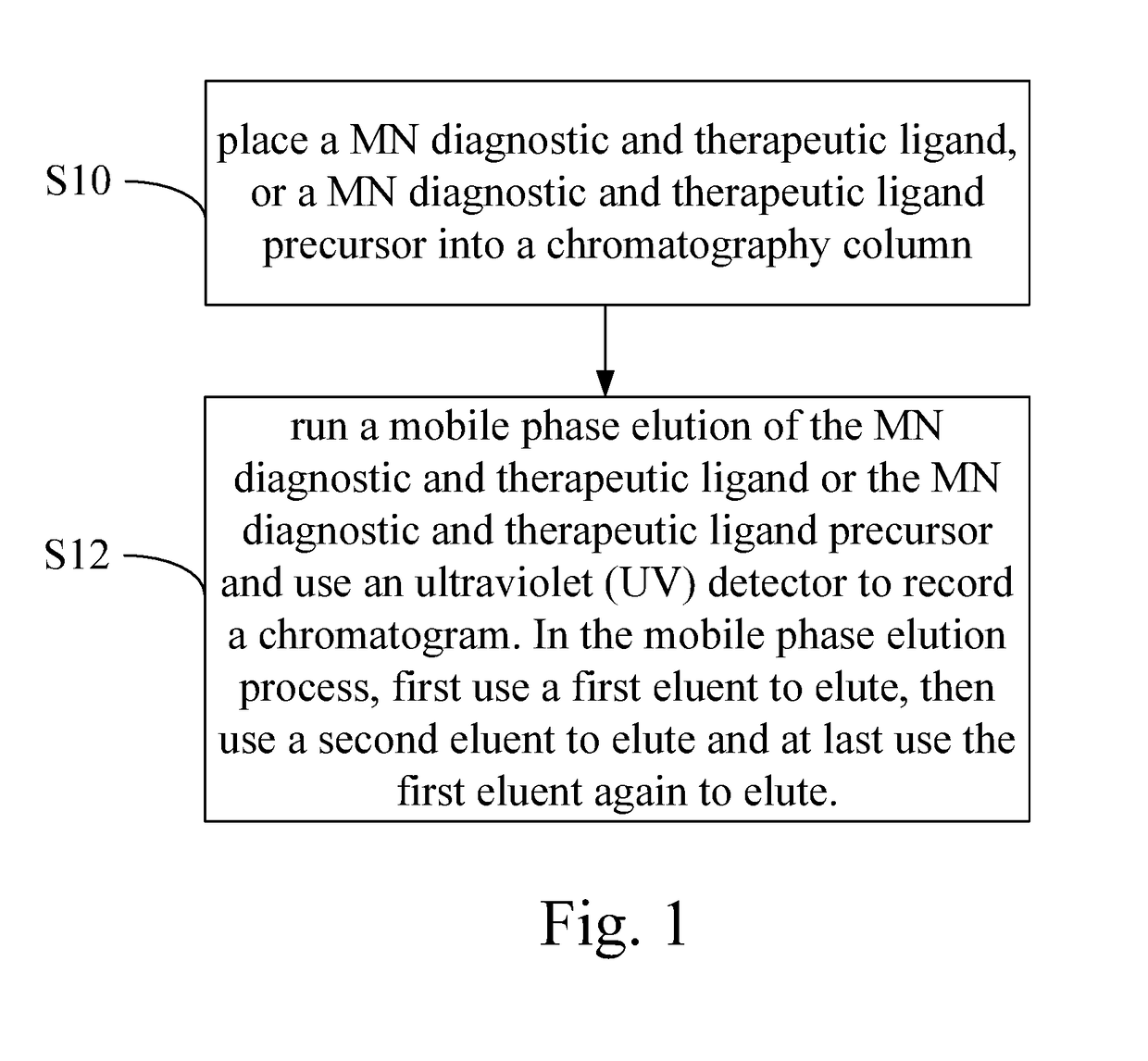
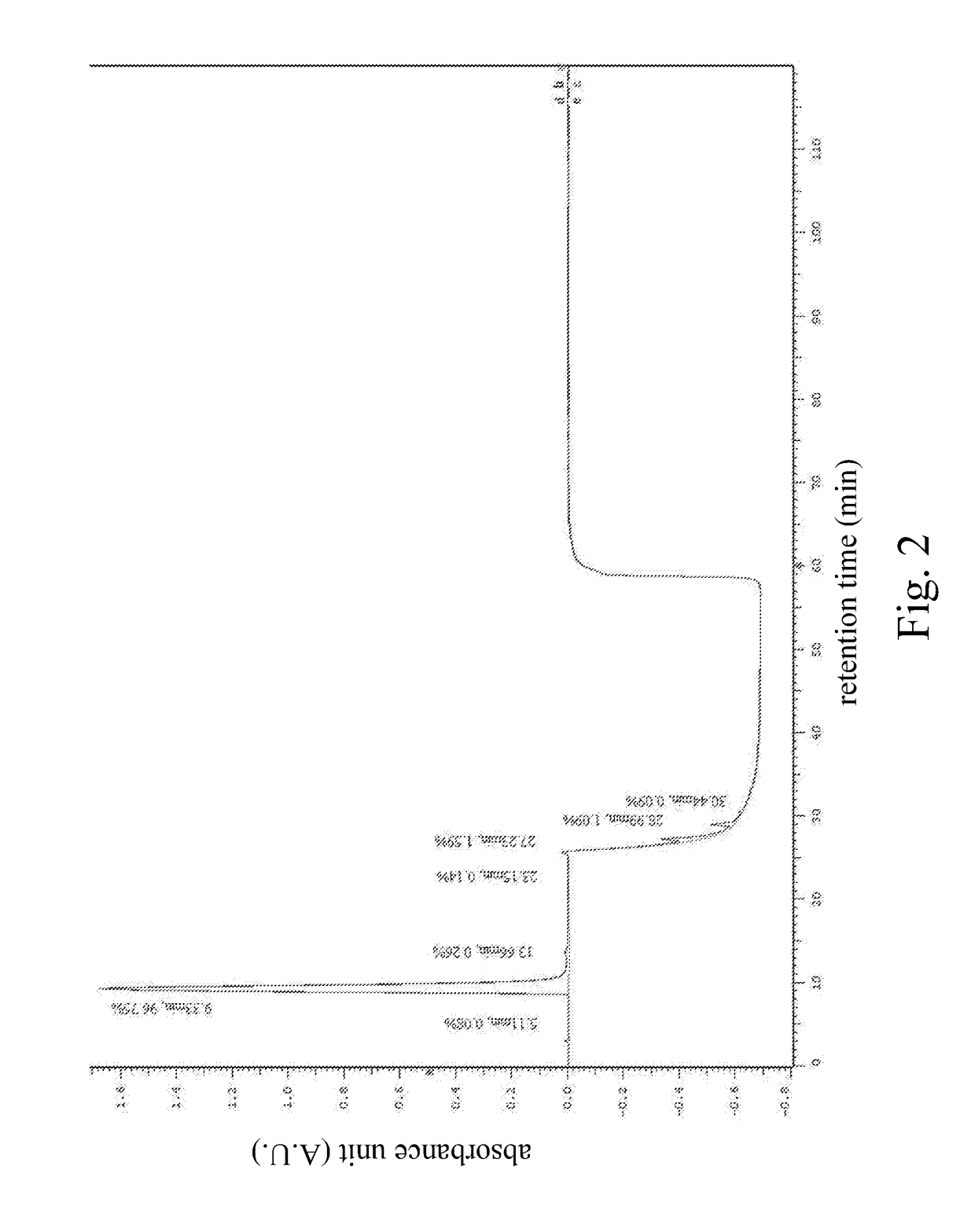
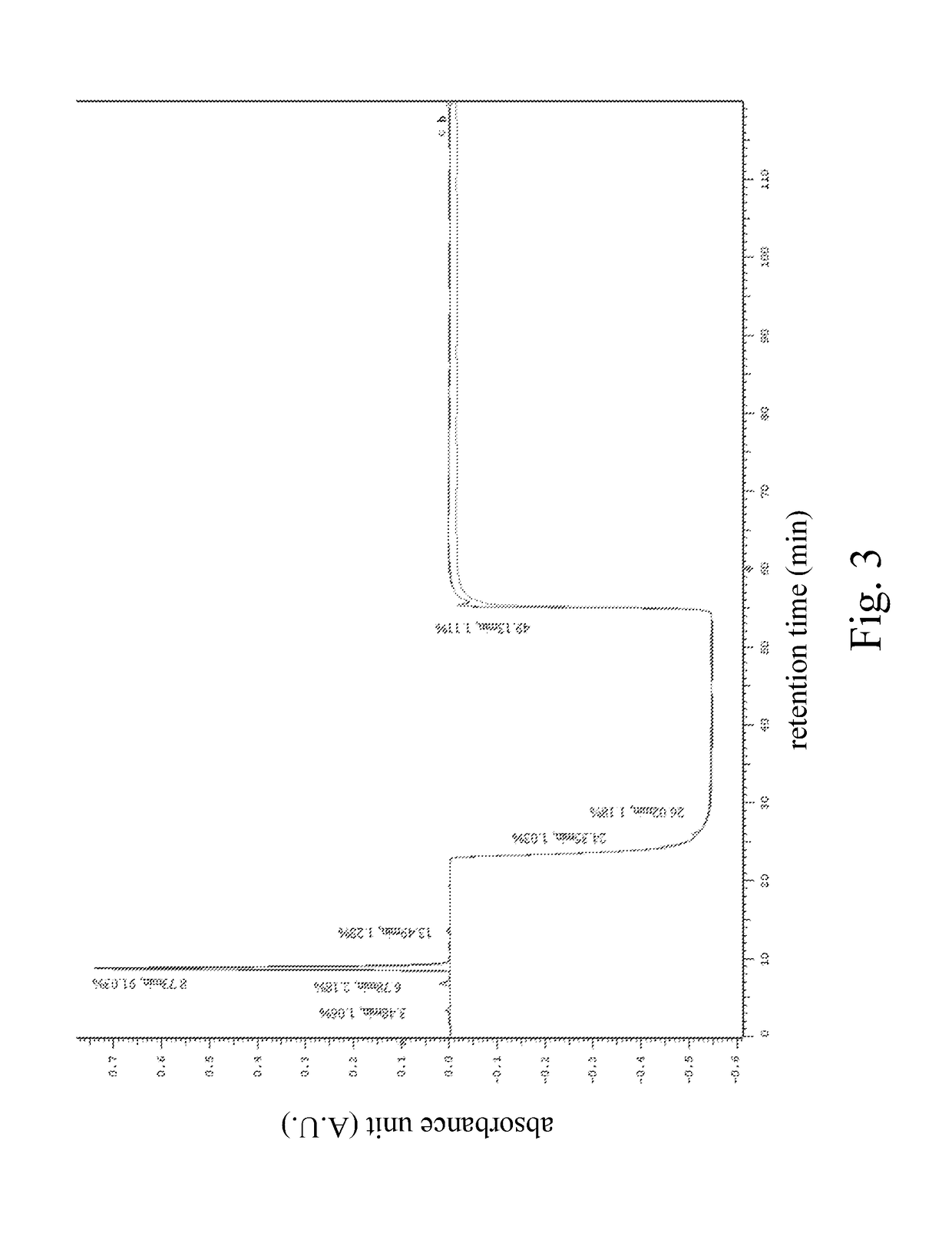
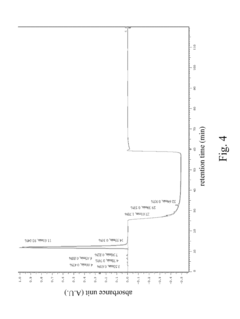
Optimize HPLC Mobile Phase for Reproducibility
SEP 19, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Mobile Phase Evolution and Optimization Goals
High-performance liquid chromatography (HPLC) has evolved significantly since its inception in the late 1960s, transforming from rudimentary separation techniques to sophisticated analytical methodologies. The evolution of mobile phase optimization represents one of the most critical aspects of this technological progression. Initially, HPLC systems utilized simple isocratic conditions with limited solvent options, primarily methanol and water. The 1980s witnessed the introduction of gradient elution techniques, expanding the capability to separate complex mixtures with varying polarities.
The advent of buffer systems in the 1990s marked a pivotal advancement, enabling better control of ionization states for improved peak shapes and retention time consistency. This period also saw the emergence of pH modifiers and ion-pairing reagents, further enhancing separation selectivity. The early 2000s brought significant innovations in mobile phase additives, including the implementation of volatile buffers compatible with mass spectrometry detection systems.
Recent developments have focused on green chemistry principles, with trends toward reduced organic solvent consumption, environmentally friendly alternatives, and miniaturization of separation systems. The introduction of ultra-high-performance liquid chromatography (UHPLC) has driven the need for mobile phases capable of withstanding higher pressures while maintaining chemical stability and reproducibility.
