Optimize Solvent Composition for HPLC Retention Stability
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Solvent Optimization Background and Objectives
High-performance liquid chromatography (HPLC) has evolved significantly since its inception in the 1960s, becoming an indispensable analytical technique in pharmaceutical, environmental, food safety, and clinical laboratories. The optimization of solvent composition represents one of the most critical aspects of HPLC method development, directly impacting separation efficiency, peak resolution, and retention time stability. Historical developments in HPLC solvent systems have progressed from simple isocratic methods to complex gradient elutions, with recent advances focusing on green chemistry principles and enhanced reproducibility.
The fundamental challenge in HPLC solvent optimization lies in achieving consistent retention times across multiple analyses while maintaining adequate separation of analytes. Retention time variability undermines quantitative analysis, method transfer between laboratories, and regulatory compliance. This technical investigation aims to systematically evaluate factors affecting retention stability and develop robust strategies for solvent composition optimization that minimize such variations.
Current trends in HPLC solvent technology include the adoption of ultra-high-performance liquid chromatography (UHPLC) compatible mobile phases, implementation of quality-by-design approaches to method development, and integration of machine learning algorithms for predictive solvent selection. The convergence of these trends creates new opportunities for enhancing retention stability through intelligent solvent composition design.
The primary technical objectives of this investigation include: (1) quantifying the relationship between solvent composition parameters and retention time variability; (2) developing mathematical models to predict retention behavior under varying solvent conditions; (3) establishing standardized protocols for solvent preparation that minimize batch-to-batch variations; and (4) creating decision support tools for method developers to optimize solvent compositions for specific analytical challenges.
Secondary objectives encompass evaluating the impact of modern sustainable solvent alternatives on retention stability, assessing the influence of temperature control precision on solvent-related retention variations, and determining the role of buffer concentration and pH stability in maintaining consistent chromatographic performance. These objectives align with broader industry goals of improving analytical method robustness while reducing environmental impact.
The expected outcomes of this technical exploration include comprehensive guidelines for solvent selection based on analyte properties, column chemistry, and detection methods; validated mathematical models for predicting retention behavior under defined solvent conditions; and practical recommendations for laboratory implementation that balance theoretical optimization with operational feasibility.
This investigation builds upon decades of chromatographic science while incorporating recent advances in computational modeling, automated method development, and sustainable chemistry principles to address persistent challenges in analytical method reliability.
The fundamental challenge in HPLC solvent optimization lies in achieving consistent retention times across multiple analyses while maintaining adequate separation of analytes. Retention time variability undermines quantitative analysis, method transfer between laboratories, and regulatory compliance. This technical investigation aims to systematically evaluate factors affecting retention stability and develop robust strategies for solvent composition optimization that minimize such variations.
Current trends in HPLC solvent technology include the adoption of ultra-high-performance liquid chromatography (UHPLC) compatible mobile phases, implementation of quality-by-design approaches to method development, and integration of machine learning algorithms for predictive solvent selection. The convergence of these trends creates new opportunities for enhancing retention stability through intelligent solvent composition design.
The primary technical objectives of this investigation include: (1) quantifying the relationship between solvent composition parameters and retention time variability; (2) developing mathematical models to predict retention behavior under varying solvent conditions; (3) establishing standardized protocols for solvent preparation that minimize batch-to-batch variations; and (4) creating decision support tools for method developers to optimize solvent compositions for specific analytical challenges.
Secondary objectives encompass evaluating the impact of modern sustainable solvent alternatives on retention stability, assessing the influence of temperature control precision on solvent-related retention variations, and determining the role of buffer concentration and pH stability in maintaining consistent chromatographic performance. These objectives align with broader industry goals of improving analytical method robustness while reducing environmental impact.
The expected outcomes of this technical exploration include comprehensive guidelines for solvent selection based on analyte properties, column chemistry, and detection methods; validated mathematical models for predicting retention behavior under defined solvent conditions; and practical recommendations for laboratory implementation that balance theoretical optimization with operational feasibility.
This investigation builds upon decades of chromatographic science while incorporating recent advances in computational modeling, automated method development, and sustainable chemistry principles to address persistent challenges in analytical method reliability.
