Analyzing Electrode Kinetics in Plasmonic Biosensor Prototypes
SEP 29, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Plasmonic Biosensor Evolution and Research Objectives
Plasmonic biosensors have evolved significantly over the past three decades, transforming from rudimentary optical sensing devices to sophisticated nanoscale detection systems. The foundational work began in the early 1990s with the development of surface plasmon resonance (SPR) technology, which exploited the interaction between electromagnetic waves and conductive surfaces to detect biomolecular binding events. This initial breakthrough established the fundamental principles that continue to underpin modern plasmonic biosensing.
The mid-2000s marked a pivotal shift with the integration of nanostructures, particularly gold and silver nanoparticles, which dramatically enhanced sensitivity through localized surface plasmon resonance (LSPR) effects. This advancement reduced detection limits from micromolar to nanomolar concentrations, expanding potential applications across medical diagnostics, environmental monitoring, and food safety testing.
Recent developments have focused on electrode kinetics optimization, a critical factor in determining biosensor performance. Understanding the electron transfer processes at the electrode-analyte interface has become essential for improving response time, sensitivity, and specificity. The interplay between plasmonic effects and electrochemical reactions represents a frontier in biosensor research, with significant implications for real-time monitoring capabilities.
Current research objectives center on several key areas. First, enhancing electrode kinetics through novel materials and surface modifications to improve charge transfer efficiency and reduce interference. Second, developing multi-modal sensing platforms that combine plasmonic and electrochemical detection methods for comprehensive analyte characterization. Third, miniaturization and integration with microfluidic systems to enable point-of-care applications with minimal sample volumes.
The field is trending toward smart biosensing systems that incorporate machine learning algorithms for signal processing and interpretation, potentially enabling predictive capabilities beyond simple detection. Additionally, there is growing interest in sustainable materials and manufacturing processes to reduce environmental impact while maintaining performance standards.
Our technical goals include developing prototype plasmonic biosensors with optimized electrode kinetics capable of detecting biomarkers at femtomolar concentrations, with response times under 60 seconds. We aim to characterize the fundamental mechanisms governing electron transfer at plasmonic interfaces and establish design principles for next-generation biosensors with enhanced stability and reproducibility under varied environmental conditions.
The convergence of nanotechnology, materials science, and electrochemistry presents unprecedented opportunities for plasmonic biosensor advancement. By systematically investigating electrode kinetics in these systems, we position ourselves at the forefront of developing highly sensitive, specific, and rapid detection platforms with transformative potential across multiple industries.
The mid-2000s marked a pivotal shift with the integration of nanostructures, particularly gold and silver nanoparticles, which dramatically enhanced sensitivity through localized surface plasmon resonance (LSPR) effects. This advancement reduced detection limits from micromolar to nanomolar concentrations, expanding potential applications across medical diagnostics, environmental monitoring, and food safety testing.
Recent developments have focused on electrode kinetics optimization, a critical factor in determining biosensor performance. Understanding the electron transfer processes at the electrode-analyte interface has become essential for improving response time, sensitivity, and specificity. The interplay between plasmonic effects and electrochemical reactions represents a frontier in biosensor research, with significant implications for real-time monitoring capabilities.
Current research objectives center on several key areas. First, enhancing electrode kinetics through novel materials and surface modifications to improve charge transfer efficiency and reduce interference. Second, developing multi-modal sensing platforms that combine plasmonic and electrochemical detection methods for comprehensive analyte characterization. Third, miniaturization and integration with microfluidic systems to enable point-of-care applications with minimal sample volumes.
The field is trending toward smart biosensing systems that incorporate machine learning algorithms for signal processing and interpretation, potentially enabling predictive capabilities beyond simple detection. Additionally, there is growing interest in sustainable materials and manufacturing processes to reduce environmental impact while maintaining performance standards.
