Maximize Selectivity in HPLC: Detector Sensitivity Focus
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Selectivity Evolution and Objectives
High-Performance Liquid Chromatography (HPLC) has evolved significantly since its inception in the 1960s, transforming from a specialized analytical technique to an essential tool across numerous scientific disciplines. The evolution of HPLC selectivity represents a critical advancement in analytical chemistry, enabling increasingly precise separation and identification of complex chemical mixtures.
The early development of HPLC focused primarily on improving basic separation capabilities through enhanced stationary phases and pumping systems. By the 1980s, the introduction of reversed-phase chromatography marked a pivotal shift, dramatically expanding the range of compounds that could be effectively analyzed. This period also witnessed the emergence of specialized columns designed to target specific molecular interactions.
The 1990s and early 2000s brought significant advancements in detector technology, with UV-Vis detectors giving way to more sensitive and selective options such as diode array, fluorescence, and mass spectrometry detectors. These technological improvements directly contributed to enhanced selectivity by providing multiple detection parameters beyond simple retention time.
Recent years have seen a paradigm shift toward integrated approaches that combine advanced column chemistry with sophisticated detection methods. The concept of orthogonal selectivity—employing complementary separation mechanisms—has gained prominence as researchers seek to maximize resolution in increasingly complex samples. This evolution reflects the growing understanding that detector sensitivity and selectivity are intrinsically linked parameters.
The primary objective in maximizing HPLC selectivity through detector sensitivity focuses on achieving the highest possible signal-to-noise ratio while maintaining the ability to distinguish between structurally similar compounds. This goal encompasses several interconnected technical challenges, including minimizing baseline noise, enhancing detector response to target analytes, and reducing interference from matrix components.
Another critical objective involves expanding the dynamic range of detection to accommodate both trace and abundant components within a single analysis. This capability is particularly valuable in fields such as pharmaceutical analysis, environmental monitoring, and metabolomics, where sample complexity continues to increase.
The ultimate aim of current research directions is to develop detection systems that combine unprecedented sensitivity with highly specific molecular recognition capabilities. This would enable the reliable identification and quantification of target compounds in complex matrices without extensive sample preparation or chromatographic optimization, representing a significant advancement in analytical efficiency and accuracy.
The early development of HPLC focused primarily on improving basic separation capabilities through enhanced stationary phases and pumping systems. By the 1980s, the introduction of reversed-phase chromatography marked a pivotal shift, dramatically expanding the range of compounds that could be effectively analyzed. This period also witnessed the emergence of specialized columns designed to target specific molecular interactions.
The 1990s and early 2000s brought significant advancements in detector technology, with UV-Vis detectors giving way to more sensitive and selective options such as diode array, fluorescence, and mass spectrometry detectors. These technological improvements directly contributed to enhanced selectivity by providing multiple detection parameters beyond simple retention time.
Recent years have seen a paradigm shift toward integrated approaches that combine advanced column chemistry with sophisticated detection methods. The concept of orthogonal selectivity—employing complementary separation mechanisms—has gained prominence as researchers seek to maximize resolution in increasingly complex samples. This evolution reflects the growing understanding that detector sensitivity and selectivity are intrinsically linked parameters.
The primary objective in maximizing HPLC selectivity through detector sensitivity focuses on achieving the highest possible signal-to-noise ratio while maintaining the ability to distinguish between structurally similar compounds. This goal encompasses several interconnected technical challenges, including minimizing baseline noise, enhancing detector response to target analytes, and reducing interference from matrix components.
Another critical objective involves expanding the dynamic range of detection to accommodate both trace and abundant components within a single analysis. This capability is particularly valuable in fields such as pharmaceutical analysis, environmental monitoring, and metabolomics, where sample complexity continues to increase.
The ultimate aim of current research directions is to develop detection systems that combine unprecedented sensitivity with highly specific molecular recognition capabilities. This would enable the reliable identification and quantification of target compounds in complex matrices without extensive sample preparation or chromatographic optimization, representing a significant advancement in analytical efficiency and accuracy.
