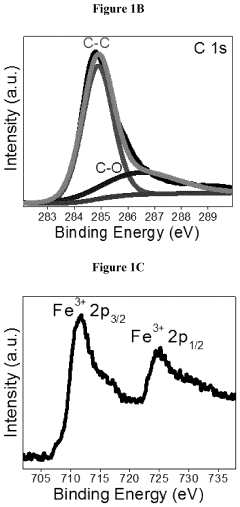
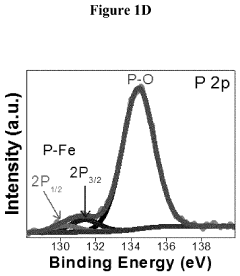
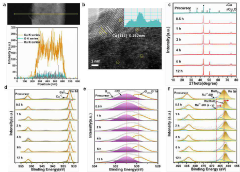
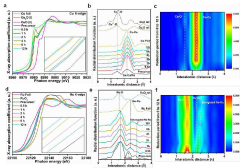
Transition Metal Phosphides as HER Catalysts: Synthesis and Performance Tuning
AUG 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
HER Catalyst Evolution
The evolution of hydrogen evolution reaction (HER) catalysts has been a journey of continuous innovation and improvement. Initially, platinum-based catalysts dominated the field due to their exceptional catalytic activity. However, the scarcity and high cost of platinum prompted researchers to explore alternative materials.
In the early stages, researchers focused on developing non-noble metal catalysts, such as nickel and cobalt-based materials. These catalysts showed promising activity but often suffered from stability issues in acidic environments. The discovery of molybdenum disulfide (MoS2) as an effective HER catalyst in the 1970s marked a significant milestone, opening up new possibilities for transition metal-based catalysts.
The next major breakthrough came with the exploration of transition metal phosphides. Researchers found that incorporating phosphorus into transition metals could significantly enhance their catalytic performance. This led to the development of various transition metal phosphides, including nickel phosphide (Ni2P), cobalt phosphide (CoP), and iron phosphide (FeP), among others.
As the field progressed, scientists began to focus on nanostructuring these materials to maximize their active surface area and expose more catalytic sites. This approach led to the creation of nanoparticles, nanosheets, and other complex nanostructures with improved catalytic performance.
The advent of advanced characterization techniques, such as in-situ spectroscopy and high-resolution electron microscopy, allowed researchers to gain deeper insights into the catalytic mechanisms of these materials. This knowledge facilitated the rational design of more efficient catalysts through strategies such as doping, defect engineering, and heterostructure formation.
Recent years have seen a shift towards multifunctional catalysts that can perform HER alongside other reactions, such as oxygen evolution or CO2 reduction. This trend aligns with the growing interest in integrated energy conversion and storage systems.
The latest frontier in HER catalyst evolution involves the application of artificial intelligence and machine learning techniques for materials discovery and optimization. These approaches are enabling researchers to rapidly screen vast numbers of potential catalyst compositions and predict their performance, accelerating the development of novel and highly efficient HER catalysts.
Throughout this evolution, the overarching goal has remained consistent: to develop catalysts that can match or exceed the performance of platinum while being more abundant, cost-effective, and durable. The journey of HER catalyst evolution continues, with ongoing efforts to push the boundaries of efficiency, stability, and scalability in the pursuit of sustainable hydrogen production technologies.
In the early stages, researchers focused on developing non-noble metal catalysts, such as nickel and cobalt-based materials. These catalysts showed promising activity but often suffered from stability issues in acidic environments. The discovery of molybdenum disulfide (MoS2) as an effective HER catalyst in the 1970s marked a significant milestone, opening up new possibilities for transition metal-based catalysts.
The next major breakthrough came with the exploration of transition metal phosphides. Researchers found that incorporating phosphorus into transition metals could significantly enhance their catalytic performance. This led to the development of various transition metal phosphides, including nickel phosphide (Ni2P), cobalt phosphide (CoP), and iron phosphide (FeP), among others.
