Alkaline stability improvement through polymer engineering
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Alkaline Stability Engineering Background and Objectives
Alkaline stability has emerged as a critical challenge in the development of advanced polymer materials for various applications, particularly in energy conversion and storage systems. The evolution of this field traces back to the early 2000s when researchers began recognizing the limitations of conventional polymers in highly alkaline environments. The degradation mechanisms observed in these conditions significantly hindered the performance and longevity of materials used in fuel cells, electrolyzers, and other electrochemical devices.
The progression of alkaline stability engineering has been marked by incremental improvements in understanding the fundamental chemical interactions between polymer structures and hydroxide ions. Initially, research focused primarily on identifying degradation pathways, with limited success in developing truly stable materials. However, the past decade has witnessed substantial advancements in polymer chemistry and materials science, enabling more sophisticated approaches to molecular design.
Current technical objectives in this field center on developing polymers that maintain structural integrity and functionality under prolonged exposure to high pH environments (typically pH > 13) and elevated temperatures (80-120°C). Specifically, researchers aim to achieve alkaline stability for operational lifetimes exceeding 10,000 hours while preserving essential properties such as ionic conductivity, mechanical strength, and processability.
The engineering challenges extend beyond mere chemical stability to encompass the complex interplay between various material properties. For instance, modifications that enhance alkaline resistance often compromise other critical characteristics like ion transport efficiency or mechanical flexibility. This necessitates a holistic approach to polymer engineering that considers the multifaceted requirements of end applications.
Recent technological trends indicate a shift toward rational molecular design strategies informed by computational modeling and high-throughput experimental techniques. These approaches enable more systematic exploration of structure-property relationships and accelerate the discovery of novel polymer architectures with superior alkaline stability.
The ultimate technical goal remains the development of a new generation of polymeric materials that can withstand aggressive alkaline conditions without sacrificing performance or economic viability. This would enable transformative advances in clean energy technologies, particularly anion exchange membrane fuel cells and electrolyzers, which offer significant advantages over their acidic counterparts in terms of cost, efficiency, and sustainability.
The progression of alkaline stability engineering has been marked by incremental improvements in understanding the fundamental chemical interactions between polymer structures and hydroxide ions. Initially, research focused primarily on identifying degradation pathways, with limited success in developing truly stable materials. However, the past decade has witnessed substantial advancements in polymer chemistry and materials science, enabling more sophisticated approaches to molecular design.
Current technical objectives in this field center on developing polymers that maintain structural integrity and functionality under prolonged exposure to high pH environments (typically pH > 13) and elevated temperatures (80-120°C). Specifically, researchers aim to achieve alkaline stability for operational lifetimes exceeding 10,000 hours while preserving essential properties such as ionic conductivity, mechanical strength, and processability.
The engineering challenges extend beyond mere chemical stability to encompass the complex interplay between various material properties. For instance, modifications that enhance alkaline resistance often compromise other critical characteristics like ion transport efficiency or mechanical flexibility. This necessitates a holistic approach to polymer engineering that considers the multifaceted requirements of end applications.
Recent technological trends indicate a shift toward rational molecular design strategies informed by computational modeling and high-throughput experimental techniques. These approaches enable more systematic exploration of structure-property relationships and accelerate the discovery of novel polymer architectures with superior alkaline stability.
The ultimate technical goal remains the development of a new generation of polymeric materials that can withstand aggressive alkaline conditions without sacrificing performance or economic viability. This would enable transformative advances in clean energy technologies, particularly anion exchange membrane fuel cells and electrolyzers, which offer significant advantages over their acidic counterparts in terms of cost, efficiency, and sustainability.
Market Analysis for Alkaline-Resistant Polymers
The alkaline-resistant polymer market is experiencing significant growth driven by increasing applications in fuel cells, water treatment systems, and industrial processes requiring high pH environments. The global market for these specialized polymers was valued at approximately $2.3 billion in 2022 and is projected to reach $3.7 billion by 2028, representing a compound annual growth rate of 8.2%. This growth trajectory is primarily fueled by the expanding clean energy sector, particularly the hydrogen economy and alkaline fuel cell technologies.
