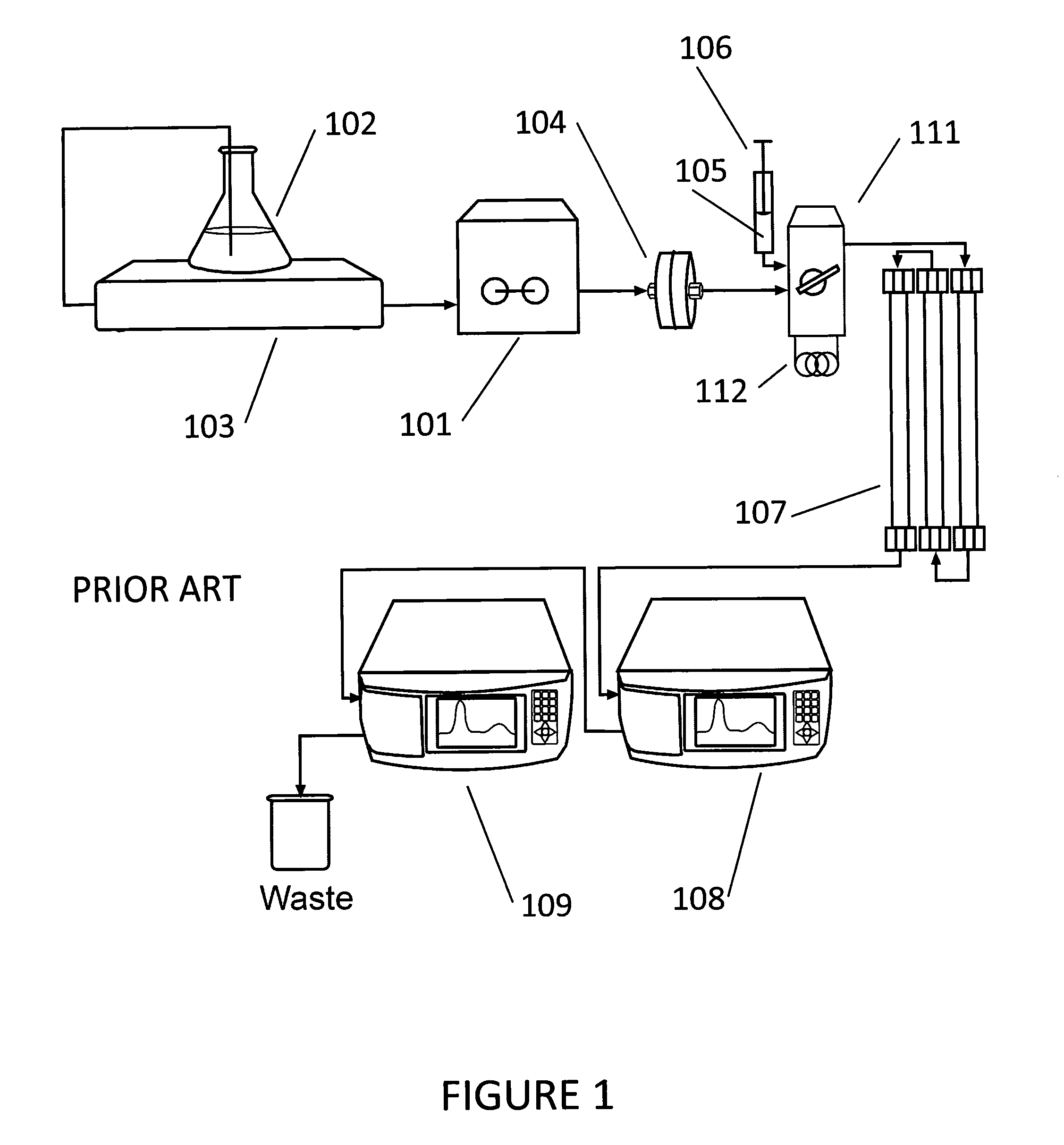
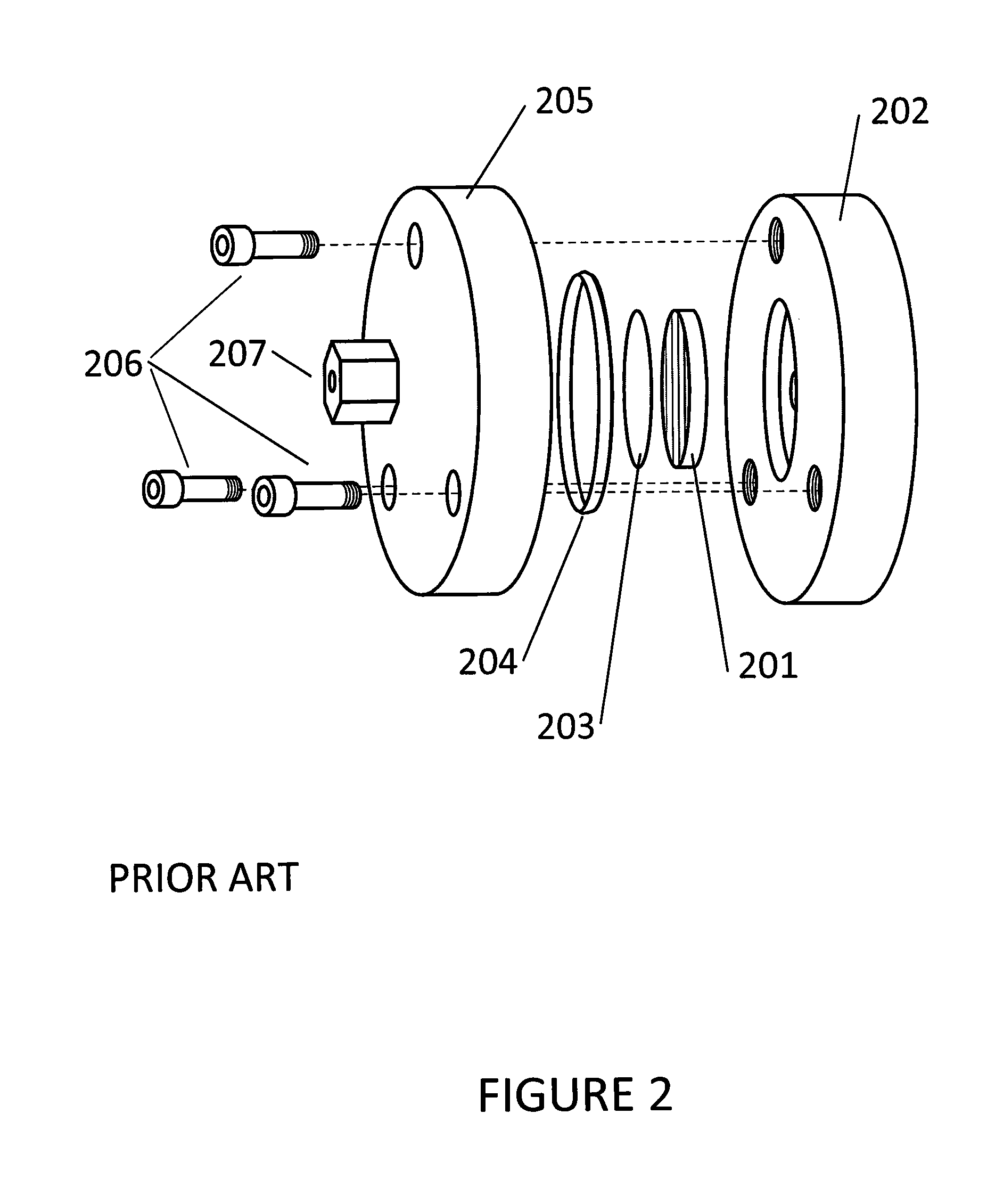
Quantify Precision in HPLC—Repeatability Testing
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Precision Quantification Background and Objectives
High-performance liquid chromatography (HPLC) has evolved significantly since its inception in the 1960s, becoming an indispensable analytical technique in pharmaceutical, environmental, food safety, and clinical laboratories. The quantification of precision in HPLC systems represents a critical aspect of analytical method validation and quality control processes across these industries. Historically, the focus on precision has intensified with increasingly stringent regulatory requirements and the growing need for reliable analytical data.
The evolution of HPLC precision quantification has paralleled advancements in instrumentation, column technology, and data processing capabilities. Early HPLC systems exhibited considerable variability, with precision often compromised by mechanical limitations and manual operations. The introduction of automated sample injectors in the 1980s marked a significant improvement, followed by computer-controlled systems that enhanced reproducibility through standardized protocols and reduced human intervention.
Current technological trends in HPLC precision assessment include the implementation of Quality by Design (QbD) principles, real-time monitoring systems, and advanced statistical tools for comprehensive uncertainty analysis. The integration of machine learning algorithms for pattern recognition and predictive maintenance represents the cutting edge of precision management in modern HPLC applications.
