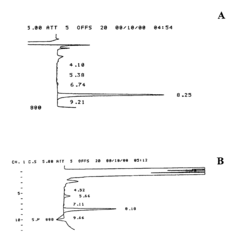
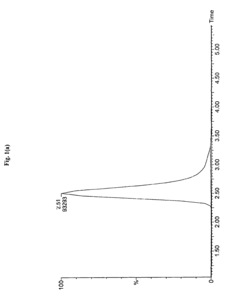
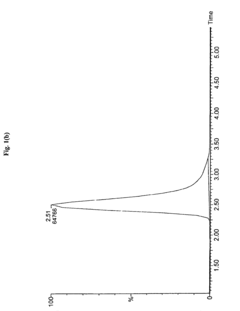
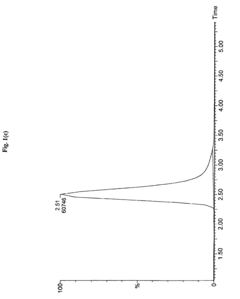
Measure Retention Factor Consistency in HPLC
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Retention Factor Background and Objectives
High-performance liquid chromatography (HPLC) has evolved significantly since its inception in the late 1960s, becoming an indispensable analytical technique in pharmaceutical, environmental, and food industries. The retention factor (k), also known as capacity factor, represents one of the most fundamental parameters in chromatographic analysis, serving as a critical indicator of separation efficiency and method reliability.
The historical development of HPLC retention factor measurement has progressed from rudimentary manual calculations to sophisticated automated systems. Early chromatographers relied on graphical methods and simple mathematical formulas to determine retention factors, often resulting in significant variability between laboratories. The 1980s and 1990s witnessed substantial improvements in instrumentation precision and computational capabilities, enabling more consistent retention factor determinations.
Recent technological advancements have introduced new challenges and opportunities in retention factor consistency. The miniaturization of HPLC systems, ultra-high-pressure capabilities, and novel stationary phase materials have expanded the application scope while simultaneously demanding higher precision in retention factor measurements. The emergence of quality-by-design approaches in analytical method development has further emphasized the importance of retention factor consistency as a critical quality attribute.
Current trends indicate a growing focus on robustness testing and system suitability parameters that directly impact retention factor consistency. The pharmaceutical industry, in particular, has established stringent guidelines for method validation that include retention factor reproducibility as a key performance indicator. Regulatory bodies increasingly scrutinize chromatographic method consistency, making retention factor reliability a compliance requirement rather than merely a scientific goal.
The primary objectives of investigating retention factor consistency in HPLC encompass several dimensions. First, to establish standardized protocols for retention factor determination that minimize inter-laboratory and inter-instrument variability. Second, to identify and quantify the critical factors affecting retention consistency, including mobile phase composition, temperature fluctuations, column aging, and sample preparation techniques. Third, to develop predictive models that can anticipate retention factor shifts under varying operational conditions.
Additionally, this technical investigation aims to explore innovative approaches for real-time monitoring and adjustment of chromatographic parameters to maintain consistent retention factors throughout analytical campaigns. The ultimate goal is to enhance the reliability of HPLC methods across different application domains, ensuring that analytical results remain comparable regardless of when or where the analysis is performed.
The historical development of HPLC retention factor measurement has progressed from rudimentary manual calculations to sophisticated automated systems. Early chromatographers relied on graphical methods and simple mathematical formulas to determine retention factors, often resulting in significant variability between laboratories. The 1980s and 1990s witnessed substantial improvements in instrumentation precision and computational capabilities, enabling more consistent retention factor determinations.
Recent technological advancements have introduced new challenges and opportunities in retention factor consistency. The miniaturization of HPLC systems, ultra-high-pressure capabilities, and novel stationary phase materials have expanded the application scope while simultaneously demanding higher precision in retention factor measurements. The emergence of quality-by-design approaches in analytical method development has further emphasized the importance of retention factor consistency as a critical quality attribute.
Current trends indicate a growing focus on robustness testing and system suitability parameters that directly impact retention factor consistency. The pharmaceutical industry, in particular, has established stringent guidelines for method validation that include retention factor reproducibility as a key performance indicator. Regulatory bodies increasingly scrutinize chromatographic method consistency, making retention factor reliability a compliance requirement rather than merely a scientific goal.