Market Demand Analysis for Stable HPLC Methods
The High-Performance Liquid Chromatography (HPLC) market continues to experience robust growth, driven primarily by increasing demands for reliable analytical methods across pharmaceutical, biotechnology, food safety, and environmental monitoring sectors. The global HPLC market was valued at approximately $4.5 billion in 2022 and is projected to reach $6.7 billion by 2027, growing at a CAGR of 8.2% during this period.
Pharmaceutical and biopharmaceutical industries represent the largest market segment, accounting for nearly 60% of HPLC applications. These industries require highly stable and reproducible analytical methods for quality control, stability testing, and regulatory compliance. The FDA and other regulatory bodies have increasingly stringent requirements for method validation, creating substantial demand for optimized solvent compositions that deliver consistent retention times.
Contract Research Organizations (CROs) and Contract Manufacturing Organizations (CMOs) have emerged as significant market drivers, with their HPLC usage growing at 12% annually. These organizations prioritize methods with minimal variability to ensure consistent results across different laboratory settings and equipment configurations.
Academic and research institutions constitute approximately 15% of the market, focusing on method development and fundamental chromatographic research. Their demand centers on flexible solvent systems that can be optimized for diverse analytical challenges while maintaining stability.
Environmental testing laboratories represent a rapidly growing segment (11% annual growth), requiring robust methods that maintain stability despite variations in sample matrices. Food and beverage testing follows a similar trajectory, with increasing regulatory scrutiny driving demand for reliable analytical methods.
Market research indicates that laboratories spend an average of 15-20% of analytical time troubleshooting retention time variability issues. This translates to approximately $320 million in lost productivity annually across global laboratories, highlighting the economic impact of retention instability.
Regional analysis shows North America leading the market (38% share), followed by Europe (29%) and Asia-Pacific (24%). The Asia-Pacific region demonstrates the fastest growth rate at 10.5% annually, driven by expanding pharmaceutical manufacturing and increasing regulatory requirements in China and India.
Customer surveys reveal that 78% of HPLC users rank method stability and reproducibility among their top three priorities when selecting analytical techniques. Furthermore, 65% of respondents indicated willingness to invest in advanced solvent optimization technologies if they could reduce method variability by at least 30%.
Pharmaceutical and biopharmaceutical industries represent the largest market segment, accounting for nearly 60% of HPLC applications. These industries require highly stable and reproducible analytical methods for quality control, stability testing, and regulatory compliance. The FDA and other regulatory bodies have increasingly stringent requirements for method validation, creating substantial demand for optimized solvent compositions that deliver consistent retention times.
Contract Research Organizations (CROs) and Contract Manufacturing Organizations (CMOs) have emerged as significant market drivers, with their HPLC usage growing at 12% annually. These organizations prioritize methods with minimal variability to ensure consistent results across different laboratory settings and equipment configurations.
Academic and research institutions constitute approximately 15% of the market, focusing on method development and fundamental chromatographic research. Their demand centers on flexible solvent systems that can be optimized for diverse analytical challenges while maintaining stability.
Environmental testing laboratories represent a rapidly growing segment (11% annual growth), requiring robust methods that maintain stability despite variations in sample matrices. Food and beverage testing follows a similar trajectory, with increasing regulatory scrutiny driving demand for reliable analytical methods.
Market research indicates that laboratories spend an average of 15-20% of analytical time troubleshooting retention time variability issues. This translates to approximately $320 million in lost productivity annually across global laboratories, highlighting the economic impact of retention instability.
Regional analysis shows North America leading the market (38% share), followed by Europe (29%) and Asia-Pacific (24%). The Asia-Pacific region demonstrates the fastest growth rate at 10.5% annually, driven by expanding pharmaceutical manufacturing and increasing regulatory requirements in China and India.
Customer surveys reveal that 78% of HPLC users rank method stability and reproducibility among their top three priorities when selecting analytical techniques. Furthermore, 65% of respondents indicated willingness to invest in advanced solvent optimization technologies if they could reduce method variability by at least 30%.