The primary goal of mobile phase optimization for reproducibility centers on achieving consistent chromatographic performance across multiple analyses, instruments, and laboratories. This encompasses several key objectives: minimizing retention time variability, ensuring consistent peak shapes, maintaining separation efficiency, and reducing system-to-system variations. Reproducibility challenges often stem from subtle changes in mobile phase composition, including variations in organic modifier concentration, buffer strength, pH fluctuations, and contamination issues.
Temperature control has emerged as a critical parameter in modern HPLC methods, with precise regulation becoming standard practice to enhance reproducibility. Additionally, the industry has recognized the importance of standardized preparation protocols and quality control measures for mobile phases, including filtration procedures, degassing techniques, and storage considerations.
Future optimization goals include the development of self-adjusting mobile phase systems capable of responding to environmental variations, implementation of machine learning algorithms for predictive maintenance of mobile phase quality, and creation of universal mobile phase formulations that maintain reproducibility across diverse column chemistries. The integration of real-time monitoring technologies for mobile phase parameters represents another frontier, potentially revolutionizing how chromatographers ensure method reproducibility in routine and research applications.
The advent of buffer systems in the 1990s marked a pivotal advancement, enabling better control of ionization states for improved peak shapes and retention time consistency. This period also saw the emergence of pH modifiers and ion-pairing reagents, further enhancing separation selectivity. The early 2000s brought significant innovations in mobile phase additives, including the implementation of volatile buffers compatible with mass spectrometry detection systems.
Recent developments have focused on green chemistry principles, with trends toward reduced organic solvent consumption, environmentally friendly alternatives, and miniaturization of separation systems. The introduction of ultra-high-performance liquid chromatography (UHPLC) has driven the need for mobile phases capable of withstanding higher pressures while maintaining chemical stability and reproducibility.
The primary goal of mobile phase optimization for reproducibility centers on achieving consistent chromatographic performance across multiple analyses, instruments, and laboratories. This encompasses several key objectives: minimizing retention time variability, ensuring consistent peak shapes, maintaining separation efficiency, and reducing system-to-system variations. Reproducibility challenges often stem from subtle changes in mobile phase composition, including variations in organic modifier concentration, buffer strength, pH fluctuations, and contamination issues.
Temperature control has emerged as a critical parameter in modern HPLC methods, with precise regulation becoming standard practice to enhance reproducibility. Additionally, the industry has recognized the importance of standardized preparation protocols and quality control measures for mobile phases, including filtration procedures, degassing techniques, and storage considerations.