The primary objectives of investigating retention factor consistency in HPLC encompass several dimensions. First, to establish standardized protocols for retention factor determination that minimize inter-laboratory and inter-instrument variability. Second, to identify and quantify the critical factors affecting retention consistency, including mobile phase composition, temperature fluctuations, column aging, and sample preparation techniques. Third, to develop predictive models that can anticipate retention factor shifts under varying operational conditions.
Additionally, this technical investigation aims to explore innovative approaches for real-time monitoring and adjustment of chromatographic parameters to maintain consistent retention factors throughout analytical campaigns. The ultimate goal is to enhance the reliability of HPLC methods across different application domains, ensuring that analytical results remain comparable regardless of when or where the analysis is performed.
Market Demand for Consistent HPLC Analysis
The global market for High-Performance Liquid Chromatography (HPLC) continues to expand significantly, driven by increasing demands for precise analytical techniques across pharmaceutical, biotechnology, food safety, environmental monitoring, and clinical diagnostics sectors. Current market valuations place the HPLC market at approximately 4.5 billion USD, with projections indicating growth to reach 6.4 billion USD by 2027, representing a compound annual growth rate of 5.2%.
Within this broader market, the demand for consistent retention factor measurement in HPLC analysis has become particularly pronounced. Pharmaceutical companies, which constitute the largest market segment at 35% of total HPLC usage, require exceptional consistency in analytical methods to meet stringent regulatory requirements for drug development and quality control processes.
Contract Research Organizations (CROs) have emerged as significant drivers of demand for consistent HPLC methodologies. The global outsourcing trend in pharmaceutical research has led to a 7.8% annual growth in CRO services, with analytical testing representing a substantial portion of this expansion. These organizations require standardized, reproducible HPLC methods that can deliver consistent results across different laboratory settings and equipment configurations.
Academic and research institutions contribute substantially to market demand, particularly as reproducibility challenges in scientific research gain increased attention. Recent surveys indicate that over 70% of researchers have experienced difficulties replicating published chromatographic methods, highlighting the critical need for improved retention factor consistency.
The food and beverage industry represents another rapidly growing segment, with increasing regulatory scrutiny driving demand for reliable analytical methods. Consistent HPLC analysis is essential for detecting contaminants, verifying nutritional content, and ensuring product safety across global supply chains.
Geographically, North America dominates the market with approximately 40% share, followed by Europe at 30% and Asia-Pacific at 25%. However, the Asia-Pacific region demonstrates the fastest growth rate at 8.3% annually, driven by expanding pharmaceutical manufacturing, contract research activities, and strengthening regulatory frameworks in countries like China and India.
Key market trends include increasing automation of HPLC systems, integration with advanced data analytics platforms, and growing demand for method transfer capabilities that ensure consistent retention factors across different instruments and laboratories. The push toward multi-laboratory standardization has created specific market opportunities for technologies that enhance method robustness and reproducibility.
Within this broader market, the demand for consistent retention factor measurement in HPLC analysis has become particularly pronounced. Pharmaceutical companies, which constitute the largest market segment at 35% of total HPLC usage, require exceptional consistency in analytical methods to meet stringent regulatory requirements for drug development and quality control processes.
Contract Research Organizations (CROs) have emerged as significant drivers of demand for consistent HPLC methodologies. The global outsourcing trend in pharmaceutical research has led to a 7.8% annual growth in CRO services, with analytical testing representing a substantial portion of this expansion. These organizations require standardized, reproducible HPLC methods that can deliver consistent results across different laboratory settings and equipment configurations.
Academic and research institutions contribute substantially to market demand, particularly as reproducibility challenges in scientific research gain increased attention. Recent surveys indicate that over 70% of researchers have experienced difficulties replicating published chromatographic methods, highlighting the critical need for improved retention factor consistency.
The food and beverage industry represents another rapidly growing segment, with increasing regulatory scrutiny driving demand for reliable analytical methods. Consistent HPLC analysis is essential for detecting contaminants, verifying nutritional content, and ensuring product safety across global supply chains.
