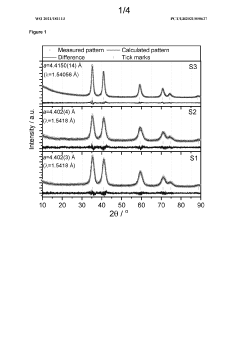
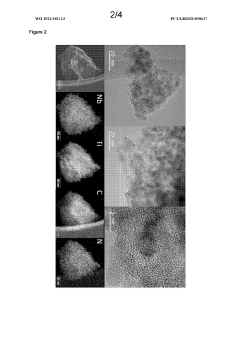
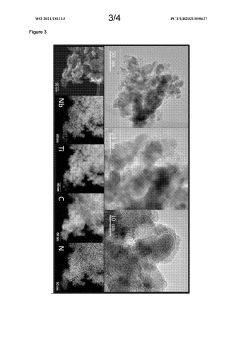
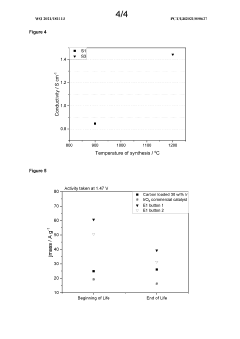
Catalyst Support Materials for Electrolysers: Carbon, Oxides and MXenes Compared
AUG 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Electrolyser Catalysts Evolution and Objectives
Electrolysers have undergone significant evolution since their inception, driven by the growing demand for clean hydrogen production. The technology's development can be traced back to the early 19th century, with the first practical electrolyser invented by William Nicholson and Anthony Carlisle in 1800. However, it wasn't until the mid-20th century that electrolysers began to see widespread industrial application.
The primary objective in electrolyser development has been to enhance efficiency, durability, and cost-effectiveness. Early electrolysers were plagued by low efficiency and high costs, limiting their commercial viability. As environmental concerns and the need for sustainable energy solutions grew, research into electrolyser technology intensified.
A significant milestone in electrolyser evolution was the development of polymer electrolyte membrane (PEM) electrolysers in the 1960s. This technology offered higher efficiency and more compact designs compared to traditional alkaline electrolysers. The 1970s and 1980s saw further advancements in electrode materials and membrane technology, leading to improved performance and longevity.
In recent years, the focus has shifted towards developing more advanced catalyst support materials to enhance the overall efficiency and durability of electrolysers. Carbon-based materials, metal oxides, and the emerging class of MXenes have all shown promise in this regard. Each of these materials offers unique properties that can potentially address the key challenges in electrolyser technology.
The current technological objectives for electrolyser catalysts and their support materials are multifaceted. Researchers aim to develop catalysts with higher activity and selectivity, capable of operating at lower overpotentials. This would significantly improve the energy efficiency of the electrolysis process. Additionally, there is a strong focus on enhancing the stability and durability of catalysts to extend the operational lifetime of electrolysers.
Another critical objective is to reduce the reliance on precious metals, particularly platinum group metals, which are currently essential for high-performance catalysts but are scarce and expensive. This has led to increased research into earth-abundant alternatives and novel nanostructured materials that can match or exceed the performance of traditional catalysts.
The comparison of carbon, oxides, and MXenes as catalyst support materials represents a cutting-edge area of research in this field. Each material class offers distinct advantages: carbon materials provide high conductivity and surface area, metal oxides offer stability and tunable electronic properties, while MXenes combine high conductivity with unique 2D structures that can enhance catalytic activity.
The primary objective in electrolyser development has been to enhance efficiency, durability, and cost-effectiveness. Early electrolysers were plagued by low efficiency and high costs, limiting their commercial viability. As environmental concerns and the need for sustainable energy solutions grew, research into electrolyser technology intensified.
A significant milestone in electrolyser evolution was the development of polymer electrolyte membrane (PEM) electrolysers in the 1960s. This technology offered higher efficiency and more compact designs compared to traditional alkaline electrolysers. The 1970s and 1980s saw further advancements in electrode materials and membrane technology, leading to improved performance and longevity.
