Hydrofluoric Acid vs Silver Nitrate: Reaction Analysis
AUG 26, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HF-AgNO3 Reaction Background and Objectives
The reaction between hydrofluoric acid (HF) and silver nitrate (AgNO3) represents a significant area of study in inorganic chemistry, with applications spanning multiple industries including semiconductor manufacturing, glass etching, and analytical chemistry. This reaction has been investigated since the early 20th century, with notable advancements occurring during the 1950s when the electronics industry began to expand rapidly.
The fundamental reaction involves the formation of silver fluoride (AgF) and nitric acid (HNO3) through an ionic exchange mechanism. Historically, this reaction gained prominence due to its unique properties compared to other halide-silver reactions, particularly the high solubility of silver fluoride compared to other silver halides.
Recent technological developments have renewed interest in this reaction, especially in the semiconductor industry where ultra-pure etching processes are required for advanced microchip fabrication. The evolution of this technology has been driven by increasingly stringent requirements for precision in nanoscale manufacturing processes, where even minor impurities or variations can significantly impact product quality.
The primary technical objective of this analysis is to comprehensively understand the kinetics, thermodynamics, and practical applications of the HF-AgNO3 reaction system. Specifically, we aim to characterize reaction rates under various conditions, identify potential side reactions, and optimize reaction parameters for industrial applications.
Secondary objectives include evaluating the safety protocols necessary for handling this reaction, as both hydrofluoric acid and silver nitrate present significant hazards. HF is particularly dangerous due to its ability to penetrate skin and cause deep tissue damage, while silver nitrate is a strong oxidizer and can cause chemical burns.
From an environmental perspective, we seek to assess the ecological impact of waste products from this reaction and develop more sustainable approaches to utilizing this chemistry in industrial settings. This includes exploring recycling methodologies for silver recovery and neutralization techniques for fluoride-containing waste streams.
The technological trajectory suggests increasing importance of this reaction in emerging fields such as flexible electronics, quantum computing components, and advanced optical materials. Understanding the fundamental science behind this reaction will enable innovation across these diverse application areas and potentially open new avenues for materials processing and manufacturing.
The fundamental reaction involves the formation of silver fluoride (AgF) and nitric acid (HNO3) through an ionic exchange mechanism. Historically, this reaction gained prominence due to its unique properties compared to other halide-silver reactions, particularly the high solubility of silver fluoride compared to other silver halides.
Recent technological developments have renewed interest in this reaction, especially in the semiconductor industry where ultra-pure etching processes are required for advanced microchip fabrication. The evolution of this technology has been driven by increasingly stringent requirements for precision in nanoscale manufacturing processes, where even minor impurities or variations can significantly impact product quality.
The primary technical objective of this analysis is to comprehensively understand the kinetics, thermodynamics, and practical applications of the HF-AgNO3 reaction system. Specifically, we aim to characterize reaction rates under various conditions, identify potential side reactions, and optimize reaction parameters for industrial applications.
Secondary objectives include evaluating the safety protocols necessary for handling this reaction, as both hydrofluoric acid and silver nitrate present significant hazards. HF is particularly dangerous due to its ability to penetrate skin and cause deep tissue damage, while silver nitrate is a strong oxidizer and can cause chemical burns.
From an environmental perspective, we seek to assess the ecological impact of waste products from this reaction and develop more sustainable approaches to utilizing this chemistry in industrial settings. This includes exploring recycling methodologies for silver recovery and neutralization techniques for fluoride-containing waste streams.
The technological trajectory suggests increasing importance of this reaction in emerging fields such as flexible electronics, quantum computing components, and advanced optical materials. Understanding the fundamental science behind this reaction will enable innovation across these diverse application areas and potentially open new avenues for materials processing and manufacturing.
Market Applications and Demand Analysis
The reaction between hydrofluoric acid (HF) and silver nitrate (AgNO₃) has significant market applications across multiple industries, driven by the unique properties of the resulting compounds and the chemical processes involved. The global market for fluoride compounds, particularly those derived from hydrofluoric acid reactions, was valued at approximately $2.1 billion in 2022 and is projected to grow at a compound annual growth rate of 4.7% through 2030.