Market Demand Analysis for High-Sensitivity HPLC Detection
The global HPLC market continues to experience robust growth, with detector sensitivity emerging as a critical differentiator in purchasing decisions. Current market valuations place the HPLC systems sector at approximately 4.5 billion USD, with a compound annual growth rate of 6.8% projected through 2028. Within this expanding market, high-sensitivity detection technologies command premium pricing and are experiencing accelerated adoption rates compared to standard detection systems.
Pharmaceutical and biotechnology sectors represent the largest demand drivers, collectively accounting for over 60% of high-sensitivity HPLC detector purchases. This demand is primarily fueled by increasingly stringent regulatory requirements for trace analysis in drug development and quality control processes. The FDA and EMA continue to lower acceptable limits for impurities, creating direct market pressure for enhanced detection capabilities.
Academic research institutions constitute another significant market segment, particularly as proteomics, metabolomics, and other -omics fields advance. These applications frequently require detection of analytes at sub-nanogram levels, driving demand for cutting-edge sensitivity in HPLC detection systems.
Environmental monitoring represents the fastest-growing application segment, with a 9.2% growth rate, as regulations worldwide tighten regarding detection limits for micropollutants in water, soil, and air samples. This sector's expansion is creating new market opportunities for portable high-sensitivity HPLC systems.
Market research indicates that end-users are willing to pay 30-40% premium pricing for detection systems offering a one-order-of-magnitude improvement in sensitivity over current standards. This price elasticity demonstrates the critical importance of detection limits in modern analytical workflows.
Regional analysis reveals that North America currently leads market consumption of high-sensitivity detectors, followed closely by Europe. However, Asia-Pacific markets, particularly China and India, are showing the most aggressive growth trajectories as their pharmaceutical and biotechnology sectors expand and regulatory frameworks mature.
Customer surveys consistently highlight three primary demands: lower detection limits without sacrificing linear range, reduced signal-to-noise ratios across diverse sample matrices, and improved sensitivity for traditionally challenging analyte classes such as non-UV absorbing compounds. These market signals provide clear direction for technology development priorities.
The competitive landscape shows increasing customer preference for integrated solutions that combine advanced detector technology with sophisticated data processing algorithms to extract meaningful signals from complex backgrounds. This trend suggests that software development may become as important as hardware improvements in addressing market demands for enhanced sensitivity.
Pharmaceutical and biotechnology sectors represent the largest demand drivers, collectively accounting for over 60% of high-sensitivity HPLC detector purchases. This demand is primarily fueled by increasingly stringent regulatory requirements for trace analysis in drug development and quality control processes. The FDA and EMA continue to lower acceptable limits for impurities, creating direct market pressure for enhanced detection capabilities.
Academic research institutions constitute another significant market segment, particularly as proteomics, metabolomics, and other -omics fields advance. These applications frequently require detection of analytes at sub-nanogram levels, driving demand for cutting-edge sensitivity in HPLC detection systems.
Environmental monitoring represents the fastest-growing application segment, with a 9.2% growth rate, as regulations worldwide tighten regarding detection limits for micropollutants in water, soil, and air samples. This sector's expansion is creating new market opportunities for portable high-sensitivity HPLC systems.
Market research indicates that end-users are willing to pay 30-40% premium pricing for detection systems offering a one-order-of-magnitude improvement in sensitivity over current standards. This price elasticity demonstrates the critical importance of detection limits in modern analytical workflows.
Regional analysis reveals that North America currently leads market consumption of high-sensitivity detectors, followed closely by Europe. However, Asia-Pacific markets, particularly China and India, are showing the most aggressive growth trajectories as their pharmaceutical and biotechnology sectors expand and regulatory frameworks mature.
Customer surveys consistently highlight three primary demands: lower detection limits without sacrificing linear range, reduced signal-to-noise ratios across diverse sample matrices, and improved sensitivity for traditionally challenging analyte classes such as non-UV absorbing compounds. These market signals provide clear direction for technology development priorities.
The competitive landscape shows increasing customer preference for integrated solutions that combine advanced detector technology with sophisticated data processing algorithms to extract meaningful signals from complex backgrounds. This trend suggests that software development may become as important as hardware improvements in addressing market demands for enhanced sensitivity.