Future optimization goals include the development of self-adjusting mobile phase systems capable of responding to environmental variations, implementation of machine learning algorithms for predictive maintenance of mobile phase quality, and creation of universal mobile phase formulations that maintain reproducibility across diverse column chemistries. The integration of real-time monitoring technologies for mobile phase parameters represents another frontier, potentially revolutionizing how chromatographers ensure method reproducibility in routine and research applications.
Market Demand for Reproducible HPLC Analysis
The global market for High-Performance Liquid Chromatography (HPLC) continues to expand significantly, driven by increasing demands for precise analytical methods across pharmaceutical, biotechnology, food safety, and environmental monitoring sectors. Current market valuations place the HPLC market at approximately 4.5 billion USD, with projections indicating growth to reach 6.7 billion USD by 2027, representing a compound annual growth rate of 5.2%.
Reproducibility in HPLC analysis has emerged as a critical market requirement, particularly in regulated industries where analytical consistency directly impacts product quality, regulatory compliance, and research validity. Pharmaceutical companies, which constitute the largest market segment for HPLC technologies, face stringent regulatory requirements that necessitate highly reproducible analytical methods to ensure batch-to-batch consistency in drug manufacturing.
Recent industry surveys reveal that over 70% of laboratory managers identify reproducibility challenges as a significant pain point in their analytical workflows. Mobile phase optimization represents one of the most frequently cited factors affecting reproducibility, with 65% of chromatographers reporting that variations in mobile phase composition directly impact their analytical results.
The economic implications of poor reproducibility are substantial. Research institutions and pharmaceutical companies report that method transfer failures and reproducibility issues can delay product development by 3-6 months, with associated costs ranging from 100,000 to 500,000 USD per incident. These figures underscore the tangible market demand for solutions that enhance HPLC reproducibility.
Contract Research Organizations (CROs) and Contract Manufacturing Organizations (CMOs) represent a rapidly growing market segment, with increasing outsourcing of analytical testing creating demand for standardized, reproducible methods that can be reliably transferred between organizations. This trend is particularly pronounced in emerging markets in Asia-Pacific, where the HPLC market is growing at 7.8% annually.
Technology providers have recognized this market need, with major instrument manufacturers increasingly focusing on integrated solutions that address reproducibility challenges. The market for automated mobile phase preparation systems has grown by 12% annually over the past five years, reflecting the industry's willingness to invest in technologies that enhance analytical consistency.
Academic research represents another significant market driver, with reproducibility in scientific publications receiving heightened scrutiny. Several high-profile studies have highlighted "reproducibility crises" across scientific disciplines, prompting funding agencies to emphasize reproducible methods in grant evaluations and creating additional market pressure for optimized HPLC protocols.
Reproducibility in HPLC analysis has emerged as a critical market requirement, particularly in regulated industries where analytical consistency directly impacts product quality, regulatory compliance, and research validity. Pharmaceutical companies, which constitute the largest market segment for HPLC technologies, face stringent regulatory requirements that necessitate highly reproducible analytical methods to ensure batch-to-batch consistency in drug manufacturing.
Recent industry surveys reveal that over 70% of laboratory managers identify reproducibility challenges as a significant pain point in their analytical workflows. Mobile phase optimization represents one of the most frequently cited factors affecting reproducibility, with 65% of chromatographers reporting that variations in mobile phase composition directly impact their analytical results.
The economic implications of poor reproducibility are substantial. Research institutions and pharmaceutical companies report that method transfer failures and reproducibility issues can delay product development by 3-6 months, with associated costs ranging from 100,000 to 500,000 USD per incident. These figures underscore the tangible market demand for solutions that enhance HPLC reproducibility.
Contract Research Organizations (CROs) and Contract Manufacturing Organizations (CMOs) represent a rapidly growing market segment, with increasing outsourcing of analytical testing creating demand for standardized, reproducible methods that can be reliably transferred between organizations. This trend is particularly pronounced in emerging markets in Asia-Pacific, where the HPLC market is growing at 7.8% annually.
Technology providers have recognized this market need, with major instrument manufacturers increasingly focusing on integrated solutions that address reproducibility challenges. The market for automated mobile phase preparation systems has grown by 12% annually over the past five years, reflecting the industry's willingness to invest in technologies that enhance analytical consistency.
Academic research represents another significant market driver, with reproducibility in scientific publications receiving heightened scrutiny. Several high-profile studies have highlighted "reproducibility crises" across scientific disciplines, prompting funding agencies to emphasize reproducible methods in grant evaluations and creating additional market pressure for optimized HPLC protocols.
Current Challenges in HPLC Mobile Phase Stability
High-performance liquid chromatography (HPLC) mobile phase stability represents one of the most critical yet challenging aspects in analytical chemistry. Despite significant advancements in HPLC instrumentation, mobile phase inconsistencies continue to plague laboratories worldwide, causing reproducibility issues that compromise data integrity and analytical reliability. Current challenges stem from multiple interconnected factors that require systematic understanding and mitigation strategies.
Temperature fluctuations within laboratory environments significantly impact mobile phase stability. Even minor temperature variations can alter solvent viscosity, affecting flow characteristics and retention times. Many laboratories lack adequate temperature control systems for both mobile phase storage and HPLC column compartments, resulting in day-to-day and seasonal variations that undermine method reproducibility.