The energy sector constitutes the largest market segment, accounting for nearly 40% of the total demand for alkaline-resistant polymers. Within this sector, alkaline fuel cells and electrolyzers represent the fastest-growing application areas due to their role in green hydrogen production and utilization. The water treatment industry follows closely, comprising about 30% of the market, where these polymers are essential for membrane technologies operating in high pH conditions.
Geographically, North America and Europe currently lead the market with combined shares of approximately 65%, attributed to their advanced research infrastructure and early adoption of hydrogen technologies. However, the Asia-Pacific region is expected to witness the highest growth rate of 10.5% annually through 2028, driven by China's aggressive investments in clean energy infrastructure and Japan's hydrogen economy initiatives.
Customer requirements are increasingly focused on polymers that can maintain structural integrity and functionality in environments with pH levels above 12 for extended periods, often exceeding 10,000 hours of operational stability. This represents a significant shift from earlier market demands that typically required stability at pH 10-11 for shorter durations.
The market is also witnessing a trend toward cost reduction, with customers seeking alkaline-resistant polymers that offer a balance between performance and affordability. Current high-performance options can cost between $200-500 per kilogram, creating a substantial barrier to widespread adoption in price-sensitive applications.
Regulatory factors are increasingly influencing market dynamics, with stricter environmental regulations favoring green hydrogen production technologies that rely heavily on alkaline-resistant polymers. Additionally, sustainability considerations are driving demand for bio-based or recyclable alkaline-resistant polymer alternatives, creating a niche but rapidly growing sub-segment within the market.
Industry analysts predict that breakthrough innovations in polymer engineering that can significantly improve alkaline stability while reducing costs could potentially double the market size within five years by unlocking new application areas currently limited by technical or economic constraints.
The energy sector constitutes the largest market segment, accounting for nearly 40% of the total demand for alkaline-resistant polymers. Within this sector, alkaline fuel cells and electrolyzers represent the fastest-growing application areas due to their role in green hydrogen production and utilization. The water treatment industry follows closely, comprising about 30% of the market, where these polymers are essential for membrane technologies operating in high pH conditions.
Geographically, North America and Europe currently lead the market with combined shares of approximately 65%, attributed to their advanced research infrastructure and early adoption of hydrogen technologies. However, the Asia-Pacific region is expected to witness the highest growth rate of 10.5% annually through 2028, driven by China's aggressive investments in clean energy infrastructure and Japan's hydrogen economy initiatives.
Customer requirements are increasingly focused on polymers that can maintain structural integrity and functionality in environments with pH levels above 12 for extended periods, often exceeding 10,000 hours of operational stability. This represents a significant shift from earlier market demands that typically required stability at pH 10-11 for shorter durations.
The market is also witnessing a trend toward cost reduction, with customers seeking alkaline-resistant polymers that offer a balance between performance and affordability. Current high-performance options can cost between $200-500 per kilogram, creating a substantial barrier to widespread adoption in price-sensitive applications.
Regulatory factors are increasingly influencing market dynamics, with stricter environmental regulations favoring green hydrogen production technologies that rely heavily on alkaline-resistant polymers. Additionally, sustainability considerations are driving demand for bio-based or recyclable alkaline-resistant polymer alternatives, creating a niche but rapidly growing sub-segment within the market.
Industry analysts predict that breakthrough innovations in polymer engineering that can significantly improve alkaline stability while reducing costs could potentially double the market size within five years by unlocking new application areas currently limited by technical or economic constraints.
Current Challenges in Polymer Alkaline Stability
Despite significant advancements in polymer engineering for alkaline environments, several critical challenges continue to impede the development of truly stable polymeric materials. The primary obstacle remains the inherent vulnerability of most polymer backbones to nucleophilic attack under high pH conditions. Hydroxide ions aggressively target electron-deficient sites within polymer structures, leading to chain scission, degradation of mechanical properties, and ultimately, complete material failure.
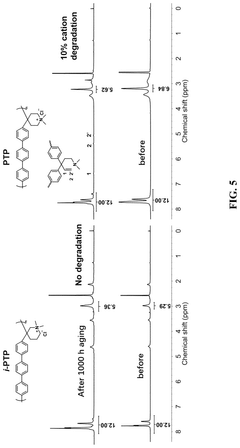
Quaternary ammonium cation-functionalized polymers, widely used in alkaline membrane fuel cells and electrolyzers, face particularly severe stability issues. These cationic groups undergo several degradation pathways including Hofmann elimination, nucleophilic substitution, and ylide formation. Current research indicates that even the most advanced materials typically maintain functionality for only 1,000-5,000 hours under operating conditions, falling significantly short of the 40,000+ hours required for commercial viability.
