Ion transport mechanisms in hydroxide conducting membranes
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Ion Transport Fundamentals and Research Objectives
Ion transport in hydroxide conducting membranes represents a critical area of research in electrochemical systems, particularly for alkaline fuel cells and electrolyzers. The fundamental mechanism involves the movement of hydroxide ions (OH-) through specialized polymer membranes, a process that differs significantly from proton transport due to the larger size and different solvation characteristics of hydroxide ions.
The historical development of this field began in the 1960s with the introduction of anion exchange membranes (AEMs), though significant progress has been limited until recent decades. The evolution of these materials has been driven by the need for alternatives to acidic proton exchange membrane systems, which often require expensive noble metal catalysts and face durability challenges in certain applications.
Hydroxide transport occurs through several mechanisms, including vehicular transport (where OH- ions move with water molecules), Grotthuss mechanism (involving proton hopping between water molecules), and structural diffusion through the membrane matrix. Understanding these transport pathways at the molecular level remains a fundamental challenge in the field.
Current research indicates that hydroxide conductivity is typically an order of magnitude lower than proton conductivity in comparable systems, presenting a significant barrier to widespread application. This conductivity gap stems from both the intrinsic mobility differences between H+ and OH- ions and the structural limitations of existing membrane materials.
The technical objectives of current research focus on several key areas: enhancing hydroxide conductivity to approach that of proton-conducting membranes (>100 mS/cm); improving chemical and mechanical stability under operating conditions; developing membranes with reduced water uptake while maintaining high ionic conductivity; and understanding the relationship between polymer architecture and ion transport properties.
Recent advances in computational modeling have provided deeper insights into the molecular-level interactions governing hydroxide transport, allowing for more rational design of membrane materials. Techniques such as molecular dynamics simulations and density functional theory calculations have become essential tools in predicting transport behavior and guiding experimental work.
The field is trending toward multifunctional membrane designs that incorporate both hydrophilic domains for ion transport and hydrophobic regions for mechanical stability. Novel cationic functional groups beyond the traditional quaternary ammonium structures are being explored to enhance both conductivity and alkaline stability.
Our research aims to develop a comprehensive understanding of the structure-property relationships governing hydroxide transport, with the ultimate goal of designing next-generation membranes that overcome current limitations in conductivity and stability for advanced electrochemical applications.
The historical development of this field began in the 1960s with the introduction of anion exchange membranes (AEMs), though significant progress has been limited until recent decades. The evolution of these materials has been driven by the need for alternatives to acidic proton exchange membrane systems, which often require expensive noble metal catalysts and face durability challenges in certain applications.
Hydroxide transport occurs through several mechanisms, including vehicular transport (where OH- ions move with water molecules), Grotthuss mechanism (involving proton hopping between water molecules), and structural diffusion through the membrane matrix. Understanding these transport pathways at the molecular level remains a fundamental challenge in the field.
Current research indicates that hydroxide conductivity is typically an order of magnitude lower than proton conductivity in comparable systems, presenting a significant barrier to widespread application. This conductivity gap stems from both the intrinsic mobility differences between H+ and OH- ions and the structural limitations of existing membrane materials.
The technical objectives of current research focus on several key areas: enhancing hydroxide conductivity to approach that of proton-conducting membranes (>100 mS/cm); improving chemical and mechanical stability under operating conditions; developing membranes with reduced water uptake while maintaining high ionic conductivity; and understanding the relationship between polymer architecture and ion transport properties.
Recent advances in computational modeling have provided deeper insights into the molecular-level interactions governing hydroxide transport, allowing for more rational design of membrane materials. Techniques such as molecular dynamics simulations and density functional theory calculations have become essential tools in predicting transport behavior and guiding experimental work.
The field is trending toward multifunctional membrane designs that incorporate both hydrophilic domains for ion transport and hydrophobic regions for mechanical stability. Novel cationic functional groups beyond the traditional quaternary ammonium structures are being explored to enhance both conductivity and alkaline stability.
Our research aims to develop a comprehensive understanding of the structure-property relationships governing hydroxide transport, with the ultimate goal of designing next-generation membranes that overcome current limitations in conductivity and stability for advanced electrochemical applications.