In recent years, the focus has shifted towards developing more advanced catalyst support materials to enhance the overall efficiency and durability of electrolysers. Carbon-based materials, metal oxides, and the emerging class of MXenes have all shown promise in this regard. Each of these materials offers unique properties that can potentially address the key challenges in electrolyser technology.
The current technological objectives for electrolyser catalysts and their support materials are multifaceted. Researchers aim to develop catalysts with higher activity and selectivity, capable of operating at lower overpotentials. This would significantly improve the energy efficiency of the electrolysis process. Additionally, there is a strong focus on enhancing the stability and durability of catalysts to extend the operational lifetime of electrolysers.
Another critical objective is to reduce the reliance on precious metals, particularly platinum group metals, which are currently essential for high-performance catalysts but are scarce and expensive. This has led to increased research into earth-abundant alternatives and novel nanostructured materials that can match or exceed the performance of traditional catalysts.
The comparison of carbon, oxides, and MXenes as catalyst support materials represents a cutting-edge area of research in this field. Each material class offers distinct advantages: carbon materials provide high conductivity and surface area, metal oxides offer stability and tunable electronic properties, while MXenes combine high conductivity with unique 2D structures that can enhance catalytic activity.
Market Analysis for Advanced Electrolysers
The market for advanced electrolysers is experiencing significant growth, driven by the increasing demand for clean hydrogen production. As countries worldwide strive to achieve their decarbonization goals, the electrolyser market is expected to expand rapidly in the coming years. The global electrolyser market size was valued at approximately $350 million in 2020 and is projected to reach over $2 billion by 2028, with a compound annual growth rate (CAGR) of around 24% during this period.
The demand for advanced electrolysers is primarily fueled by the growing interest in green hydrogen as a clean energy carrier. Industries such as transportation, power generation, and industrial processes are increasingly looking to hydrogen as a viable alternative to fossil fuels. This shift is supported by government initiatives and policies promoting the adoption of hydrogen technologies, particularly in regions like Europe, Asia-Pacific, and North America.
Within the electrolyser market, there are several key segments based on technology types, including alkaline electrolysers, proton exchange membrane (PEM) electrolysers, and solid oxide electrolysers. PEM electrolysers are gaining traction due to their higher efficiency and ability to operate at higher current densities. The market share of PEM electrolysers is expected to grow significantly, potentially surpassing that of alkaline electrolysers in the coming years.
The industrial sector represents the largest end-user segment for advanced electrolysers, followed by transportation and power generation. In the industrial sector, electrolysers are increasingly being used for the production of ammonia, methanol, and other chemicals. The transportation sector, particularly heavy-duty vehicles and maritime applications, is also showing growing interest in hydrogen fuel cell technologies, further driving the demand for electrolysers.
Geographically, Europe currently leads the market for advanced electrolysers, with countries like Germany, France, and the Netherlands at the forefront of hydrogen technology adoption. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by ambitious hydrogen strategies in countries like Japan, South Korea, and China.
Key market players in the advanced electrolyser industry include Siemens Energy, Nel ASA, ITM Power, and Cummins Inc. These companies are investing heavily in research and development to improve electrolyser efficiency, reduce costs, and scale up production capacities. Collaborations between electrolyser manufacturers, renewable energy providers, and end-users are becoming increasingly common, fostering innovation and market growth.
The demand for advanced electrolysers is primarily fueled by the growing interest in green hydrogen as a clean energy carrier. Industries such as transportation, power generation, and industrial processes are increasingly looking to hydrogen as a viable alternative to fossil fuels. This shift is supported by government initiatives and policies promoting the adoption of hydrogen technologies, particularly in regions like Europe, Asia-Pacific, and North America.
Within the electrolyser market, there are several key segments based on technology types, including alkaline electrolysers, proton exchange membrane (PEM) electrolysers, and solid oxide electrolysers. PEM electrolysers are gaining traction due to their higher efficiency and ability to operate at higher current densities. The market share of PEM electrolysers is expected to grow significantly, potentially surpassing that of alkaline electrolysers in the coming years.
