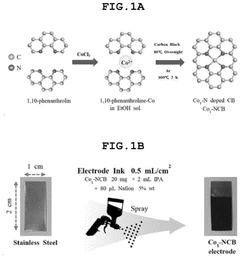
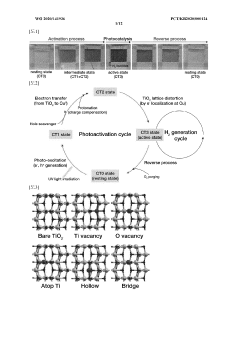
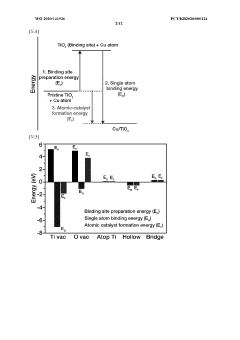
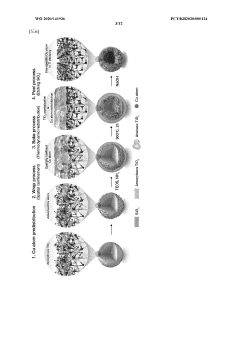
Single-Atom Catalysis for Enhanced Electrochemical Performance
OCT 15, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Single-Atom Catalysis Background and Objectives
Single-atom catalysis (SAC) represents a revolutionary frontier in heterogeneous catalysis that has emerged over the past decade. This innovative approach involves dispersing individual metal atoms onto suitable supports, maximizing atomic efficiency while delivering exceptional catalytic performance. The concept was first formally introduced in 2011, though earlier studies had observed similar phenomena without explicitly defining the field. Since then, SAC has experienced exponential growth in research interest, particularly for electrochemical applications.
The evolution of catalysis technology has progressed from traditional bulk catalysts to nanoparticles, nanoclusters, and finally to single-atom catalysts, representing the ultimate limit of size reduction. This progression has been driven by the continuous pursuit of higher catalytic efficiency, selectivity, and reduced noble metal usage. Single-atom catalysts offer unique electronic structures and coordination environments that differ significantly from their bulk counterparts, enabling novel reaction pathways and enhanced performance.
In electrochemical systems, SACs have demonstrated remarkable capabilities for reactions including oxygen reduction reaction (ORR), oxygen evolution reaction (OER), hydrogen evolution reaction (HER), and CO2 reduction. These reactions are fundamental to emerging clean energy technologies such as fuel cells, water electrolyzers, and metal-air batteries. The exceptional performance of SACs stems from their maximized atom utilization, unique electronic properties, and tunable coordination environments.
The primary objectives of research in single-atom catalysis for electrochemical applications include: developing scalable and reproducible synthesis methods for stable SACs; understanding the fundamental mechanisms governing their exceptional activity; establishing structure-property relationships to guide rational design; and demonstrating their practical implementation in real-world electrochemical devices. These objectives align with broader societal goals of sustainable energy conversion and storage.
Recent advances in characterization techniques, particularly aberration-corrected electron microscopy and X-ray absorption spectroscopy, have been instrumental in confirming the atomic dispersion of metal centers and elucidating their local coordination environments. Computational methods, especially density functional theory (DFT), have complemented experimental approaches by providing atomic-level insights into reaction mechanisms and guiding catalyst design.
Looking forward, the field aims to transition from proof-of-concept demonstrations to practical applications that can address global energy challenges. This requires overcoming persistent challenges related to stability under operating conditions, scalable synthesis, and comprehensive understanding of reaction mechanisms at the atomic level.
The evolution of catalysis technology has progressed from traditional bulk catalysts to nanoparticles, nanoclusters, and finally to single-atom catalysts, representing the ultimate limit of size reduction. This progression has been driven by the continuous pursuit of higher catalytic efficiency, selectivity, and reduced noble metal usage. Single-atom catalysts offer unique electronic structures and coordination environments that differ significantly from their bulk counterparts, enabling novel reaction pathways and enhanced performance.