Current HPLC Detector Technology Limitations
Despite significant advancements in HPLC technology, current detector systems face several critical limitations that impact selectivity optimization. Conventional UV-Vis detectors, while widely used, struggle with compounds lacking chromophores or those with similar absorption profiles, resulting in inadequate discrimination between co-eluting substances. This fundamental limitation becomes particularly problematic when analyzing complex biological samples or pharmaceutical formulations containing structurally similar compounds.
Fluorescence detectors offer improved sensitivity compared to UV-Vis systems but remain constrained by their dependence on native fluorescence properties. Many compounds require derivatization, introducing additional sample preparation steps and potential sources of error. Furthermore, the sensitivity of fluorescence detection can be compromised by matrix effects and quenching phenomena, reducing reliability in complex sample environments.
Mass spectrometry detectors, despite their exceptional sensitivity and selectivity, present significant challenges in routine analysis. Their high acquisition and operational costs limit widespread implementation, particularly in smaller laboratories or quality control environments. Additionally, MS detectors often require specialized expertise for operation and data interpretation, creating barriers to adoption in settings with limited technical resources.
Electrochemical detectors demonstrate excellent sensitivity for redox-active compounds but suffer from limited applicability across diverse chemical classes. Their selectivity is inherently restricted to electroactive substances, leaving a substantial gap in comprehensive analytical capabilities. Moreover, these detectors frequently encounter issues with electrode fouling during continuous operation, necessitating regular maintenance and calibration.
Refractive index detectors, while universal in their detection capabilities, exhibit poor sensitivity compared to other detector types. Their susceptibility to temperature fluctuations and inability to function effectively with gradient elution severely restricts their application in complex separation scenarios requiring high selectivity.
Evaporative light scattering detectors (ELSD) and charged aerosol detectors (CAD) have emerged as alternatives for non-volatile compounds but face challenges with reproducibility and quantification accuracy. Their response factors vary significantly between different compound classes, complicating quantitative analysis and method validation.
Multi-detector arrangements, designed to overcome individual detector limitations, introduce new challenges in data integration and interpretation. The complexity of aligning signals from different detection principles, each with unique response characteristics, creates difficulties in establishing unified analytical methods with optimized selectivity across diverse sample types.
Fluorescence detectors offer improved sensitivity compared to UV-Vis systems but remain constrained by their dependence on native fluorescence properties. Many compounds require derivatization, introducing additional sample preparation steps and potential sources of error. Furthermore, the sensitivity of fluorescence detection can be compromised by matrix effects and quenching phenomena, reducing reliability in complex sample environments.
Mass spectrometry detectors, despite their exceptional sensitivity and selectivity, present significant challenges in routine analysis. Their high acquisition and operational costs limit widespread implementation, particularly in smaller laboratories or quality control environments. Additionally, MS detectors often require specialized expertise for operation and data interpretation, creating barriers to adoption in settings with limited technical resources.
Electrochemical detectors demonstrate excellent sensitivity for redox-active compounds but suffer from limited applicability across diverse chemical classes. Their selectivity is inherently restricted to electroactive substances, leaving a substantial gap in comprehensive analytical capabilities. Moreover, these detectors frequently encounter issues with electrode fouling during continuous operation, necessitating regular maintenance and calibration.
Refractive index detectors, while universal in their detection capabilities, exhibit poor sensitivity compared to other detector types. Their susceptibility to temperature fluctuations and inability to function effectively with gradient elution severely restricts their application in complex separation scenarios requiring high selectivity.
Evaporative light scattering detectors (ELSD) and charged aerosol detectors (CAD) have emerged as alternatives for non-volatile compounds but face challenges with reproducibility and quantification accuracy. Their response factors vary significantly between different compound classes, complicating quantitative analysis and method validation.
Multi-detector arrangements, designed to overcome individual detector limitations, introduce new challenges in data integration and interpretation. The complexity of aligning signals from different detection principles, each with unique response characteristics, creates difficulties in establishing unified analytical methods with optimized selectivity across diverse sample types.