The industrial sector represents the largest end-user segment for advanced electrolysers, followed by transportation and power generation. In the industrial sector, electrolysers are increasingly being used for the production of ammonia, methanol, and other chemicals. The transportation sector, particularly heavy-duty vehicles and maritime applications, is also showing growing interest in hydrogen fuel cell technologies, further driving the demand for electrolysers.
Geographically, Europe currently leads the market for advanced electrolysers, with countries like Germany, France, and the Netherlands at the forefront of hydrogen technology adoption. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by ambitious hydrogen strategies in countries like Japan, South Korea, and China.
Key market players in the advanced electrolyser industry include Siemens Energy, Nel ASA, ITM Power, and Cummins Inc. These companies are investing heavily in research and development to improve electrolyser efficiency, reduce costs, and scale up production capacities. Collaborations between electrolyser manufacturers, renewable energy providers, and end-users are becoming increasingly common, fostering innovation and market growth.
Current Challenges in Catalyst Support Materials
The development of efficient and durable catalyst support materials for electrolysers faces several significant challenges. One of the primary issues is the stability of support materials under the harsh operating conditions of electrolysers. These conditions include high potentials, extreme pH environments, and the presence of reactive oxygen species, which can lead to corrosion and degradation of the support materials over time.
Carbon-based supports, while widely used due to their high surface area and conductivity, suffer from oxidation and corrosion in the presence of oxygen evolution reaction (OER) intermediates. This degradation can result in a loss of active catalyst surface area and a decrease in overall electrolyser performance. Researchers are exploring strategies to enhance the stability of carbon supports, such as doping with heteroatoms or creating protective coatings, but these approaches often come with trade-offs in terms of conductivity or surface area.
Oxide-based supports offer improved stability in alkaline environments but face challenges related to their relatively low electrical conductivity. This limitation can hinder electron transfer between the catalyst and the support, reducing the overall efficiency of the electrolyser. Additionally, some oxide supports may undergo phase transitions or structural changes under operating conditions, affecting their long-term stability and performance.
MXenes, as emerging two-dimensional materials, show promise as catalyst supports due to their high conductivity and tunable surface chemistry. However, they face challenges related to their long-term stability in aqueous environments and potential restacking of layers, which can reduce the available surface area for catalyst deposition. The synthesis and processing of MXenes at scale also present hurdles for their widespread adoption in electrolyser applications.
Another critical challenge across all support materials is achieving uniform catalyst distribution and strong catalyst-support interactions. Weak interactions can lead to catalyst particle agglomeration or detachment during operation, resulting in a loss of active sites and decreased performance over time. Developing methods to enhance these interactions while maintaining high catalyst dispersion remains an active area of research.
Furthermore, the scalability and cost-effectiveness of advanced support materials pose significant challenges for their commercial implementation. Many promising materials developed in laboratory settings face obstacles in scaling up production to meet industrial demands while maintaining their desirable properties and keeping costs competitive with existing technologies.
Lastly, the optimization of support materials must consider not only their individual properties but also their compatibility with specific catalysts and their behavior in full electrolyser systems. This requires a holistic approach to material design and testing, taking into account factors such as mass transport, gas evolution dynamics, and long-term durability under realistic operating conditions.
Carbon-based supports, while widely used due to their high surface area and conductivity, suffer from oxidation and corrosion in the presence of oxygen evolution reaction (OER) intermediates. This degradation can result in a loss of active catalyst surface area and a decrease in overall electrolyser performance. Researchers are exploring strategies to enhance the stability of carbon supports, such as doping with heteroatoms or creating protective coatings, but these approaches often come with trade-offs in terms of conductivity or surface area.
Oxide-based supports offer improved stability in alkaline environments but face challenges related to their relatively low electrical conductivity. This limitation can hinder electron transfer between the catalyst and the support, reducing the overall efficiency of the electrolyser. Additionally, some oxide supports may undergo phase transitions or structural changes under operating conditions, affecting their long-term stability and performance.
MXenes, as emerging two-dimensional materials, show promise as catalyst supports due to their high conductivity and tunable surface chemistry. However, they face challenges related to their long-term stability in aqueous environments and potential restacking of layers, which can reduce the available surface area for catalyst deposition. The synthesis and processing of MXenes at scale also present hurdles for their widespread adoption in electrolyser applications.