Our technical goals include developing prototype plasmonic biosensors with optimized electrode kinetics capable of detecting biomarkers at femtomolar concentrations, with response times under 60 seconds. We aim to characterize the fundamental mechanisms governing electron transfer at plasmonic interfaces and establish design principles for next-generation biosensors with enhanced stability and reproducibility under varied environmental conditions.
The convergence of nanotechnology, materials science, and electrochemistry presents unprecedented opportunities for plasmonic biosensor advancement. By systematically investigating electrode kinetics in these systems, we position ourselves at the forefront of developing highly sensitive, specific, and rapid detection platforms with transformative potential across multiple industries.
Market Analysis for Advanced Biosensing Technologies
The global biosensor market is experiencing unprecedented growth, driven by increasing healthcare needs, technological advancements, and the rising prevalence of chronic diseases. Currently valued at approximately $25 billion, the market is projected to reach $36 billion by 2026, with a compound annual growth rate of 7.5%. Plasmonic biosensors, particularly those utilizing advanced electrode kinetics, represent one of the fastest-growing segments within this market.
Healthcare applications dominate the biosensor market, accounting for nearly 65% of total demand. Within this sector, point-of-care testing and continuous monitoring devices are experiencing the highest growth rates due to the global shift toward personalized medicine and remote patient monitoring. The COVID-19 pandemic has further accelerated this trend, creating a surge in demand for rapid, accurate diagnostic tools that can be deployed in various settings.
The pharmaceutical and biotechnology research segment represents the second-largest market for advanced biosensing technologies, with approximately 20% market share. These industries increasingly rely on high-sensitivity plasmonic biosensors for drug discovery, protein interaction studies, and biomarker detection. The enhanced electrode kinetics in modern plasmonic biosensor prototypes offers significant advantages in terms of detection limits and response times, making them particularly valuable for research applications.
Environmental monitoring and food safety testing collectively account for about 15% of the biosensor market. These sectors are showing growing interest in plasmonic biosensing technologies due to their ability to detect contaminants at extremely low concentrations. Regulatory pressures and consumer demand for safer products are driving adoption in these areas, with annual growth rates exceeding 9%.
Regional analysis reveals that North America currently leads the biosensor market with approximately 40% share, followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is expected to show the highest growth rate over the next five years, driven by increasing healthcare expenditure, growing awareness about preventive healthcare, and substantial government investments in biotechnology infrastructure.
Key customer segments for plasmonic biosensors with advanced electrode kinetics include clinical laboratories, research institutions, pharmaceutical companies, and increasingly, primary healthcare providers. The latter represents an emerging market as miniaturization and cost reduction make these technologies more accessible for routine clinical use. Customer feedback indicates that sensitivity, reliability, and ease of integration with existing systems are the primary factors influencing purchasing decisions.
Healthcare applications dominate the biosensor market, accounting for nearly 65% of total demand. Within this sector, point-of-care testing and continuous monitoring devices are experiencing the highest growth rates due to the global shift toward personalized medicine and remote patient monitoring. The COVID-19 pandemic has further accelerated this trend, creating a surge in demand for rapid, accurate diagnostic tools that can be deployed in various settings.
The pharmaceutical and biotechnology research segment represents the second-largest market for advanced biosensing technologies, with approximately 20% market share. These industries increasingly rely on high-sensitivity plasmonic biosensors for drug discovery, protein interaction studies, and biomarker detection. The enhanced electrode kinetics in modern plasmonic biosensor prototypes offers significant advantages in terms of detection limits and response times, making them particularly valuable for research applications.
Environmental monitoring and food safety testing collectively account for about 15% of the biosensor market. These sectors are showing growing interest in plasmonic biosensing technologies due to their ability to detect contaminants at extremely low concentrations. Regulatory pressures and consumer demand for safer products are driving adoption in these areas, with annual growth rates exceeding 9%.
Regional analysis reveals that North America currently leads the biosensor market with approximately 40% share, followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is expected to show the highest growth rate over the next five years, driven by increasing healthcare expenditure, growing awareness about preventive healthcare, and substantial government investments in biotechnology infrastructure.