In electrochemical systems, SACs have demonstrated remarkable capabilities for reactions including oxygen reduction reaction (ORR), oxygen evolution reaction (OER), hydrogen evolution reaction (HER), and CO2 reduction. These reactions are fundamental to emerging clean energy technologies such as fuel cells, water electrolyzers, and metal-air batteries. The exceptional performance of SACs stems from their maximized atom utilization, unique electronic properties, and tunable coordination environments.
The primary objectives of research in single-atom catalysis for electrochemical applications include: developing scalable and reproducible synthesis methods for stable SACs; understanding the fundamental mechanisms governing their exceptional activity; establishing structure-property relationships to guide rational design; and demonstrating their practical implementation in real-world electrochemical devices. These objectives align with broader societal goals of sustainable energy conversion and storage.
Recent advances in characterization techniques, particularly aberration-corrected electron microscopy and X-ray absorption spectroscopy, have been instrumental in confirming the atomic dispersion of metal centers and elucidating their local coordination environments. Computational methods, especially density functional theory (DFT), have complemented experimental approaches by providing atomic-level insights into reaction mechanisms and guiding catalyst design.
Looking forward, the field aims to transition from proof-of-concept demonstrations to practical applications that can address global energy challenges. This requires overcoming persistent challenges related to stability under operating conditions, scalable synthesis, and comprehensive understanding of reaction mechanisms at the atomic level.
Market Analysis for Advanced Electrochemical Applications
The global market for advanced electrochemical applications is experiencing unprecedented growth, primarily driven by the integration of single-atom catalysis (SAC) technologies. Current market valuations indicate that the electrochemical energy storage and conversion sector reached approximately $112 billion in 2022, with projections suggesting a compound annual growth rate of 18.7% through 2030. This remarkable expansion is largely attributed to increasing demands for sustainable energy solutions and the superior performance characteristics offered by SAC-enhanced electrochemical systems.
The fuel cell market segment demonstrates particularly strong potential, with single-atom catalysts addressing critical cost and durability barriers that have historically limited commercial adoption. Platinum-based SACs have reduced catalyst loading requirements by up to 70% while maintaining or improving performance metrics, creating significant cost advantages for manufacturers and accelerating market penetration in transportation and stationary power applications.
Electrolyzers represent another high-growth segment, with the green hydrogen production market expected to expand at over 50% annually through 2028. Single-atom catalysts have demonstrated hydrogen evolution reaction (HER) efficiencies approaching theoretical limits, substantially reducing energy input requirements and operational costs. This performance enhancement is creating new market opportunities in industrial decarbonization efforts where economic viability was previously questionable.
Battery technologies incorporating SAC innovations are witnessing rapid commercial adoption, particularly in the electric vehicle sector. Enhanced electrode performance through single-atom catalysis has enabled faster charging rates, increased energy density, and extended cycle life—addressing key consumer concerns that have limited EV market penetration. Premium automotive manufacturers have begun incorporating SAC-enhanced battery systems in flagship models, establishing a premium market segment with higher profit margins.
Regional market analysis reveals Asia-Pacific dominance in manufacturing capacity, with China accounting for over 60% of global production capability for SAC-enhanced electrochemical components. However, North American and European markets are rapidly expanding research investments, particularly in specialized applications for aerospace, defense, and medical devices where performance requirements justify premium pricing.
Consumer electronics represents an emerging application area with substantial growth potential, as miniaturized electrochemical systems benefit significantly from the efficiency improvements offered by single-atom catalysts. Wearable technology manufacturers have demonstrated particular interest in SAC-enhanced microbatteries and supercapacitors that enable extended device operation without increasing form factors.
The fuel cell market segment demonstrates particularly strong potential, with single-atom catalysts addressing critical cost and durability barriers that have historically limited commercial adoption. Platinum-based SACs have reduced catalyst loading requirements by up to 70% while maintaining or improving performance metrics, creating significant cost advantages for manufacturers and accelerating market penetration in transportation and stationary power applications.