As the field progressed, scientists began to focus on nanostructuring these materials to maximize their active surface area and expose more catalytic sites. This approach led to the creation of nanoparticles, nanosheets, and other complex nanostructures with improved catalytic performance.
The advent of advanced characterization techniques, such as in-situ spectroscopy and high-resolution electron microscopy, allowed researchers to gain deeper insights into the catalytic mechanisms of these materials. This knowledge facilitated the rational design of more efficient catalysts through strategies such as doping, defect engineering, and heterostructure formation.
Recent years have seen a shift towards multifunctional catalysts that can perform HER alongside other reactions, such as oxygen evolution or CO2 reduction. This trend aligns with the growing interest in integrated energy conversion and storage systems.
The latest frontier in HER catalyst evolution involves the application of artificial intelligence and machine learning techniques for materials discovery and optimization. These approaches are enabling researchers to rapidly screen vast numbers of potential catalyst compositions and predict their performance, accelerating the development of novel and highly efficient HER catalysts.
Throughout this evolution, the overarching goal has remained consistent: to develop catalysts that can match or exceed the performance of platinum while being more abundant, cost-effective, and durable. The journey of HER catalyst evolution continues, with ongoing efforts to push the boundaries of efficiency, stability, and scalability in the pursuit of sustainable hydrogen production technologies.
Market Demand Analysis
The market demand for transition metal phosphides as hydrogen evolution reaction (HER) catalysts has been steadily increasing in recent years, driven by the growing interest in clean energy technologies and the push for sustainable hydrogen production. This demand is primarily fueled by the global shift towards renewable energy sources and the need for efficient water splitting catalysts in electrolyzers.
The hydrogen economy is projected to play a significant role in the future energy landscape, with the global hydrogen generation market expected to grow substantially over the next decade. Transition metal phosphides, as cost-effective alternatives to precious metal catalysts, are well-positioned to capture a considerable share of this expanding market.
In the industrial sector, there is a rising demand for high-performance HER catalysts that can operate efficiently under various pH conditions. Transition metal phosphides have shown promising results in both acidic and alkaline environments, making them attractive for a wide range of applications. This versatility has led to increased interest from manufacturers of water electrolyzers and fuel cells.
The automotive industry, particularly in the development of fuel cell electric vehicles (FCEVs), represents another significant market for HER catalysts. As major automakers invest in hydrogen-powered vehicles, the demand for efficient and durable catalysts for on-board hydrogen production is expected to rise.
In the renewable energy sector, the integration of hydrogen production with intermittent energy sources like solar and wind power has created a new market opportunity for HER catalysts. Transition metal phosphides are being explored for their potential in enhancing the efficiency of power-to-gas systems, which convert excess renewable electricity into hydrogen for storage and later use.
The research and development sector also contributes to the market demand, with academic institutions and industrial laboratories seeking novel materials for advanced energy conversion systems. This has led to increased funding for projects focused on synthesizing and optimizing transition metal phosphide catalysts.
Geographically, the market demand is particularly strong in regions with ambitious hydrogen strategies, such as the European Union, Japan, and South Korea. These regions are investing heavily in hydrogen infrastructure and technologies, creating a favorable environment for the adoption of advanced HER catalysts.
However, the market demand is not without challenges. The scalability of synthesis methods for transition metal phosphides and the long-term stability of these catalysts under industrial conditions are areas that require further development to meet commercial expectations. Additionally, competition from other emerging catalyst materials and the ongoing improvements in traditional platinum-based catalysts may impact the market penetration of transition metal phosphides.
The hydrogen economy is projected to play a significant role in the future energy landscape, with the global hydrogen generation market expected to grow substantially over the next decade. Transition metal phosphides, as cost-effective alternatives to precious metal catalysts, are well-positioned to capture a considerable share of this expanding market.
In the industrial sector, there is a rising demand for high-performance HER catalysts that can operate efficiently under various pH conditions. Transition metal phosphides have shown promising results in both acidic and alkaline environments, making them attractive for a wide range of applications. This versatility has led to increased interest from manufacturers of water electrolyzers and fuel cells.
