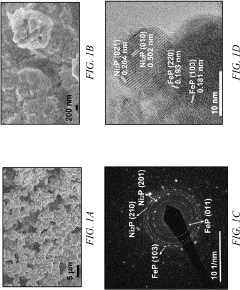
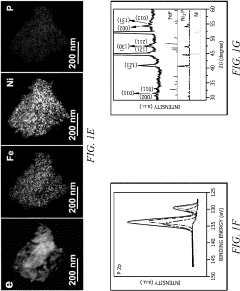
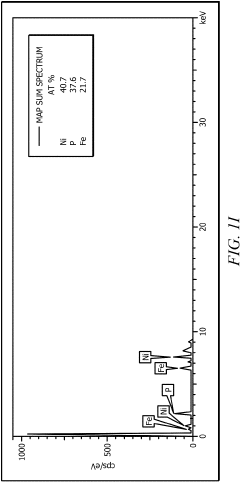
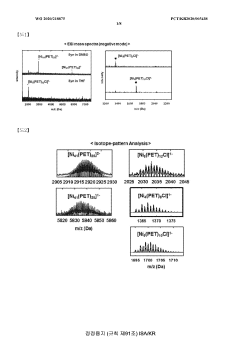
Bifunctional Catalysts for OER and HER: Opportunities and Design Constraints
AUG 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Bifunctional Catalysts Background and Objectives
Bifunctional catalysts for oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) have emerged as a critical area of research in the field of renewable energy and electrocatalysis. These catalysts play a pivotal role in water splitting, a process fundamental to the production of clean hydrogen fuel. The development of efficient bifunctional catalysts addresses the growing global demand for sustainable energy solutions and the need to reduce reliance on fossil fuels.
The evolution of bifunctional catalysts can be traced back to the early 2000s when researchers began exploring materials that could catalyze both OER and HER in a single device. This approach aimed to simplify electrolyzer designs, reduce costs, and improve overall system efficiency. Initially, noble metals such as platinum and iridium were the primary focus due to their high catalytic activity. However, their scarcity and high cost prompted a shift towards more abundant and cost-effective alternatives.
Over the past decade, significant progress has been made in developing non-precious metal-based bifunctional catalysts. Transition metal oxides, sulfides, and phosphides have shown promising results, with materials like nickel-iron layered double hydroxides and cobalt phosphides demonstrating remarkable bifunctional activity. The integration of carbon-based materials, such as graphene and carbon nanotubes, has further enhanced the performance of these catalysts by improving conductivity and surface area.
The primary objective of current research in bifunctional catalysts is to develop materials that exhibit high catalytic activity, stability, and durability for both OER and HER in alkaline media. Researchers aim to achieve low overpotentials, high current densities, and long-term operational stability under industrial conditions. Additionally, there is a focus on understanding the fundamental mechanisms of these reactions at the atomic and molecular levels to guide rational catalyst design.
Another crucial goal is to bridge the gap between laboratory-scale demonstrations and large-scale industrial applications. This involves addressing challenges related to scalability, cost-effectiveness, and integration into existing energy infrastructure. Researchers are also exploring ways to tailor catalyst properties for specific applications, such as seawater electrolysis or coupling with renewable energy sources like solar and wind power.
As the field progresses, there is an increasing emphasis on developing multifunctional catalysts that can perform beyond OER and HER, potentially catalyzing other important reactions in energy conversion and storage. This holistic approach aims to create versatile materials that can adapt to various energy scenarios and contribute to a more flexible and resilient clean energy ecosystem.
The evolution of bifunctional catalysts can be traced back to the early 2000s when researchers began exploring materials that could catalyze both OER and HER in a single device. This approach aimed to simplify electrolyzer designs, reduce costs, and improve overall system efficiency. Initially, noble metals such as platinum and iridium were the primary focus due to their high catalytic activity. However, their scarcity and high cost prompted a shift towards more abundant and cost-effective alternatives.
Over the past decade, significant progress has been made in developing non-precious metal-based bifunctional catalysts. Transition metal oxides, sulfides, and phosphides have shown promising results, with materials like nickel-iron layered double hydroxides and cobalt phosphides demonstrating remarkable bifunctional activity. The integration of carbon-based materials, such as graphene and carbon nanotubes, has further enhanced the performance of these catalysts by improving conductivity and surface area.