Current Approaches to Maximize HPLC Selectivity
01 Column selection and modification for HPLC selectivity
The choice and modification of chromatographic columns significantly impact HPLC selectivity. Various stationary phases with different chemical properties can be selected based on the analytes of interest. Column modifications, such as surface treatments or bonded phases, can enhance separation selectivity for specific compounds. Temperature control and column dimensions also play important roles in optimizing chromatographic selectivity.- Column selection and modification for HPLC selectivity: The choice and modification of chromatographic columns significantly impact HPLC selectivity. Different stationary phases (C18, phenyl, amino, etc.) offer varying separation mechanisms based on their chemical properties. Column modifications, such as end-capping or incorporating specific functional groups, can enhance selectivity for particular analytes. Temperature control and column dimensions also affect separation efficiency and selectivity in complex sample analysis.
- Mobile phase composition optimization: The composition of the mobile phase is crucial for achieving optimal HPLC selectivity. Adjusting parameters such as pH, buffer concentration, organic modifier type (acetonitrile, methanol), and gradient profiles can significantly enhance separation of structurally similar compounds. Additives like ion-pairing reagents can improve selectivity for ionizable analytes, while the ratio of aqueous to organic components affects retention and separation of compounds with different polarities.
- Advanced detection techniques for improved selectivity: Integration of specialized detection methods enhances HPLC selectivity beyond traditional UV detection. Mass spectrometry coupling (LC-MS/MS) provides structural information and high selectivity for complex mixtures. Diode array detection (DAD) offers spectral information across multiple wavelengths simultaneously. Fluorescence, electrochemical, and refractive index detectors provide selective detection for specific compound classes, improving analytical specificity in complex matrices.
- Sample preparation strategies for selectivity enhancement: Effective sample preparation techniques significantly improve HPLC selectivity by removing interfering compounds and concentrating analytes of interest. Solid-phase extraction (SPE), liquid-liquid extraction, and protein precipitation methods can be optimized for specific analyte classes. Derivatization techniques can enhance detectability and separation of structurally similar compounds by introducing chromophores or modifying functional groups, thereby improving chromatographic behavior and selectivity.
- Method development and validation for selectivity: Systematic approach to HPLC method development focuses on achieving optimal selectivity through iterative optimization. This includes screening multiple columns, mobile phase compositions, and detection parameters to identify the best separation conditions. Validation protocols specifically assess selectivity by challenging the method with closely related compounds, degradation products, and matrix components. Quality-by-design approaches incorporate multivariate analysis to understand critical parameters affecting selectivity and establish robust operating ranges.
02 Mobile phase composition optimization
The composition of the mobile phase is crucial for HPLC selectivity. By adjusting parameters such as solvent type, pH, buffer concentration, and organic modifier ratio, analysts can significantly improve the separation of complex mixtures. Gradient elution techniques allow for dynamic changes in mobile phase composition during analysis, enhancing selectivity for compounds with varying polarities.Expand Specific Solutions03 Specialized HPLC techniques for enhanced selectivity
Various specialized HPLC techniques have been developed to enhance selectivity for specific applications. These include chiral chromatography for enantiomer separation, ion-exchange chromatography for charged molecules, size-exclusion chromatography for molecular weight-based separation, and affinity chromatography for highly specific interactions. These techniques employ different separation mechanisms to achieve superior selectivity for challenging analytical problems.Expand Specific Solutions04 Detection methods and selectivity enhancement
Advanced detection methods can significantly enhance the selectivity of HPLC analyses. Mass spectrometry coupling provides both structural information and high selectivity based on mass-to-charge ratios. Diode array detectors allow for spectral analysis across multiple wavelengths simultaneously. Fluorescence and electrochemical detectors offer selective detection for specific compound classes, improving the overall selectivity of the analytical method.Expand Specific Solutions05 Method development and validation for selectivity
Systematic approaches to HPLC method development focus on optimizing selectivity through experimental design and statistical analysis. This includes screening different chromatographic conditions, evaluating critical separation parameters, and applying quality-by-design principles. Method validation protocols specifically assess selectivity by challenging the method with closely related compounds, degradation products, and matrix components to ensure reliable separation under various conditions.Expand Specific Solutions
Leading Manufacturers in HPLC Detector Industry
The HPLC selectivity and detector sensitivity market is currently in a growth phase, with increasing demand driven by pharmaceutical, environmental, and food safety applications. The global analytical instrumentation market, including HPLC systems, is estimated to exceed $5 billion annually with steady growth projections. Technologically, companies like Agilent Technologies, Thermo Fisher (through Dionex acquisition), and Waters Corporation lead innovation in detector sensitivity enhancement. These established players offer comprehensive solutions with advanced detectors (UV-Vis, fluorescence, mass spectrometry) that maximize selectivity. Emerging players like Oxford Nanoimaging are introducing novel detection technologies, while research institutions such as A*STAR and Advanced Industrial Science & Technology contribute fundamental innovations that push sensitivity boundaries beyond conventional limits.