The primary objective of repeatability testing in HPLC is to establish the degree of agreement between successive measurements obtained under identical operating conditions. This encompasses instrument precision, method precision, and system suitability testing—all critical components for ensuring reliable analytical results. Specifically, repeatability testing aims to quantify the inherent variability of the chromatographic system under controlled conditions, providing a baseline for method validation and ongoing quality assurance.
Secondary objectives include identifying sources of variability within the analytical process, establishing acceptance criteria for routine analysis, and developing strategies to minimize systematic and random errors. The ultimate goal is to ensure that analytical methods deliver consistent, accurate results regardless of when or by whom they are performed, thereby supporting critical decision-making processes in research, development, and quality control environments.
The technical objectives extend to establishing standardized protocols for precision assessment that align with international guidelines such as ICH, USP, and FDA requirements, while accommodating the specific needs of different application domains. This includes developing robust statistical frameworks for evaluating short-term and long-term precision, as well as methodologies for distinguishing between instrument-related and method-related variability factors.
The evolution of HPLC precision quantification has paralleled advancements in instrumentation, column technology, and data processing capabilities. Early HPLC systems exhibited considerable variability, with precision often compromised by mechanical limitations and manual operations. The introduction of automated sample injectors in the 1980s marked a significant improvement, followed by computer-controlled systems that enhanced reproducibility through standardized protocols and reduced human intervention.
Current technological trends in HPLC precision assessment include the implementation of Quality by Design (QbD) principles, real-time monitoring systems, and advanced statistical tools for comprehensive uncertainty analysis. The integration of machine learning algorithms for pattern recognition and predictive maintenance represents the cutting edge of precision management in modern HPLC applications.
The primary objective of repeatability testing in HPLC is to establish the degree of agreement between successive measurements obtained under identical operating conditions. This encompasses instrument precision, method precision, and system suitability testing—all critical components for ensuring reliable analytical results. Specifically, repeatability testing aims to quantify the inherent variability of the chromatographic system under controlled conditions, providing a baseline for method validation and ongoing quality assurance.
Secondary objectives include identifying sources of variability within the analytical process, establishing acceptance criteria for routine analysis, and developing strategies to minimize systematic and random errors. The ultimate goal is to ensure that analytical methods deliver consistent, accurate results regardless of when or by whom they are performed, thereby supporting critical decision-making processes in research, development, and quality control environments.
The technical objectives extend to establishing standardized protocols for precision assessment that align with international guidelines such as ICH, USP, and FDA requirements, while accommodating the specific needs of different application domains. This includes developing robust statistical frameworks for evaluating short-term and long-term precision, as well as methodologies for distinguishing between instrument-related and method-related variability factors.
Market Demand Analysis for HPLC Precision Testing
The global HPLC (High-Performance Liquid Chromatography) precision testing market has been experiencing robust growth, driven by increasing demands for analytical accuracy across pharmaceutical, biotechnology, food safety, and environmental monitoring sectors. Current market valuation stands at approximately 5.8 billion USD, with projections indicating a compound annual growth rate of 6.7% through 2028.
Pharmaceutical and biopharmaceutical industries represent the largest market segment, accounting for nearly 65% of the total demand for HPLC precision testing solutions. This dominance stems from stringent regulatory requirements imposed by authorities such as FDA, EMA, and NMPA, which mandate thorough validation of analytical methods including repeatability testing as a critical component of quality control processes.
Contract research organizations (CROs) and contract manufacturing organizations (CMOs) have emerged as significant growth drivers, as outsourcing of analytical testing continues to rise. These organizations require highly precise and reliable HPLC systems to maintain competitive advantage and meet client expectations, creating substantial demand for advanced repeatability testing solutions.
Regional analysis reveals North America currently leads the market with approximately 38% share, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region demonstrates the fastest growth trajectory, particularly in China, India, and South Korea, where expanding pharmaceutical manufacturing capabilities and increasing regulatory alignment with international standards are accelerating adoption of precision HPLC technologies.
The COVID-19 pandemic has further amplified market demand, as pharmaceutical companies and research institutions intensified their analytical testing activities for vaccine development and therapeutic research. This trend has highlighted the critical importance of repeatability in HPLC testing, creating new market opportunities for precision-focused solutions.
End-user feedback indicates growing preference for automated systems that minimize human intervention in repeatability testing, thereby reducing operator-dependent variables. Additionally, there is increasing demand for integrated software solutions that can provide comprehensive statistical analysis of repeatability data, enabling more efficient compliance with regulatory requirements.