Electrolyzers represent another high-growth segment, with the green hydrogen production market expected to expand at over 50% annually through 2028. Single-atom catalysts have demonstrated hydrogen evolution reaction (HER) efficiencies approaching theoretical limits, substantially reducing energy input requirements and operational costs. This performance enhancement is creating new market opportunities in industrial decarbonization efforts where economic viability was previously questionable.
Battery technologies incorporating SAC innovations are witnessing rapid commercial adoption, particularly in the electric vehicle sector. Enhanced electrode performance through single-atom catalysis has enabled faster charging rates, increased energy density, and extended cycle life—addressing key consumer concerns that have limited EV market penetration. Premium automotive manufacturers have begun incorporating SAC-enhanced battery systems in flagship models, establishing a premium market segment with higher profit margins.
Regional market analysis reveals Asia-Pacific dominance in manufacturing capacity, with China accounting for over 60% of global production capability for SAC-enhanced electrochemical components. However, North American and European markets are rapidly expanding research investments, particularly in specialized applications for aerospace, defense, and medical devices where performance requirements justify premium pricing.
Consumer electronics represents an emerging application area with substantial growth potential, as miniaturized electrochemical systems benefit significantly from the efficiency improvements offered by single-atom catalysts. Wearable technology manufacturers have demonstrated particular interest in SAC-enhanced microbatteries and supercapacitors that enable extended device operation without increasing form factors.
Current Status and Challenges in Single-Atom Catalysis
Single-atom catalysis (SAC) has emerged as a frontier research area in heterogeneous catalysis, offering unprecedented atom efficiency and unique catalytic properties. Currently, the field has progressed from conceptual development to practical applications, particularly in electrochemical systems. Research institutions across North America, Europe, and East Asia have established specialized laboratories dedicated to SAC development, with China and the United States leading publication output in this domain.
The synthesis of stable single-atom catalysts remains a significant challenge. Traditional methods such as wet impregnation and co-precipitation often result in metal atom aggregation during thermal treatments. Advanced techniques including atomic layer deposition (ALD) and metal-organic framework (MOF) derivation have shown promise but face scalability limitations. The metal loading in most SACs typically remains below 5 wt%, restricting their practical application in industrial settings.
Characterization presents another major hurdle. While aberration-corrected scanning transmission electron microscopy (AC-STEM) and X-ray absorption spectroscopy (XAS) have become standard tools for confirming the atomic dispersion and coordination environment of single atoms, in-situ and operando characterization under electrochemical conditions remains technically demanding. This gap limits our understanding of the dynamic structural changes and reaction mechanisms during electrochemical processes.
From a fundamental perspective, the electronic structure and catalytic mechanisms of single-atom sites are not fully understood. Density functional theory (DFT) calculations suggest that the coordination environment significantly influences the d-band center of metal atoms, thereby affecting adsorption energies and activation barriers. However, experimental validation of these theoretical predictions remains challenging, particularly under realistic electrochemical conditions.
Stability issues persist in electrochemical applications. Single metal atoms, particularly when anchored on carbon supports, can detach or aggregate during long-term operation due to potential cycling and electrolyte effects. Recent studies have shown that nitrogen-doped carbon materials provide stronger metal-support interactions, but the stability beyond 5,000 cycles remains problematic for many systems.
The scalable production of SACs represents perhaps the most significant barrier to commercialization. Current laboratory-scale synthesis methods typically produce milligram quantities, whereas industrial applications would require kilogram to ton scale production. The high cost of noble metals (Pt, Pd, Ir) commonly used in SACs further complicates economic viability, driving research toward earth-abundant alternatives like Fe, Co, and Ni, which present their own challenges in activity and stability.
Standardization of testing protocols and performance metrics is lacking across the field, making direct comparisons between different SAC systems difficult and hindering systematic progress toward practical applications in electrochemical energy conversion and storage technologies.