Cross-linking strategies, while improving dimensional stability, often introduce additional degradation pathways or reduce ionic conductivity. The trade-off between mechanical robustness and electrochemical performance represents a fundamental challenge that has yet to be fully resolved. Furthermore, many stabilization approaches that work well in laboratory settings fail to translate to real-world applications where temperature fluctuations, mechanical stress, and chemical contaminants accelerate degradation processes.
Another significant hurdle is the lack of standardized testing protocols for alkaline stability. Different research groups employ varying conditions—temperature ranges from 25°C to 120°C, KOH concentrations from 1M to 10M, and test durations from hours to months—making direct comparisons between materials nearly impossible. This inconsistency hampers systematic improvement efforts and obscures understanding of degradation mechanisms.
The scalability of synthesis methods presents additional complications. Many promising polymer architectures with enhanced alkaline stability rely on complex multi-step syntheses or expensive catalysts, limiting their commercial viability. The challenge of translating laboratory-scale success to industrial production remains largely unaddressed in current research.
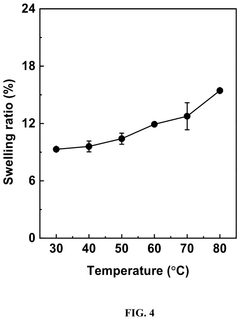
Water management within polymer systems introduces another layer of complexity. While water is necessary for ion transport, excessive water uptake leads to dimensional instability and mechanical failure. Conversely, insufficient hydration reduces ionic conductivity. Engineering polymers that maintain optimal hydration levels while resisting alkaline degradation continues to challenge researchers across the field.
Quaternary ammonium cation-functionalized polymers, widely used in alkaline membrane fuel cells and electrolyzers, face particularly severe stability issues. These cationic groups undergo several degradation pathways including Hofmann elimination, nucleophilic substitution, and ylide formation. Current research indicates that even the most advanced materials typically maintain functionality for only 1,000-5,000 hours under operating conditions, falling significantly short of the 40,000+ hours required for commercial viability.
Cross-linking strategies, while improving dimensional stability, often introduce additional degradation pathways or reduce ionic conductivity. The trade-off between mechanical robustness and electrochemical performance represents a fundamental challenge that has yet to be fully resolved. Furthermore, many stabilization approaches that work well in laboratory settings fail to translate to real-world applications where temperature fluctuations, mechanical stress, and chemical contaminants accelerate degradation processes.
Another significant hurdle is the lack of standardized testing protocols for alkaline stability. Different research groups employ varying conditions—temperature ranges from 25°C to 120°C, KOH concentrations from 1M to 10M, and test durations from hours to months—making direct comparisons between materials nearly impossible. This inconsistency hampers systematic improvement efforts and obscures understanding of degradation mechanisms.
The scalability of synthesis methods presents additional complications. Many promising polymer architectures with enhanced alkaline stability rely on complex multi-step syntheses or expensive catalysts, limiting their commercial viability. The challenge of translating laboratory-scale success to industrial production remains largely unaddressed in current research.
Water management within polymer systems introduces another layer of complexity. While water is necessary for ion transport, excessive water uptake leads to dimensional instability and mechanical failure. Conversely, insufficient hydration reduces ionic conductivity. Engineering polymers that maintain optimal hydration levels while resisting alkaline degradation continues to challenge researchers across the field.
State-of-the-Art Polymer Engineering Solutions
01 Alkaline-stable polymer compositions for industrial applications
Certain polymer compositions are specifically designed to maintain stability in alkaline environments, making them suitable for industrial applications such as battery separators, membranes, and coatings. These polymers typically contain chemical structures that resist degradation when exposed to high pH conditions. The formulations may include cross-linking agents or stabilizers that enhance their resistance to alkaline hydrolysis, allowing them to maintain structural integrity and performance characteristics in caustic environments.- Alkaline-resistant polymer compositions: Specific polymer compositions have been developed to withstand alkaline environments without degradation. These compositions often include modified backbone structures or protective functional groups that shield the polymer from hydroxide ion attack. Such polymers maintain their structural integrity and performance characteristics even when exposed to high pH conditions, making them suitable for applications in alkaline cleaning solutions, concrete additives, and industrial processes involving caustic environments.