Siemens Healthineers AG
Technical Solution: Siemens Healthineers has developed specialized HPLC systems for clinical diagnostics that focus on maximizing selectivity through their Atellica® HPLC solutions. Their approach integrates multi-modal detection systems combining UV absorption with electrochemical detection to enhance selectivity for specific biomarkers in complex biological matrices. The technology employs proprietary column chemistries optimized for clinical applications, featuring specialized stationary phases that provide improved selectivity for structurally similar compounds like isomers of vitamin D metabolites and steroid hormones[3]. Siemens' systems incorporate automated sample preparation modules that reduce matrix effects, thereby enhancing detector sensitivity while maintaining selectivity. Their advanced data processing algorithms employ machine learning techniques to distinguish between closely related compounds based on subtle differences in retention behavior and spectral characteristics. The systems also feature intelligent calibration protocols that automatically compensate for drift in detector response, ensuring consistent selectivity across large batches of clinical samples.
Strengths: Specialized solutions for clinical diagnostics with validated methods for specific biomarkers; integrated sample preparation enhancing overall workflow efficiency; advanced data processing algorithms for complex biological matrices. Weaknesses: Systems primarily optimized for clinical applications rather than general analytical purposes; proprietary consumables may increase operational costs; limited flexibility for method development outside predefined clinical assays.
Agilent Technologies, Inc.
Technical Solution: Agilent Technologies has developed advanced HPLC detector systems focused on maximizing selectivity through their InfinityLab LC series. Their technology employs multi-signal detection approaches combining diode array detection (DAD) with mass spectrometry (MS) to significantly enhance selectivity. The InfinityLab LC/MSD iQ system integrates intelligent diagnostics and maintenance features that optimize detector sensitivity while maintaining high selectivity parameters. Agilent's ZORBAX RRHD (Rapid Resolution High Definition) column technology further enhances selectivity by providing superior peak resolution and reduced peak tailing, allowing for better separation of structurally similar compounds. Their OpenLab CDS software platform incorporates advanced algorithms for peak deconvolution and spectral analysis, enabling improved selectivity in complex sample matrices[1]. Agilent has also pioneered the development of Intelligent System Emulation Technology (ISET) that maintains consistent selectivity across different instrument platforms, ensuring method transferability while preserving sensitivity parameters.
Strengths: Industry-leading integration of multiple detection technologies (UV-Vis, MS, fluorescence) providing orthogonal selectivity; comprehensive software solutions for data analysis and method optimization. Weaknesses: Higher cost compared to basic HPLC systems; complex systems may require specialized training for optimal utilization; some advanced features may be unnecessary for routine analyses.
Key Innovations in Detector Sensitivity Enhancement
High performance liquid chromatography method for analyzing imaging agent, precursor of imaging agent, or intermediate of imaging agent
PatentInactiveUS20170115259A1
Innovation
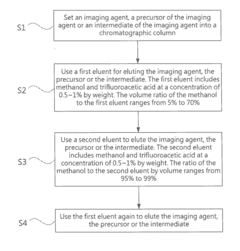
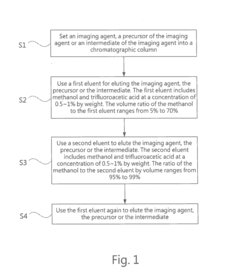
- A high performance liquid chromatography (HPLC) method using a solution with a high proportion of methanol for elution and gradient elution with eluents containing 0.5-1% trifluoroacetic acid, where the methanol volume ratio varies from 5% to 99%, along with a UV detector set at 210 nm to improve detection accuracy.