The synthesis of stable single-atom catalysts remains a significant challenge. Traditional methods such as wet impregnation and co-precipitation often result in metal atom aggregation during thermal treatments. Advanced techniques including atomic layer deposition (ALD) and metal-organic framework (MOF) derivation have shown promise but face scalability limitations. The metal loading in most SACs typically remains below 5 wt%, restricting their practical application in industrial settings.
Characterization presents another major hurdle. While aberration-corrected scanning transmission electron microscopy (AC-STEM) and X-ray absorption spectroscopy (XAS) have become standard tools for confirming the atomic dispersion and coordination environment of single atoms, in-situ and operando characterization under electrochemical conditions remains technically demanding. This gap limits our understanding of the dynamic structural changes and reaction mechanisms during electrochemical processes.
From a fundamental perspective, the electronic structure and catalytic mechanisms of single-atom sites are not fully understood. Density functional theory (DFT) calculations suggest that the coordination environment significantly influences the d-band center of metal atoms, thereby affecting adsorption energies and activation barriers. However, experimental validation of these theoretical predictions remains challenging, particularly under realistic electrochemical conditions.
Stability issues persist in electrochemical applications. Single metal atoms, particularly when anchored on carbon supports, can detach or aggregate during long-term operation due to potential cycling and electrolyte effects. Recent studies have shown that nitrogen-doped carbon materials provide stronger metal-support interactions, but the stability beyond 5,000 cycles remains problematic for many systems.
The scalable production of SACs represents perhaps the most significant barrier to commercialization. Current laboratory-scale synthesis methods typically produce milligram quantities, whereas industrial applications would require kilogram to ton scale production. The high cost of noble metals (Pt, Pd, Ir) commonly used in SACs further complicates economic viability, driving research toward earth-abundant alternatives like Fe, Co, and Ni, which present their own challenges in activity and stability.
Standardization of testing protocols and performance metrics is lacking across the field, making direct comparisons between different SAC systems difficult and hindering systematic progress toward practical applications in electrochemical energy conversion and storage technologies.
Current Single-Atom Catalyst Design Strategies
01 Metal-based single-atom catalysts for electrochemical applications
Metal-based single-atom catalysts have emerged as promising materials for various electrochemical applications due to their maximized atom utilization and unique electronic properties. These catalysts feature isolated metal atoms anchored on support materials, offering enhanced catalytic activity and selectivity compared to traditional nanoparticle catalysts. The atomically dispersed active sites provide optimal exposure to reactants, leading to improved electrochemical performance in applications such as fuel cells, water splitting, and CO2 reduction.- Metal-based single-atom catalysts for electrochemical applications: Metal-based single-atom catalysts (SACs) have emerged as promising materials for electrochemical applications due to their maximized atom utilization efficiency and unique electronic properties. These catalysts feature isolated metal atoms anchored on various supports, offering enhanced catalytic activity and selectivity compared to traditional nanoparticle catalysts. The electronic structure of the metal atoms can be tuned by the surrounding environment, leading to optimized adsorption energies for reactants and improved reaction kinetics in electrochemical processes.
- Carbon-supported single-atom catalysts for energy conversion: Carbon-based materials serve as excellent supports for single-atom catalysts in electrochemical energy conversion applications. These supports include graphene, carbon nanotubes, porous carbon, and carbon nitride materials, which provide high surface area and good electrical conductivity. The strong interaction between metal atoms and carbon supports prevents aggregation during electrochemical reactions, maintaining the single-atom nature of the catalyst. These catalysts demonstrate superior performance in reactions such as hydrogen evolution, oxygen reduction, and CO2 reduction, making them valuable for fuel cells and electrolyzers.