Key customer segments for plasmonic biosensors with advanced electrode kinetics include clinical laboratories, research institutions, pharmaceutical companies, and increasingly, primary healthcare providers. The latter represents an emerging market as miniaturization and cost reduction make these technologies more accessible for routine clinical use. Customer feedback indicates that sensitivity, reliability, and ease of integration with existing systems are the primary factors influencing purchasing decisions.
Electrode Kinetics Challenges in Plasmonic Biosensors
The development of plasmonic biosensors has been significantly hindered by several electrode kinetics challenges that affect their performance, reliability, and commercial viability. These challenges stem from the complex interplay between the electrode surface, the biological recognition elements, and the target analytes in the sensing environment.
One of the primary challenges is the electron transfer rate limitation at the electrode-solution interface. The kinetics of electron transfer processes are often slower than desired, leading to reduced sensitivity and increased response times. This is particularly problematic in real-time monitoring applications where rapid detection is essential, such as point-of-care diagnostics and environmental monitoring systems.
Surface fouling represents another significant obstacle, as biomolecules and other contaminants can adsorb non-specifically onto electrode surfaces, gradually degrading the electrode performance over time. This fouling effect alters the electrode kinetics by creating physical barriers that impede electron transfer and change the electrochemical properties of the interface.
The stability of the electrode-biomolecule interface poses additional challenges. Many biological recognition elements (antibodies, enzymes, DNA) undergo conformational changes or denaturation when immobilized on electrode surfaces, affecting their binding affinity and specificity. The orientation and density of these biomolecules on the electrode surface directly impact the kinetics of the biorecognition events.
Mass transport limitations further complicate electrode kinetics in plasmonic biosensors. The diffusion of analytes to the sensing surface can become rate-limiting, especially in viscous biological media or when targeting low-concentration analytes. This creates a situation where the sensor response is controlled not by the intrinsic electrode kinetics but by mass transport phenomena.
The integration of plasmonic elements with electrochemical components introduces unique challenges related to electromagnetic field effects on electrode kinetics. The localized surface plasmon resonance (LSPR) generates intense electromagnetic fields that can alter the energetics of redox reactions at the electrode surface, creating complex kinetic behaviors that are difficult to model and predict.
Temperature fluctuations significantly impact electrode kinetics, with even small variations causing substantial changes in reaction rates according to the Arrhenius equation. This temperature dependence creates challenges for maintaining consistent performance across different operating environments and complicates the calibration of plasmonic biosensor systems.
Finally, the miniaturization of plasmonic biosensors for portable applications introduces additional electrode kinetics challenges related to increased surface-to-volume ratios, altered diffusion profiles, and enhanced surface effects. These factors collectively make the electrode kinetics in miniaturized systems behave differently from their macroscale counterparts, requiring specialized approaches for optimization.
One of the primary challenges is the electron transfer rate limitation at the electrode-solution interface. The kinetics of electron transfer processes are often slower than desired, leading to reduced sensitivity and increased response times. This is particularly problematic in real-time monitoring applications where rapid detection is essential, such as point-of-care diagnostics and environmental monitoring systems.
Surface fouling represents another significant obstacle, as biomolecules and other contaminants can adsorb non-specifically onto electrode surfaces, gradually degrading the electrode performance over time. This fouling effect alters the electrode kinetics by creating physical barriers that impede electron transfer and change the electrochemical properties of the interface.
The stability of the electrode-biomolecule interface poses additional challenges. Many biological recognition elements (antibodies, enzymes, DNA) undergo conformational changes or denaturation when immobilized on electrode surfaces, affecting their binding affinity and specificity. The orientation and density of these biomolecules on the electrode surface directly impact the kinetics of the biorecognition events.
Mass transport limitations further complicate electrode kinetics in plasmonic biosensors. The diffusion of analytes to the sensing surface can become rate-limiting, especially in viscous biological media or when targeting low-concentration analytes. This creates a situation where the sensor response is controlled not by the intrinsic electrode kinetics but by mass transport phenomena.
