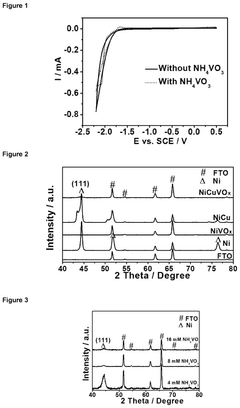
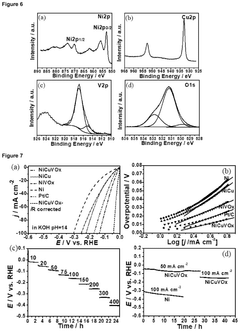
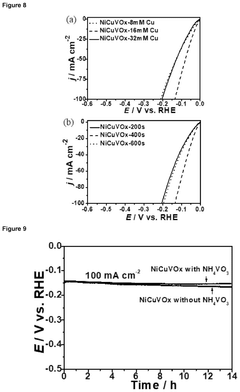
How to Benchmark Catalyst Activity for the Hydrogen Evolution Reaction (HER)
AUG 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
HER Catalyst Benchmarking: Background and Objectives
The Hydrogen Evolution Reaction (HER) is a critical process in electrochemical water splitting, playing a pivotal role in the production of clean hydrogen fuel. As global efforts to transition towards sustainable energy sources intensify, the development of efficient and cost-effective HER catalysts has become a focal point of research. The benchmarking of catalyst activity for HER is essential to evaluate and compare the performance of different materials, guiding the design and optimization of next-generation catalysts.
The primary objective of HER catalyst benchmarking is to establish standardized methods for assessing catalytic activity, enabling reliable comparisons across various research efforts. This standardization is crucial for accelerating progress in the field and facilitating the translation of laboratory discoveries into practical applications. By providing a common framework for evaluation, benchmarking allows researchers to identify promising catalysts and understand the fundamental principles governing their performance.
Historically, the development of HER catalysts has evolved from noble metal-based materials, such as platinum, to earth-abundant alternatives. This shift has been driven by the need for scalable and economically viable solutions for large-scale hydrogen production. The benchmarking process has accordingly adapted to encompass a wider range of materials and to consider factors beyond mere catalytic activity, including stability, scalability, and cost-effectiveness.
Current benchmarking approaches typically involve electrochemical measurements to determine key parameters such as overpotential, Tafel slope, and exchange current density. These metrics provide insights into the catalytic efficiency and reaction kinetics. However, the complexity of the HER process and the diversity of catalyst materials necessitate a multifaceted approach to benchmarking, incorporating advanced characterization techniques and theoretical modeling.
As the field progresses, there is a growing recognition of the need to standardize testing conditions and reporting practices. This includes considerations such as electrode preparation, electrolyte composition, and measurement protocols. Standardization efforts aim to address discrepancies in reported performance metrics and enhance the reproducibility of results across different laboratories.
Looking forward, the objectives of HER catalyst benchmarking are expanding to include in-situ and operando studies, which provide deeper insights into catalyst behavior under realistic operating conditions. Additionally, there is an increasing focus on integrating benchmarking practices with computational methods to accelerate catalyst discovery and optimization. These advancements are expected to drive the development of more efficient, durable, and scalable HER catalysts, ultimately contributing to the realization of a hydrogen-based clean energy economy.
The primary objective of HER catalyst benchmarking is to establish standardized methods for assessing catalytic activity, enabling reliable comparisons across various research efforts. This standardization is crucial for accelerating progress in the field and facilitating the translation of laboratory discoveries into practical applications. By providing a common framework for evaluation, benchmarking allows researchers to identify promising catalysts and understand the fundamental principles governing their performance.
Historically, the development of HER catalysts has evolved from noble metal-based materials, such as platinum, to earth-abundant alternatives. This shift has been driven by the need for scalable and economically viable solutions for large-scale hydrogen production. The benchmarking process has accordingly adapted to encompass a wider range of materials and to consider factors beyond mere catalytic activity, including stability, scalability, and cost-effectiveness.