Market Analysis of Hydroxide Conducting Membrane Applications
The hydroxide conducting membrane market is experiencing significant growth driven by the increasing demand for clean energy solutions and sustainable technologies. Currently valued at approximately 320 million USD, this market is projected to grow at a compound annual growth rate of 8.7% through 2030, potentially reaching 650 million USD by the end of the decade. This growth trajectory is primarily fueled by the expanding applications in fuel cells, particularly anion exchange membrane fuel cells (AEMFCs), which offer cost advantages over traditional proton exchange membrane technologies.
The energy sector represents the largest application segment, accounting for roughly 45% of the total market share. Within this sector, fuel cells for stationary power generation and transportation applications dominate the demand landscape. The automotive industry's shift toward hydrogen-powered vehicles has created a substantial market opportunity, with major manufacturers investing in AEMFC technology for next-generation vehicles.
Water electrolysis for green hydrogen production constitutes another rapidly growing application segment, currently representing approximately 30% of the market. As global initiatives for carbon neutrality accelerate, the demand for efficient hydroxide conducting membranes in electrolyzers is expected to increase substantially, with projections suggesting a doubling of market share by 2028.
Geographically, North America leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (25%). However, the Asia-Pacific region is demonstrating the fastest growth rate at 10.2% annually, driven by aggressive clean energy policies in China, Japan, and South Korea. These countries have implemented substantial subsidies and research funding for hydrogen technologies, creating favorable market conditions.
The industrial sector applications, including chemical processing and wastewater treatment, represent smaller but steadily growing segments at 15% and 10% of the market respectively. These applications benefit from the selective ion transport properties of hydroxide conducting membranes, enabling more efficient separation processes and reduced energy consumption in industrial operations.
Customer demand is increasingly focused on membranes with improved hydroxide conductivity, mechanical stability, and chemical durability. End-users are willing to pay premium prices for membranes that demonstrate longer operational lifetimes and consistent performance under variable conditions, indicating a market shift toward higher-value products rather than cost-minimized solutions.
The energy sector represents the largest application segment, accounting for roughly 45% of the total market share. Within this sector, fuel cells for stationary power generation and transportation applications dominate the demand landscape. The automotive industry's shift toward hydrogen-powered vehicles has created a substantial market opportunity, with major manufacturers investing in AEMFC technology for next-generation vehicles.
Water electrolysis for green hydrogen production constitutes another rapidly growing application segment, currently representing approximately 30% of the market. As global initiatives for carbon neutrality accelerate, the demand for efficient hydroxide conducting membranes in electrolyzers is expected to increase substantially, with projections suggesting a doubling of market share by 2028.
Geographically, North America leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (25%). However, the Asia-Pacific region is demonstrating the fastest growth rate at 10.2% annually, driven by aggressive clean energy policies in China, Japan, and South Korea. These countries have implemented substantial subsidies and research funding for hydrogen technologies, creating favorable market conditions.
The industrial sector applications, including chemical processing and wastewater treatment, represent smaller but steadily growing segments at 15% and 10% of the market respectively. These applications benefit from the selective ion transport properties of hydroxide conducting membranes, enabling more efficient separation processes and reduced energy consumption in industrial operations.
Customer demand is increasingly focused on membranes with improved hydroxide conductivity, mechanical stability, and chemical durability. End-users are willing to pay premium prices for membranes that demonstrate longer operational lifetimes and consistent performance under variable conditions, indicating a market shift toward higher-value products rather than cost-minimized solutions.
Current Challenges in Hydroxide Conducting Membrane Technology
Despite significant advancements in hydroxide conducting membrane technology, several critical challenges continue to impede widespread commercial adoption and optimal performance. The most persistent issue remains chemical stability, as hydroxide ions are highly nucleophilic and can degrade polymer backbones through nucleophilic substitution reactions. This degradation is particularly pronounced at elevated temperatures and high pH environments, limiting operational longevity and reliability in practical applications.
Conductivity limitations represent another major hurdle. Current hydroxide conducting membranes typically achieve only 10-30% of the conductivity exhibited by their proton-conducting counterparts under comparable conditions. This performance gap stems from the inherently lower mobility of hydroxide ions compared to protons, as well as suboptimal water management within membrane structures.
Water management presents a complex challenge with significant implications for membrane performance. Hydroxide conductivity is heavily dependent on water content, yet excessive water uptake leads to mechanical instability and dimensional changes. Conversely, insufficient hydration severely restricts ion transport. Achieving this delicate balance remains elusive in current membrane designs.