The integration of plasmonic elements with electrochemical components introduces unique challenges related to electromagnetic field effects on electrode kinetics. The localized surface plasmon resonance (LSPR) generates intense electromagnetic fields that can alter the energetics of redox reactions at the electrode surface, creating complex kinetic behaviors that are difficult to model and predict.
Temperature fluctuations significantly impact electrode kinetics, with even small variations causing substantial changes in reaction rates according to the Arrhenius equation. This temperature dependence creates challenges for maintaining consistent performance across different operating environments and complicates the calibration of plasmonic biosensor systems.
Finally, the miniaturization of plasmonic biosensors for portable applications introduces additional electrode kinetics challenges related to increased surface-to-volume ratios, altered diffusion profiles, and enhanced surface effects. These factors collectively make the electrode kinetics in miniaturized systems behave differently from their macroscale counterparts, requiring specialized approaches for optimization.
Current Electrode Kinetics Optimization Approaches
01 Surface plasmon resonance (SPR) biosensor designs
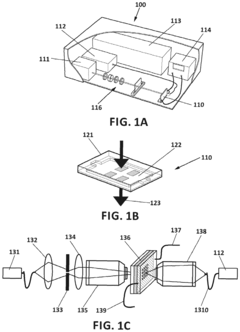
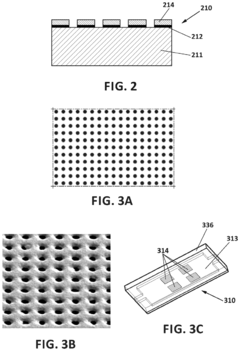
Surface plasmon resonance technology forms the basis for many advanced biosensor prototypes. These designs utilize the interaction between electromagnetic waves and conductive surfaces to detect biomolecular interactions with high sensitivity. Various configurations include prism-coupled systems, waveguide structures, and nanostructured surfaces that enhance the plasmonic effect. These designs optimize the electrode-analyte interface to improve detection limits and response times for biological and chemical sensing applications.- Plasmonic biosensor design and fabrication: Plasmonic biosensors utilize surface plasmon resonance (SPR) phenomena for highly sensitive detection of biomolecules. These prototypes incorporate specialized electrode designs and nanomaterials to enhance sensitivity. The fabrication process typically involves deposition of noble metal films (gold or silver) on substrates, followed by surface functionalization for specific target binding. Advanced manufacturing techniques ensure precise control over the sensor architecture, which is critical for optimizing the plasmonic response and electrode kinetics.
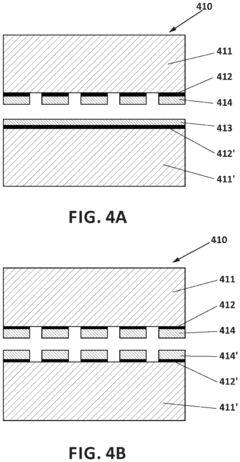
- Electrode kinetics optimization in biosensing applications: Electrode kinetics play a crucial role in determining the performance of plasmonic biosensors. Optimization involves controlling electron transfer rates at the electrode-solution interface, which affects the sensor's response time and detection limits. Various approaches include surface modification with conductive polymers, incorporation of nanomaterials to increase effective surface area, and tailoring the electrode composition to enhance charge transfer efficiency. These improvements lead to faster response times and improved sensitivity for detecting biomolecular interactions.
- Integration of plasmonic elements with electrochemical systems: The integration of plasmonic structures with electrochemical detection systems creates powerful hybrid sensing platforms. These systems combine the advantages of optical plasmonic sensing with electrochemical detection methods, allowing for multi-modal analysis of complex biological samples. The electrode design must accommodate both plasmonic field enhancement and efficient electron transfer. This integration enables simultaneous monitoring of binding events through optical signals and electrochemical reactions, providing complementary information about analyte interactions.