Market research reveals that customers are willing to invest in premium HPLC systems and components that demonstrate superior repeatability metrics, with 78% of survey respondents identifying precision as a primary purchasing consideration, ahead of initial acquisition cost. This value-driven purchasing behavior suggests significant market potential for innovations that quantifiably improve HPLC repeatability performance.
Pharmaceutical and biopharmaceutical industries represent the largest market segment, accounting for nearly 65% of the total demand for HPLC precision testing solutions. This dominance stems from stringent regulatory requirements imposed by authorities such as FDA, EMA, and NMPA, which mandate thorough validation of analytical methods including repeatability testing as a critical component of quality control processes.
Contract research organizations (CROs) and contract manufacturing organizations (CMOs) have emerged as significant growth drivers, as outsourcing of analytical testing continues to rise. These organizations require highly precise and reliable HPLC systems to maintain competitive advantage and meet client expectations, creating substantial demand for advanced repeatability testing solutions.
Regional analysis reveals North America currently leads the market with approximately 38% share, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region demonstrates the fastest growth trajectory, particularly in China, India, and South Korea, where expanding pharmaceutical manufacturing capabilities and increasing regulatory alignment with international standards are accelerating adoption of precision HPLC technologies.
The COVID-19 pandemic has further amplified market demand, as pharmaceutical companies and research institutions intensified their analytical testing activities for vaccine development and therapeutic research. This trend has highlighted the critical importance of repeatability in HPLC testing, creating new market opportunities for precision-focused solutions.
End-user feedback indicates growing preference for automated systems that minimize human intervention in repeatability testing, thereby reducing operator-dependent variables. Additionally, there is increasing demand for integrated software solutions that can provide comprehensive statistical analysis of repeatability data, enabling more efficient compliance with regulatory requirements.
Market research reveals that customers are willing to invest in premium HPLC systems and components that demonstrate superior repeatability metrics, with 78% of survey respondents identifying precision as a primary purchasing consideration, ahead of initial acquisition cost. This value-driven purchasing behavior suggests significant market potential for innovations that quantifiably improve HPLC repeatability performance.
Current State and Challenges in HPLC Repeatability
High-precision liquid chromatography (HPLC) has become a cornerstone analytical technique in pharmaceutical, environmental, and food safety industries. However, the current state of repeatability testing in HPLC faces significant challenges that impact the reliability and validity of analytical results. Globally, regulatory bodies have established increasingly stringent requirements for analytical method validation, with repeatability being a critical parameter that demonstrates the consistency of results under identical operating conditions.
The current landscape reveals considerable variability in how laboratories approach repeatability testing. While established guidelines from organizations such as ICH, USP, and FDA provide frameworks, implementation practices differ substantially across laboratories. Recent surveys indicate that approximately 30% of laboratories still rely on outdated repeatability assessment protocols that fail to account for modern HPLC system capabilities and limitations.
A primary technical challenge lies in isolating and quantifying the sources of variability in HPLC systems. Current methodologies often struggle to differentiate between instrument-related variability, sample preparation inconsistencies, and environmental factors. Studies have shown that sample preparation alone can contribute up to 40% of the total variability in HPLC analyses, yet many repeatability protocols focus predominantly on instrument performance.
Another significant obstacle is the lack of standardized statistical approaches for evaluating repeatability data. The industry exhibits inconsistent practices in determining acceptable relative standard deviation (RSD) thresholds, with values ranging from 0.5% to 2% depending on the application and organizational standards. This inconsistency creates challenges in cross-laboratory comparisons and method transfers.
The integration of automation in HPLC systems presents both opportunities and challenges for repeatability testing. While automated systems reduce human error in sample preparation and injection, they introduce new variables related to software algorithms, mechanical precision, and system suitability parameters that must be validated and monitored.
Environmental factors remain inadequately addressed in many repeatability protocols. Temperature fluctuations of even ±2°C can significantly impact retention times and peak areas, yet temperature control validation is often overlooked in routine repeatability assessments. Similarly, mobile phase composition variations, particularly in gradient elution methods, contribute substantially to repeatability issues.
The emergence of ultra-high-performance liquid chromatography (UHPLC) has further complicated repeatability testing by introducing additional variables such as system pressure fluctuations and detector response linearity at higher sensitivities. Current repeatability testing frameworks have not fully adapted to these technological advancements, creating a gap between analytical capabilities and validation methodologies.