- Membrane polymers with enhanced alkaline stability: Specialized membrane polymers have been engineered to maintain functionality in alkaline conditions. These membranes incorporate chemical modifications that prevent hydrolysis and degradation in high pH environments. The enhanced stability allows for extended operational lifetimes in applications such as alkaline fuel cells, electrolysis systems, and water treatment processes where membranes are exposed to caustic solutions while maintaining ion selectivity and mechanical properties.
- Coating polymers resistant to alkaline degradation: Protective coating polymers have been formulated to resist degradation in alkaline environments. These coatings typically incorporate cross-linking agents, stabilizing additives, or modified polymer structures that prevent saponification and chain scission under alkaline conditions. Such coatings provide long-term protection for substrates exposed to high pH environments, including concrete structures, industrial equipment, and alkaline cleaning processes.
- Alkaline-stable polymer additives for industrial applications: Specialized polymer additives have been developed to maintain functionality in alkaline industrial processes. These additives include dispersants, flocculants, rheology modifiers, and processing aids that resist degradation in high pH environments. The enhanced alkaline stability allows these polymers to perform effectively in applications such as paper manufacturing, textile processing, water treatment, and mineral processing where alkaline conditions are prevalent.
- Biodegradable polymers with improved alkaline stability: Environmentally friendly polymer formulations have been created with enhanced resistance to alkaline hydrolysis. These biodegradable polymers incorporate structural modifications or protective additives that slow degradation in high pH environments while maintaining their ability to eventually break down through natural processes. Such polymers are particularly valuable in applications where both alkaline stability and environmental sustainability are required, including agricultural products, packaging materials, and consumer goods.
02 Modified polyolefins with enhanced alkaline stability
Polyolefins can be modified to improve their stability in alkaline conditions through various chemical treatments and additives. These modifications often involve the incorporation of functional groups that resist alkaline attack or the addition of stabilizing compounds that neutralize the effects of alkaline substances. The resulting polymers exhibit improved resistance to degradation, swelling, and dissolution when exposed to high pH environments, making them suitable for applications in alkaline batteries, cleaning solutions, and industrial processes.Expand Specific Solutions03 Alkaline-resistant polymer membranes and films
Specialized polymer membranes and films can be formulated to withstand alkaline conditions while maintaining their barrier properties and mechanical strength. These materials often incorporate alkaline-resistant monomers or are treated with surface modifiers to enhance their stability. The membranes may be used in filtration systems, fuel cells, and other applications where exposure to alkaline solutions is common. Their design typically balances permeability, selectivity, and chemical resistance to ensure optimal performance in challenging pH environments.Expand Specific Solutions04 Hydrogels and superabsorbent polymers with alkaline stability
Hydrogels and superabsorbent polymers can be engineered to maintain their absorption capacity and structural integrity in alkaline environments. These materials typically contain specially designed cross-linking networks that resist degradation under high pH conditions. The formulations may include alkaline-resistant monomers and stabilizing additives that protect the polymer backbone from hydrolysis. Applications include absorbent products used in industrial spill control, agricultural soil amendments, and personal care products that may encounter alkaline conditions.Expand Specific Solutions05 Coating polymers with enhanced alkaline resistance
Protective coating polymers can be formulated to resist degradation in alkaline environments through the incorporation of specific chemical structures and additives. These coatings typically contain monomers with low susceptibility to alkaline hydrolysis and may include stabilizers that neutralize alkaline compounds. The resulting materials provide durable protection for substrates exposed to high pH conditions, such as concrete structures, industrial equipment, and alkaline cleaning processes. Their formulation balances adhesion properties, flexibility, and chemical resistance to ensure long-term performance.Expand Specific Solutions
Leading Organizations in Alkaline-Stable Polymer Research
The alkaline stability improvement through polymer engineering field is currently in a growth phase, with an estimated market size of $2-3 billion and expanding at 7-9% annually. The competitive landscape features established chemical giants like BASF, DuPont, and Eastman Chemical leading commercial applications, while academic institutions such as the National University of Singapore and Rensselaer Polytechnic Institute drive fundamental research. Technology maturity varies across applications, with Solvay Specialty Polymers and Clariant International demonstrating advanced commercial solutions for fuel cells and water treatment membranes, while companies like Nippon Shokubai and Resonac Holdings focus on emerging applications in energy storage. The field is characterized by increasing industry-academic collaborations, with companies like EMS-INVENTA and Arkema France actively developing next-generation alkaline-stable polymers for sustainable applications.