- Novel materials for enhanced plasmonic biosensor performance: Advanced materials are being developed to improve the performance of plasmonic biosensor electrodes. These include nanostructured metals, conductive polymers, graphene, and other 2D materials that offer superior electrical conductivity and biocompatibility. The incorporation of these materials enhances electrode kinetics by facilitating faster electron transfer and reducing interference. Additionally, composite materials combining plasmonic metals with other functional components can provide multifunctional capabilities, such as simultaneous sensing and catalysis, leading to more versatile biosensor platforms.
- Real-time monitoring and signal processing in plasmonic biosensors: Advanced signal processing techniques are essential for extracting meaningful data from plasmonic biosensor measurements. These methods involve real-time monitoring of electrode kinetics and plasmonic responses, often using integrated microelectronics and sophisticated algorithms. Techniques such as impedance spectroscopy, cyclic voltammetry, and differential pulse analysis can be combined with optical measurements to enhance detection sensitivity and specificity. The integration of these analytical approaches with machine learning algorithms enables more accurate interpretation of complex biosensor signals and reduces false positives in diagnostic applications.
02 Electrode materials and surface modifications for enhanced kinetics
The choice of electrode materials and surface modifications significantly impacts biosensor performance. Noble metals like gold and silver are commonly used for their excellent plasmonic properties, while surface modifications with nanomaterials or functional groups can enhance electrode kinetics. These modifications improve electron transfer rates, reduce interference, and increase specificity for target analytes. Techniques such as electrodeposition, self-assembled monolayers, and nanostructuring are employed to optimize the electrode-electrolyte interface.Expand Specific Solutions03 Integration of microfluidics with plasmonic biosensors
Microfluidic systems integrated with plasmonic biosensors enable precise control over sample delivery and reaction conditions. These systems facilitate rapid mixing, reduced sample volumes, and controlled flow rates that enhance electrode kinetics and improve detection sensitivity. Lab-on-chip designs incorporate multiple sensing elements, reference electrodes, and sample preparation steps on a single platform, making them suitable for point-of-care diagnostics and high-throughput screening applications.Expand Specific Solutions04 Signal processing and data analysis methods
Advanced signal processing techniques are crucial for extracting meaningful information from plasmonic biosensor responses. These methods include real-time data filtering, baseline correction, and multivariate analysis to improve signal-to-noise ratios and detection limits. Machine learning algorithms can be applied to analyze complex electrode kinetics patterns and distinguish specific binding events from non-specific interactions. Digital signal processing techniques help compensate for environmental variations and enhance the reproducibility of biosensor measurements.Expand Specific Solutions05 Multiplexed detection systems and array configurations
Multiplexed plasmonic biosensor systems enable simultaneous detection of multiple analytes, significantly increasing throughput and efficiency. These systems utilize electrode arrays with different recognition elements or spatially separated sensing regions. Advanced electrode configurations incorporate reference and counter electrodes to improve measurement accuracy and stability. Imaging-based detection methods allow for spatial mapping of binding events across the sensor surface, providing additional information about electrode kinetics and binding dynamics.Expand Specific Solutions
Leading Research Groups and Commercial Entities
The plasmonic biosensor electrode kinetics market is in a growth phase, characterized by increasing research intensity and technological advancements. The competitive landscape features established healthcare giants like Roche Diagnostics, PHC Holdings, and F. Hoffmann-La Roche alongside academic powerhouses such as University of California, Queen Mary University of London, and Chinese Academy of Sciences. The market is experiencing cross-sector collaboration between pharmaceutical companies, medical device manufacturers, and research institutions. Technology maturity varies significantly, with companies like Roche demonstrating advanced commercial applications while university-affiliated research teams focus on prototype development. The presence of government laboratories (Naval Research Laboratory, Korea Research Institute of Standards & Science) indicates strategic importance, suggesting potential market expansion beyond current biomedical applications into broader sensing technologies.