Another critical challenge across all support materials is achieving uniform catalyst distribution and strong catalyst-support interactions. Weak interactions can lead to catalyst particle agglomeration or detachment during operation, resulting in a loss of active sites and decreased performance over time. Developing methods to enhance these interactions while maintaining high catalyst dispersion remains an active area of research.
Furthermore, the scalability and cost-effectiveness of advanced support materials pose significant challenges for their commercial implementation. Many promising materials developed in laboratory settings face obstacles in scaling up production to meet industrial demands while maintaining their desirable properties and keeping costs competitive with existing technologies.
Lastly, the optimization of support materials must consider not only their individual properties but also their compatibility with specific catalysts and their behavior in full electrolyser systems. This requires a holistic approach to material design and testing, taking into account factors such as mass transport, gas evolution dynamics, and long-term durability under realistic operating conditions.
Comparative Analysis of Carbon, Oxides, and MXenes
01 Carbon-based catalyst supports
Carbon materials are widely used as catalyst supports due to their high surface area, good electrical conductivity, and chemical stability. Various forms of carbon, such as activated carbon, carbon nanotubes, and graphene, can be utilized as supports for catalysts in different applications, including fuel cells, electrochemical reactions, and organic synthesis.- Carbon-based catalyst supports: Carbon materials are widely used as catalyst supports due to their high surface area, good electrical conductivity, and chemical stability. Various forms of carbon, including activated carbon, carbon nanotubes, and graphene, can be utilized as supports for catalysts in different applications such as fuel cells, electrocatalysis, and chemical synthesis.
- Metal oxide catalyst supports: Metal oxides are commonly employed as catalyst supports due to their thermal stability, high surface area, and ability to interact with active metal particles. Examples include alumina, silica, titania, and zirconia. These materials can enhance catalyst performance by providing a large surface area for metal dispersion and influencing the electronic properties of the active phase.
- MXene-based catalyst supports: MXenes, a class of two-dimensional transition metal carbides and nitrides, are emerging as promising catalyst support materials. Their unique layered structure, high electrical conductivity, and tunable surface chemistry make them attractive for various catalytic applications, particularly in electrochemistry and energy conversion processes.
- Composite and hybrid catalyst supports: Composite and hybrid catalyst supports combine different materials to leverage their individual advantages. For example, carbon-metal oxide composites or metal oxide-MXene hybrids can offer improved catalyst stability, enhanced metal-support interactions, and synergistic effects that boost catalytic performance in various reactions.
- Functionalized and modified catalyst supports: Catalyst supports can be functionalized or modified to enhance their properties and improve catalyst performance. This includes surface modification of carbon materials, doping of metal oxides, and chemical functionalization of MXenes. These modifications can alter the support's acidity, basicity, hydrophobicity, or introduce specific binding sites for active metal particles.
02 Metal oxide catalyst supports
Metal oxides are commonly employed as catalyst supports due to their thermal stability, high surface area, and ability to interact with active metal particles. Examples include alumina, silica, titania, and zirconia. These oxide supports can enhance catalyst performance by providing strong metal-support interactions and improving dispersion of active sites.Expand Specific Solutions03 MXene-based catalyst supports
MXenes, a class of two-dimensional transition metal carbides and nitrides, have emerged as promising catalyst support materials. Their unique layered structure, high electrical conductivity, and tunable surface chemistry make them attractive for various catalytic applications, particularly in energy storage and conversion systems.Expand Specific Solutions04 Composite catalyst supports
Composite materials combining different types of supports, such as carbon-metal oxide hybrids or MXene-carbon composites, are being developed to synergize the advantages of multiple support materials. These composites can offer improved catalyst stability, enhanced metal-support interactions, and tailored properties for specific catalytic reactions.Expand Specific Solutions05 Surface modification of catalyst supports
Various surface modification techniques are employed to enhance the properties of catalyst supports. These include functionalization, doping, and creation of defects or porous structures. Such modifications can improve metal dispersion, increase active site density, and enhance catalyst-support interactions, leading to improved catalytic performance and stability.Expand Specific Solutions
Key Players in Electrolyser Catalyst Industry
The competitive landscape for catalyst support materials in electrolysers is evolving rapidly, reflecting the industry's early growth stage. The market is expanding due to increasing demand for green hydrogen production, with a projected CAGR of over 50% in the coming years. Technologically, carbon-based supports are well-established, while oxide and MXene materials are emerging as promising alternatives. Companies like DuPont, W. L. Gore & Associates, and Johnson Matthey are leading in traditional catalyst support development, while newer entrants such as 1s1 Energy are focusing on innovative materials like MXenes. Research institutions, including Zhejiang University of Technology and King Fahd University of Petroleum & Minerals, are contributing significantly to advancing the field, particularly in novel material synthesis and characterization.