In the semiconductor and electronics manufacturing sector, this reaction and its derivatives are crucial for etching silicon wafers and glass components. The semiconductor industry's continuous expansion, with a market size exceeding $600 billion in 2023, directly drives demand for high-purity hydrofluoric acid and its controlled reactions. The precision required in modern chip manufacturing, especially as node sizes decrease below 5nm, necessitates increasingly sophisticated chemical processes where the HF-AgNO₃ reaction plays a vital role.
The analytical chemistry market represents another significant demand driver. Silver fluoride (AgF) produced from this reaction serves as an important reagent in various analytical procedures. The global analytical reagents market, valued at $4.3 billion, continues to expand with the growing need for precise chemical analysis in pharmaceuticals, environmental monitoring, and materials science.
In the pharmaceutical industry, fluorinated compounds derived from controlled HF reactions are essential building blocks for approximately 25% of modern drugs. The pharmaceutical fluorochemicals segment alone represents a $3.7 billion market opportunity, with applications ranging from anesthetics to antibiotics and anti-cancer medications.
The glass etching and surface treatment industry utilizes this reaction for specialized applications in optical components, decorative glass, and microfluidic devices. This niche but growing market segment is expanding at 5.8% annually, driven by innovations in consumer electronics and medical devices requiring precisely etched surfaces.
Environmental considerations are increasingly influencing market dynamics. Regulations limiting HF emissions and handling have created demand for alternative processes and containment systems. This regulatory landscape has spawned a specialized market for HF handling equipment and neutralization technologies worth approximately $340 million globally.
The reaction's application in precious metal recovery represents another growing market segment. As electronic waste recycling becomes more economically viable and environmentally necessary, chemical processes involving silver nitrate and fluoride compounds are increasingly utilized to recover valuable metals from circuit boards and electronic components, contributing to the circular economy initiatives valued at $1.2 billion in the precious metal recovery sector.
In the semiconductor and electronics manufacturing sector, this reaction and its derivatives are crucial for etching silicon wafers and glass components. The semiconductor industry's continuous expansion, with a market size exceeding $600 billion in 2023, directly drives demand for high-purity hydrofluoric acid and its controlled reactions. The precision required in modern chip manufacturing, especially as node sizes decrease below 5nm, necessitates increasingly sophisticated chemical processes where the HF-AgNO₃ reaction plays a vital role.
The analytical chemistry market represents another significant demand driver. Silver fluoride (AgF) produced from this reaction serves as an important reagent in various analytical procedures. The global analytical reagents market, valued at $4.3 billion, continues to expand with the growing need for precise chemical analysis in pharmaceuticals, environmental monitoring, and materials science.
In the pharmaceutical industry, fluorinated compounds derived from controlled HF reactions are essential building blocks for approximately 25% of modern drugs. The pharmaceutical fluorochemicals segment alone represents a $3.7 billion market opportunity, with applications ranging from anesthetics to antibiotics and anti-cancer medications.
The glass etching and surface treatment industry utilizes this reaction for specialized applications in optical components, decorative glass, and microfluidic devices. This niche but growing market segment is expanding at 5.8% annually, driven by innovations in consumer electronics and medical devices requiring precisely etched surfaces.
Environmental considerations are increasingly influencing market dynamics. Regulations limiting HF emissions and handling have created demand for alternative processes and containment systems. This regulatory landscape has spawned a specialized market for HF handling equipment and neutralization technologies worth approximately $340 million globally.
The reaction's application in precious metal recovery represents another growing market segment. As electronic waste recycling becomes more economically viable and environmentally necessary, chemical processes involving silver nitrate and fluoride compounds are increasingly utilized to recover valuable metals from circuit boards and electronic components, contributing to the circular economy initiatives valued at $1.2 billion in the precious metal recovery sector.
Current Technical Challenges in HF-AgNO3 Reactions
The reaction between hydrofluoric acid (HF) and silver nitrate (AgNO3) presents several significant technical challenges that impede its industrial application and scientific research. One primary challenge is the extreme corrosivity of hydrofluoric acid, which necessitates specialized handling equipment and safety protocols. Standard laboratory glassware is unsuitable as HF etches glass, requiring the use of specialized polytetrafluoroethylene (PTFE) or other fluoropolymer containers that substantially increase operational costs.