The Regents of the University of California
Technical Solution: The University of California has developed advanced plasmonic biosensor platforms that utilize nanostructured electrodes with precisely controlled surface chemistry for enhanced electrode kinetics. Their approach combines surface plasmon resonance (SPR) with electrochemical techniques to create dual-mode sensing capabilities. The research teams have pioneered the use of gold nanoparticle arrays with optimized morphologies that significantly enhance the local electromagnetic field, improving sensitivity by up to two orders of magnitude compared to conventional electrodes. Their electrode designs incorporate self-assembled monolayers with tailored functional groups that facilitate controlled biomolecule immobilization while maintaining optimal electron transfer rates. Recent developments include integration of microfluidic systems with these plasmonic electrodes to enable real-time monitoring of binding kinetics with minimal sample volumes.
Strengths: Exceptional sensitivity through optimized nanostructure design; multidisciplinary approach combining plasmonics, electrochemistry, and surface chemistry; strong publication record in high-impact journals. Weaknesses: Complex fabrication processes may limit scalability; higher production costs compared to conventional biosensors; requires specialized equipment for both fabrication and analysis.
Queen Mary University of London
Technical Solution: Queen Mary University of London has developed innovative plasmonic biosensor prototypes focusing on electrode kinetics optimization through nanomaterial engineering. Their research utilizes gold nanostructured electrodes with precisely controlled morphologies to enhance surface plasmon generation while maintaining efficient electron transfer. A key innovation is their development of hybrid electrodes incorporating both metallic nanostructures and conductive polymers, creating a synergistic effect that amplifies both plasmonic and electrochemical signals. Their electrode designs feature carefully engineered surface roughness at the nanoscale, which has been demonstrated to increase the effective surface area while creating plasmonic "hot spots" that significantly enhance local electromagnetic fields. Recent publications have shown their sensors achieving detection limits in the femtomolar range for various biomarkers, with exceptional reproducibility and stability over extended periods.
Strengths: Innovative hybrid material approach combining plasmonic and electrochemical properties; excellent sensitivity and reproducibility; strong focus on practical clinical applications. Weaknesses: Some designs require complex nanofabrication techniques that may challenge mass production; potential long-term stability issues in complex biological matrices.
Breakthrough Patents in Plasmonic Biosensor Electrodes
Sensing devices and techniques using 3-d arrays based on surface plasmon excitations
PatentInactiveUS20120105853A1
Innovation
- A 3-dimensional composite metallodielectric nanoresonant array is developed, featuring a substrate with a dielectric material and a metallic film forming nanoholes, which supports both propagating surface plasmon polariton waves and localized surface plasmon resonant modes, enhancing the electric field through oblique metal deposition and holographic lithography, allowing for improved sensing capabilities.
Device and method for detecting biomarkers
PatentInactiveEP3121587A1
Innovation
- A biosensor system utilizing localized surface plasmon resonance (LSPR) with nanostructured surfaces that change refractive index in response to biomarkers, allowing for sensitive and specific detection of CTCs and miRs without the need for specialized personnel, using biofunctionalized nanostructures and a microfluidic cell for sample interaction.
Nanofabrication Techniques for Enhanced Electrode Performance
Nanofabrication techniques play a pivotal role in developing high-performance electrodes for plasmonic biosensor prototypes. The evolution of these techniques has significantly enhanced electrode kinetics, enabling more sensitive and reliable biosensing applications. Traditional lithography methods have been supplemented with advanced approaches that offer precise control over electrode geometry and surface properties at the nanoscale.
Electron beam lithography (EBL) represents one of the most precise fabrication methods, capable of creating electrode features below 10 nm. This technique has proven particularly valuable for plasmonic biosensors where the electrode-analyte interface critically influences detection sensitivity. The high-resolution patterning enables the creation of optimized electrode architectures that maximize surface plasmon resonance effects while maintaining excellent electrochemical properties.