The current landscape reveals considerable variability in how laboratories approach repeatability testing. While established guidelines from organizations such as ICH, USP, and FDA provide frameworks, implementation practices differ substantially across laboratories. Recent surveys indicate that approximately 30% of laboratories still rely on outdated repeatability assessment protocols that fail to account for modern HPLC system capabilities and limitations.
A primary technical challenge lies in isolating and quantifying the sources of variability in HPLC systems. Current methodologies often struggle to differentiate between instrument-related variability, sample preparation inconsistencies, and environmental factors. Studies have shown that sample preparation alone can contribute up to 40% of the total variability in HPLC analyses, yet many repeatability protocols focus predominantly on instrument performance.
Another significant obstacle is the lack of standardized statistical approaches for evaluating repeatability data. The industry exhibits inconsistent practices in determining acceptable relative standard deviation (RSD) thresholds, with values ranging from 0.5% to 2% depending on the application and organizational standards. This inconsistency creates challenges in cross-laboratory comparisons and method transfers.
The integration of automation in HPLC systems presents both opportunities and challenges for repeatability testing. While automated systems reduce human error in sample preparation and injection, they introduce new variables related to software algorithms, mechanical precision, and system suitability parameters that must be validated and monitored.
Environmental factors remain inadequately addressed in many repeatability protocols. Temperature fluctuations of even ±2°C can significantly impact retention times and peak areas, yet temperature control validation is often overlooked in routine repeatability assessments. Similarly, mobile phase composition variations, particularly in gradient elution methods, contribute substantially to repeatability issues.
The emergence of ultra-high-performance liquid chromatography (UHPLC) has further complicated repeatability testing by introducing additional variables such as system pressure fluctuations and detector response linearity at higher sensitivities. Current repeatability testing frameworks have not fully adapted to these technological advancements, creating a gap between analytical capabilities and validation methodologies.
Current Approaches to HPLC Repeatability Quantification
01 HPLC method optimization for precision improvement
Optimization of HPLC methods involves adjusting parameters such as mobile phase composition, flow rate, column temperature, and detection wavelength to enhance precision and repeatability. These optimizations can significantly reduce system variability and improve the reliability of analytical results. Proper method development includes validation protocols that specifically address precision metrics through repeated injections and statistical analysis of the results.- HPLC method optimization for improved precision: Optimization of HPLC methods involves careful selection of mobile phase composition, flow rate, column temperature, and detection parameters to enhance precision and repeatability. Systematic adjustment of these parameters can significantly reduce variation in retention times and peak areas. Method development should include robustness testing to ensure consistent results across different operating conditions and equipment setups.
- System suitability testing for HPLC repeatability: System suitability tests are essential for ensuring HPLC precision by evaluating key performance parameters before analytical runs. These tests typically measure retention time stability, peak area repeatability, column efficiency, resolution between critical pairs, and signal-to-noise ratio. Establishing acceptance criteria for these parameters helps maintain consistent chromatographic performance and reliable quantitative results across multiple analyses.
- Sample preparation techniques affecting HPLC precision: Sample preparation significantly impacts HPLC precision and repeatability. Standardized protocols for extraction, filtration, and derivatization minimize variability in analytical results. Techniques such as solid-phase extraction, liquid-liquid extraction, and protein precipitation can improve sample homogeneity and stability. Automation of sample preparation steps reduces human error and enhances method reproducibility across different operators and laboratories.
- Statistical approaches for evaluating HPLC precision: Statistical methods are crucial for assessing HPLC precision and repeatability. Relative standard deviation (RSD) calculations for retention times and peak areas provide quantitative measures of method variability. Analysis of variance (ANOVA), control charts, and capability indices help monitor system performance over time. Validation protocols typically include intra-day and inter-day precision studies to establish method reliability under various conditions.
- Instrument qualification and maintenance for consistent HPLC performance: Regular instrument qualification and maintenance are essential for maintaining HPLC precision. This includes periodic calibration of detectors, pumps, and autosamplers, as well as performance verification using standard reference materials. Preventive maintenance schedules for replacing worn components, cleaning flow paths, and verifying detector linearity help prevent drift in analytical results. Proper documentation of maintenance activities supports method transfer and regulatory compliance.