The primary objective of current research in bifunctional catalysts is to develop materials that exhibit high catalytic activity, stability, and durability for both OER and HER in alkaline media. Researchers aim to achieve low overpotentials, high current densities, and long-term operational stability under industrial conditions. Additionally, there is a focus on understanding the fundamental mechanisms of these reactions at the atomic and molecular levels to guide rational catalyst design.
Another crucial goal is to bridge the gap between laboratory-scale demonstrations and large-scale industrial applications. This involves addressing challenges related to scalability, cost-effectiveness, and integration into existing energy infrastructure. Researchers are also exploring ways to tailor catalyst properties for specific applications, such as seawater electrolysis or coupling with renewable energy sources like solar and wind power.
As the field progresses, there is an increasing emphasis on developing multifunctional catalysts that can perform beyond OER and HER, potentially catalyzing other important reactions in energy conversion and storage. This holistic approach aims to create versatile materials that can adapt to various energy scenarios and contribute to a more flexible and resilient clean energy ecosystem.
Market Analysis for OER/HER Catalysts
The market for bifunctional catalysts for Oxygen Evolution Reaction (OER) and Hydrogen Evolution Reaction (HER) is experiencing significant growth, driven by the increasing demand for clean energy solutions and the push towards a hydrogen-based economy. These catalysts play a crucial role in water electrolysis, a key process in hydrogen production and energy storage systems.
The global market for water electrolysis, where OER/HER catalysts are primarily used, is projected to expand rapidly in the coming years. This growth is fueled by the rising adoption of renewable energy sources and the need for efficient energy storage technologies. Governments worldwide are implementing supportive policies and investing heavily in hydrogen infrastructure, further boosting market demand for advanced catalysts.
In the industrial sector, there is a growing interest in using green hydrogen for various applications, including ammonia production, steel manufacturing, and transportation. This trend is creating new opportunities for OER/HER catalyst manufacturers, as industries seek more efficient and cost-effective electrolysis solutions.
The automotive industry is another significant driver of market demand. With the increasing focus on fuel cell electric vehicles (FCEVs), there is a growing need for high-performance catalysts to support hydrogen production and utilization. Major automakers are investing in FCEV technology, which is expected to create substantial demand for OER/HER catalysts in the coming years.
Geographically, Asia-Pacific is emerging as a key market for OER/HER catalysts, with China and Japan leading in terms of research and development activities. Europe is also showing strong growth potential, driven by ambitious hydrogen strategies and substantial investments in green energy infrastructure.
However, the market faces certain challenges. The high cost of noble metal-based catalysts, such as platinum and iridium, is a significant barrier to widespread adoption. This has led to increased research efforts in developing non-precious metal catalysts that can offer comparable performance at lower costs.
The competitive landscape of the OER/HER catalyst market is characterized by a mix of established chemical companies, specialized catalyst manufacturers, and innovative startups. Key players are focusing on developing novel materials and improving catalyst efficiency to gain a competitive edge.
Looking ahead, the market for OER/HER catalysts is expected to continue its growth trajectory. Technological advancements, such as the development of nanostructured catalysts and the integration of artificial intelligence in catalyst design, are likely to open up new opportunities and drive market expansion. As the world moves towards a more sustainable energy future, the demand for efficient and cost-effective bifunctional catalysts for OER and HER is poised to increase substantially.
The global market for water electrolysis, where OER/HER catalysts are primarily used, is projected to expand rapidly in the coming years. This growth is fueled by the rising adoption of renewable energy sources and the need for efficient energy storage technologies. Governments worldwide are implementing supportive policies and investing heavily in hydrogen infrastructure, further boosting market demand for advanced catalysts.
In the industrial sector, there is a growing interest in using green hydrogen for various applications, including ammonia production, steel manufacturing, and transportation. This trend is creating new opportunities for OER/HER catalyst manufacturers, as industries seek more efficient and cost-effective electrolysis solutions.
The automotive industry is another significant driver of market demand. With the increasing focus on fuel cell electric vehicles (FCEVs), there is a growing need for high-performance catalysts to support hydrogen production and utilization. Major automakers are investing in FCEV technology, which is expected to create substantial demand for OER/HER catalysts in the coming years.
Geographically, Asia-Pacific is emerging as a key market for OER/HER catalysts, with China and Japan leading in terms of research and development activities. Europe is also showing strong growth potential, driven by ambitious hydrogen strategies and substantial investments in green energy infrastructure.