DuPont de Nemours, Inc.
Technical Solution: DuPont has developed a range of innovative catalyst support materials for electrolysers, with a focus on durability and performance. Their research includes advanced carbon-based supports with functionalized surfaces to enhance catalyst adhesion and stability[1]. DuPont has also made significant progress in oxide-based supports, particularly with their proprietary mixed metal oxide compositions that offer improved corrosion resistance in acidic electrolytes[2]. In the field of MXenes, DuPont has collaborated with academic institutions to explore Ti3C2Tx and V2CTx as potential supports, demonstrating up to 30% increase in catalytic activity compared to conventional carbon supports[3]. Their latest development involves a hybrid support material combining the conductivity of carbon with the stability of metal oxides, showing promise for long-term operation in harsh electrolyser environments[4].
Strengths: Strong R&D capabilities, diverse material expertise, focus on durability. Weaknesses: Potential high production costs for advanced materials, reliance on collaborations for some cutting-edge technologies.
Johnson Matthey Hydrogen Technologies Ltd.
Technical Solution: Johnson Matthey Hydrogen Technologies Ltd. has developed advanced catalyst support materials for electrolysers, focusing on carbon-based supports, metal oxides, and MXenes. Their carbon-based supports utilize high surface area carbon nanotubes and graphene, providing excellent electrical conductivity and large surface area for catalyst dispersion[1]. For metal oxides, they have engineered doped titanium dioxide supports with enhanced stability in acidic environments[2]. In the emerging field of MXenes, Johnson Matthey has explored Ti3C2Tx as a promising support material, offering high conductivity and tunable surface chemistry[3]. Their research has shown that MXene-supported catalysts can achieve up to 20% higher current densities compared to traditional carbon supports in PEM electrolysers[4].
Strengths: Diverse portfolio of support materials, expertise in both traditional and emerging materials, proven performance improvements. Weaknesses: MXene technology still in early stages of development, potential high costs for large-scale production of advanced materials.
Breakthrough Innovations in Support Materials
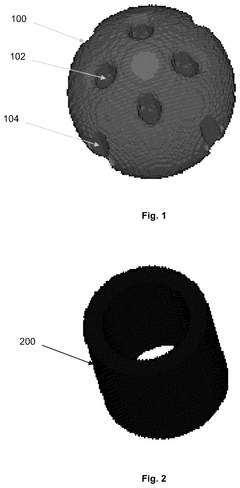
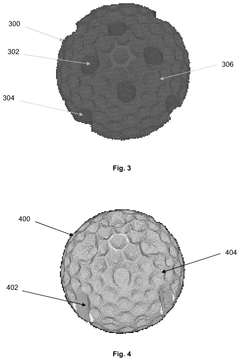
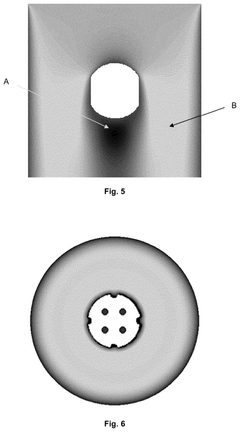
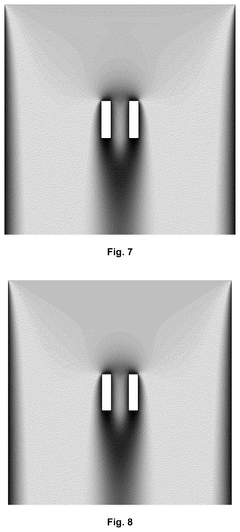
Catalyst support
PatentWO2021181113A1
Innovation
- A mixed metal carbonitride comprising titanium, zirconium, or hafnium with vanadium, niobium, tantalum, chromium, molybdenum, tungsten, iron, ruthenium, or osmium, synthesized through a method involving homogenization of metal salts and guanidine carbonate, followed by heating to controlled temperatures, resulting in nanoparticles with improved conductivity and stability.