- Single-atom catalysts for water splitting and hydrogen production: Single-atom catalysts have shown exceptional performance in electrochemical water splitting for hydrogen production. By precisely controlling the coordination environment of single metal atoms, these catalysts can achieve optimal binding energies for hydrogen and oxygen intermediates, enhancing both the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER). The atomically dispersed active sites provide uniform catalytic centers with maximized exposure to reactants, resulting in higher intrinsic activity and improved stability compared to conventional catalysts, while requiring significantly lower amounts of precious metals.
- Novel synthesis methods for single-atom catalysts: Advanced synthesis methods have been developed to prepare single-atom catalysts with high metal loading and uniform distribution. These techniques include atomic layer deposition, wet chemistry approaches, high-temperature atom trapping, electrochemical deposition, and photochemical reduction. The synthesis strategies focus on creating strong metal-support interactions to prevent aggregation during both synthesis and electrochemical operation. Recent innovations have enabled the preparation of single-atom catalysts with loadings exceeding traditional limits, making them more practical for large-scale applications while maintaining their unique electrochemical performance advantages.
- Characterization and performance enhancement of single-atom catalysts: Advanced characterization techniques and performance enhancement strategies are crucial for developing high-performance single-atom catalysts. Techniques such as aberration-corrected electron microscopy, X-ray absorption spectroscopy, and scanning tunneling microscopy enable precise identification of atomic structures and coordination environments. Performance enhancement strategies include electronic structure modulation through support selection, introduction of defects, heteroatom doping, and creation of dual-atom or cluster sites. These approaches have led to significant improvements in activity, selectivity, and stability of single-atom catalysts for various electrochemical reactions, pushing performance metrics closer to commercial viability.
02 Carbon-supported single-atom catalysts for energy conversion
Carbon-based materials serve as excellent supports for single-atom catalysts in electrochemical energy conversion processes. These supports, including graphene, carbon nanotubes, and porous carbon structures, provide high surface area, good electrical conductivity, and strong metal-support interactions that stabilize the isolated metal atoms. The resulting catalysts demonstrate enhanced electrochemical performance in oxygen reduction reactions, hydrogen evolution reactions, and other energy conversion processes, making them valuable for sustainable energy technologies.Expand Specific Solutions03 Single-atom catalysts for electrocatalytic water splitting
Single-atom catalysts have shown exceptional performance in electrocatalytic water splitting reactions, including both hydrogen evolution reaction (HER) and oxygen evolution reaction (OER). The atomically dispersed active sites offer optimized adsorption energies for reaction intermediates, leading to lower overpotentials and higher current densities. These catalysts can be designed with specific coordination environments to enhance their stability and activity under the harsh conditions of water electrolysis, contributing to more efficient hydrogen production technologies.Expand Specific Solutions04 Novel synthesis methods for single-atom catalysts
Advanced synthesis strategies have been developed to prepare single-atom catalysts with controlled loading, uniform distribution, and strong metal-support interactions. These methods include atomic layer deposition, wet chemistry approaches, high-temperature atom trapping, and electrochemical deposition techniques. The precise control over the synthesis process enables the creation of catalysts with optimized coordination environments and electronic structures, leading to enhanced electrochemical performance and stability during long-term operation.Expand Specific Solutions05 Characterization and performance evaluation of single-atom catalysts
Advanced characterization techniques and performance evaluation methods are crucial for understanding the structure-property relationships in single-atom catalysts. Techniques such as X-ray absorption spectroscopy, aberration-corrected electron microscopy, and in-situ/operando spectroscopy provide insights into the atomic structure, oxidation state, and coordination environment of the catalytic sites. Electrochemical performance is evaluated through various metrics including onset potential, Tafel slope, exchange current density, and stability tests, allowing for systematic improvement of catalyst design for specific applications.Expand Specific Solutions
Key Industry Players and Research Institutions
Single-atom catalysis for enhanced electrochemical performance is emerging as a transformative technology in the early commercialization phase. The market is projected to grow significantly, driven by increasing demand for efficient energy conversion and storage solutions. Leading research institutions like KIST, Dalian Institute of Chemical Physics, and Tsinghua University are advancing fundamental understanding, while companies such as SK Innovation and Beijing Photosynthetic Hydrogen Energy Technology are working toward commercial applications. The technology is approaching maturity with key players from South Korea, China, and the US collaborating across academia and industry to overcome challenges in catalyst stability and scalable production methods, positioning single-atom catalysis as a critical enabler for next-generation electrochemical systems.