However, the market faces certain challenges. The high cost of noble metal-based catalysts, such as platinum and iridium, is a significant barrier to widespread adoption. This has led to increased research efforts in developing non-precious metal catalysts that can offer comparable performance at lower costs.
The competitive landscape of the OER/HER catalyst market is characterized by a mix of established chemical companies, specialized catalyst manufacturers, and innovative startups. Key players are focusing on developing novel materials and improving catalyst efficiency to gain a competitive edge.
Looking ahead, the market for OER/HER catalysts is expected to continue its growth trajectory. Technological advancements, such as the development of nanostructured catalysts and the integration of artificial intelligence in catalyst design, are likely to open up new opportunities and drive market expansion. As the world moves towards a more sustainable energy future, the demand for efficient and cost-effective bifunctional catalysts for OER and HER is poised to increase substantially.
Current Challenges in Bifunctional Catalyst Development
The development of bifunctional catalysts for both the oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) faces several significant challenges. One of the primary obstacles is achieving optimal performance for both reactions simultaneously. OER and HER typically require different catalytic properties, making it difficult to design a single material that excels in both processes.
Material stability presents another major hurdle. Catalysts must withstand harsh operating conditions, including extreme pH levels and high current densities, without degrading or losing their catalytic activity. This is particularly challenging for bifunctional catalysts, as they need to maintain their dual functionality over extended periods.
Scalability and cost-effectiveness remain critical issues in bifunctional catalyst development. Many high-performance catalysts rely on expensive noble metals or complex nanostructures, limiting their potential for large-scale industrial applications. Researchers are striving to develop catalysts using earth-abundant elements, but achieving comparable performance to noble metal-based catalysts is challenging.
The optimization of catalyst morphology and structure poses another significant challenge. The ideal catalyst should have a high surface area, numerous active sites, and efficient mass transport properties. However, balancing these factors while maintaining stability and conductivity is a complex task that requires careful design and engineering.
Understanding the fundamental mechanisms of bifunctional catalysis is crucial yet challenging. The interplay between OER and HER at the molecular level is not fully elucidated, making it difficult to rationally design catalysts with improved performance. Advanced in-situ and operando characterization techniques are needed to gain deeper insights into the catalytic processes.
Bridging the gap between laboratory-scale research and industrial implementation remains a significant challenge. Many promising catalysts demonstrate excellent performance in controlled laboratory conditions but fail to maintain their efficiency when scaled up or integrated into real-world systems. Addressing this challenge requires collaborative efforts between researchers and industry partners.
The development of standardized testing protocols and benchmarking methods is another area that needs attention. Currently, there is a lack of uniformity in how bifunctional catalysts are evaluated, making it difficult to compare results across different studies and assess the true potential of new materials.
In conclusion, while significant progress has been made in bifunctional catalyst research, overcoming these challenges will be crucial for advancing the field and realizing the potential of these materials in practical applications such as water splitting and renewable energy storage.
Material stability presents another major hurdle. Catalysts must withstand harsh operating conditions, including extreme pH levels and high current densities, without degrading or losing their catalytic activity. This is particularly challenging for bifunctional catalysts, as they need to maintain their dual functionality over extended periods.
Scalability and cost-effectiveness remain critical issues in bifunctional catalyst development. Many high-performance catalysts rely on expensive noble metals or complex nanostructures, limiting their potential for large-scale industrial applications. Researchers are striving to develop catalysts using earth-abundant elements, but achieving comparable performance to noble metal-based catalysts is challenging.
The optimization of catalyst morphology and structure poses another significant challenge. The ideal catalyst should have a high surface area, numerous active sites, and efficient mass transport properties. However, balancing these factors while maintaining stability and conductivity is a complex task that requires careful design and engineering.
Understanding the fundamental mechanisms of bifunctional catalysis is crucial yet challenging. The interplay between OER and HER at the molecular level is not fully elucidated, making it difficult to rationally design catalysts with improved performance. Advanced in-situ and operando characterization techniques are needed to gain deeper insights into the catalytic processes.
Bridging the gap between laboratory-scale research and industrial implementation remains a significant challenge. Many promising catalysts demonstrate excellent performance in controlled laboratory conditions but fail to maintain their efficiency when scaled up or integrated into real-world systems. Addressing this challenge requires collaborative efforts between researchers and industry partners.
The development of standardized testing protocols and benchmarking methods is another area that needs attention. Currently, there is a lack of uniformity in how bifunctional catalysts are evaluated, making it difficult to compare results across different studies and assess the true potential of new materials.