Carbonate formation poses a unique challenge specific to hydroxide conducting systems. When exposed to atmospheric carbon dioxide, hydroxide ions readily convert to carbonate and bicarbonate species, which exhibit substantially lower mobility. This phenomenon, known as "carbonation poisoning," can reduce conductivity by up to 60% under ambient conditions, necessitating complex system designs to minimize air exposure.
The alkaline environment required for hydroxide conduction also accelerates degradation of not only the membrane but also other device components, including catalysts and support materials. This system-level incompatibility complicates integration into practical devices and increases overall system costs.
Manufacturing scalability remains problematic, with many high-performance laboratory materials utilizing complex synthesis routes involving multiple steps and hazardous reagents. The transition from laboratory demonstration to industrial-scale production has proven challenging, with significant performance losses often observed during scale-up processes.
Characterization techniques for hydroxide conducting membranes are less developed than those for proton conductors, making it difficult to accurately measure and understand fundamental transport mechanisms. This knowledge gap hinders rational design approaches and slows progress toward optimized materials.
Cost considerations further complicate commercial viability, with current high-performance materials often requiring expensive fluorinated polymers or complex synthetic procedures. The development of cost-effective alternatives that maintain performance metrics remains an active research challenge.
Conductivity limitations represent another major hurdle. Current hydroxide conducting membranes typically achieve only 10-30% of the conductivity exhibited by their proton-conducting counterparts under comparable conditions. This performance gap stems from the inherently lower mobility of hydroxide ions compared to protons, as well as suboptimal water management within membrane structures.
Water management presents a complex challenge with significant implications for membrane performance. Hydroxide conductivity is heavily dependent on water content, yet excessive water uptake leads to mechanical instability and dimensional changes. Conversely, insufficient hydration severely restricts ion transport. Achieving this delicate balance remains elusive in current membrane designs.
Carbonate formation poses a unique challenge specific to hydroxide conducting systems. When exposed to atmospheric carbon dioxide, hydroxide ions readily convert to carbonate and bicarbonate species, which exhibit substantially lower mobility. This phenomenon, known as "carbonation poisoning," can reduce conductivity by up to 60% under ambient conditions, necessitating complex system designs to minimize air exposure.
The alkaline environment required for hydroxide conduction also accelerates degradation of not only the membrane but also other device components, including catalysts and support materials. This system-level incompatibility complicates integration into practical devices and increases overall system costs.
Manufacturing scalability remains problematic, with many high-performance laboratory materials utilizing complex synthesis routes involving multiple steps and hazardous reagents. The transition from laboratory demonstration to industrial-scale production has proven challenging, with significant performance losses often observed during scale-up processes.
Characterization techniques for hydroxide conducting membranes are less developed than those for proton conductors, making it difficult to accurately measure and understand fundamental transport mechanisms. This knowledge gap hinders rational design approaches and slows progress toward optimized materials.
Cost considerations further complicate commercial viability, with current high-performance materials often requiring expensive fluorinated polymers or complex synthetic procedures. The development of cost-effective alternatives that maintain performance metrics remains an active research challenge.
Contemporary Ion Transport Models and Solutions
01 Anion exchange membranes for hydroxide conduction
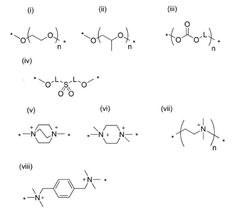
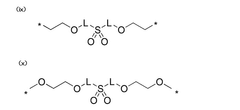
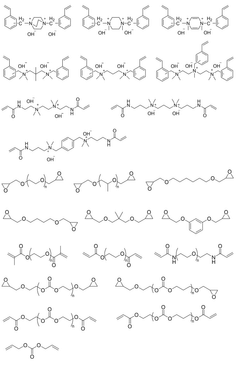
Anion exchange membranes (AEMs) are specifically designed for hydroxide ion transport in electrochemical systems. These membranes contain positively charged functional groups that facilitate the movement of negatively charged hydroxide ions. The transport mechanism involves hydroxide ions hopping between water molecules and functional groups within the membrane structure, allowing for efficient ion conduction while maintaining membrane stability in alkaline environments.- Polymer-based hydroxide conducting membranes: Polymer-based membranes are developed for efficient hydroxide ion conduction. These membranes typically incorporate quaternary ammonium functional groups or other cationic sites that facilitate hydroxide ion transport. The polymer backbone provides structural stability while the functional groups create ion channels for hydroxide movement. These membranes often balance mechanical strength with ion conductivity through careful polymer selection and cross-linking strategies.