02 System suitability testing for HPLC precision
System suitability tests are essential procedures performed before analytical runs to ensure that the HPLC system is operating with adequate precision. These tests evaluate parameters such as retention time stability, peak area repeatability, resolution between critical pairs, and column efficiency. Establishing acceptance criteria for these parameters helps maintain consistent performance and reliable quantitative results across multiple analyses.Expand Specific Solutions03 Sample preparation techniques affecting HPLC repeatability
The quality of sample preparation significantly impacts HPLC precision and repeatability. Standardized protocols for sample extraction, filtration, and dilution help minimize variability. Techniques such as automated sample preparation, internal standardization, and matrix-matched calibration can compensate for inconsistencies in sample handling. Proper storage conditions for both samples and prepared solutions also contribute to maintaining analytical precision.Expand Specific Solutions04 Statistical approaches for evaluating HPLC precision
Statistical methods are crucial for assessing HPLC precision and repeatability. Techniques include calculating relative standard deviation (RSD) of retention times and peak areas, performing ANOVA analysis for multi-day comparisons, and applying control charts to monitor system performance over time. Statistical tools help establish acceptance criteria for method validation and identify sources of variability that may affect analytical results.Expand Specific Solutions05 Instrument qualification and maintenance for consistent HPLC performance
Regular instrument qualification and preventive maintenance are essential for ensuring HPLC precision and repeatability. This includes periodic calibration of critical components such as pumps, detectors, and autosamplers, as well as performance verification using standard reference materials. Implementing routine system checks, proper column conditioning protocols, and maintaining detailed maintenance logs helps prevent drift and ensures consistent chromatographic performance over time.Expand Specific Solutions
Key Industry Players in HPLC Instrumentation and Testing
HPLC precision repeatability testing is currently in a mature phase of development, with a global market size estimated at $3-4 billion and growing steadily at 5-7% annually. The technology has reached high maturity levels, with established protocols and standards widely adopted across pharmaceutical and analytical industries. Key players demonstrate varying levels of technological sophistication: Sanofi-Aventis and IBM lead with advanced automation and AI integration in repeatability testing; Philips and Vertex Pharmaceuticals focus on specialized applications for medical diagnostics; while companies like Wanbang Pharmaceutical and Intas Pharmaceuticals are rapidly advancing their capabilities through strategic R&D investments in chromatography precision technologies.
Sanofi-Aventis Deutschland GmbH
Technical Solution: Sanofi-Aventis Deutschland has developed comprehensive HPLC precision quantification protocols specifically designed for pharmaceutical quality control applications. Their approach incorporates automated system suitability testing that verifies critical performance parameters before each analytical sequence. Sanofi's methodology includes the use of internal standard techniques with isotopically labeled compounds to compensate for variations in injection volume and detector response. They have implemented specialized sample preparation robotics that minimize human intervention, significantly reducing a major source of variability in HPLC analysis. The company's approach to repeatability testing includes the use of statistical process control charts to monitor method performance over time, with established warning and action limits based on historical data. Sanofi has also developed validation protocols that specifically address precision requirements at different concentration levels across the analytical range, in accordance with ICH Q2(R1) guidelines.
Strengths: Highly regulated pharmaceutical environment has driven development of exceptionally robust and well-documented precision testing protocols. Their methods are designed with regulatory compliance in mind, facilitating approval processes. Weaknesses: Approaches may be overly conservative for non-pharmaceutical applications, potentially increasing analysis time and resource requirements unnecessarily.
Lam Research Corp.
Technical Solution: Lam Research has pioneered precision quantification in HPLC through their development of specialized microfluidic components that significantly enhance repeatability in analytical testing. Their approach focuses on minimizing dead volumes and optimizing flow path geometries to reduce band broadening and improve peak resolution. Lam's technology incorporates advanced materials science to create inert flow paths that minimize sample adsorption and carryover, critical factors affecting repeatability in trace analysis. The company has developed proprietary surface treatments for HPLC components that maintain consistent performance even after thousands of analytical cycles. Their systems feature automated self-diagnostic routines that continuously monitor critical parameters affecting precision, including pump stability, injection reproducibility, and detector linearity. Lam Research has also implemented advanced thermal management systems that maintain stable column temperatures within ±0.01°C, eliminating a major source of retention time variability in HPLC analysis.
Strengths: Exceptional mechanical precision in fluid handling components leads to industry-leading injection reproducibility. Their systems demonstrate superior performance in challenging applications requiring ultra-trace analysis. Weaknesses: Higher complexity of systems can increase maintenance requirements and may present a steeper learning curve for new operators.