Temperature control represents another critical challenge in HF-AgNO3 reactions. The reaction exhibits high sensitivity to temperature fluctuations, with optimal reaction efficiency occurring within a narrow temperature range. Deviations as small as ±2°C can significantly alter reaction kinetics, yield, and product purity. This necessitates sophisticated temperature monitoring and control systems, particularly challenging when scaling up to industrial production levels.
The formation of insoluble silver fluoride (AgF) precipitates during the reaction creates additional complications. These precipitates can adhere to reaction vessel walls and instrumentation, causing measurement inaccuracies and potential equipment damage. The removal and recovery of these precipitates require additional processing steps, increasing production complexity and cost while potentially introducing contaminants.
Concentration control presents a persistent technical hurdle. The reaction's stoichiometry is highly sensitive to the precise concentrations of both reactants. Excess HF can lead to unwanted side reactions and equipment degradation, while excess AgNO3 results in wastage of this expensive reagent. Real-time concentration monitoring remains technically challenging due to the corrosive environment.
Safety concerns constitute perhaps the most significant challenge. HF exposure presents severe health risks, including deep tissue burns and potential cardiac complications from systemic fluoride toxicity. Current engineering controls and personal protective equipment provide inadequate protection for workers in large-scale operations, necessitating extensive automation that increases capital expenditure.
Waste management poses environmental challenges, as reaction byproducts contain both fluoride compounds and silver residues. Current treatment methods are energy-intensive and costly, with regulatory compliance becoming increasingly stringent worldwide. The development of more efficient waste treatment protocols remains an active area of research.
Analytical monitoring capabilities represent another technical limitation. Real-time analysis of reaction progress is hampered by the corrosive nature of the reaction medium, which damages conventional sensors and probes. Non-invasive monitoring techniques show promise but currently lack the precision required for tight process control in industrial settings.
Temperature control represents another critical challenge in HF-AgNO3 reactions. The reaction exhibits high sensitivity to temperature fluctuations, with optimal reaction efficiency occurring within a narrow temperature range. Deviations as small as ±2°C can significantly alter reaction kinetics, yield, and product purity. This necessitates sophisticated temperature monitoring and control systems, particularly challenging when scaling up to industrial production levels.
The formation of insoluble silver fluoride (AgF) precipitates during the reaction creates additional complications. These precipitates can adhere to reaction vessel walls and instrumentation, causing measurement inaccuracies and potential equipment damage. The removal and recovery of these precipitates require additional processing steps, increasing production complexity and cost while potentially introducing contaminants.
Concentration control presents a persistent technical hurdle. The reaction's stoichiometry is highly sensitive to the precise concentrations of both reactants. Excess HF can lead to unwanted side reactions and equipment degradation, while excess AgNO3 results in wastage of this expensive reagent. Real-time concentration monitoring remains technically challenging due to the corrosive environment.
Safety concerns constitute perhaps the most significant challenge. HF exposure presents severe health risks, including deep tissue burns and potential cardiac complications from systemic fluoride toxicity. Current engineering controls and personal protective equipment provide inadequate protection for workers in large-scale operations, necessitating extensive automation that increases capital expenditure.
Waste management poses environmental challenges, as reaction byproducts contain both fluoride compounds and silver residues. Current treatment methods are energy-intensive and costly, with regulatory compliance becoming increasingly stringent worldwide. The development of more efficient waste treatment protocols remains an active area of research.
Analytical monitoring capabilities represent another technical limitation. Real-time analysis of reaction progress is hampered by the corrosive nature of the reaction medium, which damages conventional sensors and probes. Non-invasive monitoring techniques show promise but currently lack the precision required for tight process control in industrial settings.
Existing Methodologies for HF-AgNO3 Reactions
01 Formation of silver fluoride
When hydrofluoric acid reacts with silver nitrate, silver fluoride (AgF) is formed as the primary product. This reaction involves the exchange of ions where the fluoride ion from hydrofluoric acid combines with the silver ion from silver nitrate. The reaction also produces nitric acid as a byproduct. Silver fluoride is a water-soluble compound that has applications in various chemical processes and can be used as a fluorinating agent.- Formation of silver fluoride and nitric acid: When hydrofluoric acid (HF) reacts with silver nitrate (AgNO3), the primary products formed are silver fluoride (AgF) and nitric acid (HNO3). This is a double displacement reaction where the silver cation from silver nitrate combines with the fluoride anion from hydrofluoric acid, while the hydrogen cation from hydrofluoric acid combines with the nitrate anion from silver nitrate. The reaction proceeds rapidly in aqueous solution and is often used in laboratory settings for the preparation of silver fluoride.