- Composite and hybrid membrane structures: Composite membranes combine organic polymers with inorganic materials to enhance hydroxide conductivity. These hybrid structures often incorporate metal oxides, nanoparticles, or layered materials that create additional ion transport pathways or improve membrane stability in alkaline environments. The interface between organic and inorganic components plays a crucial role in facilitating hydroxide ion movement while maintaining membrane integrity under operating conditions.
- Water-mediated ion transport mechanisms: Water molecules play a critical role in hydroxide ion transport through membranes. The mechanism involves hydrogen bonding networks where hydroxide ions hop between water molecules (Grotthuss mechanism) or move through hydrated channels. Membrane hydration levels significantly impact conductivity, with optimal water content balancing ion mobility against excessive swelling. Controlling water management is essential for maintaining consistent hydroxide conductivity across varying operating conditions.
- Temperature and pressure effects on ion transport: Temperature and pressure significantly influence hydroxide ion conductivity in membranes. Higher temperatures generally increase ion mobility but can also accelerate membrane degradation or cause dehydration. Pressure effects can alter membrane morphology and water content, thereby affecting ion transport pathways. Understanding these relationships helps optimize membrane performance under various operating conditions, particularly in electrochemical devices like fuel cells and electrolyzers.
- Stability and durability enhancements: Improving the chemical stability of hydroxide conducting membranes is crucial for long-term operation. Strategies include developing degradation-resistant polymer backbones, incorporating stabilizing additives, and designing protective functional groups that shield ion-conducting sites from nucleophilic attack. Advanced cross-linking methods and composite structures help maintain mechanical integrity while preserving ion transport capabilities under harsh alkaline conditions and repeated hydration/dehydration cycles.
02 Polymer-based hydroxide conducting membranes
Polymer-based membranes incorporate specific chemical structures to enhance hydroxide conductivity. These membranes typically use quaternary ammonium groups or other cationic functionalities attached to polymer backbones to create pathways for hydroxide ion transport. The polymer chemistry can be tailored to optimize the balance between mechanical strength, chemical stability, and ion conductivity, with hydrophilic-hydrophobic microphase separation often playing a key role in creating effective ion transport channels.Expand Specific Solutions03 Water-mediated hydroxide transport mechanisms
Water plays a crucial role in hydroxide ion transport through membranes. The transport mechanism involves hydroxide ions moving through hydrogen-bonded networks of water molecules via the Grotthuss mechanism, where proton transfer occurs between adjacent water molecules, effectively moving the hydroxide ion in the opposite direction. Membrane hydration levels significantly impact conductivity, with higher water content generally leading to improved hydroxide mobility but potentially compromising mechanical stability.Expand Specific Solutions04 Composite and inorganic hydroxide conducting materials
Composite and inorganic materials offer alternative approaches to hydroxide conduction. These materials often combine polymers with inorganic components such as metal oxides or layered double hydroxides to enhance conductivity and stability. The hydroxide transport in these systems can occur through interfacial pathways between organic and inorganic phases, through structural defects in crystalline materials, or via specially designed nanochannels that facilitate ion movement while blocking other species.Expand Specific Solutions05 Stability and degradation mechanisms in hydroxide conducting membranes
A critical challenge in hydroxide conducting membranes is their chemical stability in highly alkaline environments. Common degradation mechanisms include nucleophilic attack by hydroxide ions on the cationic functional groups, leading to decreased ion exchange capacity and conductivity over time. Research focuses on developing more stable cationic groups, protective strategies such as steric hindrance around vulnerable sites, and membrane architectures that maintain conductivity while minimizing exposure to degradation pathways.Expand Specific Solutions
Leading Research Institutions and Industrial Stakeholders
The ion transport mechanisms in hydroxide conducting membranes market is currently in a growth phase, with increasing demand driven by clean energy applications. The global market size is expanding rapidly, projected to reach significant value as hydrogen technologies gain traction. Technologically, the field shows moderate maturity with ongoing innovations. Key players demonstrate varying levels of expertise: Air Liquide, Air Products & Chemicals, and Johnson Matthey lead with established commercial solutions; Dalian Institute of Chemical Physics and University of Maryland contribute significant academic research; while IBM, Samsung SDI, and Hyundai-Kia are investing in next-generation membrane technologies. Research institutions like AIST and CNRS are advancing fundamental understanding, creating a competitive landscape balanced between established industrial players and emerging technology developers.