Dalian Institute of Chemical Physics of CAS
Technical Solution: Dalian Institute of Chemical Physics (DICP) has pioneered single-atom catalysis (SAC) for electrochemical applications, developing atomically dispersed metal catalysts on various supports. Their innovative approach involves anchoring isolated metal atoms (primarily Pt, Pd, Fe, and Co) on nitrogen-doped carbon materials to create M-N-C structures with maximized atom utilization. DICP researchers have demonstrated remarkable activity for oxygen reduction reaction (ORR), hydrogen evolution reaction (HER), and CO2 reduction, achieving near 100% atom efficiency compared to conventional nanoparticle catalysts. Their proprietary synthesis methods include atomic layer deposition and high-temperature pyrolysis techniques that ensure strong metal-support interactions, preventing aggregation during electrochemical cycling. Recent breakthroughs include developing SACs with oxygen vacancy-rich supports that demonstrate exceptional stability for over 10,000 cycles in fuel cell testing environments.
Strengths: Maximized atomic efficiency (nearly 100% atom utilization) significantly reduces precious metal loading while maintaining high catalytic activity. Their synthesis methods create exceptionally stable catalysts with strong metal-support interactions. Weaknesses: Scale-up challenges remain for industrial production, and some of their advanced synthesis methods require specialized equipment and precise control conditions that may limit commercial viability.
University of Science & Technology of China
Technical Solution: The University of Science & Technology of China (USTC) has developed innovative single-atom catalysts (SACs) specifically engineered for electrochemical applications. Their research team has pioneered a defect-engineering approach to create highly active single-atom sites on various supports. USTC's proprietary synthesis method involves creating controlled defects in graphene and carbon nitride materials that serve as anchoring points for isolated metal atoms, preventing aggregation during electrochemical processes. Their most advanced catalysts feature atomically dispersed transition metals (Fe, Co, Ni) coordinated with nitrogen in specific configurations that optimize the electronic structure for target reactions. These catalysts have demonstrated exceptional performance for oxygen evolution reaction (OER) and nitrogen reduction reaction (NRR), achieving current densities comparable to precious metal catalysts but at significantly reduced costs. USTC researchers have further enhanced performance by developing dual-functional SACs that can simultaneously catalyze multiple reactions, making them particularly valuable for integrated electrochemical systems like unitized regenerative fuel cells.
Strengths: Their defect-engineering approach creates highly stable single-atom sites with tunable electronic properties that can be optimized for specific reactions. The dual-functional capability allows for simplified system design in complex electrochemical applications. Weaknesses: The creation of precisely controlled defects requires sophisticated synthesis techniques that may be challenging to scale up, and some of their most effective catalysts still rely on specialized support materials with limited commercial availability.
Critical Patents and Breakthroughs in SAC Technology
Single-atom catalyst and method of preparing same
PatentPendingEP4550480A2
Innovation
- A single-atom catalyst (SAC) is developed, comprising a nitrogen-doped carbon structure and a single-atom metal, such as cobalt, that forms a coordination bond with nitrogen atoms. This catalyst extends the optimal pH range and prevents hydroxyl radical adsorption to electrodes.
Single-atom catalyst and method for forming same
PatentWO2020141936A1
Innovation
- A single atom catalyst is developed, comprising a support with a first metal oxide and a second metal atom, formed by creating sacrificial nanoparticles, coating them with the first metal oxide, adsorbing the second metal atom, and heating to spatially confine the metal atoms within the oxide, allowing for improved catalytic and photocatalytic performance.