National University of Singapore
Technical Solution: The National University of Singapore (NUS) has pioneered groundbreaking research in alkaline-stable polymers through their Advanced Materials Research Group. Their approach centers on molecular engineering of aromatic polymers with sterically protected cationic groups. NUS researchers have developed novel synthesis routes for poly(arylene ether)s with pendant quaternary ammonium groups positioned to minimize β-hydrogen elimination—a primary degradation pathway in alkaline environments. Their work has demonstrated polymers retaining over 95% conductivity after 1000 hours in 1M KOH at 80°C. NUS has also created innovative cross-linked networks using click chemistry that forms alkaline-resistant triazole linkages, significantly enhancing mechanical stability while maintaining ion transport properties. Their recent publications have showcased hybrid materials incorporating graphene oxide nanosheets that create tortuous pathways for hydroxide ions, reducing direct attack on vulnerable polymer sites while maintaining excellent ionic conductivity.
Strengths: Cutting-edge fundamental research capabilities; strong international collaboration network; access to advanced characterization facilities. Weaknesses: Limited manufacturing scale-up capabilities; dependency on external funding; challenges in technology transfer to industrial applications.
Solvay Specialty Polymers USA LLC
Technical Solution: Solvay has developed advanced perfluorinated sulfonic acid (PFSA) polymers with enhanced alkaline stability through strategic modification of polymer backbones and side chains. Their approach involves incorporating bulky aromatic groups that shield vulnerable sites from hydroxide attack while maintaining ion conductivity. Solvay's proprietary cross-linking technology creates three-dimensional polymer networks that significantly improve mechanical stability in alkaline environments. Their research has demonstrated membranes retaining over 80% conductivity after 1000 hours in 1M KOH at 80°C, representing a substantial improvement over conventional materials. Solvay has also pioneered the development of hydrocarbon-based polymers with quaternary ammonium functional groups specifically engineered to resist degradation in high pH environments.
Strengths: Industry-leading expertise in fluoropolymers with exceptional chemical resistance; established manufacturing infrastructure for specialty polymers; extensive intellectual property portfolio. Weaknesses: Higher production costs compared to hydrocarbon alternatives; environmental concerns regarding fluorinated materials; potential regulatory challenges for certain applications.
Critical Patents and Innovations in Alkaline Stability
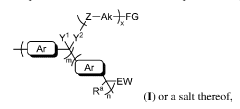
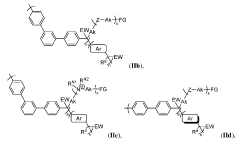
Highly alkali-stable cationic polymers and their preparation methods and applications
PatentActiveUS12240924B2
Innovation
- The preparation method involves changing the connection position of piperidinium cations to the C-3 position of aromatic units, reducing the influence of electron-withdrawing and conformational restriction effects, and enhancing the steric hindrance to prevent Hoffman elimination, thereby improving alkaline stability.
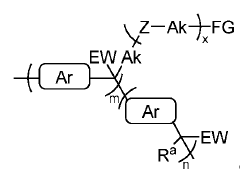
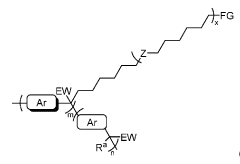
Anion-solvating polymer membranes
PatentWO2021150994A1
Innovation
- Development of anion-solvating polymer membranes with hydrophilic graft chains and specific electron-withdrawing moieties that enhance alkaline stability and conductivity, allowing for operation in 6M KOH at 120°C with improved mechanical robustness and chemical stability.
Sustainability Aspects of Alkaline-Stable Materials
The development of alkaline-stable polymers represents a significant advancement in sustainable materials science. These materials offer considerable environmental benefits compared to traditional alternatives, particularly in applications such as anion exchange membranes for fuel cells and electrolyzers. By enabling efficient alkaline-based energy conversion systems, these polymers contribute to reducing carbon emissions and fossil fuel dependence, aligning with global sustainability goals.