The automotive industry, particularly in the development of fuel cell electric vehicles (FCEVs), represents another significant market for HER catalysts. As major automakers invest in hydrogen-powered vehicles, the demand for efficient and durable catalysts for on-board hydrogen production is expected to rise.
In the renewable energy sector, the integration of hydrogen production with intermittent energy sources like solar and wind power has created a new market opportunity for HER catalysts. Transition metal phosphides are being explored for their potential in enhancing the efficiency of power-to-gas systems, which convert excess renewable electricity into hydrogen for storage and later use.
The research and development sector also contributes to the market demand, with academic institutions and industrial laboratories seeking novel materials for advanced energy conversion systems. This has led to increased funding for projects focused on synthesizing and optimizing transition metal phosphide catalysts.
Geographically, the market demand is particularly strong in regions with ambitious hydrogen strategies, such as the European Union, Japan, and South Korea. These regions are investing heavily in hydrogen infrastructure and technologies, creating a favorable environment for the adoption of advanced HER catalysts.
However, the market demand is not without challenges. The scalability of synthesis methods for transition metal phosphides and the long-term stability of these catalysts under industrial conditions are areas that require further development to meet commercial expectations. Additionally, competition from other emerging catalyst materials and the ongoing improvements in traditional platinum-based catalysts may impact the market penetration of transition metal phosphides.
TMP Synthesis Challenges
The synthesis of transition metal phosphides (TMPs) as hydrogen evolution reaction (HER) catalysts presents several significant challenges that researchers must overcome to optimize their performance and scalability. One of the primary difficulties lies in controlling the composition and stoichiometry of the final product. The precise ratio of transition metal to phosphorus is crucial for achieving optimal catalytic activity, yet maintaining this balance during synthesis can be complex due to the varying reactivity of precursors and the potential for multiple phases to form.
Another major challenge is the control of particle size and morphology. The catalytic activity of TMPs is heavily influenced by their surface area and exposed active sites. Achieving uniform nanostructures with high surface-to-volume ratios is essential for maximizing catalytic efficiency. However, the high-temperature conditions often required for TMP synthesis can lead to particle agglomeration and growth, reducing the overall surface area and, consequently, the catalytic performance.
The choice of synthesis method also presents challenges. While solid-state reactions can produce bulk TMPs, they often result in large particles with limited surface area. Solution-based methods, such as solvothermal synthesis, offer better control over particle size and morphology but may introduce impurities or result in incomplete reactions. Balancing reaction conditions to achieve complete conversion while maintaining desired nanostructures remains a significant hurdle.
Furthermore, the incorporation of dopants or the creation of multi-metal phosphides to enhance catalytic activity introduces additional complexity to the synthesis process. Achieving homogeneous distribution of dopants or maintaining the desired ratios in multi-metal systems requires careful precursor selection and reaction control.
The stability of TMPs during synthesis and subsequent processing is another critical challenge. Many TMPs are sensitive to oxidation, which can occur rapidly upon exposure to air or moisture. This necessitates careful handling and storage procedures, as well as the development of protective strategies to maintain the integrity of the catalysts.
Scalability of synthesis methods poses a significant challenge for industrial applications. While laboratory-scale syntheses can produce high-quality TMPs, translating these processes to large-scale production while maintaining performance and cost-effectiveness is complex. Issues such as heat and mass transfer limitations, reactor design, and process control become increasingly important at larger scales.
Lastly, the environmental impact and sustainability of TMP synthesis methods are growing concerns. Many current synthesis routes involve the use of toxic precursors or generate hazardous by-products. Developing green synthesis methods that utilize benign reagents and minimize waste production is crucial for the widespread adoption of TMP catalysts in sustainable hydrogen production technologies.