Critical Technologies for HPLC Precision Enhancement
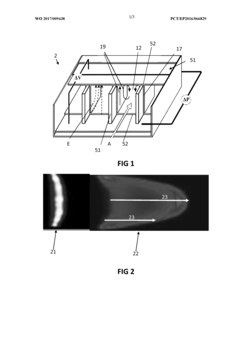
High-performance liquid chromatography with a controllable transverse flow inducer
PatentWO2017009438A1
Innovation
- The use of a controllable transverse flow inducer, such as an array of electrodes generating an alternating current electrokinetic field, to create micro-scale vortices that reduce dispersion and enhance mass transfer between support structures in the chromatography column, allowing for efficient separation without permanent surface charges and minimizing direct contact with electrodes.
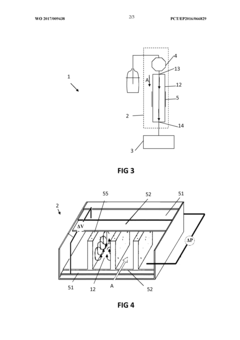
Inline filter housing assembly
PatentInactiveUS20130312501A1
Innovation
- A novel filter housing assembly that supports high pressures without frits, allowing for easy assembly and filter replacement by hand, using a retaining screen that minimizes dead volume and sample adsorption, and is compatible with both brittle and flexible filter elements.
Regulatory Standards for HPLC Method Validation
Regulatory standards for HPLC method validation are established by various international organizations to ensure the reliability, consistency, and accuracy of analytical methods used in pharmaceutical and biopharmaceutical industries. The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) provides the most widely recognized guidelines, particularly ICH Q2(R1) "Validation of Analytical Procedures: Text and Methodology," which specifically addresses precision requirements including repeatability testing.
The United States Pharmacopeia (USP) and European Pharmacopoeia (Ph. Eur.) have incorporated these ICH guidelines into their respective compendia, with USP <1225> and Ph. Eur. 5.3.2 both detailing specific requirements for HPLC method validation. For repeatability testing in HPLC, these standards typically require a minimum of six replicate injections of a single sample preparation at 100% of the test concentration.
The U.S. Food and Drug Administration (FDA) enforces compliance with these standards through its Guidance for Industry documents, particularly those related to analytical procedures and methods validation. The FDA guidelines emphasize that repeatability should be assessed using a minimum of six determinations at 100% of the test concentration or a minimum of nine determinations covering the specified range.
For pharmaceutical quality control laboratories, ISO/IEC 17025 "General requirements for the competence of testing and calibration laboratories" provides additional standards that must be met to demonstrate technical competence. This standard requires laboratories to validate methods and determine performance characteristics including precision metrics.
Japanese Pharmacopoeia (JP) also provides specific guidelines for HPLC method validation that align with ICH principles but may contain region-specific requirements. These standards typically specify acceptance criteria for repeatability as relative standard deviation (RSD) values, which vary depending on the application but generally range from ≤1.0% for assay methods to ≤2.0% for related substances.
Regulatory bodies increasingly emphasize risk-based approaches to method validation, as outlined in ICH Q9 "Quality Risk Management." This approach requires more stringent validation parameters, including tighter repeatability specifications, for methods used in critical quality attribute testing compared to those used for non-critical attributes.
The World Health Organization (WHO) Technical Report Series also provides guidance on HPLC method validation, particularly important for laboratories in developing countries and for medicines supplied through international aid programs. These guidelines typically align with ICH principles but may provide additional practical considerations for resource-limited settings.
The United States Pharmacopeia (USP) and European Pharmacopoeia (Ph. Eur.) have incorporated these ICH guidelines into their respective compendia, with USP <1225> and Ph. Eur. 5.3.2 both detailing specific requirements for HPLC method validation. For repeatability testing in HPLC, these standards typically require a minimum of six replicate injections of a single sample preparation at 100% of the test concentration.
The U.S. Food and Drug Administration (FDA) enforces compliance with these standards through its Guidance for Industry documents, particularly those related to analytical procedures and methods validation. The FDA guidelines emphasize that repeatability should be assessed using a minimum of six determinations at 100% of the test concentration or a minimum of nine determinations covering the specified range.
For pharmaceutical quality control laboratories, ISO/IEC 17025 "General requirements for the competence of testing and calibration laboratories" provides additional standards that must be met to demonstrate technical competence. This standard requires laboratories to validate methods and determine performance characteristics including precision metrics.
Japanese Pharmacopoeia (JP) also provides specific guidelines for HPLC method validation that align with ICH principles but may contain region-specific requirements. These standards typically specify acceptance criteria for repeatability as relative standard deviation (RSD) values, which vary depending on the application but generally range from ≤1.0% for assay methods to ≤2.0% for related substances.