In conclusion, while significant progress has been made in bifunctional catalyst research, overcoming these challenges will be crucial for advancing the field and realizing the potential of these materials in practical applications such as water splitting and renewable energy storage.
State-of-the-Art Bifunctional Catalyst Solutions
01 Bifunctional catalysts for chemical reactions
Bifunctional catalysts are designed to perform two distinct catalytic functions simultaneously. These catalysts often combine acidic and basic sites or metal and acid/base functionalities on a single support. They can enhance reaction efficiency, selectivity, and yield in various chemical processes, including hydrogenation, dehydrogenation, and isomerization reactions.- Bifunctional catalysts for chemical reactions: Bifunctional catalysts are designed to perform multiple catalytic functions simultaneously, enhancing reaction efficiency and selectivity. These catalysts often combine acidic and basic sites or different metal centers to facilitate complex transformations in a single step. They are particularly useful in organic synthesis, petrochemical processes, and environmental applications.
- Nanostructured bifunctional catalysts: Nanostructured materials are increasingly used as bifunctional catalysts due to their high surface area and unique properties. These catalysts often incorporate metal nanoparticles or nanocomposites, which can be tailored to exhibit both redox and acid-base properties. The nanostructure allows for better dispersion of active sites and improved catalyst stability.
- Bifunctional catalysts for energy applications: In the energy sector, bifunctional catalysts play a crucial role in processes such as fuel cells, water splitting, and CO2 conversion. These catalysts often combine hydrogen evolution and oxygen evolution capabilities or can simultaneously reduce CO2 and produce valuable chemicals. The design of these catalysts focuses on maximizing energy efficiency and selectivity.
- Heterogeneous bifunctional catalysts: Heterogeneous bifunctional catalysts are solid materials that provide multiple active sites on their surface. These catalysts are often based on metal oxides, zeolites, or supported metal complexes. They offer advantages such as easy separation from reaction mixtures, reusability, and the ability to work under diverse reaction conditions. Applications include biomass conversion, fine chemical synthesis, and petroleum refining.
- Characterization and optimization of bifunctional catalysts: Advanced characterization techniques are essential for understanding the structure-activity relationships of bifunctional catalysts. Methods such as spectroscopy, microscopy, and computational modeling are used to analyze the nature of active sites, their distribution, and synergistic effects. This knowledge guides the optimization of catalyst composition, morphology, and preparation methods to enhance catalytic activity and selectivity.
02 Nanostructured bifunctional catalysts
Nanostructured materials are increasingly used as bifunctional catalysts due to their high surface area and unique properties. These catalysts can be synthesized using various methods, including sol-gel processes, hydrothermal synthesis, and templating approaches. Nanostructured bifunctional catalysts often exhibit improved catalytic activity, stability, and selectivity compared to their bulk counterparts.Expand Specific Solutions03 Bifunctional catalysts for energy applications
Bifunctional catalysts play a crucial role in energy-related applications, such as fuel cells, electrolyzers, and batteries. These catalysts can facilitate both oxygen reduction and evolution reactions, or combine hydrogen evolution and oxidation functionalities. The development of efficient bifunctional catalysts is essential for improving the performance and cost-effectiveness of various energy conversion and storage devices.Expand Specific Solutions04 Supported bifunctional catalysts
Supported bifunctional catalysts consist of active catalytic species dispersed on high-surface-area supports. Common support materials include metal oxides, zeolites, and carbon-based materials. The support not only provides a large surface area for catalytic reactions but can also contribute to the overall catalytic activity through synergistic effects with the active species. Proper selection and modification of support materials can significantly enhance the performance of bifunctional catalysts.Expand Specific Solutions05 Characterization and optimization of bifunctional catalysts
Advanced characterization techniques are employed to understand the structure-activity relationships of bifunctional catalysts. These include spectroscopic methods, microscopy, and in-situ/operando studies. Optimization of bifunctional catalysts involves tuning the composition, morphology, and surface properties to achieve desired catalytic performance. Computational methods and high-throughput screening approaches are increasingly used to accelerate the discovery and development of novel bifunctional catalysts.Expand Specific Solutions
Key Players in Electrocatalyst Research and Industry
The research on bifunctional catalysts for OER and HER is in a rapidly evolving phase, with significant market potential due to the growing interest in renewable energy technologies. The global market for these catalysts is expanding, driven by the increasing demand for efficient water splitting and hydrogen production systems. Technologically, the field is progressing from early-stage research to more advanced development, with companies and institutions like Zhengzhou University, Indian Institute of Technology Madras, and University of Houston leading the way. The competition is intensifying as more players enter the market, including specialized firms like Zn2H2, Inc., which is developing innovative zinc-based hydrogen generation technology. The maturity of bifunctional catalysts varies, with some approaches nearing commercialization while others remain in the experimental stage.