- Purification and recovery methods for reaction products: Various methods are employed for the purification and recovery of products from the reaction between hydrofluoric acid and silver nitrate. These include crystallization techniques to isolate silver fluoride, distillation processes to separate nitric acid, and specialized filtration methods to remove impurities. Some processes involve the use of additional reagents to facilitate separation, while others employ temperature and pressure control to optimize product recovery. These purification methods are crucial for obtaining high-purity reaction products for industrial and research applications.
- Industrial applications of the reaction products: The products of the reaction between hydrofluoric acid and silver nitrate find numerous industrial applications. Silver fluoride is used in electronics manufacturing, particularly in the production of conductive pastes and films. It also serves as a fluorinating agent in organic synthesis and as an antibacterial agent in certain medical applications. The nitric acid byproduct is utilized in metal etching, oxidation processes, and as a reagent in various chemical syntheses. These applications leverage the unique properties of both reaction products across multiple industries.
- Safety considerations and handling procedures: The reaction between hydrofluoric acid and silver nitrate involves hazardous materials that require specific safety protocols. Hydrofluoric acid is highly corrosive and can cause severe tissue damage upon contact, while silver nitrate is a strong oxidizer and can cause chemical burns. Specialized equipment, including acid-resistant containers and personal protective gear, is necessary when conducting this reaction. Neutralization procedures for spills and waste disposal methods are also critical considerations. Various patents describe improved handling techniques and safety systems designed to minimize risks associated with this reaction.
- Modified reaction conditions and catalysts: Research has focused on modifying reaction conditions and introducing catalysts to improve the efficiency and selectivity of the reaction between hydrofluoric acid and silver nitrate. Variations in temperature, pressure, concentration, and reaction time can significantly affect the yield and purity of products. Some processes incorporate catalysts or additives to accelerate the reaction or direct it toward specific products. Alternative reaction media, such as non-aqueous solvents or ionic liquids, have also been explored to enhance reaction performance and facilitate product separation.
02 Purification and recovery methods
Various methods are employed for the purification and recovery of products from the reaction between hydrofluoric acid and silver nitrate. These include crystallization techniques, filtration processes, and solvent extraction methods. The purification steps are crucial to obtain high-purity silver fluoride and to separate it from unreacted starting materials and other byproducts. Recovery methods also focus on reclaiming any unreacted silver compounds due to the high value of silver.Expand Specific Solutions03 Industrial applications of reaction products
The products from the reaction between hydrofluoric acid and silver nitrate have various industrial applications. Silver fluoride is used in organic synthesis as a fluorinating agent, in the preparation of certain pharmaceuticals, and in specialized glass etching processes. The reaction is also relevant in metal surface treatment, semiconductor manufacturing, and in the production of certain catalysts. These applications leverage the unique properties of silver fluoride and other reaction products.Expand Specific Solutions04 Reaction conditions and optimization
The reaction between hydrofluoric acid and silver nitrate is influenced by various conditions that can be optimized for better yield and product quality. These include temperature control, concentration of reactants, reaction time, and pH levels. The reaction is typically conducted in controlled environments due to the corrosive nature of hydrofluoric acid. Optimization strategies focus on maximizing the conversion rate while minimizing side reactions and ensuring safety during the process.Expand Specific Solutions05 Safety considerations and handling procedures
Due to the hazardous nature of both hydrofluoric acid and silver compounds, specific safety considerations and handling procedures are necessary when conducting this reaction. These include the use of appropriate personal protective equipment, specialized containment systems, neutralization protocols for spills, and proper waste disposal methods. The reaction should be performed in well-ventilated areas with access to safety showers and eyewash stations. Training in handling these materials is essential to prevent accidents and exposure.Expand Specific Solutions
Key Industry Players and Research Institutions
The hydrofluoric acid and silver nitrate reaction market is in a growth phase, with increasing applications in chemical analysis, materials science, and industrial processes. The global market for these reagents is expanding, driven by demand in electronics manufacturing, glass etching, and analytical chemistry. Technologically, companies like Toagosei, The Chemours Co., and Solvay SA lead in production of high-purity hydrofluoric acid, while Glaxo Group and Abbott Laboratories have developed advanced applications in pharmaceutical research. Academic institutions such as Central South University and King Fahd University contribute significant research on reaction mechanisms and safety protocols. The industry faces challenges in handling toxicity and environmental concerns, spurring innovation in safer methodologies and containment systems from companies like Halliburton and Fujifilm.