Another major challenge is the control of particle size and morphology. The catalytic activity of TMPs is heavily influenced by their surface area and exposed active sites. Achieving uniform nanostructures with high surface-to-volume ratios is essential for maximizing catalytic efficiency. However, the high-temperature conditions often required for TMP synthesis can lead to particle agglomeration and growth, reducing the overall surface area and, consequently, the catalytic performance.
The choice of synthesis method also presents challenges. While solid-state reactions can produce bulk TMPs, they often result in large particles with limited surface area. Solution-based methods, such as solvothermal synthesis, offer better control over particle size and morphology but may introduce impurities or result in incomplete reactions. Balancing reaction conditions to achieve complete conversion while maintaining desired nanostructures remains a significant hurdle.
Furthermore, the incorporation of dopants or the creation of multi-metal phosphides to enhance catalytic activity introduces additional complexity to the synthesis process. Achieving homogeneous distribution of dopants or maintaining the desired ratios in multi-metal systems requires careful precursor selection and reaction control.
The stability of TMPs during synthesis and subsequent processing is another critical challenge. Many TMPs are sensitive to oxidation, which can occur rapidly upon exposure to air or moisture. This necessitates careful handling and storage procedures, as well as the development of protective strategies to maintain the integrity of the catalysts.
Scalability of synthesis methods poses a significant challenge for industrial applications. While laboratory-scale syntheses can produce high-quality TMPs, translating these processes to large-scale production while maintaining performance and cost-effectiveness is complex. Issues such as heat and mass transfer limitations, reactor design, and process control become increasingly important at larger scales.
Lastly, the environmental impact and sustainability of TMP synthesis methods are growing concerns. Many current synthesis routes involve the use of toxic precursors or generate hazardous by-products. Developing green synthesis methods that utilize benign reagents and minimize waste production is crucial for the widespread adoption of TMP catalysts in sustainable hydrogen production technologies.
Current TMP Synthesis
01 Catalytic performance of transition metal phosphides
Transition metal phosphides exhibit excellent catalytic performance in various applications. They show high activity and selectivity in reactions such as hydrodesulfurization, hydrodenitrogenation, and hydrogenation. The unique electronic structure and surface properties of these materials contribute to their enhanced catalytic abilities.- Catalytic performance of transition metal phosphides: Transition metal phosphides exhibit excellent catalytic performance in various applications. They show high activity and selectivity in reactions such as hydrodesulfurization, hydrodenitrogenation, and hydrogen evolution. The unique electronic structure and surface properties of these materials contribute to their enhanced catalytic abilities.
- Electrochemical performance in energy storage devices: Transition metal phosphides demonstrate promising electrochemical performance in energy storage applications. They are used as electrode materials in batteries and supercapacitors, offering high capacity, good cycling stability, and improved rate capability. The unique structure and composition of these materials contribute to their enhanced electrochemical properties.
- Magnetic properties of transition metal phosphides: Certain transition metal phosphides exhibit interesting magnetic properties, making them suitable for various applications in magnetism and spintronics. These materials can display ferromagnetic, antiferromagnetic, or paramagnetic behavior depending on their composition and structure. The magnetic properties can be tuned by controlling the synthesis conditions and doping.
- Optical and electronic properties: Transition metal phosphides possess unique optical and electronic properties that make them attractive for optoelectronic applications. These materials can exhibit tunable bandgaps, high carrier mobility, and strong light absorption or emission characteristics. Their properties can be tailored through composition control and nanostructuring.
- Synthesis methods and performance optimization: Various synthesis methods have been developed to produce transition metal phosphides with controlled composition, structure, and morphology. These include solid-state reactions, solution-based methods, and vapor deposition techniques. The performance of these materials can be optimized by controlling particle size, introducing defects, and creating composite structures.