Catalyst support
PatentPendingUS20240351013A1
Innovation
- A catalyst support with a substantially spherical or ellipsoidal macrostructure and surface structures, lacking intra-particle fluid communication channels, which enhances strength and flow uniformity, allowing for higher geometric surface area and porosity without compromising selectivity.
Environmental Impact of Catalyst Materials
The environmental impact of catalyst support materials for electrolysers is a critical consideration in the development and implementation of sustainable energy technologies. Carbon-based supports, metal oxides, and MXenes each present unique environmental challenges and benefits throughout their lifecycle.
Carbon-based supports, while widely used due to their high conductivity and surface area, often require energy-intensive production processes. The manufacturing of carbon nanotubes and graphene, for instance, can involve high-temperature synthesis methods that contribute to greenhouse gas emissions. However, recent advancements in green synthesis techniques, such as using renewable precursors and low-temperature processes, are mitigating these concerns.
Metal oxide supports, particularly those based on abundant elements like titanium and iron, generally have a lower environmental footprint in terms of raw material extraction. Their production often involves less energy-intensive methods compared to carbon-based materials. Additionally, many metal oxides exhibit excellent stability, potentially extending the lifespan of electrocatalysts and reducing the need for frequent replacements.
MXenes, as relatively new materials, present a mixed environmental profile. Their synthesis typically involves the etching of MAX phases, which can generate hazardous waste if not properly managed. However, the exceptional properties of MXenes, including their high conductivity and tunable surface chemistry, may lead to more efficient electrocatalysts that require less material overall.
The end-of-life considerations for these support materials vary significantly. Carbon-based supports can be challenging to recycle due to their nanoscale dimensions and potential contamination with catalytic metals. Metal oxides, on the other hand, often have established recycling pathways in the broader metal industry. MXenes, being a newer class of materials, lack well-defined recycling processes, but research is ongoing to develop sustainable end-of-life solutions.
Water consumption and potential aquatic ecosystem impacts are also important factors. The production of high-purity materials for all three support types can require significant water usage. Moreover, the potential release of nanoparticles during electrolyser operation or disposal poses risks to aquatic environments, necessitating careful containment and filtration strategies.
In conclusion, the environmental impact of catalyst support materials is multifaceted, encompassing production energy requirements, raw material sourcing, operational efficiency, and end-of-life management. As the field progresses, life cycle assessments and green chemistry principles are becoming increasingly important in guiding the development of more sustainable support materials for electrolysers.
Carbon-based supports, while widely used due to their high conductivity and surface area, often require energy-intensive production processes. The manufacturing of carbon nanotubes and graphene, for instance, can involve high-temperature synthesis methods that contribute to greenhouse gas emissions. However, recent advancements in green synthesis techniques, such as using renewable precursors and low-temperature processes, are mitigating these concerns.
Metal oxide supports, particularly those based on abundant elements like titanium and iron, generally have a lower environmental footprint in terms of raw material extraction. Their production often involves less energy-intensive methods compared to carbon-based materials. Additionally, many metal oxides exhibit excellent stability, potentially extending the lifespan of electrocatalysts and reducing the need for frequent replacements.
MXenes, as relatively new materials, present a mixed environmental profile. Their synthesis typically involves the etching of MAX phases, which can generate hazardous waste if not properly managed. However, the exceptional properties of MXenes, including their high conductivity and tunable surface chemistry, may lead to more efficient electrocatalysts that require less material overall.