Halliburton Energy Services, Inc.
Technical Solution: Halliburton has developed specialized treatment processes for handling hydrofluoric acid (HF) and silver nitrate reactions in oil field applications. Their technology focuses on using controlled reactions between HF and silver compounds to create temporary plugging agents in well stimulation operations. The company has engineered proprietary chemical formulations that leverage the precipitation reaction between HF and silver nitrate to form insoluble silver fluoride barriers that can temporarily seal off specific zones during hydraulic fracturing. This approach allows for more targeted acid treatments in carbonate formations while minimizing damage to wellbore infrastructure. Halliburton's system includes precise delivery mechanisms that control the timing and location of the reaction, preventing premature precipitation while ensuring effective placement of treatment fluids.
Strengths: Provides selective zonal isolation during acid treatments, reducing overall acid volumes needed and improving stimulation efficiency. The temporary nature of the plugging allows for subsequent production without additional intervention. Weaknesses: Requires careful handling of hazardous materials (HF) and precise control of reaction conditions to prevent equipment damage. The cost of silver compounds makes this approach more expensive than conventional methods.
Toagosei Co., Ltd.
Technical Solution: Toagosei has developed advanced manufacturing processes centered on the controlled reaction between hydrofluoric acid and silver nitrate for producing high-purity fluorochemicals. Their proprietary technology utilizes a multi-stage reaction system that carefully manages the highly exothermic reaction between HF and AgNO3 to synthesize specialty fluorinated compounds for semiconductor manufacturing. The company has engineered specialized corrosion-resistant reaction vessels with precise temperature control systems that prevent the formation of dangerous hydrogen fluoride gas while maximizing yield. Toagosei's process incorporates a unique recovery system that captures and recycles silver from the reaction byproducts, significantly reducing raw material costs and environmental impact. Their technology enables the production of ultra-high purity fluorinated compounds with impurity levels below 1 ppb, critical for advanced electronics manufacturing applications.
Strengths: Achieves exceptional product purity necessary for semiconductor applications while maximizing silver recovery and reuse. The closed-loop system minimizes environmental impact and worker exposure to hazardous materials. Weaknesses: Requires sophisticated equipment with specialized corrosion-resistant materials, resulting in high capital costs. The process demands rigorous safety protocols and continuous monitoring due to the hazardous nature of hydrofluoric acid.
Critical Reaction Mechanisms and Kinetics Analysis
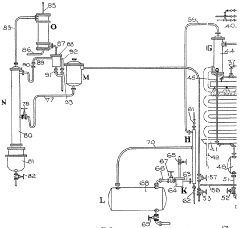
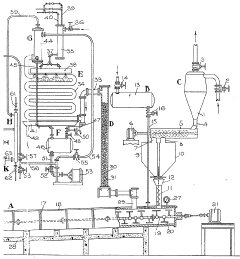
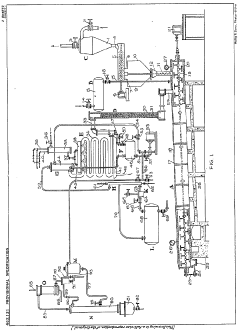
Improvements in or relating to the manufacture of hydrofluoric acid
PatentInactiveGB462131A
Innovation
- A continuous process under superatmospheric pressure reacts a fluoride with an acid, followed by preliminary cooling and condensation to separate dilute and concentrated hydrofluoric acid, using a reactor and condensers to achieve high yields and purity.
Production of nanostructures
PatentActiveUS20140251086A1
Innovation
- A method involving a reaction mixture with a solvent containing at least two hydroxyl groups, a metal-containing reagent, a templating agent, and a seed-promoting agent (SPA) that includes halide anions, where the mixture is heated at specific temperatures to produce silver nanowires with controlled morphology.