From a lifecycle perspective, alkaline-stable polymers demonstrate promising sustainability profiles. Many research efforts focus on developing these materials from renewable resources rather than petroleum-based feedstocks, significantly reducing their carbon footprint. The extended operational lifetime achieved through improved alkaline stability directly translates to reduced material consumption and waste generation, as fewer replacement components are needed over time.
Water management represents another critical sustainability aspect of these materials. Alkaline-stable polymers with optimized water uptake properties contribute to more efficient water utilization in electrochemical systems, addressing water scarcity concerns in certain regions. Additionally, the elimination of precious metal catalysts in alkaline environments provides substantial sustainability advantages, reducing dependence on scarce resources and environmentally damaging mining operations.
The recyclability and end-of-life management of alkaline-stable polymers remain active areas of research. Current efforts focus on designing these materials with circular economy principles in mind, incorporating degradable linkages or recoverable components that facilitate material reclamation and reprocessing. This approach minimizes waste and reduces the environmental impact associated with disposal.
Energy efficiency improvements represent perhaps the most significant sustainability contribution of alkaline-stable polymers. By enabling more efficient electrochemical energy conversion and storage systems, these materials support the broader transition to renewable energy sources. The enhanced performance and durability of alkaline-stable polymer-based systems result in higher energy conversion efficiencies and lower overall energy consumption throughout their operational lifetime.
Regulatory frameworks increasingly recognize the sustainability benefits of these advanced materials. Several jurisdictions have implemented policies that incentivize the development and adoption of sustainable polymer technologies, including alkaline-stable variants. These policies typically focus on reducing environmental impacts, promoting circular economy principles, and supporting the transition to renewable energy systems.
From a lifecycle perspective, alkaline-stable polymers demonstrate promising sustainability profiles. Many research efforts focus on developing these materials from renewable resources rather than petroleum-based feedstocks, significantly reducing their carbon footprint. The extended operational lifetime achieved through improved alkaline stability directly translates to reduced material consumption and waste generation, as fewer replacement components are needed over time.
Water management represents another critical sustainability aspect of these materials. Alkaline-stable polymers with optimized water uptake properties contribute to more efficient water utilization in electrochemical systems, addressing water scarcity concerns in certain regions. Additionally, the elimination of precious metal catalysts in alkaline environments provides substantial sustainability advantages, reducing dependence on scarce resources and environmentally damaging mining operations.
The recyclability and end-of-life management of alkaline-stable polymers remain active areas of research. Current efforts focus on designing these materials with circular economy principles in mind, incorporating degradable linkages or recoverable components that facilitate material reclamation and reprocessing. This approach minimizes waste and reduces the environmental impact associated with disposal.
Energy efficiency improvements represent perhaps the most significant sustainability contribution of alkaline-stable polymers. By enabling more efficient electrochemical energy conversion and storage systems, these materials support the broader transition to renewable energy sources. The enhanced performance and durability of alkaline-stable polymer-based systems result in higher energy conversion efficiencies and lower overall energy consumption throughout their operational lifetime.
Regulatory frameworks increasingly recognize the sustainability benefits of these advanced materials. Several jurisdictions have implemented policies that incentivize the development and adoption of sustainable polymer technologies, including alkaline-stable variants. These policies typically focus on reducing environmental impacts, promoting circular economy principles, and supporting the transition to renewable energy systems.
Performance Metrics and Testing Protocols
Standardized performance metrics and testing protocols are essential for evaluating the alkaline stability of polymers in a consistent and reproducible manner. The field currently employs several key methodologies to quantify stability performance, with accelerated aging tests being among the most common. These tests typically involve immersing polymer samples in concentrated alkaline solutions (often 1-6M KOH or NaOH) at elevated temperatures (60-80°C) for extended periods, simulating years of operational degradation within weeks.