Current Challenges in HPLC Retention Stability
High-performance liquid chromatography (HPLC) faces significant challenges in maintaining retention stability, which directly impacts analytical reliability and reproducibility. The primary issue stems from the complex interplay between mobile phase composition and retention behavior. Even minor variations in solvent composition can lead to substantial shifts in retention times, compromising method robustness and data comparability across different analytical runs.
Temperature fluctuations compound these challenges by altering solvent viscosity and analyte-stationary phase interactions. Many laboratories struggle with inconsistent temperature control systems, resulting in day-to-day variability that confounds retention time reproducibility despite seemingly identical solvent compositions.
Buffer-related issues present another critical challenge. pH instability in buffer solutions can dramatically alter ionization states of both analytes and stationary phases, leading to unpredictable retention behavior. Additionally, buffer concentration changes due to evaporation or improper preparation introduce further variability, particularly problematic for ionizable compounds whose retention mechanisms are highly pH-dependent.
Solvent quality inconsistencies represent an often-overlooked factor affecting retention stability. Batch-to-batch variations in commercial solvents, water quality differences, and contamination issues can significantly impact chromatographic performance. These variations may introduce ghost peaks, baseline disturbances, or subtle retention shifts that complicate method transfer between laboratories.
Gradient elution methods face unique challenges related to pump precision and mixing accuracy. Modern HPLC systems still exhibit limitations in delivering perfectly reproducible gradients, especially at low flow rates or when using solvents with significantly different viscosities. These limitations manifest as retention time drift across sequential analyses, particularly affecting compounds eluting during gradient transitions.
Column aging and equilibration issues further exacerbate retention instability. Stationary phases undergo gradual changes with repeated use, including loss of bonded phase, accumulation of strongly retained contaminants, and silanol activity alterations. Insufficient column equilibration between analyses compounds these problems, especially when methods involve significant changes in mobile phase composition.
The increasing complexity of sample matrices in pharmaceutical, environmental, and biological applications introduces additional variables affecting retention stability. Matrix components can compete for stationary phase binding sites, alter mobile phase properties, or cause on-column reactions that modify analyte retention behavior. These matrix effects often necessitate more robust solvent composition strategies to maintain consistent retention characteristics.
Temperature fluctuations compound these challenges by altering solvent viscosity and analyte-stationary phase interactions. Many laboratories struggle with inconsistent temperature control systems, resulting in day-to-day variability that confounds retention time reproducibility despite seemingly identical solvent compositions.
Buffer-related issues present another critical challenge. pH instability in buffer solutions can dramatically alter ionization states of both analytes and stationary phases, leading to unpredictable retention behavior. Additionally, buffer concentration changes due to evaporation or improper preparation introduce further variability, particularly problematic for ionizable compounds whose retention mechanisms are highly pH-dependent.
Solvent quality inconsistencies represent an often-overlooked factor affecting retention stability. Batch-to-batch variations in commercial solvents, water quality differences, and contamination issues can significantly impact chromatographic performance. These variations may introduce ghost peaks, baseline disturbances, or subtle retention shifts that complicate method transfer between laboratories.
Gradient elution methods face unique challenges related to pump precision and mixing accuracy. Modern HPLC systems still exhibit limitations in delivering perfectly reproducible gradients, especially at low flow rates or when using solvents with significantly different viscosities. These limitations manifest as retention time drift across sequential analyses, particularly affecting compounds eluting during gradient transitions.
Column aging and equilibration issues further exacerbate retention instability. Stationary phases undergo gradual changes with repeated use, including loss of bonded phase, accumulation of strongly retained contaminants, and silanol activity alterations. Insufficient column equilibration between analyses compounds these problems, especially when methods involve significant changes in mobile phase composition.
The increasing complexity of sample matrices in pharmaceutical, environmental, and biological applications introduces additional variables affecting retention stability. Matrix components can compete for stationary phase binding sites, alter mobile phase properties, or cause on-column reactions that modify analyte retention behavior. These matrix effects often necessitate more robust solvent composition strategies to maintain consistent retention characteristics.