Focused ion beam (FIB) milling offers complementary capabilities for electrode fabrication, allowing for direct writing and modification of electrode surfaces. This technique enables the creation of three-dimensional nanostructures that can enhance electrode kinetics through increased surface area and optimized geometry. Recent advancements in FIB technology have reduced beam damage, preserving the electrochemical properties of the fabricated electrodes.
Self-assembly techniques have emerged as cost-effective alternatives for large-scale electrode fabrication. Block copolymer lithography and colloidal lithography enable the creation of regular nanopatterns across large areas, facilitating the mass production of high-performance electrodes. These bottom-up approaches are particularly valuable for creating electrodes with controlled porosity and surface roughness, factors that significantly impact electrode kinetics in biosensing applications.
Surface modification techniques complement physical nanofabrication methods by tailoring the chemical properties of electrode surfaces. Atomic layer deposition (ALD) enables the conformal coating of electrodes with ultrathin films of functional materials, enhancing stability and electrochemical performance. Plasma treatment and chemical functionalization further modify surface energy and reactivity, optimizing electrode-analyte interactions.
Hybrid fabrication approaches combining multiple techniques have shown particular promise for plasmonic biosensor electrodes. For instance, template-assisted electrodeposition combines the precision of lithographic patterning with the versatility of electrochemical deposition, enabling the creation of complex electrode architectures with tailored composition and morphology. These hybrid approaches often achieve superior electrode kinetics compared to electrodes fabricated using single techniques.
Recent innovations in nanoimprint lithography and direct laser writing have further expanded the toolbox for electrode fabrication. These techniques offer high throughput and versatility, enabling rapid prototyping and optimization of electrode designs. The ability to quickly iterate through different electrode configurations accelerates the development cycle for plasmonic biosensor prototypes, facilitating the optimization of electrode kinetics for specific sensing applications.
Electron beam lithography (EBL) represents one of the most precise fabrication methods, capable of creating electrode features below 10 nm. This technique has proven particularly valuable for plasmonic biosensors where the electrode-analyte interface critically influences detection sensitivity. The high-resolution patterning enables the creation of optimized electrode architectures that maximize surface plasmon resonance effects while maintaining excellent electrochemical properties.
Focused ion beam (FIB) milling offers complementary capabilities for electrode fabrication, allowing for direct writing and modification of electrode surfaces. This technique enables the creation of three-dimensional nanostructures that can enhance electrode kinetics through increased surface area and optimized geometry. Recent advancements in FIB technology have reduced beam damage, preserving the electrochemical properties of the fabricated electrodes.
Self-assembly techniques have emerged as cost-effective alternatives for large-scale electrode fabrication. Block copolymer lithography and colloidal lithography enable the creation of regular nanopatterns across large areas, facilitating the mass production of high-performance electrodes. These bottom-up approaches are particularly valuable for creating electrodes with controlled porosity and surface roughness, factors that significantly impact electrode kinetics in biosensing applications.
Surface modification techniques complement physical nanofabrication methods by tailoring the chemical properties of electrode surfaces. Atomic layer deposition (ALD) enables the conformal coating of electrodes with ultrathin films of functional materials, enhancing stability and electrochemical performance. Plasma treatment and chemical functionalization further modify surface energy and reactivity, optimizing electrode-analyte interactions.
Hybrid fabrication approaches combining multiple techniques have shown particular promise for plasmonic biosensor electrodes. For instance, template-assisted electrodeposition combines the precision of lithographic patterning with the versatility of electrochemical deposition, enabling the creation of complex electrode architectures with tailored composition and morphology. These hybrid approaches often achieve superior electrode kinetics compared to electrodes fabricated using single techniques.
Recent innovations in nanoimprint lithography and direct laser writing have further expanded the toolbox for electrode fabrication. These techniques offer high throughput and versatility, enabling rapid prototyping and optimization of electrode designs. The ability to quickly iterate through different electrode configurations accelerates the development cycle for plasmonic biosensor prototypes, facilitating the optimization of electrode kinetics for specific sensing applications.