Chemical degradation of mobile phase components presents another substantial challenge. Buffers commonly used in HPLC mobile phases, such as phosphates and acetates, can undergo gradual pH shifts due to microbial growth, absorption of atmospheric carbon dioxide, or chemical reactions between components. These subtle changes often go undetected until significant retention time shifts or peak shape deteriorations manifest in chromatograms.
Solvent evaporation and subsequent concentration changes represent a persistent issue, particularly with volatile organic components like acetonitrile and methanol. Inadequate sealing of solvent reservoirs allows preferential evaporation of more volatile components, gradually altering the mobile phase composition during extended analytical runs or between calibrations.
Contamination from various sources further complicates mobile phase stability. Particulate matter, dissolved gases, metal ions from storage containers, and leachables from tubing or filters can all introduce variables that affect chromatographic performance. Modern ultra-high-performance liquid chromatography (UHPLC) systems, with their enhanced sensitivity, are particularly susceptible to these contamination effects.
Preparation inconsistencies between analysts represent a significant human factor challenge. Variations in weighing, dissolution techniques, pH adjustment methods, and filtration protocols introduce subtle differences in mobile phase composition. These differences become particularly problematic in multi-shift operations or when methods transfer between laboratories.
Inadequate documentation and standardization of mobile phase preparation procedures exacerbate reproducibility issues. Many laboratories lack detailed standard operating procedures that specify critical parameters such as water quality requirements, precise buffer preparation steps, and appropriate storage conditions and expiration dates for prepared mobile phases.
The increasing complexity of modern HPLC methods, particularly those involving gradient elution with multiple solvents or complex buffer systems, multiplies the potential stability issues. Each additional component introduces new variables that must be controlled to maintain consistent chromatographic performance across analytical batches.
Temperature fluctuations within laboratory environments significantly impact mobile phase stability. Even minor temperature variations can alter solvent viscosity, affecting flow characteristics and retention times. Many laboratories lack adequate temperature control systems for both mobile phase storage and HPLC column compartments, resulting in day-to-day and seasonal variations that undermine method reproducibility.
Chemical degradation of mobile phase components presents another substantial challenge. Buffers commonly used in HPLC mobile phases, such as phosphates and acetates, can undergo gradual pH shifts due to microbial growth, absorption of atmospheric carbon dioxide, or chemical reactions between components. These subtle changes often go undetected until significant retention time shifts or peak shape deteriorations manifest in chromatograms.
Solvent evaporation and subsequent concentration changes represent a persistent issue, particularly with volatile organic components like acetonitrile and methanol. Inadequate sealing of solvent reservoirs allows preferential evaporation of more volatile components, gradually altering the mobile phase composition during extended analytical runs or between calibrations.
Contamination from various sources further complicates mobile phase stability. Particulate matter, dissolved gases, metal ions from storage containers, and leachables from tubing or filters can all introduce variables that affect chromatographic performance. Modern ultra-high-performance liquid chromatography (UHPLC) systems, with their enhanced sensitivity, are particularly susceptible to these contamination effects.
Preparation inconsistencies between analysts represent a significant human factor challenge. Variations in weighing, dissolution techniques, pH adjustment methods, and filtration protocols introduce subtle differences in mobile phase composition. These differences become particularly problematic in multi-shift operations or when methods transfer between laboratories.
Inadequate documentation and standardization of mobile phase preparation procedures exacerbate reproducibility issues. Many laboratories lack detailed standard operating procedures that specify critical parameters such as water quality requirements, precise buffer preparation steps, and appropriate storage conditions and expiration dates for prepared mobile phases.
The increasing complexity of modern HPLC methods, particularly those involving gradient elution with multiple solvents or complex buffer systems, multiplies the potential stability issues. Each additional component introduces new variables that must be controlled to maintain consistent chromatographic performance across analytical batches.
Established Strategies for Mobile Phase Reproducibility
01 Mobile phase composition control
Precise control of mobile phase composition is critical for HPLC reproducibility. This includes maintaining consistent solvent ratios, pH levels, and buffer concentrations. Automated systems for mobile phase preparation can minimize human error and ensure batch-to-batch consistency. Temperature-controlled solvent reservoirs and precise mixing technologies help maintain stable mobile phase conditions throughout analytical runs.- Mobile phase composition control for improved reproducibility: Controlling the composition of the mobile phase is crucial for HPLC reproducibility. This includes precise mixing of solvents, maintaining consistent pH levels, and using high-purity reagents. Automated systems for mobile phase preparation can minimize human error and ensure consistent composition across multiple analyses. Temperature control of the mobile phase also contributes to reproducibility by preventing solvent expansion or contraction that could affect separation efficiency.
- Automated mobile phase delivery systems: Automated systems for mobile phase delivery improve reproducibility by eliminating manual handling errors. These systems include precise pumps, degassing units, and mixing chambers that ensure consistent flow rates and composition. Some advanced systems incorporate real-time monitoring of mobile phase parameters and automatic adjustment capabilities to maintain optimal conditions throughout the analysis. Programmable gradient controllers allow for reproducible gradient profiles across multiple runs.