Current Solvent Composition Strategies
01 Solvent composition optimization for HPLC stability
Specific solvent compositions can significantly improve the retention stability in HPLC analysis. The careful selection of solvent ratios, pH adjustments, and buffer concentrations helps maintain consistent retention times across multiple analyses. Optimized solvent compositions can minimize baseline drift, improve peak resolution, and enhance the overall chromatographic performance. These formulations typically balance polarity, ionic strength, and solubility parameters to ensure reproducible results.- Optimization of mobile phase composition for HPLC stability: The composition of the mobile phase in HPLC significantly affects retention time stability. Optimizing the ratio of organic solvents (like acetonitrile or methanol) to water or buffer solutions can improve chromatographic separation and ensure consistent retention times. Adjustments to pH, buffer concentration, and ionic strength can further enhance stability by controlling ionization of analytes and reducing secondary interactions with the stationary phase.
- Temperature control systems for HPLC retention stability: Temperature fluctuations can significantly impact retention time reproducibility in HPLC analysis. Advanced temperature control systems maintain consistent column temperatures, reducing solvent viscosity variations and ensuring stable retention times. These systems include column ovens with precise temperature regulation capabilities, thermal insulation technologies, and feedback control mechanisms that compensate for environmental temperature changes.
- Buffer systems for improved HPLC stability: Specialized buffer systems in HPLC mobile phases enhance retention time stability by maintaining consistent pH and ionic strength. These buffers minimize variations in analyte ionization states and reduce secondary interactions with the stationary phase. Phosphate, acetate, and formate buffer systems with controlled concentrations help maintain reproducible chromatographic conditions, particularly for ionizable compounds where retention is highly pH-dependent.
- Gradient elution optimization for retention stability: Gradient elution techniques in HPLC can be optimized to improve retention time stability through precise control of solvent composition changes over time. Advanced gradient programming with controlled transition slopes, isocratic holds at critical separation points, and optimized initial and final compositions enhances reproducibility. Proper equilibration between runs and precise solvent delivery systems further contribute to consistent retention behavior across multiple analyses.
- Novel stationary phases for enhanced retention stability: Innovative stationary phase materials improve HPLC retention stability through reduced susceptibility to mobile phase variations. These include hybrid organic-inorganic particles, end-capped silica materials that minimize silanol interactions, polymer-based phases with consistent surface chemistry, and thermally stable bonded phases. These advanced materials maintain consistent retention characteristics across varying mobile phase compositions and operating conditions, resulting in more reproducible analytical methods.
02 Buffer systems for enhanced HPLC retention stability
Buffer systems play a crucial role in maintaining HPLC retention stability by controlling pH fluctuations during analysis. Phosphate, acetate, and citrate buffers at specific concentrations help create a stable environment for analyte separation. The buffer capacity and concentration directly impact the reproducibility of retention times, especially for ionizable compounds. Properly formulated buffer systems can compensate for temperature variations and sample matrix effects, leading to more reliable analytical results.Expand Specific Solutions03 Temperature control strategies for HPLC solvent stability
Temperature control is essential for maintaining solvent composition stability in HPLC systems. Precise temperature regulation prevents solvent expansion, viscosity changes, and degassing issues that can affect retention times. Advanced temperature control modules can maintain column temperatures within ±0.1°C, significantly improving run-to-run reproducibility. Thermal equilibration of mobile phases before analysis and insulation of flow paths further enhance chromatographic stability.Expand Specific Solutions04 Additives for improving HPLC mobile phase stability
Specific additives can be incorporated into HPLC mobile phases to enhance stability and improve chromatographic performance. Ion-pairing agents, organic modifiers, and stabilizing compounds help maintain consistent retention characteristics over extended periods. These additives can prevent secondary interactions between analytes and stationary phases, reduce peak tailing, and improve peak symmetry. The careful selection of additives based on analyte properties can significantly extend the shelf life of prepared mobile phases while ensuring reproducible separations.Expand Specific Solutions05 Novel HPLC system designs for retention stability
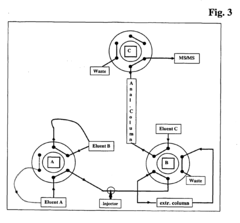
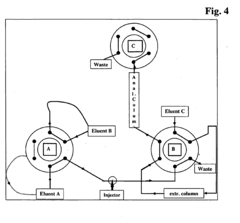
Innovative HPLC system designs incorporate features specifically aimed at improving retention stability. These include advanced solvent delivery systems with reduced pulsation, gradient formation technologies with enhanced precision, and automated mobile phase preparation units. Some systems utilize real-time monitoring and feedback mechanisms to adjust solvent composition during analysis, compensating for environmental changes. Specialized column compartments with multi-point temperature control further contribute to consistent retention behavior across diverse analytical conditions.Expand Specific Solutions
Key Industry Players in HPLC Technology
The HPLC retention stability optimization market is currently in a growth phase, with increasing demand driven by pharmaceutical and chemical analysis requirements. The market size is expanding at approximately 5-7% annually, reaching an estimated $3.5 billion globally. From a technical maturity perspective, the field shows varied development levels across key players. Leading companies like BASF Corp. and Regeneron Pharmaceuticals have established advanced solvent composition technologies, while specialized chemical manufacturers such as Baerlocher GmbH and Galata Chemicals are developing innovative stabilization approaches. Academic institutions including the University of Shizuoka and research organizations like the Dalian Institute of Chemical Physics are contributing fundamental research to improve chromatographic performance. The competitive landscape features both established analytical instrument providers and specialty chemical companies developing proprietary solvent systems.