University of Houston
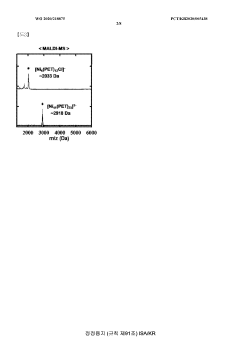
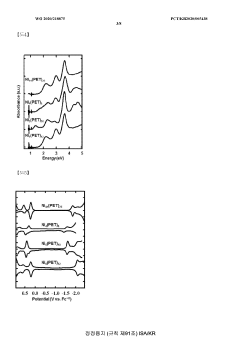
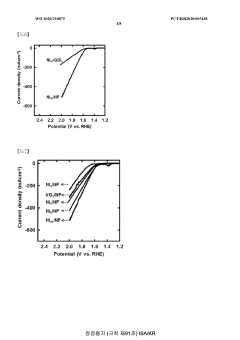
Technical Solution: The University of Houston has developed a novel bifunctional catalyst for both OER and HER based on transition metal phosphides. Their approach involves doping cobalt phosphide with iron to create a highly efficient and stable catalyst. The Fe-doped CoP nanoparticles are synthesized on carbon fiber paper, creating a 3D porous structure that enhances catalytic activity. This catalyst demonstrates excellent performance in both alkaline and acidic media, with low overpotentials for OER and HER. The team has also investigated the mechanism of the bifunctional activity, revealing that the iron doping modifies the electronic structure of CoP, optimizing the binding energies of reaction intermediates[1][3].
Strengths: High efficiency in both OER and HER, stability in various pH conditions, and cost-effective materials. Weaknesses: Potential scalability issues for large-scale production and long-term stability in industrial applications.
Kyushu University
Technical Solution: Kyushu University has developed an innovative approach to bifunctional catalysts for OER and HER using layered double hydroxides (LDHs) as a platform. Their research focuses on creating highly active and stable catalysts through the incorporation of multiple transition metals into the LDH structure. The team has successfully synthesized NiFe-based LDHs with controlled composition and morphology, achieving exceptional bifunctional performance. The catalysts demonstrate low overpotentials for both OER and HER in alkaline media, with values as low as 250 mV for OER and 120 mV for HER at 10 mA cm^-2. Furthermore, they have investigated the synergistic effects between different metal species in the LDH structure, optimizing the electronic configuration and surface properties to enhance catalytic activity[5][6].
Strengths: High catalytic activity, tunable composition, and excellent stability in alkaline conditions. Weaknesses: Limited performance in acidic media and potential challenges in large-scale synthesis.
Core Innovations in OER/HER Bifunctional Catalysts
High Performance Bifunctional Porous Non-Noble Metal Phosphide Catalyst for Overall Water Splitting
PatentPendingUS20230272542A1
Innovation
- A bifunctional electrocatalyst comprising primarily metallic phosphides, specifically iron phosphide (FeP) and dinickel phosphide (Ni2P), is grown on a three-dimensional conductive substrate using a thermal phosphidation method, enabling simultaneous promotion of HER and OER at the cathode and anode, respectively, with a uniform distribution on both electrodes.
Nickel nanocluster bifunctional catalyst for oxygen and hydrogen evolution reactions, and method for producing same
PatentWO2020218875A1
Innovation
- Development of a nickel nanocluster bifunctional catalyst with a specific composition of 41 nickel atoms and 25 organic thiol-based ligands, which is synthesized using a controlled method involving a nickel precursor, organic thiol-based ligand, and reducing agent in a polar aprotic solvent, enhancing stability and uniformity while reducing costs.
Environmental Impact of Catalyst Materials
The environmental impact of catalyst materials used in bifunctional catalysts for OER and HER is a critical consideration in the development and implementation of these technologies. The production, use, and disposal of these materials can have significant implications for ecosystems, resource depletion, and overall sustainability.