Geographically, North America dominates the market with approximately 40% share, followed by Europe at 30% and Asia-Pacific at 25%. However, the Asia-Pacific region demonstrates the fastest growth rate at 8.3% annually, driven by expanding pharmaceutical manufacturing, contract research activities, and strengthening regulatory frameworks in countries like China and India.
Key market trends include increasing automation of HPLC systems, integration with advanced data analytics platforms, and growing demand for method transfer capabilities that ensure consistent retention factors across different instruments and laboratories. The push toward multi-laboratory standardization has created specific market opportunities for technologies that enhance method robustness and reproducibility.
Current Challenges in Retention Factor Measurement
Despite significant advancements in HPLC technology, achieving consistent retention factor (k) measurements remains a persistent challenge in analytical chemistry. Instrument-to-instrument variability presents a fundamental obstacle, as even identical HPLC systems from the same manufacturer can produce different retention factors for the same compound under seemingly identical conditions. This variability stems from subtle differences in flow path geometry, detector cell volumes, and connection tubing dimensions that are difficult to standardize across instruments.
Temperature control represents another critical challenge affecting retention factor consistency. Even minor fluctuations of ±0.5°C can significantly alter retention behavior, particularly for temperature-sensitive compounds. Many laboratories struggle with inadequate temperature control systems or environmental variations that introduce unpredictable shifts in retention measurements.
Mobile phase composition inconsistencies further complicate retention factor reproducibility. Variations in solvent quality between manufacturers, batches, or even storage conditions can alter selectivity and retention characteristics. The challenge is magnified when dealing with gradient elution methods, where minute differences in gradient formation and mixing efficiency between systems can dramatically affect retention behavior.
Column-to-column variability remains perhaps the most significant hurdle to consistent retention factor measurements. Despite advances in manufacturing processes, stationary phases still exhibit batch-to-batch variations in surface chemistry, particle size distribution, and packing density. These differences manifest as shifts in retention factors that cannot be fully compensated for through method adjustments.
Method transfer between laboratories introduces additional complexity, as differences in data processing algorithms and integration parameters can yield different retention factor values even from identical chromatograms. The lack of standardized approaches to baseline correction and peak integration contributes significantly to inter-laboratory variability.
Aging effects on both columns and instruments present long-term challenges for retention factor consistency. Column performance inevitably changes over time due to chemical degradation, contamination, and mechanical stress. Similarly, pump performance, detector sensitivity, and system plumbing characteristics evolve with use, creating temporal drift in retention measurements that complicates long-term studies and method validation.
The increasing complexity of sample matrices in modern applications introduces matrix-related challenges to retention factor consistency. Co-eluting compounds, ion-suppression effects, and matrix-induced changes to mobile phase properties can all alter retention behavior in ways that are difficult to predict or control, particularly in biological, environmental, and food analysis applications.
Temperature control represents another critical challenge affecting retention factor consistency. Even minor fluctuations of ±0.5°C can significantly alter retention behavior, particularly for temperature-sensitive compounds. Many laboratories struggle with inadequate temperature control systems or environmental variations that introduce unpredictable shifts in retention measurements.
Mobile phase composition inconsistencies further complicate retention factor reproducibility. Variations in solvent quality between manufacturers, batches, or even storage conditions can alter selectivity and retention characteristics. The challenge is magnified when dealing with gradient elution methods, where minute differences in gradient formation and mixing efficiency between systems can dramatically affect retention behavior.
Column-to-column variability remains perhaps the most significant hurdle to consistent retention factor measurements. Despite advances in manufacturing processes, stationary phases still exhibit batch-to-batch variations in surface chemistry, particle size distribution, and packing density. These differences manifest as shifts in retention factors that cannot be fully compensated for through method adjustments.
Method transfer between laboratories introduces additional complexity, as differences in data processing algorithms and integration parameters can yield different retention factor values even from identical chromatograms. The lack of standardized approaches to baseline correction and peak integration contributes significantly to inter-laboratory variability.
Aging effects on both columns and instruments present long-term challenges for retention factor consistency. Column performance inevitably changes over time due to chemical degradation, contamination, and mechanical stress. Similarly, pump performance, detector sensitivity, and system plumbing characteristics evolve with use, creating temporal drift in retention measurements that complicates long-term studies and method validation.