- Solvent degassing techniques for stable baselines: Effective degassing of mobile phases prevents air bubble formation that can disrupt flow and detection, leading to improved baseline stability and reproducibility. Various degassing techniques include vacuum degassing, helium sparging, and ultrasonic treatment. Online degassing systems integrated into HPLC instruments provide continuous removal of dissolved gases during analysis. Proper degassing minimizes detector noise and improves quantitative precision in chromatographic separations.
- Buffer preparation and stability considerations: Proper buffer preparation and maintenance significantly impact HPLC reproducibility. Factors include using analytical grade reagents, precise pH adjustment, appropriate buffer concentration, and regular replacement of buffers to prevent microbial growth or chemical degradation. Buffer selection should consider compatibility with the analytes, column, and detection method. Some systems incorporate buffer monitoring capabilities to alert users when buffer conditions drift outside acceptable parameters, ensuring consistent chromatographic performance.
- Quality control protocols for mobile phase management: Implementing comprehensive quality control protocols for mobile phase management enhances reproducibility across multiple analyses. These protocols include regular system suitability testing, standardized procedures for mobile phase preparation, proper storage conditions, and documentation of all parameters. Some approaches involve the use of internal standards to normalize variations in chromatographic conditions. Advanced quality control systems may incorporate artificial intelligence to predict and prevent potential issues affecting mobile phase reproducibility.
02 Temperature regulation systems
Temperature fluctuations significantly impact HPLC reproducibility by affecting mobile phase viscosity, analyte retention, and column efficiency. Advanced temperature regulation systems maintain consistent conditions throughout the chromatographic process. These systems include column compartment temperature control, pre-column mobile phase heating, and thermal stabilization of instrument components to minimize variations in separation performance.Expand Specific Solutions03 Degassing and gas management techniques
Dissolved gases in mobile phases can cause baseline instability, detector noise, and flow rate inconsistencies. Effective degassing techniques include vacuum degassing, helium sparging, and ultrasonic treatments. Integrated degassing units remove dissolved gases continuously during operation, while specialized flow path designs prevent gas bubble formation. These approaches ensure consistent mobile phase delivery and improve detector response reproducibility.Expand Specific Solutions04 Automated mobile phase preparation systems
Automated systems for mobile phase preparation eliminate variability associated with manual processes. These systems incorporate precise solvent dispensing, mixing, and quality verification steps. Real-time monitoring of critical parameters ensures consistent mobile phase properties across analytical batches. Integration with laboratory information management systems enables traceability and standardization of preparation protocols, enhancing overall method reproducibility.Expand Specific Solutions05 Quality control and validation protocols
Comprehensive quality control protocols for mobile phases include verification of chemical purity, water quality, pH accuracy, and filtration effectiveness. System suitability tests assess chromatographic performance before analytical runs. Regular validation of mobile phase preparation procedures identifies potential sources of variability. Implementation of statistical process control techniques enables continuous monitoring of method reproducibility and early detection of systematic deviations.Expand Specific Solutions
Leading Manufacturers and Research Institutions in HPLC Technology
The HPLC mobile phase optimization market is currently in a growth phase, with increasing demand for reproducible chromatographic methods across pharmaceutical and research sectors. The global market size for analytical chromatography is estimated at $10-12 billion, with HPLC optimization solutions representing a significant segment. Leading pharmaceutical companies like Janssen Pharmaceutica, Bristol Myers Squibb, and Amgen are driving innovation in this field, while specialized equipment manufacturers such as Siemens and analytical service providers like Cellzome offer complementary technologies. Academic institutions including Shanghai Jiao Tong University and Kunming University of Science & Technology contribute to fundamental research. The technology has reached moderate maturity with established protocols, but continuous innovation focuses on improving reproducibility, automation, and integration with advanced analytical systems.
Amgen, Inc.
Technical Solution: Amgen has developed a comprehensive approach to HPLC mobile phase optimization focusing on reproducibility through their Quality by Design (QbD) methodology. Their system employs automated mobile phase preparation with precise pH control (±0.02 units) and buffer concentration management. They utilize a dual-path gradient system with quaternary pumping capabilities that allows for real-time adjustments of mobile phase composition. Amgen's approach incorporates temperature-controlled solvent cabinets (±0.5°C) to minimize thermal fluctuations that can affect retention times. Their method development platform includes Design of Experiments (DoE) software that systematically evaluates critical method parameters including organic modifier percentage, buffer concentration, pH, and column temperature to establish a robust design space. Additionally, they implement inline degassing systems and particle filters to prevent air bubble formation and column contamination, which are common sources of irreproducibility in HPLC analyses.
Strengths: Superior reproducibility with reported RSD values <0.5% for retention times across multiple laboratories and instruments. The automated mobile phase preparation minimizes human error. Weaknesses: The comprehensive system requires significant capital investment and specialized training for analysts. The approach may be overly complex for routine analyses where extreme precision is not required.
Janssen Pharmaceuticals, Inc.