Many bifunctional catalysts for OER and HER rely on precious metals such as platinum, iridium, and ruthenium. The mining and extraction of these metals often involve energy-intensive processes and can lead to habitat destruction, soil erosion, and water pollution. Furthermore, the limited availability of these resources raises concerns about long-term sustainability and potential supply chain disruptions.
To address these issues, researchers are increasingly focusing on developing catalysts based on earth-abundant elements. Transition metal compounds, such as nickel, iron, and cobalt-based materials, have shown promising performance while potentially reducing environmental impact. However, the synthesis of these materials may still involve the use of hazardous chemicals or energy-intensive processes, which must be carefully managed.
The lifecycle of catalyst materials is another important aspect to consider. The durability and stability of catalysts directly influence their environmental footprint. More stable catalysts require less frequent replacement, reducing waste generation and the need for additional resource extraction. Efforts to improve catalyst longevity through innovative design and protective coatings can significantly mitigate environmental impacts.
Nanomaterials used in advanced catalysts present unique environmental challenges. While they offer enhanced catalytic activity due to their high surface area, the potential release of nanoparticles into the environment raises concerns about ecotoxicity and bioaccumulation. Proper containment and end-of-life management strategies are essential to prevent unintended environmental consequences.
Water consumption and quality are also important factors to consider. Some catalyst materials may leach ions or compounds into water during operation, potentially affecting aquatic ecosystems. Additionally, the production of catalysts often requires significant amounts of water, which can strain local water resources, especially in water-scarce regions.
As the field progresses, there is a growing emphasis on green chemistry principles in catalyst design and synthesis. This includes the use of less toxic precursors, room-temperature synthesis methods, and the exploration of bio-inspired catalysts. These approaches aim to minimize the environmental footprint of catalyst production while maintaining or improving catalytic performance.
In conclusion, the environmental impact of catalyst materials for OER and HER is a multifaceted issue that requires careful consideration throughout the research, development, and implementation stages. Balancing performance requirements with environmental sustainability will be crucial for the widespread adoption of these technologies in clean energy applications.
Many bifunctional catalysts for OER and HER rely on precious metals such as platinum, iridium, and ruthenium. The mining and extraction of these metals often involve energy-intensive processes and can lead to habitat destruction, soil erosion, and water pollution. Furthermore, the limited availability of these resources raises concerns about long-term sustainability and potential supply chain disruptions.
To address these issues, researchers are increasingly focusing on developing catalysts based on earth-abundant elements. Transition metal compounds, such as nickel, iron, and cobalt-based materials, have shown promising performance while potentially reducing environmental impact. However, the synthesis of these materials may still involve the use of hazardous chemicals or energy-intensive processes, which must be carefully managed.
The lifecycle of catalyst materials is another important aspect to consider. The durability and stability of catalysts directly influence their environmental footprint. More stable catalysts require less frequent replacement, reducing waste generation and the need for additional resource extraction. Efforts to improve catalyst longevity through innovative design and protective coatings can significantly mitigate environmental impacts.
Nanomaterials used in advanced catalysts present unique environmental challenges. While they offer enhanced catalytic activity due to their high surface area, the potential release of nanoparticles into the environment raises concerns about ecotoxicity and bioaccumulation. Proper containment and end-of-life management strategies are essential to prevent unintended environmental consequences.
Water consumption and quality are also important factors to consider. Some catalyst materials may leach ions or compounds into water during operation, potentially affecting aquatic ecosystems. Additionally, the production of catalysts often requires significant amounts of water, which can strain local water resources, especially in water-scarce regions.
As the field progresses, there is a growing emphasis on green chemistry principles in catalyst design and synthesis. This includes the use of less toxic precursors, room-temperature synthesis methods, and the exploration of bio-inspired catalysts. These approaches aim to minimize the environmental footprint of catalyst production while maintaining or improving catalytic performance.
In conclusion, the environmental impact of catalyst materials for OER and HER is a multifaceted issue that requires careful consideration throughout the research, development, and implementation stages. Balancing performance requirements with environmental sustainability will be crucial for the widespread adoption of these technologies in clean energy applications.
Scalability and Cost Analysis of Bifunctional Catalysts
The scalability and cost analysis of bifunctional catalysts for OER and HER is a critical aspect in determining their practical viability for large-scale applications. As research progresses in this field, it is essential to evaluate the potential for scaling up production and the associated economic factors.