Safety Protocols and Hazard Mitigation Strategies
The handling of hydrofluoric acid (HF) and silver nitrate (AgNO₃) requires comprehensive safety protocols due to their hazardous properties. When these chemicals react, they form silver fluoride and nitric acid, presenting multiple safety concerns that demand rigorous mitigation strategies.
Personal protective equipment (PPE) constitutes the first line of defense when working with these chemicals. For HF, specialized protection is mandatory, including chemical-resistant gloves (neoprene or butyl rubber), face shields with chemical splash goggles, acid-resistant lab coats, and closed-toe shoes. Silver nitrate handling requires similar protection, with emphasis on preventing skin contact to avoid staining and potential burns.
Engineering controls represent critical safety infrastructure for these reactions. All operations involving HF must be conducted in properly functioning fume hoods with face velocities of 80-120 feet per minute. Secondary containment systems are essential for both chemicals, with specialized HF spill kits containing calcium gluconate readily accessible. Emergency eyewash stations and safety showers must be located within 10 seconds of travel time from the work area.
Administrative controls include mandatory specialized training for all personnel handling HF, with certification renewal required annually. Standard Operating Procedures (SOPs) must detail exact reaction protocols, including precise mixing sequences to prevent dangerous exothermic reactions. A buddy system is imperative when working with HF, ensuring immediate assistance in case of exposure.
Emergency response protocols must address the unique hazards of each chemical. For HF exposure, immediate application of calcium gluconate gel (2.5%) to affected skin areas is critical to prevent deep tissue damage. Silver nitrate exposures require thorough rinsing with water and sodium chloride solution to neutralize the silver ions through precipitation as silver chloride.
Waste management procedures must comply with local regulations, with separate containment systems for HF and silver-containing waste streams. Neutralization of HF waste typically involves careful treatment with calcium hydroxide, while silver waste requires recovery processes to prevent environmental contamination.
Monitoring and surveillance systems should include regular testing of engineering controls, documented safety inspections, and exposure monitoring where appropriate. Medical surveillance programs should be implemented for personnel regularly working with HF, including baseline and periodic medical evaluations focusing on potential effects on bone density and kidney function.
Communication protocols must ensure clear hazard information through proper labeling, safety data sheets, and visible warning signs in areas where these chemicals are used or stored. Regular safety briefings and mock emergency drills reinforce proper response procedures among laboratory personnel.
Personal protective equipment (PPE) constitutes the first line of defense when working with these chemicals. For HF, specialized protection is mandatory, including chemical-resistant gloves (neoprene or butyl rubber), face shields with chemical splash goggles, acid-resistant lab coats, and closed-toe shoes. Silver nitrate handling requires similar protection, with emphasis on preventing skin contact to avoid staining and potential burns.
Engineering controls represent critical safety infrastructure for these reactions. All operations involving HF must be conducted in properly functioning fume hoods with face velocities of 80-120 feet per minute. Secondary containment systems are essential for both chemicals, with specialized HF spill kits containing calcium gluconate readily accessible. Emergency eyewash stations and safety showers must be located within 10 seconds of travel time from the work area.
Administrative controls include mandatory specialized training for all personnel handling HF, with certification renewal required annually. Standard Operating Procedures (SOPs) must detail exact reaction protocols, including precise mixing sequences to prevent dangerous exothermic reactions. A buddy system is imperative when working with HF, ensuring immediate assistance in case of exposure.
Emergency response protocols must address the unique hazards of each chemical. For HF exposure, immediate application of calcium gluconate gel (2.5%) to affected skin areas is critical to prevent deep tissue damage. Silver nitrate exposures require thorough rinsing with water and sodium chloride solution to neutralize the silver ions through precipitation as silver chloride.
Waste management procedures must comply with local regulations, with separate containment systems for HF and silver-containing waste streams. Neutralization of HF waste typically involves careful treatment with calcium hydroxide, while silver waste requires recovery processes to prevent environmental contamination.
Monitoring and surveillance systems should include regular testing of engineering controls, documented safety inspections, and exposure monitoring where appropriate. Medical surveillance programs should be implemented for personnel regularly working with HF, including baseline and periodic medical evaluations focusing on potential effects on bone density and kidney function.