02 Electrochemical performance in energy storage devices
Transition metal phosphides demonstrate promising electrochemical performance in energy storage applications. They are used as electrode materials in batteries and supercapacitors, offering high capacity, good cycling stability, and improved rate capability. The incorporation of these materials enhances the overall performance of energy storage devices.Expand Specific Solutions03 Magnetic properties and applications
Certain transition metal phosphides exhibit interesting magnetic properties, making them suitable for various magnetic applications. These materials can display ferromagnetic, antiferromagnetic, or paramagnetic behavior depending on their composition and structure. Their magnetic properties are utilized in areas such as magnetic recording media and magnetic sensors.Expand Specific Solutions04 Synthesis methods and performance optimization
Various synthesis methods are employed to produce transition metal phosphides with optimized performance. These include solid-state reactions, solution-based methods, and vapor deposition techniques. The synthesis conditions and parameters significantly influence the morphology, composition, and ultimately the performance of the resulting materials.Expand Specific Solutions05 Nanostructured transition metal phosphides
Nanostructured forms of transition metal phosphides, such as nanoparticles, nanowires, and nanosheets, exhibit enhanced performance compared to their bulk counterparts. The increased surface area and unique properties at the nanoscale lead to improved catalytic activity, electrochemical performance, and magnetic properties. These nanostructures find applications in various fields, including catalysis, energy storage, and sensing.Expand Specific Solutions
Key Players in TMP HER
The field of Transition Metal Phosphides as HER Catalysts is in a rapidly developing stage, with significant research efforts focused on synthesis and performance optimization. The market for these catalysts is expanding, driven by the growing demand for efficient hydrogen production technologies. While the technology is still maturing, it shows promising potential for commercial applications. Key players in this field include academic institutions like King Abdullah University of Science & Technology, Korea Advanced Institute of Science & Technology, and Zhejiang University, as well as research organizations such as the Agency for Science, Technology & Research. Industrial involvement is also evident, with companies like PetroChina Co., Ltd. and Toyota Motor Europe NV/SA showing interest in this technology, indicating its potential for future commercialization and industrial applications.
King Abdullah University of Science & Technology
Technical Solution: KAUST has developed a novel approach for synthesizing transition metal phosphides (TMPs) as efficient hydrogen evolution reaction (HER) catalysts. Their method involves a two-step process: first, creating metal-organic frameworks (MOFs) as precursors, then converting them to TMPs through phosphidation. This approach allows for precise control over the composition and morphology of the catalysts[1]. They have successfully synthesized various TMPs, including Ni2P, CoP, and FeP, demonstrating excellent HER performance in acidic media. The university has also explored doping strategies to further enhance catalytic activity, such as incorporating trace amounts of platinum into Ni2P nanoparticles, which significantly improved HER performance[2].
Strengths: Precise control over catalyst composition and morphology, high catalytic activity, and innovative doping strategies. Weaknesses: Potential scalability issues for large-scale production and the need for further optimization for alkaline conditions.
Zhejiang University
Technical Solution: Zhejiang University has made significant strides in the development of transition metal phosphides for HER catalysis. Their research focuses on creating hierarchical nanostructures to maximize the active surface area of TMPs. They have successfully synthesized 3D porous Ni2P nanosheets with exceptional HER performance in both acidic and alkaline media[3]. The university has also explored the use of bimetallic phosphides, such as NiCoP, to leverage synergistic effects between different transition metals. Their innovative approach includes the incorporation of carbon-based materials, like graphene, to enhance the conductivity and stability of TMP catalysts[4].
Strengths: High-performance catalysts in both acidic and alkaline conditions, innovative nanostructure design, and successful integration with carbon-based materials. Weaknesses: Potential high production costs and the need for further long-term stability studies.
TMP HER Mechanisms
Transition metal phosphide supported on carbon nanosheets
PatentActiveUS11982006B2
Innovation
- Development of an electrocatalyst comprising nanoparticles of transition metal phosphide supported on ultrathin, interconnected carbon nanosheets, where the metal phosphide nanoparticles have a diameter of 1-30 nm and are dispersed on carbon nanosheets with a thickness of less than 10 nm, enhancing electrochemical activity and stability.