Inline filter housing assembly
PatentInactiveUS20130312501A1
Innovation
- A novel filter housing assembly that supports high pressures without frits, allowing for easy assembly and filter replacement by hand, using a retaining screen that minimizes dead volume and sample adsorption, and is compatible with both brittle and flexible filter elements.
Method Validation Protocols for Enhanced Selectivity
Method validation protocols are essential for ensuring the reliability and reproducibility of HPLC methods with enhanced selectivity. These protocols must be systematically designed to verify that the analytical method consistently delivers accurate results with optimal selectivity across various operational conditions.
The validation process begins with specificity testing, which confirms the method's ability to unambiguously assess the analyte in the presence of expected components. This involves analyzing samples containing known impurities, degradation products, and matrix components to demonstrate that these substances do not interfere with the detection of the target analyte.
Linearity assessment forms another critical component, evaluating the method's ability to obtain test results proportional to analyte concentration across the specified range. For selectivity-focused methods, this assessment should be conducted at concentrations near the detection limit to verify that selectivity is maintained even at low analyte levels.
Precision validation, encompassing repeatability, intermediate precision, and reproducibility, ensures consistent selectivity under varying conditions. Repeatability tests involve multiple analyses of the same sample under identical conditions, while intermediate precision evaluates variations introduced by different analysts, instruments, or days. Reproducibility examines method performance across different laboratories.
Limit of detection (LOD) and limit of quantitation (LOQ) determinations are particularly important for selectivity enhancement. These parameters should be established using signal-to-noise ratio approaches or statistical methods based on the standard deviation of the response and the slope of the calibration curve.
Robustness testing evaluates the method's capacity to maintain selectivity despite small, deliberate variations in method parameters. Critical factors to examine include mobile phase composition, pH, column temperature, flow rate, and detection wavelength. This testing identifies which parameters most significantly impact selectivity and establishes acceptable operational ranges.
System suitability tests should be incorporated into the validation protocol to verify adequate chromatographic performance before analysis. These tests typically include parameters such as resolution, tailing factor, theoretical plate count, and retention factor, all of which directly influence selectivity.
Finally, the validation protocol should include stability testing of analytical solutions to determine the timeframe within which analyses can be performed without compromising selectivity. This involves analyzing standard and sample solutions at defined intervals to detect any changes in chromatographic behavior or response.
The validation process begins with specificity testing, which confirms the method's ability to unambiguously assess the analyte in the presence of expected components. This involves analyzing samples containing known impurities, degradation products, and matrix components to demonstrate that these substances do not interfere with the detection of the target analyte.
Linearity assessment forms another critical component, evaluating the method's ability to obtain test results proportional to analyte concentration across the specified range. For selectivity-focused methods, this assessment should be conducted at concentrations near the detection limit to verify that selectivity is maintained even at low analyte levels.
Precision validation, encompassing repeatability, intermediate precision, and reproducibility, ensures consistent selectivity under varying conditions. Repeatability tests involve multiple analyses of the same sample under identical conditions, while intermediate precision evaluates variations introduced by different analysts, instruments, or days. Reproducibility examines method performance across different laboratories.
Limit of detection (LOD) and limit of quantitation (LOQ) determinations are particularly important for selectivity enhancement. These parameters should be established using signal-to-noise ratio approaches or statistical methods based on the standard deviation of the response and the slope of the calibration curve.
Robustness testing evaluates the method's capacity to maintain selectivity despite small, deliberate variations in method parameters. Critical factors to examine include mobile phase composition, pH, column temperature, flow rate, and detection wavelength. This testing identifies which parameters most significantly impact selectivity and establishes acceptable operational ranges.
System suitability tests should be incorporated into the validation protocol to verify adequate chromatographic performance before analysis. These tests typically include parameters such as resolution, tailing factor, theoretical plate count, and retention factor, all of which directly influence selectivity.
Finally, the validation protocol should include stability testing of analytical solutions to determine the timeframe within which analyses can be performed without compromising selectivity. This involves analyzing standard and sample solutions at defined intervals to detect any changes in chromatographic behavior or response.