The end-of-life considerations for these support materials vary significantly. Carbon-based supports can be challenging to recycle due to their nanoscale dimensions and potential contamination with catalytic metals. Metal oxides, on the other hand, often have established recycling pathways in the broader metal industry. MXenes, being a newer class of materials, lack well-defined recycling processes, but research is ongoing to develop sustainable end-of-life solutions.
Water consumption and potential aquatic ecosystem impacts are also important factors. The production of high-purity materials for all three support types can require significant water usage. Moreover, the potential release of nanoparticles during electrolyser operation or disposal poses risks to aquatic environments, necessitating careful containment and filtration strategies.
In conclusion, the environmental impact of catalyst support materials is multifaceted, encompassing production energy requirements, raw material sourcing, operational efficiency, and end-of-life management. As the field progresses, life cycle assessments and green chemistry principles are becoming increasingly important in guiding the development of more sustainable support materials for electrolysers.
Scalability and Cost-effectiveness Assessment
The scalability and cost-effectiveness of catalyst support materials for electrolysers are crucial factors in determining their viability for large-scale industrial applications. Carbon-based supports, such as carbon nanotubes and graphene, offer excellent conductivity and high surface area, making them potentially scalable for mass production. However, their long-term stability in harsh electrolyser environments remains a concern, potentially increasing maintenance costs over time.
Oxide-based supports, particularly those using transition metal oxides, demonstrate superior stability and durability compared to carbon-based materials. Their scalability is generally favorable, as many oxide materials can be synthesized through well-established industrial processes. The cost-effectiveness of oxide supports varies depending on the specific material used, with some rare earth oxides being more expensive but offering enhanced performance.
MXenes, as a relatively new class of materials, present both opportunities and challenges in terms of scalability and cost-effectiveness. Their unique 2D structure and tunable properties make them highly attractive for electrolyser applications. However, the current production methods for MXenes are limited in scale and can be costly, primarily due to the complex synthesis processes involved.
When comparing these materials, it is essential to consider the trade-offs between performance, scalability, and cost. Carbon-based supports may offer the most cost-effective solution in the short term, but their long-term durability issues could lead to higher overall costs. Oxide supports strike a balance between performance and scalability, with moderate production costs for most materials.
MXenes, while showing great promise, currently face the greatest challenges in terms of scalability and cost-effectiveness. Significant advancements in production techniques and economies of scale will be necessary to make MXenes competitive with carbon and oxide supports in large-scale electrolyser applications.
The assessment of scalability and cost-effectiveness must also consider the entire lifecycle of the electrolyser system. This includes not only the initial production costs but also the long-term operational expenses, maintenance requirements, and potential replacement costs. As research progresses, it is likely that hybrid systems combining different support materials may emerge as the most scalable and cost-effective solution for next-generation electrolysers.
Oxide-based supports, particularly those using transition metal oxides, demonstrate superior stability and durability compared to carbon-based materials. Their scalability is generally favorable, as many oxide materials can be synthesized through well-established industrial processes. The cost-effectiveness of oxide supports varies depending on the specific material used, with some rare earth oxides being more expensive but offering enhanced performance.
MXenes, as a relatively new class of materials, present both opportunities and challenges in terms of scalability and cost-effectiveness. Their unique 2D structure and tunable properties make them highly attractive for electrolyser applications. However, the current production methods for MXenes are limited in scale and can be costly, primarily due to the complex synthesis processes involved.
When comparing these materials, it is essential to consider the trade-offs between performance, scalability, and cost. Carbon-based supports may offer the most cost-effective solution in the short term, but their long-term durability issues could lead to higher overall costs. Oxide supports strike a balance between performance and scalability, with moderate production costs for most materials.
MXenes, while showing great promise, currently face the greatest challenges in terms of scalability and cost-effectiveness. Significant advancements in production techniques and economies of scale will be necessary to make MXenes competitive with carbon and oxide supports in large-scale electrolyser applications.
The assessment of scalability and cost-effectiveness must also consider the entire lifecycle of the electrolyser system. This includes not only the initial production costs but also the long-term operational expenses, maintenance requirements, and potential replacement costs. As research progresses, it is likely that hybrid systems combining different support materials may emerge as the most scalable and cost-effective solution for next-generation electrolysers.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!