Sustainability Impact of Single-Atom Catalysis
Single-atom catalysis (SAC) represents a significant advancement in sustainable technologies, offering profound environmental benefits across multiple dimensions. The atomically dispersed nature of active metal sites in SACs dramatically increases atom utilization efficiency to nearly 100%, compared to traditional catalysts where only surface atoms participate in reactions. This exceptional efficiency translates directly to reduced metal consumption, addressing critical concerns regarding the depletion of precious and rare earth metals.
The environmental footprint of SAC technology extends beyond resource conservation. These catalysts demonstrate remarkable performance in environmental remediation applications, particularly in water purification processes where they effectively degrade persistent organic pollutants and remove heavy metal contaminants. Their high activity at ambient temperatures reduces energy requirements, contributing to lower carbon emissions in industrial processes.
In the energy sector, SACs are revolutionizing electrochemical systems by enabling more efficient hydrogen evolution reactions, oxygen reduction reactions, and CO2 conversion. These capabilities directly support the transition to renewable energy systems by improving the efficiency of fuel cells, electrolyzers, and energy storage technologies. The enhanced selectivity of SACs also minimizes unwanted by-products, reducing waste generation in chemical manufacturing processes.
Life cycle assessments of SAC-based technologies reveal significant reductions in environmental impact compared to conventional catalytic systems. The extended catalyst lifetime and stability of many SAC formulations reduce replacement frequency and associated manufacturing emissions. Furthermore, their high activity under mild conditions decreases the energy intensity of various industrial processes, contributing to overall carbon footprint reduction.
Economic sustainability benefits accompany these environmental advantages. The reduced metal loading in SACs offers cost advantages despite their sophisticated preparation methods, particularly as manufacturing scales increase and techniques mature. Industries adopting SAC technologies can potentially achieve compliance with increasingly stringent environmental regulations while maintaining economic competitiveness.
Looking forward, the integration of SACs into circular economy frameworks presents promising opportunities. Research into recovery and regeneration methods for spent SACs could further enhance their sustainability profile by enabling metal recycling and reuse. As manufacturing processes continue to evolve, the environmental impact of SAC production itself is expected to decrease, creating a more holistically sustainable technology platform for next-generation electrochemical applications.
The environmental footprint of SAC technology extends beyond resource conservation. These catalysts demonstrate remarkable performance in environmental remediation applications, particularly in water purification processes where they effectively degrade persistent organic pollutants and remove heavy metal contaminants. Their high activity at ambient temperatures reduces energy requirements, contributing to lower carbon emissions in industrial processes.
In the energy sector, SACs are revolutionizing electrochemical systems by enabling more efficient hydrogen evolution reactions, oxygen reduction reactions, and CO2 conversion. These capabilities directly support the transition to renewable energy systems by improving the efficiency of fuel cells, electrolyzers, and energy storage technologies. The enhanced selectivity of SACs also minimizes unwanted by-products, reducing waste generation in chemical manufacturing processes.
Life cycle assessments of SAC-based technologies reveal significant reductions in environmental impact compared to conventional catalytic systems. The extended catalyst lifetime and stability of many SAC formulations reduce replacement frequency and associated manufacturing emissions. Furthermore, their high activity under mild conditions decreases the energy intensity of various industrial processes, contributing to overall carbon footprint reduction.
Economic sustainability benefits accompany these environmental advantages. The reduced metal loading in SACs offers cost advantages despite their sophisticated preparation methods, particularly as manufacturing scales increase and techniques mature. Industries adopting SAC technologies can potentially achieve compliance with increasingly stringent environmental regulations while maintaining economic competitiveness.
Looking forward, the integration of SACs into circular economy frameworks presents promising opportunities. Research into recovery and regeneration methods for spent SACs could further enhance their sustainability profile by enabling metal recycling and reuse. As manufacturing processes continue to evolve, the environmental impact of SAC production itself is expected to decrease, creating a more holistically sustainable technology platform for next-generation electrochemical applications.