Sample Preparation Techniques for Improved Detection
Sample preparation represents a critical pre-analytical phase in HPLC analysis that significantly impacts detector sensitivity and overall method selectivity. Effective sample preparation techniques can enhance analyte concentration, remove interfering substances, and improve chromatographic performance. The selection of appropriate sample preparation methods depends on sample matrix complexity, target analyte properties, and detection requirements.
Solid-phase extraction (SPE) remains one of the most versatile sample preparation techniques for HPLC analysis. Modern SPE materials offer selective retention mechanisms including reversed-phase, normal-phase, ion-exchange, and mixed-mode interactions. Advanced polymer-based SPE sorbents demonstrate superior recovery rates compared to traditional silica-based materials, with recovery improvements of 15-30% for complex biological samples. Automated SPE systems further enhance reproducibility by minimizing human error and standardizing extraction parameters.
Liquid-liquid extraction (LLE) techniques have evolved with the development of supported liquid extraction (SLE) and dispersive liquid-liquid microextraction (DLLME). These approaches require minimal solvent volumes while achieving concentration factors of 100-1000 times for trace analytes. The integration of salting-out assisted liquid-liquid extraction (SALLE) with HPLC has shown particular promise for polar compounds in aqueous matrices, improving detection limits by up to two orders of magnitude.
Protein precipitation techniques are essential for biological sample preparation, with recent advances in precipitation agents and protocols significantly reducing matrix effects. Combining protein precipitation with phospholipid removal technologies has demonstrated a 40-60% reduction in ion suppression effects during LC-MS/MS analysis, directly enhancing detector response for target analytes in complex biological matrices.
Microextraction techniques including solid-phase microextraction (SPME), stir bar sorptive extraction (SBSE), and thin-film microextraction (TFME) offer solvent-free or solvent-minimized approaches with exceptional concentration capabilities. These techniques can achieve enrichment factors exceeding 1000-fold while simultaneously removing interfering matrix components. Novel SPME coatings incorporating nanomaterials have shown particular promise for selective extraction of challenging analytes.
Sample filtration technologies have advanced significantly with the development of specialized membrane materials that minimize analyte adsorption while effectively removing particulates. Modern filtration systems incorporating multiple filtration mechanisms can remove both particulate matter and dissolved interferents, protecting analytical columns and reducing background noise in detection systems.
Solid-phase extraction (SPE) remains one of the most versatile sample preparation techniques for HPLC analysis. Modern SPE materials offer selective retention mechanisms including reversed-phase, normal-phase, ion-exchange, and mixed-mode interactions. Advanced polymer-based SPE sorbents demonstrate superior recovery rates compared to traditional silica-based materials, with recovery improvements of 15-30% for complex biological samples. Automated SPE systems further enhance reproducibility by minimizing human error and standardizing extraction parameters.
Liquid-liquid extraction (LLE) techniques have evolved with the development of supported liquid extraction (SLE) and dispersive liquid-liquid microextraction (DLLME). These approaches require minimal solvent volumes while achieving concentration factors of 100-1000 times for trace analytes. The integration of salting-out assisted liquid-liquid extraction (SALLE) with HPLC has shown particular promise for polar compounds in aqueous matrices, improving detection limits by up to two orders of magnitude.
Protein precipitation techniques are essential for biological sample preparation, with recent advances in precipitation agents and protocols significantly reducing matrix effects. Combining protein precipitation with phospholipid removal technologies has demonstrated a 40-60% reduction in ion suppression effects during LC-MS/MS analysis, directly enhancing detector response for target analytes in complex biological matrices.
Microextraction techniques including solid-phase microextraction (SPME), stir bar sorptive extraction (SBSE), and thin-film microextraction (TFME) offer solvent-free or solvent-minimized approaches with exceptional concentration capabilities. These techniques can achieve enrichment factors exceeding 1000-fold while simultaneously removing interfering matrix components. Novel SPME coatings incorporating nanomaterials have shown particular promise for selective extraction of challenging analytes.
Sample filtration technologies have advanced significantly with the development of specialized membrane materials that minimize analyte adsorption while effectively removing particulates. Modern filtration systems incorporating multiple filtration mechanisms can remove both particulate matter and dissolved interferents, protecting analytical columns and reducing background noise in detection systems.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!