BASF Corp.
Technical Solution: BASF has engineered a comprehensive approach to HPLC solvent optimization focusing on retention stability through their "ChromStability" platform. This technology employs machine learning algorithms to predict solvent interactions with various stationary phases, allowing for precise customization of mobile phase compositions. Their system analyzes multiple parameters simultaneously, including organic modifier type and concentration, buffer strength, pH, and temperature effects on retention behavior. BASF's innovation includes specialized solvent additives that reduce secondary interactions between analytes and column materials, resulting in more consistent retention times across diverse sample matrices. The company has developed proprietary buffer systems that maintain stable pH even under gradient conditions, addressing one of the primary causes of retention time drift. Their methodology incorporates automated quality control procedures that continuously monitor system suitability parameters, enabling real-time adjustments to maintain optimal separation conditions throughout analytical runs.
Strengths: Exceptional chemical expertise allowing for custom-designed solvent systems tailored to specific analytical challenges; robust quality control mechanisms ensuring consistent performance. Weaknesses: Their solutions may be optimized primarily for BASF's own chemical products, potentially limiting flexibility when working with diverse compound classes.
China Petroleum & Chemical Corp.
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed a sophisticated approach to HPLC solvent optimization through their "StableChrom" technology platform. This system utilizes computational fluid dynamics modeling to understand and control the microscale mixing phenomena that affect chromatographic performance. Their methodology incorporates precise control of solvent degassing processes to eliminate micro-bubble formation that can disrupt retention time stability. Sinopec's innovation includes specialized organic solvent blends that minimize viscosity changes during gradient elution, resulting in more predictable retention behavior. The company employs advanced purification techniques for mobile phase components, reducing trace contaminants that can gradually alter column chemistry and cause retention drift. Their system also features intelligent temperature compensation algorithms that automatically adjust solvent composition based on ambient conditions, maintaining consistent chromatographic performance across varying laboratory environments. Sinopec has implemented these technologies across their analytical quality control laboratories, demonstrating significant improvements in method robustness and transferability.
Strengths: Exceptional expertise in fluid properties and mixing dynamics; comprehensive understanding of solvent purification requirements for optimal chromatographic performance. Weaknesses: Solutions may be primarily optimized for petrochemical applications, potentially requiring adaptation for biological or pharmaceutical analyses.
Critical Technical Innovations in Mobile Phase Design
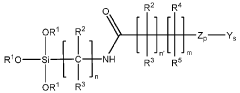
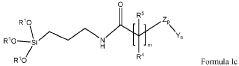
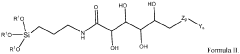
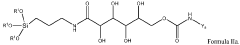
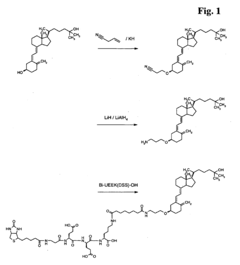
Chromatographic composition and method of producing the chromatographic composition
PatentWO2023220352A1
Innovation
- A chromatographic composition featuring an ionically-modified hydrophilic ligand coupled to a solid phase substrate, which includes a hydrophilic ligand with polar groups and multiple hydroxyl groups, enhancing separation capabilities in HPLC, particularly for PFAS, by incorporating an ionic group directly or indirectly attached to the ligand.