Dalian Institute of Chemical Physics of CAS
Technical Solution: Dalian Institute of Chemical Physics (DICP) has developed innovative hydroxide exchange membranes (HEMs) with quaternary ammonium functional groups that facilitate efficient hydroxide ion transport. Their approach involves synthesizing polymer backbones with high chemical stability against nucleophilic attack, addressing the traditional degradation issues in alkaline environments. DICP researchers have pioneered the use of radiation-grafted membranes where stable polymer backbones are first formed and then functionalized, creating distinct hydrophilic channels for ion transport while maintaining mechanical integrity. Their recent breakthrough includes developing membranes with microphase-separated morphology that creates well-connected ion transport pathways, achieving hydroxide conductivity exceeding 100 mS/cm at 80°C while maintaining stability for over 1000 hours in alkaline conditions. DICP has also explored incorporating metal-organic frameworks (MOFs) into membrane matrices to enhance ion transport and stability simultaneously.
Strengths: Superior alkaline stability compared to conventional membranes, with demonstrated long-term durability in harsh conditions. Their microphase separation approach achieves exceptional hydroxide conductivity without sacrificing mechanical properties. Weaknesses: Some of their advanced membranes require complex synthesis procedures that may challenge commercial scalability, and performance at lower temperatures (below 60°C) sometimes lags behind competing technologies.
Asahi Kasei Corp.
Technical Solution: Asahi Kasei has developed proprietary chlor-alkali ion exchange membranes that leverage their expertise in hydroxide ion transport mechanisms. Their flagship technology centers on perfluorinated membranes with specially designed side chains containing quaternary ammonium groups that facilitate hydroxide conductivity while resisting degradation. Asahi's approach involves creating precisely controlled membrane morphology with interconnected hydrophilic domains that form efficient ion transport channels while maintaining dimensional stability. Their membranes incorporate reinforcement layers that prevent excessive swelling while maintaining high mechanical strength under operating conditions. A key innovation is their multi-layer membrane structure that optimizes both ion conductivity and gas crossover prevention, critical for applications in electrolyzers and fuel cells. Asahi Kasei has recently developed advanced membranes with hydroxide conductivity exceeding 80 mS/cm at 80°C while demonstrating stability for over 2000 hours in concentrated alkaline solutions, representing significant improvements over previous generations.
Strengths: Exceptional manufacturing consistency and quality control, with proven scalability for industrial applications. Their multi-layer approach effectively balances the competing requirements of conductivity, mechanical strength, and chemical stability. Weaknesses: Higher production costs compared to some emerging membrane technologies, and their perfluorinated materials face increasing environmental scrutiny despite performance advantages.
Critical Patents and Scientific Breakthroughs
High-performance hydroxide exchange membrane
PatentPendingUS20230317997A1
Innovation
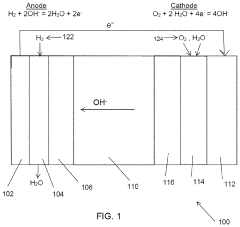
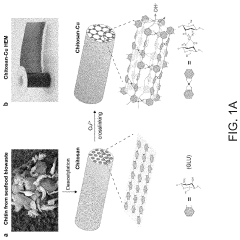
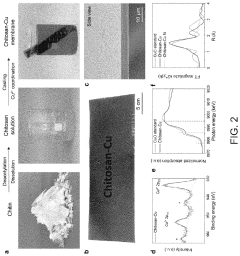
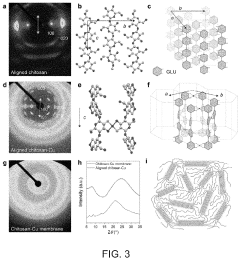
- A chitosan-based ion exchange membrane is developed by crosslinking chitosan molecular chains with multivalent cations, such as copper, to form a trigonal crystal structure with hexagonal nanochannels, enhancing ion transport and mechanical strength while maintaining stability in alkaline environments.