The increasing complexity of sample matrices in modern applications introduces matrix-related challenges to retention factor consistency. Co-eluting compounds, ion-suppression effects, and matrix-induced changes to mobile phase properties can all alter retention behavior in ways that are difficult to predict or control, particularly in biological, environmental, and food analysis applications.
Existing Methods for Retention Factor Consistency
01 Mobile phase composition optimization for HPLC retention factor consistency
The composition of the mobile phase in HPLC significantly affects retention factor consistency. Optimizing parameters such as pH, buffer concentration, organic solvent ratio, and ionic strength can improve reproducibility of retention factors. Careful selection and preparation of mobile phase components helps maintain consistent chromatographic conditions across multiple analyses, reducing variability in retention times and improving quantitative reliability.- Mobile phase composition optimization for HPLC retention factor consistency: The composition of the mobile phase in HPLC significantly affects retention factor consistency. Optimizing parameters such as pH, buffer concentration, organic solvent ratio, and additives can improve reproducibility of retention times. Careful selection and preparation of mobile phase components helps maintain consistent chromatographic conditions across multiple analyses, ensuring reliable retention factor values.
- Temperature control systems for HPLC retention factor stability: Temperature fluctuations can significantly impact retention factor consistency in HPLC analyses. Advanced temperature control systems that maintain precise column temperatures throughout the analytical process help minimize variations in retention factors. These systems include column ovens with accurate temperature regulation capabilities and thermal stabilization mechanisms that prevent temperature gradients within the column.
- Column conditioning and equilibration procedures: Proper column conditioning and equilibration are essential for achieving consistent retention factors in HPLC analyses. Standardized protocols for column preparation, including initial washing, equilibration with mobile phase, and regular conditioning between analyses, help maintain stable chromatographic conditions. These procedures ensure that the stationary phase maintains consistent interaction properties with analytes across multiple injections.
- Reference standard methods for retention factor calibration: The use of reference standards for calibrating and monitoring retention factor consistency improves the reliability of HPLC analyses. Internal standards with known retention behaviors can be incorporated into samples to normalize retention factors across different analytical runs. Regular system suitability testing with standard compounds helps identify and correct variations in chromatographic performance that might affect retention factor consistency.
- Advanced data processing algorithms for retention factor normalization: Sophisticated data processing algorithms can compensate for minor variations in experimental conditions that affect retention factors. These computational approaches include retention time alignment algorithms, normalization techniques based on internal standards, and statistical methods for identifying and correcting systematic shifts in retention behavior. Machine learning approaches can also be employed to predict and adjust for variations in retention factors across different analytical runs.
02 Temperature control systems for HPLC retention factor stability
Temperature fluctuations can significantly impact retention factor consistency in HPLC analysis. Implementing precise temperature control systems for both the column and mobile phase helps maintain stable retention factors. Advanced temperature regulation mechanisms with feedback controls ensure that thermal conditions remain constant throughout analytical runs, reducing retention time drift and improving method reproducibility.Expand Specific Solutions03 Column conditioning and equilibration protocols
Proper column conditioning and equilibration are essential for consistent retention factors in HPLC analysis. Standardized protocols for column preparation, including sufficient equilibration time with mobile phase, regular cleaning procedures, and controlled flow rates during conditioning, help establish stable column chemistry. These practices ensure reproducible interactions between analytes and stationary phase, leading to consistent retention behavior across multiple analyses.Expand Specific Solutions04 Internal standard calibration methods for retention factor normalization
Internal standard calibration techniques can compensate for variations in HPLC retention factors. By incorporating reference compounds with known retention characteristics, analysts can normalize retention data across different analytical runs. This approach allows for the calculation of relative retention factors that remain consistent despite minor fluctuations in chromatographic conditions, enhancing the reliability of qualitative and quantitative analyses.Expand Specific Solutions05 Automated system suitability testing for retention factor monitoring
Automated system suitability testing protocols can continuously monitor and verify HPLC retention factor consistency. These systems employ specialized software algorithms to track retention parameters in real-time, flagging deviations that exceed predetermined thresholds. Regular system performance checks with standard reference materials help identify and correct issues affecting retention factor stability before they impact analytical results.Expand Specific Solutions
Key Industry Players in HPLC Instrumentation
The HPLC retention factor consistency measurement market is currently in a growth phase, with increasing demand driven by pharmaceutical quality control requirements. The global market is expanding steadily as analytical testing becomes more stringent in drug development and manufacturing. Leading players include established analytical instrument providers like Agilent Technologies, who dominate with comprehensive HPLC solutions, alongside pharmaceutical companies with significant internal capabilities such as Merck Patent GmbH, Bristol Myers Squibb, and Daiichi Sankyo. The technology has reached maturity in standard applications but continues to evolve for complex separations, with companies like Janssen Pharmaceutica and F. Hoffmann-La Roche investing in advanced methodologies to enhance reproducibility and precision for challenging compounds, particularly in biologics analysis.