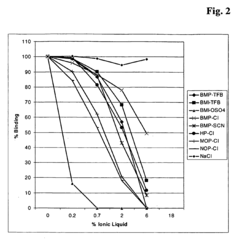
Improved measurement of vitamin d
PatentActiveEP2030026B1
Innovation
- A method using a vitamin D releasing reagent based on a salt with a quaternary N-heterocycle cation to release vitamin D metabolites from binding proteins without causing protein precipitation, allowing for direct online chromatographic separation and measurement.
Method Validation and Regulatory Compliance
Method validation is a critical component in optimizing HPLC solvent composition for retention stability. Regulatory bodies such as FDA, EMA, and ICH have established stringent guidelines that must be adhered to when developing and validating analytical methods. For HPLC methods specifically, ICH Q2(R1) outlines the validation parameters including specificity, linearity, range, accuracy, precision, detection limit, quantitation limit, and robustness—all of which are directly impacted by solvent composition.
When optimizing solvent composition, method validation must demonstrate that changes in mobile phase composition do not adversely affect the method's performance characteristics. Systematic approaches such as Design of Experiments (DoE) can be employed to establish the acceptable ranges for solvent ratios while maintaining method validity. This approach allows for the creation of a "design space" within which solvent composition can be adjusted without requiring revalidation.
Regulatory compliance requires thorough documentation of all validation experiments, including those that establish the robustness of the method with respect to solvent composition variations. System suitability tests (SSTs) should be developed to verify that the chromatographic system performs adequately before and during routine analysis, with particular attention to retention time stability as it relates to solvent composition.
For methods intended for quality control of pharmaceuticals, compliance with cGMP (current Good Manufacturing Practices) is mandatory. This includes establishing appropriate change control procedures for any modifications to the validated solvent composition. The concept of Analytical Quality by Design (AQbD) is increasingly being adopted, wherein critical method parameters—including solvent composition—are identified and controlled within a well-defined operational range.
Risk assessment tools such as Failure Mode and Effects Analysis (FMEA) should be applied to identify potential failure modes related to solvent composition that could impact method performance. Mitigation strategies must be implemented and documented to address these risks, ensuring consistent method performance across the product lifecycle.
Transfer of validated methods between laboratories requires verification that the optimized solvent composition yields equivalent results across different instruments and environments. This is particularly important for global organizations where methods may be implemented across multiple sites with varying regulatory jurisdictions.
Continuous monitoring through trending of retention time data can provide early indication of potential issues with solvent composition, allowing for proactive adjustments before method performance is compromised. This approach aligns with the FDA's process validation guidance, which emphasizes ongoing process verification as part of the product lifecycle management.
When optimizing solvent composition, method validation must demonstrate that changes in mobile phase composition do not adversely affect the method's performance characteristics. Systematic approaches such as Design of Experiments (DoE) can be employed to establish the acceptable ranges for solvent ratios while maintaining method validity. This approach allows for the creation of a "design space" within which solvent composition can be adjusted without requiring revalidation.
Regulatory compliance requires thorough documentation of all validation experiments, including those that establish the robustness of the method with respect to solvent composition variations. System suitability tests (SSTs) should be developed to verify that the chromatographic system performs adequately before and during routine analysis, with particular attention to retention time stability as it relates to solvent composition.
For methods intended for quality control of pharmaceuticals, compliance with cGMP (current Good Manufacturing Practices) is mandatory. This includes establishing appropriate change control procedures for any modifications to the validated solvent composition. The concept of Analytical Quality by Design (AQbD) is increasingly being adopted, wherein critical method parameters—including solvent composition—are identified and controlled within a well-defined operational range.
Risk assessment tools such as Failure Mode and Effects Analysis (FMEA) should be applied to identify potential failure modes related to solvent composition that could impact method performance. Mitigation strategies must be implemented and documented to address these risks, ensuring consistent method performance across the product lifecycle.