Catalyst for hydrogen evolution reaction and preparing method of the same
PatentActiveKR1020220111344A
Innovation
- A catalyst is developed using a transition metal matrix, such as copper oxide, with adsorbed noble metal atoms like ruthenium, achieved through a cation exchange strategy, providing a cost-effective alternative with enhanced HER performance.
Sustainability Aspects
The sustainability aspects of transition metal phosphides (TMPs) as hydrogen evolution reaction (HER) catalysts are crucial considerations in their development and application. These materials offer promising alternatives to precious metal catalysts, potentially reducing the environmental impact and cost of hydrogen production. However, their sustainability profile encompasses various dimensions that require careful examination.
The synthesis of TMPs often involves energy-intensive processes and the use of hazardous precursors, which can raise environmental concerns. Efforts to develop greener synthesis routes, such as low-temperature methods and the use of renewable precursors, are ongoing to mitigate these issues. Additionally, the scalability of TMP production is a key factor in their long-term sustainability, as large-scale hydrogen production will require substantial quantities of catalysts.
The durability and stability of TMP catalysts in operational conditions are essential for their sustainable use. Prolonged catalyst lifetimes reduce the need for frequent replacements, minimizing waste generation and resource consumption. Research into enhancing the stability of TMPs in acidic and alkaline environments is crucial for improving their overall sustainability profile.
Recycling and end-of-life management of TMP catalysts present both challenges and opportunities. Developing efficient recovery and recycling processes for these materials can significantly reduce their environmental footprint and improve resource utilization. This aspect is particularly important given the potential scarcity of some transition metals used in these catalysts.
The energy efficiency of HER processes using TMP catalysts is another critical sustainability factor. Improvements in catalytic activity and reduced overpotentials contribute to more efficient hydrogen production, potentially lowering the overall energy requirements and associated carbon emissions of the process.
Life cycle assessments (LCAs) of TMP-based HER systems are essential to comprehensively evaluate their sustainability. These assessments should consider raw material extraction, synthesis, use phase, and end-of-life management to provide a holistic view of the environmental impacts and potential benefits compared to conventional catalysts.
The potential for TMPs to enable large-scale, renewable hydrogen production could have far-reaching sustainability implications. By facilitating the transition to a hydrogen-based economy, these catalysts could contribute to reducing global dependence on fossil fuels and mitigating climate change impacts. However, this potential must be balanced against the environmental costs of their production and use.
The synthesis of TMPs often involves energy-intensive processes and the use of hazardous precursors, which can raise environmental concerns. Efforts to develop greener synthesis routes, such as low-temperature methods and the use of renewable precursors, are ongoing to mitigate these issues. Additionally, the scalability of TMP production is a key factor in their long-term sustainability, as large-scale hydrogen production will require substantial quantities of catalysts.
The durability and stability of TMP catalysts in operational conditions are essential for their sustainable use. Prolonged catalyst lifetimes reduce the need for frequent replacements, minimizing waste generation and resource consumption. Research into enhancing the stability of TMPs in acidic and alkaline environments is crucial for improving their overall sustainability profile.
Recycling and end-of-life management of TMP catalysts present both challenges and opportunities. Developing efficient recovery and recycling processes for these materials can significantly reduce their environmental footprint and improve resource utilization. This aspect is particularly important given the potential scarcity of some transition metals used in these catalysts.
The energy efficiency of HER processes using TMP catalysts is another critical sustainability factor. Improvements in catalytic activity and reduced overpotentials contribute to more efficient hydrogen production, potentially lowering the overall energy requirements and associated carbon emissions of the process.
Life cycle assessments (LCAs) of TMP-based HER systems are essential to comprehensively evaluate their sustainability. These assessments should consider raw material extraction, synthesis, use phase, and end-of-life management to provide a holistic view of the environmental impacts and potential benefits compared to conventional catalysts.
The potential for TMPs to enable large-scale, renewable hydrogen production could have far-reaching sustainability implications. By facilitating the transition to a hydrogen-based economy, these catalysts could contribute to reducing global dependence on fossil fuels and mitigating climate change impacts. However, this potential must be balanced against the environmental costs of their production and use.