Hydroxy ion conductive membrane, method for producing hydroxy ion conductive membrane, membrane electrode assembly, hydrogen production method, and hydrogen production system
PatentWO2025070387A1
Innovation
- A hydroxy ion conductive membrane comprising a porous substrate with a hydroxy ion conductive polymer containing a specific component derived from a polyfunctional polymerizable monomer, ensuring a thickness of 5 μm to 50 μm, high water retention capacity, and a specific elemental composition, which is produced by impregnating a porous substrate with a polymerizable monomer-containing liquid and polymerizing it to form a three-dimensional network structure.
Environmental Impact and Sustainability Considerations
The development of hydroxide conducting membranes represents a significant advancement in clean energy technologies, particularly for fuel cells and electrolyzers. When evaluating these ion transport mechanisms, environmental impact and sustainability considerations become paramount factors that influence both research directions and commercial applications.
Hydroxide conducting membranes offer substantial environmental benefits compared to traditional acidic systems. The alkaline environment enables the use of non-precious metal catalysts, reducing dependence on scarce platinum group metals whose mining operations cause significant ecological disruption and generate substantial carbon emissions. This transition toward earth-abundant catalysts aligns with circular economy principles and reduces the environmental footprint of energy conversion devices.
Water management in hydroxide conducting membranes presents both challenges and opportunities from a sustainability perspective. These membranes typically require careful hydration control, which impacts system efficiency and lifetime. However, recent advancements in membrane design have improved water retention capabilities, reducing the energy requirements for maintaining optimal operating conditions and consequently lowering the overall environmental impact of systems employing these membranes.
The manufacturing processes for hydroxide conducting membranes merit careful environmental assessment. Current production methods often involve fluorinated polymers and organic solvents that pose potential environmental hazards. Research trends indicate growing interest in green synthesis routes utilizing bio-based polymers and environmentally benign processing techniques. These approaches significantly reduce toxic waste generation and align with sustainable chemistry principles.
End-of-life considerations for hydroxide conducting membranes remain an underdeveloped area requiring further attention. The durability challenges these membranes face, particularly degradation through carbonate formation, not only affect performance but also influence replacement frequency and waste generation. Emerging research on recyclable membrane materials and regeneration protocols shows promise for extending useful lifetimes and creating closed-loop material cycles.
Carbon footprint analysis of the entire lifecycle of hydroxide conducting membrane technologies reveals potential climate benefits when compared to conventional energy systems. While membrane production carries environmental costs, the operational phase typically demonstrates significant greenhouse gas reductions, especially when powered by renewable energy sources. This favorable balance supports their role in decarbonization strategies across multiple sectors.
Policy frameworks increasingly recognize the environmental attributes of hydroxide conducting membrane technologies, with several jurisdictions implementing incentives for clean energy solutions with demonstrable sustainability credentials. These policy drivers accelerate both research investment and market adoption, creating positive feedback loops for environmental innovation in this field.
Hydroxide conducting membranes offer substantial environmental benefits compared to traditional acidic systems. The alkaline environment enables the use of non-precious metal catalysts, reducing dependence on scarce platinum group metals whose mining operations cause significant ecological disruption and generate substantial carbon emissions. This transition toward earth-abundant catalysts aligns with circular economy principles and reduces the environmental footprint of energy conversion devices.
Water management in hydroxide conducting membranes presents both challenges and opportunities from a sustainability perspective. These membranes typically require careful hydration control, which impacts system efficiency and lifetime. However, recent advancements in membrane design have improved water retention capabilities, reducing the energy requirements for maintaining optimal operating conditions and consequently lowering the overall environmental impact of systems employing these membranes.
The manufacturing processes for hydroxide conducting membranes merit careful environmental assessment. Current production methods often involve fluorinated polymers and organic solvents that pose potential environmental hazards. Research trends indicate growing interest in green synthesis routes utilizing bio-based polymers and environmentally benign processing techniques. These approaches significantly reduce toxic waste generation and align with sustainable chemistry principles.