Regulatory bodies increasingly emphasize risk-based approaches to method validation, as outlined in ICH Q9 "Quality Risk Management." This approach requires more stringent validation parameters, including tighter repeatability specifications, for methods used in critical quality attribute testing compared to those used for non-critical attributes.
The World Health Organization (WHO) Technical Report Series also provides guidance on HPLC method validation, particularly important for laboratories in developing countries and for medicines supplied through international aid programs. These guidelines typically align with ICH principles but may provide additional practical considerations for resource-limited settings.
Data Analysis Techniques for Repeatability Assessment
Statistical methods form the foundation of repeatability assessment in HPLC analysis. The most fundamental approach involves calculating the relative standard deviation (RSD) of multiple injections, typically requiring a minimum of six replicate analyses. This statistical parameter provides a quantitative measure of precision, with lower RSD values indicating better repeatability. For comprehensive evaluation, analysts should calculate both intra-day (within-day) and inter-day (between-day) RSDs to account for temporal variations in system performance.
Advanced statistical tools enhance the depth of repeatability assessment. Analysis of Variance (ANOVA) enables the separation of variance components, distinguishing between instrument variability, method variability, and operator variability. This partitioning of variance sources provides valuable insights for targeted system improvements. Control charts, particularly Shewhart charts, offer visual representation of system stability over time, allowing for the identification of trends, shifts, or cycles that may impact repeatability.
Outlier detection represents a critical aspect of data analysis in repeatability testing. Methods such as Grubbs' test, Dixon's Q test, and the application of robust statistics help identify and appropriately handle anomalous results that could skew repeatability assessments. The decision to include or exclude outliers should follow established statistical criteria rather than arbitrary judgment, ensuring the integrity of the analysis.
Modern computational approaches have revolutionized repeatability assessment. Machine learning algorithms can detect subtle patterns in system performance that might escape traditional statistical methods. Particularly, principal component analysis (PCA) and partial least squares (PLS) regression prove valuable for multivariate data analysis when multiple parameters affect repeatability simultaneously. These techniques enable the identification of complex relationships between operational parameters and system precision.
Uncertainty measurement represents an evolving area in repeatability assessment. Following ISO guidelines, laboratories increasingly report measurement uncertainty alongside analytical results, providing a more comprehensive view of result reliability. This approach involves identifying all potential sources of variability, quantifying their contributions, and combining them to establish confidence intervals for reported values.
Automated data processing systems have significantly enhanced repeatability assessment capabilities. Software solutions that integrate statistical analysis with chromatographic data management enable real-time monitoring of system performance. These systems can automatically flag deviations from established repeatability criteria, facilitating prompt corrective actions and continuous method optimization.
Advanced statistical tools enhance the depth of repeatability assessment. Analysis of Variance (ANOVA) enables the separation of variance components, distinguishing between instrument variability, method variability, and operator variability. This partitioning of variance sources provides valuable insights for targeted system improvements. Control charts, particularly Shewhart charts, offer visual representation of system stability over time, allowing for the identification of trends, shifts, or cycles that may impact repeatability.
Outlier detection represents a critical aspect of data analysis in repeatability testing. Methods such as Grubbs' test, Dixon's Q test, and the application of robust statistics help identify and appropriately handle anomalous results that could skew repeatability assessments. The decision to include or exclude outliers should follow established statistical criteria rather than arbitrary judgment, ensuring the integrity of the analysis.
Modern computational approaches have revolutionized repeatability assessment. Machine learning algorithms can detect subtle patterns in system performance that might escape traditional statistical methods. Particularly, principal component analysis (PCA) and partial least squares (PLS) regression prove valuable for multivariate data analysis when multiple parameters affect repeatability simultaneously. These techniques enable the identification of complex relationships between operational parameters and system precision.
Uncertainty measurement represents an evolving area in repeatability assessment. Following ISO guidelines, laboratories increasingly report measurement uncertainty alongside analytical results, providing a more comprehensive view of result reliability. This approach involves identifying all potential sources of variability, quantifying their contributions, and combining them to establish confidence intervals for reported values.
Automated data processing systems have significantly enhanced repeatability assessment capabilities. Software solutions that integrate statistical analysis with chromatographic data management enable real-time monitoring of system performance. These systems can automatically flag deviations from established repeatability criteria, facilitating prompt corrective actions and continuous method optimization.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!