Economic Feasibility
The economic feasibility of transition metal phosphides as HER catalysts is a critical factor in their potential widespread adoption and commercialization. These materials have shown promising performance in hydrogen evolution reaction (HER) catalysis, but their economic viability must be carefully evaluated.
One of the primary advantages of transition metal phosphides is their relatively low cost compared to traditional noble metal catalysts like platinum. The use of earth-abundant elements such as iron, cobalt, and nickel as the base metals significantly reduces material costs. Additionally, phosphorus is an inexpensive and readily available element, further contributing to the cost-effectiveness of these catalysts.
The synthesis methods for transition metal phosphides have been extensively studied and optimized, leading to more efficient and scalable production processes. Techniques such as phosphidation of metal precursors, solution-based methods, and solid-state reactions have been developed, allowing for potential large-scale manufacturing. This scalability is crucial for reducing production costs and making these catalysts economically competitive.
However, the economic feasibility also depends on the long-term stability and durability of these catalysts under operating conditions. While many transition metal phosphides have demonstrated good stability, further improvements may be necessary to ensure their longevity in industrial applications. The cost of potential catalyst replacement or regeneration must be factored into the overall economic assessment.
The performance of transition metal phosphides in terms of catalytic activity and efficiency directly impacts their economic viability. Higher catalytic activity translates to lower catalyst loadings, reducing material costs and improving overall system efficiency. Ongoing research in performance tuning, such as nanostructuring and doping, has shown promising results in enhancing catalytic activity, potentially improving the economic attractiveness of these materials.
When considering economic feasibility, it is essential to evaluate the entire hydrogen production system, including infrastructure requirements and operational costs. While transition metal phosphides may offer cost advantages as catalysts, the overall economics of hydrogen production depend on factors such as electricity costs, system efficiency, and scale of operation.
In conclusion, transition metal phosphides show promising economic feasibility as HER catalysts due to their low material costs, scalable synthesis methods, and improving performance. However, further advancements in stability, efficiency, and large-scale production techniques are necessary to fully realize their economic potential in industrial hydrogen production applications.
One of the primary advantages of transition metal phosphides is their relatively low cost compared to traditional noble metal catalysts like platinum. The use of earth-abundant elements such as iron, cobalt, and nickel as the base metals significantly reduces material costs. Additionally, phosphorus is an inexpensive and readily available element, further contributing to the cost-effectiveness of these catalysts.
The synthesis methods for transition metal phosphides have been extensively studied and optimized, leading to more efficient and scalable production processes. Techniques such as phosphidation of metal precursors, solution-based methods, and solid-state reactions have been developed, allowing for potential large-scale manufacturing. This scalability is crucial for reducing production costs and making these catalysts economically competitive.
However, the economic feasibility also depends on the long-term stability and durability of these catalysts under operating conditions. While many transition metal phosphides have demonstrated good stability, further improvements may be necessary to ensure their longevity in industrial applications. The cost of potential catalyst replacement or regeneration must be factored into the overall economic assessment.
The performance of transition metal phosphides in terms of catalytic activity and efficiency directly impacts their economic viability. Higher catalytic activity translates to lower catalyst loadings, reducing material costs and improving overall system efficiency. Ongoing research in performance tuning, such as nanostructuring and doping, has shown promising results in enhancing catalytic activity, potentially improving the economic attractiveness of these materials.
When considering economic feasibility, it is essential to evaluate the entire hydrogen production system, including infrastructure requirements and operational costs. While transition metal phosphides may offer cost advantages as catalysts, the overall economics of hydrogen production depend on factors such as electricity costs, system efficiency, and scale of operation.
In conclusion, transition metal phosphides show promising economic feasibility as HER catalysts due to their low material costs, scalable synthesis methods, and improving performance. However, further advancements in stability, efficiency, and large-scale production techniques are necessary to fully realize their economic potential in industrial hydrogen production applications.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!