Technical Solution: Janssen Pharmaceuticals has implemented their "Robust Mobile Phase Engineering" platform focused on enhancing HPLC reproducibility through systematic control of mobile phase variables. Their approach begins with water quality standardization using validated purification systems that produce consistent resistivity (>18.2 MΩ·cm) and total organic carbon levels (<5 ppb). They employ a unique buffer preparation strategy using certified reference materials for pH calibration and standardized weighing protocols with precision balances (±0.1 mg). A key innovation is their "solvent quality verification system" that uses UV spectroscopy and refractive index measurements to assess batch-to-batch consistency of organic modifiers before use. Janssen's platform includes specialized mobile phase vessels with minimal headspace design to reduce evaporation and atmospheric gas absorption. They have also developed proprietary algorithms for gradient formation that compensate for system dwell volume variations between instruments, ensuring consistent separation profiles across different HPLC systems.
Strengths: Highly effective at minimizing day-to-day variability with documented improvements in retention time reproducibility (RSD reduced from >1% to <0.3%). Their comprehensive approach addresses multiple sources of variability simultaneously. Weaknesses: Implementation requires significant method transfer validation when adopting existing methods. The system demands rigorous adherence to standardized protocols, potentially increasing analyst workload.
Key Innovations in Buffer Systems and Solvent Mixtures
Liquid feeding device and fluid chromatograph
PatentWO2018198234A1
Innovation
- A liquid feeding device with a pump head connected to a cooling unit that absorbs and cools the heat of the mobile phase to a consistent temperature, using a flat plate-shaped flow path with a high surface-to-volume ratio and strain detection to control the plunger speed, ensuring accurate pressure detection and compensation for thermal expansion.
High performance liquid chromatography method for analysis of MN diagnostic and therapeutic ligand and precursor
PatentInactiveUS20180059072A1
Innovation
- A HPLC method utilizing a high ratio of acetonitrile in the mobile phase to wash out low-polarity impurities and employing gradient elution with a UV detector at 210 nm to improve detection accuracy, ensuring accurate analysis and reducing residual impurities in the column.
Quality Control Standards for Analytical Chromatography
Quality control standards for analytical chromatography represent a critical framework for ensuring the reliability and reproducibility of HPLC analyses across laboratories and industries. These standards encompass comprehensive guidelines that address system suitability, method validation, and ongoing performance verification to maintain analytical integrity.
The pharmaceutical industry has established particularly rigorous standards through regulatory bodies such as the FDA, EMA, and ICH. These organizations mandate specific performance parameters including retention time reproducibility (typically <1% RSD), peak area precision (<2% RSD), and resolution factors (>1.5 between critical pairs) that must be consistently achieved. For mobile phase optimization, these standards require documented evidence of robustness across minor variations in mobile phase composition (±2% organic modifier) and pH (±0.2 units).
USP <621> provides detailed specifications for chromatographic systems, stipulating that mobile phase preparation must follow validated procedures with documented traceability of reagents. This includes verification of solvent quality (HPLC-grade or higher), proper degassing protocols, and filtration requirements (typically 0.45μm or finer). The standard also addresses storage conditions, with prepared mobile phases requiring stability documentation if used beyond 24 hours.
ASTM E1657 outlines specific practices for mobile phase qualification, including procedures for verifying batch-to-batch consistency through retention time mapping of standard compounds. This standard recommends periodic testing of critical separation parameters using certified reference materials to detect subtle changes in mobile phase performance over time.
ISO/IEC 17025 accreditation requirements extend beyond technical specifications to include comprehensive documentation of mobile phase preparation, including personnel training records, equipment calibration, and environmental controls. These standards mandate uncertainty calculations that account for all variables affecting mobile phase performance.
Industry best practices have evolved to include system suitability tests (SSTs) that must be performed before analytical runs, with acceptance criteria specifically designed to verify mobile phase performance. These typically include measurements of theoretical plates (>2000), tailing factors (<2.0), and signal-to-noise ratios (>10:1) that directly reflect mobile phase quality and consistency.
Modern quality control approaches increasingly incorporate statistical process control (SPC) methodologies, with control charts monitoring critical mobile phase parameters over time to detect trends before they impact analytical results. This proactive approach allows laboratories to maintain reproducibility by addressing potential issues before they affect analytical outcomes.
The pharmaceutical industry has established particularly rigorous standards through regulatory bodies such as the FDA, EMA, and ICH. These organizations mandate specific performance parameters including retention time reproducibility (typically <1% RSD), peak area precision (<2% RSD), and resolution factors (>1.5 between critical pairs) that must be consistently achieved. For mobile phase optimization, these standards require documented evidence of robustness across minor variations in mobile phase composition (±2% organic modifier) and pH (±0.2 units).
USP <621> provides detailed specifications for chromatographic systems, stipulating that mobile phase preparation must follow validated procedures with documented traceability of reagents. This includes verification of solvent quality (HPLC-grade or higher), proper degassing protocols, and filtration requirements (typically 0.45μm or finer). The standard also addresses storage conditions, with prepared mobile phases requiring stability documentation if used beyond 24 hours.
ASTM E1657 outlines specific practices for mobile phase qualification, including procedures for verifying batch-to-batch consistency through retention time mapping of standard compounds. This standard recommends periodic testing of critical separation parameters using certified reference materials to detect subtle changes in mobile phase performance over time.
ISO/IEC 17025 accreditation requirements extend beyond technical specifications to include comprehensive documentation of mobile phase preparation, including personnel training records, equipment calibration, and environmental controls. These standards mandate uncertainty calculations that account for all variables affecting mobile phase performance.