End-of-life considerations for hydroxide conducting membranes remain an underdeveloped area requiring further attention. The durability challenges these membranes face, particularly degradation through carbonate formation, not only affect performance but also influence replacement frequency and waste generation. Emerging research on recyclable membrane materials and regeneration protocols shows promise for extending useful lifetimes and creating closed-loop material cycles.
Carbon footprint analysis of the entire lifecycle of hydroxide conducting membrane technologies reveals potential climate benefits when compared to conventional energy systems. While membrane production carries environmental costs, the operational phase typically demonstrates significant greenhouse gas reductions, especially when powered by renewable energy sources. This favorable balance supports their role in decarbonization strategies across multiple sectors.
Policy frameworks increasingly recognize the environmental attributes of hydroxide conducting membrane technologies, with several jurisdictions implementing incentives for clean energy solutions with demonstrable sustainability credentials. These policy drivers accelerate both research investment and market adoption, creating positive feedback loops for environmental innovation in this field.
Scalability and Manufacturing Challenges
The scaling of hydroxide conducting membrane production from laboratory to industrial scale presents significant challenges that must be addressed for widespread commercial adoption. Current manufacturing processes for these membranes often involve complex chemical synthesis routes that work well at small scales but face reproducibility issues when scaled up. The precision required in controlling polymer chain length, functional group distribution, and membrane morphology becomes increasingly difficult to maintain in larger batch processes.
Material consistency represents a major hurdle in mass production. Small variations in raw materials or processing conditions can lead to significant performance differences in ion transport capabilities. This inconsistency is particularly problematic for hydroxide conducting membranes, where even minor changes in the microstructure can dramatically alter hydroxide conductivity and membrane stability.
Cost factors also present substantial barriers to commercialization. The specialized monomers and functionalization agents required for high-performance hydroxide conducting membranes remain expensive, with limited economies of scale currently available. Production methods often involve multiple steps with hazardous chemicals and precise reaction conditions, necessitating sophisticated equipment and safety measures that drive up manufacturing costs.
Environmental and safety considerations further complicate large-scale production. Many synthesis routes utilize toxic solvents or reagents that pose challenges for worker safety and environmental compliance. Developing greener synthesis routes that maintain membrane performance while reducing hazardous waste remains an active area of research but has not yet yielded fully satisfactory solutions for industrial implementation.
Quality control presents another significant challenge. Current analytical techniques for characterizing ion transport mechanisms in membranes are often time-consuming and require specialized equipment, making real-time quality assessment during production difficult. The development of rapid, inline characterization methods would significantly enhance manufacturing capabilities but remains technically challenging.
The transition from batch to continuous manufacturing processes represents a promising direction for overcoming scalability issues. Continuous flow chemistry and roll-to-roll processing techniques could potentially improve consistency and reduce costs, but adapting current synthesis protocols to these manufacturing paradigms requires substantial engineering innovation and process redesign.
Material consistency represents a major hurdle in mass production. Small variations in raw materials or processing conditions can lead to significant performance differences in ion transport capabilities. This inconsistency is particularly problematic for hydroxide conducting membranes, where even minor changes in the microstructure can dramatically alter hydroxide conductivity and membrane stability.
Cost factors also present substantial barriers to commercialization. The specialized monomers and functionalization agents required for high-performance hydroxide conducting membranes remain expensive, with limited economies of scale currently available. Production methods often involve multiple steps with hazardous chemicals and precise reaction conditions, necessitating sophisticated equipment and safety measures that drive up manufacturing costs.
Environmental and safety considerations further complicate large-scale production. Many synthesis routes utilize toxic solvents or reagents that pose challenges for worker safety and environmental compliance. Developing greener synthesis routes that maintain membrane performance while reducing hazardous waste remains an active area of research but has not yet yielded fully satisfactory solutions for industrial implementation.
Quality control presents another significant challenge. Current analytical techniques for characterizing ion transport mechanisms in membranes are often time-consuming and require specialized equipment, making real-time quality assessment during production difficult. The development of rapid, inline characterization methods would significantly enhance manufacturing capabilities but remains technically challenging.
The transition from batch to continuous manufacturing processes represents a promising direction for overcoming scalability issues. Continuous flow chemistry and roll-to-roll processing techniques could potentially improve consistency and reduce costs, but adapting current synthesis protocols to these manufacturing paradigms requires substantial engineering innovation and process redesign.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!