Regulatory Considerations for Clinical Biosensor Implementation
The implementation of plasmonic biosensor prototypes in clinical settings necessitates careful navigation through complex regulatory frameworks established by various health authorities worldwide. The FDA in the United States requires extensive validation of biosensor performance metrics, with particular emphasis on electrode kinetics stability across different biological matrices. These regulations mandate comprehensive documentation of electrode degradation patterns and their potential impact on diagnostic accuracy over the device's intended lifecycle.
European regulatory bodies under the EU Medical Device Regulation (MDR) impose additional requirements specifically addressing the nanoscale interactions at electrode surfaces. Manufacturers must demonstrate that plasmonic resonance shifts remain consistent and interpretable under varying physiological conditions. The electrode kinetics must maintain stability within defined tolerance limits, with particular attention to potential interference from commonly prescribed medications or biological compounds.
ISO 13485 certification represents another critical regulatory hurdle, requiring robust quality management systems that specifically address electrode manufacturing consistency. The standard emphasizes process validation for electrode preparation, coating procedures, and surface modification techniques that directly influence kinetic performance. Manufacturers must implement statistical process control methods to monitor electrode quality parameters throughout production.
Regulatory bodies increasingly demand real-world performance data beyond controlled laboratory conditions. This includes post-market surveillance plans specifically monitoring electrode degradation patterns in clinical use. The kinetic behavior of electrodes under actual patient conditions must be systematically tracked and analyzed to identify any deviation from expected performance characteristics.
Data privacy regulations intersect with biosensor implementation when considering the handling of patient information generated through electrode kinetic measurements. HIPAA in the US and GDPR in Europe establish strict requirements for data security, storage, and transmission protocols. Manufacturers must demonstrate that their electrode kinetic analysis algorithms maintain patient privacy while delivering clinically relevant information.
Emerging regulatory trends indicate movement toward harmonized international standards specifically addressing nanoscale biosensing technologies. The International Medical Device Regulators Forum (IMDRF) is developing guidance documents that will likely establish consistent performance requirements for electrode kinetics across major markets. These developments suggest manufacturers should design validation protocols that anticipate increasingly stringent requirements for demonstrating long-term electrode stability and performance reproducibility.
European regulatory bodies under the EU Medical Device Regulation (MDR) impose additional requirements specifically addressing the nanoscale interactions at electrode surfaces. Manufacturers must demonstrate that plasmonic resonance shifts remain consistent and interpretable under varying physiological conditions. The electrode kinetics must maintain stability within defined tolerance limits, with particular attention to potential interference from commonly prescribed medications or biological compounds.
ISO 13485 certification represents another critical regulatory hurdle, requiring robust quality management systems that specifically address electrode manufacturing consistency. The standard emphasizes process validation for electrode preparation, coating procedures, and surface modification techniques that directly influence kinetic performance. Manufacturers must implement statistical process control methods to monitor electrode quality parameters throughout production.
Regulatory bodies increasingly demand real-world performance data beyond controlled laboratory conditions. This includes post-market surveillance plans specifically monitoring electrode degradation patterns in clinical use. The kinetic behavior of electrodes under actual patient conditions must be systematically tracked and analyzed to identify any deviation from expected performance characteristics.
Data privacy regulations intersect with biosensor implementation when considering the handling of patient information generated through electrode kinetic measurements. HIPAA in the US and GDPR in Europe establish strict requirements for data security, storage, and transmission protocols. Manufacturers must demonstrate that their electrode kinetic analysis algorithms maintain patient privacy while delivering clinically relevant information.
Emerging regulatory trends indicate movement toward harmonized international standards specifically addressing nanoscale biosensing technologies. The International Medical Device Regulators Forum (IMDRF) is developing guidance documents that will likely establish consistent performance requirements for electrode kinetics across major markets. These developments suggest manufacturers should design validation protocols that anticipate increasingly stringent requirements for demonstrating long-term electrode stability and performance reproducibility.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!