One of the primary considerations in scaling bifunctional catalysts is the availability and cost of raw materials. Many high-performance catalysts rely on precious metals or rare earth elements, which can significantly impact the overall cost structure. As production scales up, the demand for these materials may increase, potentially leading to supply chain challenges and price fluctuations.
Manufacturing processes play a crucial role in the scalability of bifunctional catalysts. Current laboratory-scale synthesis methods may not be directly transferable to industrial-scale production. Developing efficient, reproducible, and cost-effective manufacturing techniques is essential for successful commercialization. This may involve optimizing reaction conditions, exploring alternative synthesis routes, or adapting existing industrial processes to accommodate the unique requirements of bifunctional catalysts.
The stability and durability of bifunctional catalysts under real-world operating conditions are vital factors affecting their long-term cost-effectiveness. Catalysts that degrade quickly or lose activity over time may require frequent replacement, increasing operational costs. Improving catalyst stability through innovative design strategies and materials engineering can significantly enhance the economic viability of large-scale implementations.
Energy efficiency is another critical aspect of scalability and cost analysis. Bifunctional catalysts that can operate at lower overpotentials and achieve higher current densities can reduce overall energy consumption, leading to improved economic performance in industrial applications. Balancing catalyst performance with energy efficiency is crucial for optimizing the cost-benefit ratio.
The integration of bifunctional catalysts into existing or new electrolyzer designs presents both opportunities and challenges for scalability. Adapting current electrolyzer architectures to accommodate bifunctional catalysts may require significant engineering efforts and capital investments. However, successful integration could lead to simplified system designs and reduced overall costs.
Environmental considerations and regulatory compliance also factor into the scalability and cost analysis of bifunctional catalysts. As production scales up, ensuring environmentally friendly manufacturing processes and minimizing the use of toxic materials become increasingly important. Compliance with regulations and obtaining necessary certifications can impact both the timeline and cost of scaling up production.
In conclusion, the scalability and cost analysis of bifunctional catalysts for OER and HER involves a complex interplay of factors, including raw material availability, manufacturing processes, catalyst stability, energy efficiency, system integration, and environmental considerations. Addressing these challenges will be crucial for realizing the full potential of bifunctional catalysts in large-scale applications and driving the transition towards sustainable hydrogen production technologies.
One of the primary considerations in scaling bifunctional catalysts is the availability and cost of raw materials. Many high-performance catalysts rely on precious metals or rare earth elements, which can significantly impact the overall cost structure. As production scales up, the demand for these materials may increase, potentially leading to supply chain challenges and price fluctuations.
Manufacturing processes play a crucial role in the scalability of bifunctional catalysts. Current laboratory-scale synthesis methods may not be directly transferable to industrial-scale production. Developing efficient, reproducible, and cost-effective manufacturing techniques is essential for successful commercialization. This may involve optimizing reaction conditions, exploring alternative synthesis routes, or adapting existing industrial processes to accommodate the unique requirements of bifunctional catalysts.
The stability and durability of bifunctional catalysts under real-world operating conditions are vital factors affecting their long-term cost-effectiveness. Catalysts that degrade quickly or lose activity over time may require frequent replacement, increasing operational costs. Improving catalyst stability through innovative design strategies and materials engineering can significantly enhance the economic viability of large-scale implementations.
Energy efficiency is another critical aspect of scalability and cost analysis. Bifunctional catalysts that can operate at lower overpotentials and achieve higher current densities can reduce overall energy consumption, leading to improved economic performance in industrial applications. Balancing catalyst performance with energy efficiency is crucial for optimizing the cost-benefit ratio.
The integration of bifunctional catalysts into existing or new electrolyzer designs presents both opportunities and challenges for scalability. Adapting current electrolyzer architectures to accommodate bifunctional catalysts may require significant engineering efforts and capital investments. However, successful integration could lead to simplified system designs and reduced overall costs.
Environmental considerations and regulatory compliance also factor into the scalability and cost analysis of bifunctional catalysts. As production scales up, ensuring environmentally friendly manufacturing processes and minimizing the use of toxic materials become increasingly important. Compliance with regulations and obtaining necessary certifications can impact both the timeline and cost of scaling up production.
In conclusion, the scalability and cost analysis of bifunctional catalysts for OER and HER involves a complex interplay of factors, including raw material availability, manufacturing processes, catalyst stability, energy efficiency, system integration, and environmental considerations. Addressing these challenges will be crucial for realizing the full potential of bifunctional catalysts in large-scale applications and driving the transition towards sustainable hydrogen production technologies.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!