Bristol Myers Squibb Co.
Technical Solution: Bristol Myers Squibb has developed a sophisticated approach to HPLC retention factor consistency through their Analytical Control Strategy platform. Their technology incorporates real-time mobile phase monitoring using inline refractive index detection to verify composition consistency before reaching the analytical column. BMS's Method Robustness Enhancement Protocol systematically evaluates the impact of minor variations in critical parameters on retention factors, establishing acceptable operating ranges that guarantee consistent results. Their Column Management System tracks individual column performance over time, allowing predictive maintenance before retention inconsistencies occur. BMS has pioneered the use of dual-wavelength detection for continuous monitoring of column chemistry changes that might affect retention. Their Method Transfer Assessment Tool quantitatively evaluates retention factor consistency when methods are transferred between different instruments or laboratories, providing statistical confidence in analytical equivalence. Additionally, BMS has implemented automated system suitability testing with trend analysis to detect gradual shifts in retention factors before they exceed acceptance criteria.
Strengths: Comprehensive approach addressing both preventive and monitoring aspects of retention consistency; strong focus on method transfer reliability between different sites; sophisticated statistical tools for trend analysis and early intervention. Weaknesses: Systems optimized primarily for pharmaceutical applications; significant computational resources required for full implementation; some components require specialized analytical expertise beyond typical HPLC users.
Merck Patent GmbH
Technical Solution: Merck Patent GmbH has pioneered innovative approaches to HPLC retention factor consistency through their Chromolith® monolithic column technology. Unlike traditional particle-packed columns, these monolithic silica columns provide more consistent flow characteristics and reduced backpressure, resulting in more stable retention factors across varying flow rates. Their SmartSeal™ technology prevents mobile phase evaporation during extended analyses, eliminating concentration changes that affect retention. Merck's ZipTip® sample preparation technology removes matrix interferents that can alter column chemistry and retention behavior over time. Their ChromSwordAuto® software incorporates retention factor monitoring with automated method adjustment capabilities, continuously optimizing separation parameters to maintain consistent retention. Additionally, Merck has developed specialized reference standard mixtures specifically designed for monitoring retention factor drift in different application areas, allowing analysts to implement standardized system suitability protocols.
Strengths: Monolithic column technology provides superior mechanical stability and consistent performance over extended use; comprehensive approach addressing multiple sources of retention variability; strong integration between consumables and software. Weaknesses: Monolithic columns offer fewer stationary phase chemistry options compared to traditional columns; higher initial investment costs; some technologies require specialized training for optimal implementation.
Critical Innovations in HPLC Retention Measurement
Preparation of samples for LC-MS/MS using magnetic particles
PatentActiveUS7815803B2
Innovation
- The use of functionalized magnetic particles with a hydrophobic surface for extracting low molecular weight compounds from complex biological samples, allowing for reversible binding and efficient enrichment of analytes, even in the presence of abundant lipids, peptides, and proteins, with minimal particle quantities required.
Graphically representing a number of measurement data sets
PatentInactiveUS20110029256A1
Innovation
- A method for graphically representing measurement data sets by selecting a subset and distinguishing them using different graphical styles, such as varying transparency, color, or line sharpness, allowing for a primary data set to be highlighted and secondary sets to be represented differently, facilitating analysis and comparison.