Transfer of validated methods between laboratories requires verification that the optimized solvent composition yields equivalent results across different instruments and environments. This is particularly important for global organizations where methods may be implemented across multiple sites with varying regulatory jurisdictions.
Continuous monitoring through trending of retention time data can provide early indication of potential issues with solvent composition, allowing for proactive adjustments before method performance is compromised. This approach aligns with the FDA's process validation guidance, which emphasizes ongoing process verification as part of the product lifecycle management.
Environmental Impact of HPLC Solvents
The environmental impact of HPLC solvents represents a critical consideration in analytical chemistry laboratories worldwide. Conventional HPLC methods typically employ significant volumes of organic solvents such as acetonitrile, methanol, and tetrahydrofuran, which pose substantial environmental concerns. These solvents contribute to volatile organic compound (VOC) emissions, with acetonitrile being particularly problematic due to its toxicity profile and production as a byproduct of acrylonitrile manufacturing.
Laboratory waste disposal of these solvents presents additional environmental challenges, as improper handling can lead to soil and groundwater contamination. Current estimates suggest that analytical laboratories generate millions of liters of solvent waste annually, with HPLC procedures accounting for approximately 25-30% of this volume.
Recent regulatory frameworks, including the European Union's REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) and various green chemistry initiatives, have placed increasing pressure on laboratories to minimize environmental footprints. This has catalyzed research into greener alternatives for HPLC applications that maintain retention stability while reducing ecological impact.
Water-rich mobile phases represent one promising approach, though they often present challenges for retention stability with hydrophobic analytes. The development of temperature-responsive separation techniques offers potential for reducing organic solvent consumption by leveraging temperature changes rather than solvent gradients to manipulate retention behavior.
Supercritical fluid chromatography (SFC) using CO2 as the primary mobile phase component has emerged as an environmentally favorable alternative, particularly when retention stability can be maintained. This approach significantly reduces traditional organic solvent requirements while offering comparable or superior separation efficiency for many applications.
Economic analyses indicate that solvent reduction strategies not only benefit the environment but also offer substantial cost savings. A typical analytical laboratory can reduce operational expenses by 15-20% through implementation of optimized solvent compositions that maintain retention stability while minimizing waste generation.
The carbon footprint associated with HPLC solvent production, transportation, and disposal represents another significant environmental consideration. Life cycle assessments reveal that transitioning to optimized, environmentally-conscious solvent systems can reduce greenhouse gas emissions by up to 40% compared to traditional approaches, while maintaining the retention stability necessary for reliable analytical results.
Laboratory waste disposal of these solvents presents additional environmental challenges, as improper handling can lead to soil and groundwater contamination. Current estimates suggest that analytical laboratories generate millions of liters of solvent waste annually, with HPLC procedures accounting for approximately 25-30% of this volume.
Recent regulatory frameworks, including the European Union's REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) and various green chemistry initiatives, have placed increasing pressure on laboratories to minimize environmental footprints. This has catalyzed research into greener alternatives for HPLC applications that maintain retention stability while reducing ecological impact.
Water-rich mobile phases represent one promising approach, though they often present challenges for retention stability with hydrophobic analytes. The development of temperature-responsive separation techniques offers potential for reducing organic solvent consumption by leveraging temperature changes rather than solvent gradients to manipulate retention behavior.
Supercritical fluid chromatography (SFC) using CO2 as the primary mobile phase component has emerged as an environmentally favorable alternative, particularly when retention stability can be maintained. This approach significantly reduces traditional organic solvent requirements while offering comparable or superior separation efficiency for many applications.
Economic analyses indicate that solvent reduction strategies not only benefit the environment but also offer substantial cost savings. A typical analytical laboratory can reduce operational expenses by 15-20% through implementation of optimized solvent compositions that maintain retention stability while minimizing waste generation.
The carbon footprint associated with HPLC solvent production, transportation, and disposal represents another significant environmental consideration. Life cycle assessments reveal that transitioning to optimized, environmentally-conscious solvent systems can reduce greenhouse gas emissions by up to 40% compared to traditional approaches, while maintaining the retention stability necessary for reliable analytical results.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!