Communication protocols must ensure clear hazard information through proper labeling, safety data sheets, and visible warning signs in areas where these chemicals are used or stored. Regular safety briefings and mock emergency drills reinforce proper response procedures among laboratory personnel.
Environmental Impact and Waste Management Considerations
The reaction between hydrofluoric acid and silver nitrate presents significant environmental challenges that require careful consideration. When these chemicals interact, they produce silver fluoride and nitric acid, both of which pose distinct environmental hazards. The fluoride compounds can persist in soil and water systems, potentially affecting aquatic ecosystems and contaminating groundwater sources. Similarly, nitric acid contributes to soil acidification and can disrupt natural pH balances in affected environments.
Waste management for this reaction requires specialized protocols due to the corrosive nature of hydrofluoric acid and the toxicity of silver compounds. Industrial facilities utilizing this reaction must implement advanced treatment systems including neutralization processes, precipitation techniques, and ion exchange methods to remove harmful components before discharge. The silver content in waste streams represents both an environmental concern and a resource recovery opportunity, as proper extraction can reduce pollution while reclaiming valuable materials.
Regulatory frameworks governing these chemicals vary globally but generally include strict disposal guidelines. In the United States, the Environmental Protection Agency classifies hydrofluoric acid wastes as hazardous under the Resource Conservation and Recovery Act, mandating specific handling procedures. The European Union's REACH regulations similarly impose stringent controls on fluoride-containing waste streams and heavy metal disposal, including silver compounds.
Life cycle assessment studies indicate that the environmental footprint of processes involving these reactions extends beyond immediate waste management concerns. The production of both hydrofluoric acid and silver nitrate involves energy-intensive processes with associated carbon emissions. Furthermore, mining activities for fluorite (calcium fluoride) and silver ore contribute to habitat disruption and potential acid mine drainage issues.
Best practices for minimizing environmental impact include implementing closed-loop systems that recycle reaction components, substituting less hazardous alternatives where technically feasible, and employing real-time monitoring systems to detect and prevent accidental releases. Advanced treatment technologies such as membrane filtration and electrochemical recovery systems have demonstrated promising results in reducing discharge concentrations below regulatory thresholds.
The economic implications of proper environmental management for this reaction are substantial, with waste treatment costs often representing a significant percentage of overall operational expenses. However, these costs must be weighed against the potential liabilities associated with environmental contamination, including remediation expenses and regulatory penalties that can far exceed preventive measures.
Waste management for this reaction requires specialized protocols due to the corrosive nature of hydrofluoric acid and the toxicity of silver compounds. Industrial facilities utilizing this reaction must implement advanced treatment systems including neutralization processes, precipitation techniques, and ion exchange methods to remove harmful components before discharge. The silver content in waste streams represents both an environmental concern and a resource recovery opportunity, as proper extraction can reduce pollution while reclaiming valuable materials.
Regulatory frameworks governing these chemicals vary globally but generally include strict disposal guidelines. In the United States, the Environmental Protection Agency classifies hydrofluoric acid wastes as hazardous under the Resource Conservation and Recovery Act, mandating specific handling procedures. The European Union's REACH regulations similarly impose stringent controls on fluoride-containing waste streams and heavy metal disposal, including silver compounds.
Life cycle assessment studies indicate that the environmental footprint of processes involving these reactions extends beyond immediate waste management concerns. The production of both hydrofluoric acid and silver nitrate involves energy-intensive processes with associated carbon emissions. Furthermore, mining activities for fluorite (calcium fluoride) and silver ore contribute to habitat disruption and potential acid mine drainage issues.
Best practices for minimizing environmental impact include implementing closed-loop systems that recycle reaction components, substituting less hazardous alternatives where technically feasible, and employing real-time monitoring systems to detect and prevent accidental releases. Advanced treatment technologies such as membrane filtration and electrochemical recovery systems have demonstrated promising results in reducing discharge concentrations below regulatory thresholds.
The economic implications of proper environmental management for this reaction are substantial, with waste treatment costs often representing a significant percentage of overall operational expenses. However, these costs must be weighed against the potential liabilities associated with environmental contamination, including remediation expenses and regulatory penalties that can far exceed preventive measures.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!