Regulatory Standards for HPLC Method Validation
Regulatory standards for HPLC method validation are critical frameworks that ensure the reliability, reproducibility, and accuracy of analytical methods used in pharmaceutical, food, and environmental testing industries. These standards are established by various international regulatory bodies including the International Conference on Harmonization (ICH), the United States Pharmacopeia (USP), the European Pharmacopoeia (EP), and the Food and Drug Administration (FDA).
The ICH Q2(R1) guideline, "Validation of Analytical Procedures: Text and Methodology," serves as the cornerstone document for HPLC method validation. It outlines specific parameters that must be evaluated, including accuracy, precision, specificity, detection limit, quantitation limit, linearity, range, and robustness. For retention factor consistency specifically, the guideline emphasizes system suitability testing as a crucial component.
FDA's Guidance for Industry on Analytical Procedures and Methods Validation provides additional requirements for chromatographic methods, stipulating that retention factor (k') values should typically exceed 2.0 to ensure adequate separation from the void volume. The guidance also recommends monitoring retention factor consistency through control charts and establishing appropriate acceptance criteria based on method requirements.
The USP <621> Chromatography chapter details specific requirements for system suitability testing, including retention factor evaluation. It recommends that retention factors should be consistent within ±15% of established values during method validation and routine analysis. This standard is particularly relevant when measuring retention factor consistency in HPLC methods.
For laboratories seeking ISO/IEC 17025 accreditation, additional requirements apply to method validation and verification processes. These standards mandate documented procedures for evaluating chromatographic parameters, including retention factor consistency, and require ongoing monitoring through quality control samples.
AOAC International guidelines provide sector-specific validation requirements, particularly relevant for food and environmental testing applications. Their Appendix F outlines chromatographic method validation parameters, including specific criteria for retention time and retention factor reproducibility.
Regulatory bodies increasingly emphasize the implementation of Quality by Design (QbD) principles in analytical method development and validation. This approach requires systematic evaluation of critical method parameters affecting retention factor consistency, including mobile phase composition, pH, temperature, and flow rate.
Recent updates to regulatory standards have incorporated advances in automated method development and validation technologies. These updates acknowledge the value of retention modeling software and automated robustness testing in establishing and maintaining consistent retention factors across different analytical conditions.
The ICH Q2(R1) guideline, "Validation of Analytical Procedures: Text and Methodology," serves as the cornerstone document for HPLC method validation. It outlines specific parameters that must be evaluated, including accuracy, precision, specificity, detection limit, quantitation limit, linearity, range, and robustness. For retention factor consistency specifically, the guideline emphasizes system suitability testing as a crucial component.
FDA's Guidance for Industry on Analytical Procedures and Methods Validation provides additional requirements for chromatographic methods, stipulating that retention factor (k') values should typically exceed 2.0 to ensure adequate separation from the void volume. The guidance also recommends monitoring retention factor consistency through control charts and establishing appropriate acceptance criteria based on method requirements.
The USP <621> Chromatography chapter details specific requirements for system suitability testing, including retention factor evaluation. It recommends that retention factors should be consistent within ±15% of established values during method validation and routine analysis. This standard is particularly relevant when measuring retention factor consistency in HPLC methods.
For laboratories seeking ISO/IEC 17025 accreditation, additional requirements apply to method validation and verification processes. These standards mandate documented procedures for evaluating chromatographic parameters, including retention factor consistency, and require ongoing monitoring through quality control samples.
AOAC International guidelines provide sector-specific validation requirements, particularly relevant for food and environmental testing applications. Their Appendix F outlines chromatographic method validation parameters, including specific criteria for retention time and retention factor reproducibility.
Regulatory bodies increasingly emphasize the implementation of Quality by Design (QbD) principles in analytical method development and validation. This approach requires systematic evaluation of critical method parameters affecting retention factor consistency, including mobile phase composition, pH, temperature, and flow rate.
Recent updates to regulatory standards have incorporated advances in automated method development and validation technologies. These updates acknowledge the value of retention modeling software and automated robustness testing in establishing and maintaining consistent retention factors across different analytical conditions.