Industry best practices have evolved to include system suitability tests (SSTs) that must be performed before analytical runs, with acceptance criteria specifically designed to verify mobile phase performance. These typically include measurements of theoretical plates (>2000), tailing factors (<2.0), and signal-to-noise ratios (>10:1) that directly reflect mobile phase quality and consistency.
Modern quality control approaches increasingly incorporate statistical process control (SPC) methodologies, with control charts monitoring critical mobile phase parameters over time to detect trends before they impact analytical results. This proactive approach allows laboratories to maintain reproducibility by addressing potential issues before they affect analytical outcomes.
Environmental Considerations in Sustainable HPLC Methods
The environmental impact of HPLC methods has become increasingly important as laboratories worldwide adopt more sustainable practices. Traditional HPLC mobile phases often contain harmful organic solvents such as acetonitrile and methanol, which pose significant environmental and health risks when improperly disposed. A sustainable approach to HPLC mobile phase optimization must balance analytical performance with environmental responsibility.
Green chemistry principles are now being integrated into HPLC method development, focusing on reducing solvent consumption and replacing toxic solvents with more environmentally benign alternatives. Water-rich mobile phases, when analytically feasible, significantly reduce the environmental footprint of HPLC analyses. The implementation of recycling systems for mobile phases can decrease solvent waste by up to 90% in certain applications, though careful monitoring is required to maintain reproducibility.
Temperature-responsive polymers represent an innovative approach to sustainable HPLC, allowing for separation using primarily aqueous mobile phases with minimal organic modifiers. These systems utilize temperature changes rather than organic solvents to modify selectivity, dramatically reducing environmental impact while maintaining analytical performance. However, their application remains limited to specific compound classes and requires specialized column technology.
Supercritical fluid chromatography (SFC) using CO2 as the primary mobile phase component offers a greener alternative to traditional HPLC for many applications. CO2 is non-toxic, non-flammable, and can be sourced as a by-product from industrial processes, making it environmentally preferable to conventional organic solvents. SFC methods have demonstrated comparable reproducibility to HPLC for many analytes while significantly reducing hazardous waste generation.
Miniaturization through micro and nano-HPLC systems provides another pathway to environmental sustainability. These systems require substantially lower volumes of mobile phase—often 100-1000 times less than conventional HPLC—while maintaining or even improving analytical performance. The reduced solvent consumption directly translates to decreased waste generation and lower disposal costs, though initial investment in specialized equipment is required.
Laboratory waste management practices must evolve alongside analytical methods. Proper collection, treatment, and disposal of HPLC mobile phase waste are essential components of sustainable laboratory operations. Advanced treatment technologies such as photocatalytic degradation and advanced oxidation processes can render HPLC waste less environmentally harmful before discharge, though these add operational complexity and cost.
Regulatory frameworks increasingly encourage or mandate sustainable practices in analytical laboratories. The EU's REACH regulations and similar initiatives worldwide are driving the adoption of greener HPLC methods. Laboratories that proactively implement sustainable mobile phase strategies not only reduce their environmental impact but also position themselves advantageously for future regulatory compliance.
Green chemistry principles are now being integrated into HPLC method development, focusing on reducing solvent consumption and replacing toxic solvents with more environmentally benign alternatives. Water-rich mobile phases, when analytically feasible, significantly reduce the environmental footprint of HPLC analyses. The implementation of recycling systems for mobile phases can decrease solvent waste by up to 90% in certain applications, though careful monitoring is required to maintain reproducibility.
Temperature-responsive polymers represent an innovative approach to sustainable HPLC, allowing for separation using primarily aqueous mobile phases with minimal organic modifiers. These systems utilize temperature changes rather than organic solvents to modify selectivity, dramatically reducing environmental impact while maintaining analytical performance. However, their application remains limited to specific compound classes and requires specialized column technology.
Supercritical fluid chromatography (SFC) using CO2 as the primary mobile phase component offers a greener alternative to traditional HPLC for many applications. CO2 is non-toxic, non-flammable, and can be sourced as a by-product from industrial processes, making it environmentally preferable to conventional organic solvents. SFC methods have demonstrated comparable reproducibility to HPLC for many analytes while significantly reducing hazardous waste generation.
Miniaturization through micro and nano-HPLC systems provides another pathway to environmental sustainability. These systems require substantially lower volumes of mobile phase—often 100-1000 times less than conventional HPLC—while maintaining or even improving analytical performance. The reduced solvent consumption directly translates to decreased waste generation and lower disposal costs, though initial investment in specialized equipment is required.
Laboratory waste management practices must evolve alongside analytical methods. Proper collection, treatment, and disposal of HPLC mobile phase waste are essential components of sustainable laboratory operations. Advanced treatment technologies such as photocatalytic degradation and advanced oxidation processes can render HPLC waste less environmentally harmful before discharge, though these add operational complexity and cost.
Regulatory frameworks increasingly encourage or mandate sustainable practices in analytical laboratories. The EU's REACH regulations and similar initiatives worldwide are driving the adoption of greener HPLC methods. Laboratories that proactively implement sustainable mobile phase strategies not only reduce their environmental impact but also position themselves advantageously for future regulatory compliance.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!