Scale-up and Commercialization Roadmap
The commercialization of single-atom catalysis (SAC) technology for electrochemical applications requires a systematic approach to overcome the transition from laboratory-scale research to industrial production. Currently, most SAC synthesis methods are limited to small-scale production, typically yielding milligram quantities suitable only for academic research. The primary challenge lies in maintaining atomic dispersion during scale-up while ensuring cost-effectiveness and reproducibility.
A phased commercialization roadmap begins with process optimization focusing on continuous flow synthesis methods that can maintain precise control over atomic dispersion while increasing production volume. Microfluidic reactors and continuous precipitation techniques show particular promise for maintaining uniform catalyst properties at larger scales. These approaches must be coupled with in-line quality control systems utilizing advanced characterization techniques such as in-situ XAS or operando spectroscopy.
The second phase involves pilot production, targeting gram to kilogram scale manufacturing while maintaining performance metrics comparable to laboratory samples. This phase requires significant investment in specialized equipment and process validation protocols. Industry partnerships with established catalyst manufacturers can accelerate this transition by leveraging existing production infrastructure and quality management systems.
Economic viability represents a critical milestone, with current production costs estimated at $5,000-10,000 per gram for high-quality SACs. Cost reduction strategies must focus on precursor selection, recycling of noble metals, and process intensification. The target price point for commercial viability in high-value applications such as fuel cells and electrolyzers is approximately $500-1,000 per gram, requiring a 10-fold reduction in production costs.
Regulatory considerations and standardization will become increasingly important as production scales increase. Developing industry standards for SAC characterization, performance testing, and safety protocols will facilitate market acceptance. Collaboration with standards organizations such as ISO and ASTM to establish testing protocols specific to single-atom catalysts will be essential.
Market entry strategy should initially target high-value, low-volume applications where performance advantages outweigh cost considerations. Premium segments in electrocatalysis for green hydrogen production, pharmaceutical synthesis, and specialized sensing applications represent ideal entry points. As production scales and costs decrease, expansion into broader markets such as automotive catalytic converters and industrial chemical processing becomes feasible, potentially reaching commercial maturity within 5-7 years.
A phased commercialization roadmap begins with process optimization focusing on continuous flow synthesis methods that can maintain precise control over atomic dispersion while increasing production volume. Microfluidic reactors and continuous precipitation techniques show particular promise for maintaining uniform catalyst properties at larger scales. These approaches must be coupled with in-line quality control systems utilizing advanced characterization techniques such as in-situ XAS or operando spectroscopy.
The second phase involves pilot production, targeting gram to kilogram scale manufacturing while maintaining performance metrics comparable to laboratory samples. This phase requires significant investment in specialized equipment and process validation protocols. Industry partnerships with established catalyst manufacturers can accelerate this transition by leveraging existing production infrastructure and quality management systems.
Economic viability represents a critical milestone, with current production costs estimated at $5,000-10,000 per gram for high-quality SACs. Cost reduction strategies must focus on precursor selection, recycling of noble metals, and process intensification. The target price point for commercial viability in high-value applications such as fuel cells and electrolyzers is approximately $500-1,000 per gram, requiring a 10-fold reduction in production costs.
Regulatory considerations and standardization will become increasingly important as production scales increase. Developing industry standards for SAC characterization, performance testing, and safety protocols will facilitate market acceptance. Collaboration with standards organizations such as ISO and ASTM to establish testing protocols specific to single-atom catalysts will be essential.
Market entry strategy should initially target high-value, low-volume applications where performance advantages outweigh cost considerations. Premium segments in electrocatalysis for green hydrogen production, pharmaceutical synthesis, and specialized sensing applications represent ideal entry points. As production scales and costs decrease, expansion into broader markets such as automotive catalytic converters and industrial chemical processing becomes feasible, potentially reaching commercial maturity within 5-7 years.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!