Quality Control Strategies for HPLC Analysis
Quality control in HPLC analysis requires systematic approaches to ensure consistent and reliable results. Establishing robust quality control strategies begins with the implementation of system suitability tests (SSTs) that verify the chromatographic system's performance before sample analysis. These tests typically include parameters such as retention factor consistency, resolution, tailing factor, and theoretical plate count, which must meet predetermined acceptance criteria.
The development of control charts represents a cornerstone of HPLC quality control, enabling analysts to monitor retention factor consistency over time. By plotting retention times or retention factors against analysis date, laboratories can identify trends, shifts, or cyclical patterns that may indicate system deterioration or methodological issues. Upper and lower control limits, typically set at ±3 standard deviations from the mean, provide objective criteria for intervention.
Reference standard protocols constitute another critical component of quality control strategies. The use of certified reference materials with known retention characteristics allows for regular calibration and verification of the HPLC system. These standards should be analyzed at defined intervals—daily, weekly, or at the beginning of each analytical batch—depending on the criticality of the analysis and regulatory requirements.
Method validation parameters directly impact retention factor consistency and must be thoroughly evaluated. Specificity, linearity, accuracy, precision, detection limit, quantitation limit, and robustness assessments provide comprehensive evidence of method reliability. Particularly for retention factor consistency, robustness testing that deliberately varies critical parameters (mobile phase composition, pH, temperature, flow rate) helps establish acceptable operating ranges.
Preventive maintenance schedules represent a proactive quality control measure. Regular replacement of consumables (frits, filters, guard columns), inspection of pump seals, detector lamps, and autosampler components helps prevent retention time drift and system failures. Documentation of maintenance activities creates a traceable history that can be correlated with retention factor performance.
Electronic data management systems enhance quality control by automating data acquisition, processing, and reporting. Modern chromatography data systems can flag out-of-specification results, track system suitability trends, and generate compliance reports. Integration with laboratory information management systems (LIMS) further strengthens data integrity and facilitates comprehensive quality oversight across multiple instruments and analysts.
Training programs ensure that analysts understand the critical factors affecting retention consistency. Standard operating procedures (SOPs) should detail proper column conditioning, equilibration protocols, sample preparation techniques, and system operation guidelines. Regular competency assessments verify that personnel maintain the necessary skills to execute methods consistently.
The development of control charts represents a cornerstone of HPLC quality control, enabling analysts to monitor retention factor consistency over time. By plotting retention times or retention factors against analysis date, laboratories can identify trends, shifts, or cyclical patterns that may indicate system deterioration or methodological issues. Upper and lower control limits, typically set at ±3 standard deviations from the mean, provide objective criteria for intervention.
Reference standard protocols constitute another critical component of quality control strategies. The use of certified reference materials with known retention characteristics allows for regular calibration and verification of the HPLC system. These standards should be analyzed at defined intervals—daily, weekly, or at the beginning of each analytical batch—depending on the criticality of the analysis and regulatory requirements.
Method validation parameters directly impact retention factor consistency and must be thoroughly evaluated. Specificity, linearity, accuracy, precision, detection limit, quantitation limit, and robustness assessments provide comprehensive evidence of method reliability. Particularly for retention factor consistency, robustness testing that deliberately varies critical parameters (mobile phase composition, pH, temperature, flow rate) helps establish acceptable operating ranges.
Preventive maintenance schedules represent a proactive quality control measure. Regular replacement of consumables (frits, filters, guard columns), inspection of pump seals, detector lamps, and autosampler components helps prevent retention time drift and system failures. Documentation of maintenance activities creates a traceable history that can be correlated with retention factor performance.
Electronic data management systems enhance quality control by automating data acquisition, processing, and reporting. Modern chromatography data systems can flag out-of-specification results, track system suitability trends, and generate compliance reports. Integration with laboratory information management systems (LIMS) further strengthens data integrity and facilitates comprehensive quality oversight across multiple instruments and analysts.
Training programs ensure that analysts understand the critical factors affecting retention consistency. Standard operating procedures (SOPs) should detail proper column conditioning, equilibration protocols, sample preparation techniques, and system operation guidelines. Regular competency assessments verify that personnel maintain the necessary skills to execute methods consistently.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!