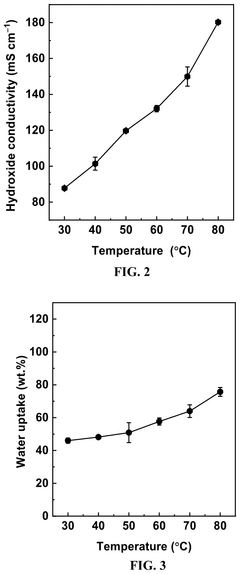
Ion exchange capacity (IEC) measurements serve as a primary indicator of polymer stability, tracking the retention of functional groups over time. A stable polymer should maintain at least 80% of its initial IEC after exposure to alkaline conditions. Complementary to IEC, conductivity retention measurements directly assess the polymer's ability to maintain ion transport capabilities despite alkaline exposure, with high-performing materials exhibiting less than 20% conductivity loss after 1000 hours in 1M KOH at 80°C.
Mechanical property evaluation constitutes another critical dimension of stability assessment. Tensile strength, elongation at break, and dimensional stability tests reveal how alkaline environments affect the physical integrity of polymer membranes. Advanced materials should retain at least 70% of their mechanical properties after extended alkaline exposure to be considered viable for long-term applications.
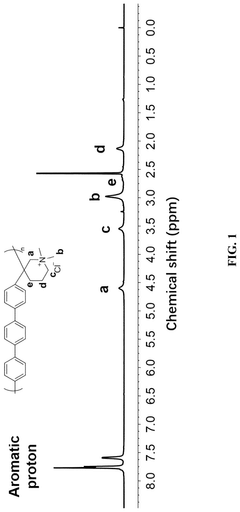
Spectroscopic analysis techniques, including FTIR, NMR, and Raman spectroscopy, provide molecular-level insights into degradation mechanisms by identifying chemical changes in polymer structures. These techniques help researchers pinpoint vulnerable bonds and functional groups, guiding targeted polymer engineering efforts.
Electrochemical impedance spectroscopy (EIS) offers a comprehensive evaluation of membrane performance in operating conditions, measuring resistance changes over time under alkaline exposure. This technique is particularly valuable for assessing real-world performance degradation patterns.
Standardization efforts have been advancing through organizations like the International Electrotechnical Commission (IEC) and ASTM International, which are developing protocols specifically for alkaline membrane testing. However, the field still lacks universally accepted testing standards, creating challenges when comparing results across different research groups.
Emerging accelerated testing methodologies utilizing higher hydroxide concentrations and temperatures must be carefully validated against real-world degradation patterns to ensure their predictive accuracy. The development of in-situ and operando characterization techniques represents the frontier of testing protocols, enabling real-time monitoring of degradation processes during actual device operation.
Ion exchange capacity (IEC) measurements serve as a primary indicator of polymer stability, tracking the retention of functional groups over time. A stable polymer should maintain at least 80% of its initial IEC after exposure to alkaline conditions. Complementary to IEC, conductivity retention measurements directly assess the polymer's ability to maintain ion transport capabilities despite alkaline exposure, with high-performing materials exhibiting less than 20% conductivity loss after 1000 hours in 1M KOH at 80°C.
Mechanical property evaluation constitutes another critical dimension of stability assessment. Tensile strength, elongation at break, and dimensional stability tests reveal how alkaline environments affect the physical integrity of polymer membranes. Advanced materials should retain at least 70% of their mechanical properties after extended alkaline exposure to be considered viable for long-term applications.
Spectroscopic analysis techniques, including FTIR, NMR, and Raman spectroscopy, provide molecular-level insights into degradation mechanisms by identifying chemical changes in polymer structures. These techniques help researchers pinpoint vulnerable bonds and functional groups, guiding targeted polymer engineering efforts.
Electrochemical impedance spectroscopy (EIS) offers a comprehensive evaluation of membrane performance in operating conditions, measuring resistance changes over time under alkaline exposure. This technique is particularly valuable for assessing real-world performance degradation patterns.
Standardization efforts have been advancing through organizations like the International Electrotechnical Commission (IEC) and ASTM International, which are developing protocols specifically for alkaline membrane testing. However, the field still lacks universally accepted testing standards, creating challenges when comparing results across different research groups.
Emerging accelerated testing methodologies utilizing higher hydroxide concentrations and temperatures must be carefully validated against real-world degradation patterns to ensure their predictive accuracy. The development of in-situ and operando characterization techniques represents the frontier of testing protocols, enabling real-time monitoring of degradation processes during actual device operation.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!