global supply chain and lifecycle analysis of carbon tetrachloride products.
Environmental regulations have also driven innovation in alternative technologies and substances. Many industries that previously relied on carbon tetrachloride have been forced to develop safer alternatives, leading to advancements in green chemistry and sustainable production processes.
The impact of these regulations extends beyond the immediate production and use of carbon tetrachloride. They have influenced waste management practices, recycling technologies, and remediation efforts for contaminated sites. Environmental agencies worldwide have established guidelines for the proper disposal and treatment of carbon tetrachloride waste, ensuring minimal environmental impact throughout the product's lifecycle.
As global environmental concerns continue to grow, it is likely that regulations surrounding carbon tetrachloride will become even more stringent. Future policies may focus on complete phase-out of remaining uses, stricter monitoring of environmental levels, and enhanced requirements for lifecycle assessments. These evolving regulations will continue to shape the comprehensive lifecycle analysis of carbon tetrachloride products, driving further innovations in sustainable chemistry and environmental protection.
The Montreal Protocol, signed in 1987, marked a turning point in the regulation of carbon tetrachloride. This international treaty aimed to phase out the production of ozone-depleting substances, including carbon tetrachloride. As a result, the production and use of carbon tetrachloride for non-feedstock purposes have been severely restricted in many countries.
In the United States, the Environmental Protection Agency (EPA) has implemented stringent regulations under the Clean Air Act and the Toxic Substances Control Act. These regulations mandate strict controls on the production, use, and disposal of carbon tetrachloride. The EPA has also established reporting requirements for facilities that handle this substance, ensuring better tracking and management throughout its lifecycle.
The European Union has adopted similar measures through the REACH (Registration, Evaluation, Authorization, and Restriction of Chemicals) regulation. REACH requires manufacturers and importers to assess and manage the risks associated with carbon tetrachloride and other chemicals. This comprehensive approach covers the entire lifecycle of the substance, from production to end-of-life disposal.
Developing countries have also begun to implement stricter regulations on carbon tetrachloride. China, for instance, has introduced a national plan to phase out the production and consumption of ozone-depleting substances, including carbon tetrachloride. This shift in policy has significant implications for the global supply chain and lifecycle analysis of carbon tetrachloride products.
Environmental regulations have also driven innovation in alternative technologies and substances. Many industries that previously relied on carbon tetrachloride have been forced to develop safer alternatives, leading to advancements in green chemistry and sustainable production processes.
The impact of these regulations extends beyond the immediate production and use of carbon tetrachloride. They have influenced waste management practices, recycling technologies, and remediation efforts for contaminated sites. Environmental agencies worldwide have established guidelines for the proper disposal and treatment of carbon tetrachloride waste, ensuring minimal environmental impact throughout the product's lifecycle.
As global environmental concerns continue to grow, it is likely that regulations surrounding carbon tetrachloride will become even more stringent. Future policies may focus on complete phase-out of remaining uses, stricter monitoring of environmental levels, and enhanced requirements for lifecycle assessments. These evolving regulations will continue to shape the comprehensive lifecycle analysis of carbon tetrachloride products, driving further innovations in sustainable chemistry and environmental protection.
Sustainable Alternatives
In the pursuit of sustainable alternatives to carbon tetrachloride products, several promising approaches have emerged. One key strategy involves the development of environmentally friendly substitutes that maintain similar functionality while reducing environmental impact. For instance, hydrofluoroethers (HFEs) have shown potential as replacements in various applications, offering lower ozone depletion potential and global warming impact.
Another avenue focuses on process optimization and redesign to minimize or eliminate the need for carbon tetrachloride. This includes exploring alternative synthesis routes for chemicals traditionally produced using carbon tetrachloride as a feedstock or solvent. Advanced catalytic processes and green chemistry principles are being applied to develop more sustainable production methods.
Recycling and circular economy approaches are gaining traction as sustainable alternatives. Closed-loop systems that capture and reuse carbon tetrachloride within industrial processes can significantly reduce overall consumption and emissions. Additionally, technologies for the safe destruction or conversion of carbon tetrachloride waste into less harmful substances are being developed and implemented.
Biobased alternatives represent a growing area of research. Scientists are investigating natural compounds and bio-derived materials that could potentially replace carbon tetrachloride in certain applications. These alternatives often offer the added benefit of being renewable and biodegradable.
Technological advancements in materials science are also contributing to sustainable alternatives. Novel materials with similar properties to carbon tetrachloride-based products but with improved environmental profiles are being engineered. This includes the development of advanced polymers and composite materials that can replace traditional carbon tetrachloride-containing products in industries such as electronics and aerospace.
Policy and regulatory frameworks play a crucial role in driving the adoption of sustainable alternatives. Incentives for research and development of green technologies, coupled with stricter regulations on carbon tetrachloride use, are accelerating the transition to more sustainable practices across industries.
As the global focus on sustainability intensifies, collaborative efforts between academia, industry, and government agencies are fostering innovation in this space. International initiatives and knowledge-sharing platforms are facilitating the rapid development and dissemination of sustainable alternatives to carbon tetrachloride products, paving the way for a more environmentally responsible future.
Another avenue focuses on process optimization and redesign to minimize or eliminate the need for carbon tetrachloride. This includes exploring alternative synthesis routes for chemicals traditionally produced using carbon tetrachloride as a feedstock or solvent. Advanced catalytic processes and green chemistry principles are being applied to develop more sustainable production methods.
Recycling and circular economy approaches are gaining traction as sustainable alternatives. Closed-loop systems that capture and reuse carbon tetrachloride within industrial processes can significantly reduce overall consumption and emissions. Additionally, technologies for the safe destruction or conversion of carbon tetrachloride waste into less harmful substances are being developed and implemented.
Biobased alternatives represent a growing area of research. Scientists are investigating natural compounds and bio-derived materials that could potentially replace carbon tetrachloride in certain applications. These alternatives often offer the added benefit of being renewable and biodegradable.
Technological advancements in materials science are also contributing to sustainable alternatives. Novel materials with similar properties to carbon tetrachloride-based products but with improved environmental profiles are being engineered. This includes the development of advanced polymers and composite materials that can replace traditional carbon tetrachloride-containing products in industries such as electronics and aerospace.
Policy and regulatory frameworks play a crucial role in driving the adoption of sustainable alternatives. Incentives for research and development of green technologies, coupled with stricter regulations on carbon tetrachloride use, are accelerating the transition to more sustainable practices across industries.
As the global focus on sustainability intensifies, collaborative efforts between academia, industry, and government agencies are fostering innovation in this space. International initiatives and knowledge-sharing platforms are facilitating the rapid development and dissemination of sustainable alternatives to carbon tetrachloride products, paving the way for a more environmentally responsible future.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!