Current benchmarking approaches typically involve electrochemical measurements to determine key parameters such as overpotential, Tafel slope, and exchange current density. These metrics provide insights into the catalytic efficiency and reaction kinetics. However, the complexity of the HER process and the diversity of catalyst materials necessitate a multifaceted approach to benchmarking, incorporating advanced characterization techniques and theoretical modeling.
As the field progresses, there is a growing recognition of the need to standardize testing conditions and reporting practices. This includes considerations such as electrode preparation, electrolyte composition, and measurement protocols. Standardization efforts aim to address discrepancies in reported performance metrics and enhance the reproducibility of results across different laboratories.
Looking forward, the objectives of HER catalyst benchmarking are expanding to include in-situ and operando studies, which provide deeper insights into catalyst behavior under realistic operating conditions. Additionally, there is an increasing focus on integrating benchmarking practices with computational methods to accelerate catalyst discovery and optimization. These advancements are expected to drive the development of more efficient, durable, and scalable HER catalysts, ultimately contributing to the realization of a hydrogen-based clean energy economy.
Market Analysis for HER Catalysts
The market for hydrogen evolution reaction (HER) catalysts is experiencing significant growth, driven by the increasing demand for clean energy solutions and the global push towards a hydrogen-based economy. As the world seeks to reduce carbon emissions and transition away from fossil fuels, hydrogen production through water electrolysis has emerged as a promising technology. This has created a substantial market opportunity for efficient and cost-effective HER catalysts.
The HER catalyst market is closely tied to the broader hydrogen production industry, which is projected to grow exponentially in the coming years. Major industrial players, including chemical manufacturers, energy companies, and automotive firms, are investing heavily in hydrogen technologies, creating a robust demand for high-performance catalysts. Additionally, government initiatives and funding programs aimed at promoting hydrogen as a clean energy carrier are further stimulating market growth.
Currently, platinum-based catalysts dominate the HER market due to their superior catalytic activity and stability. However, the high cost and scarcity of platinum have led to intensive research efforts to develop alternative catalysts based on earth-abundant materials. This has opened up new market segments for non-precious metal catalysts, such as those based on nickel, molybdenum, and cobalt compounds.
The market for HER catalysts can be segmented based on material type, application, and geography. Material-wise, the market includes precious metal catalysts, non-precious metal catalysts, and hybrid systems. Application segments encompass industrial hydrogen production, fuel cells, and research and development. Geographically, North America, Europe, and Asia-Pacific are the key regions driving market growth, with China emerging as a major player in both production and consumption.
Key market trends include the development of nanostructured catalysts to enhance surface area and catalytic activity, the integration of catalysts with novel support materials to improve stability and conductivity, and the exploration of bio-inspired catalysts mimicking natural hydrogen-producing enzymes. These innovations are expected to expand the market and potentially disrupt current pricing structures.
The competitive landscape of the HER catalyst market is characterized by a mix of established chemical companies, specialized catalyst manufacturers, and innovative startups. Collaborations between academic institutions and industry players are common, fostering rapid technology transfer and commercialization of new catalyst designs. As benchmarking methods for catalyst activity become more standardized, competition is likely to intensify, driving further innovation and cost reduction in the sector.
The HER catalyst market is closely tied to the broader hydrogen production industry, which is projected to grow exponentially in the coming years. Major industrial players, including chemical manufacturers, energy companies, and automotive firms, are investing heavily in hydrogen technologies, creating a robust demand for high-performance catalysts. Additionally, government initiatives and funding programs aimed at promoting hydrogen as a clean energy carrier are further stimulating market growth.
Currently, platinum-based catalysts dominate the HER market due to their superior catalytic activity and stability. However, the high cost and scarcity of platinum have led to intensive research efforts to develop alternative catalysts based on earth-abundant materials. This has opened up new market segments for non-precious metal catalysts, such as those based on nickel, molybdenum, and cobalt compounds.
The market for HER catalysts can be segmented based on material type, application, and geography. Material-wise, the market includes precious metal catalysts, non-precious metal catalysts, and hybrid systems. Application segments encompass industrial hydrogen production, fuel cells, and research and development. Geographically, North America, Europe, and Asia-Pacific are the key regions driving market growth, with China emerging as a major player in both production and consumption.
Key market trends include the development of nanostructured catalysts to enhance surface area and catalytic activity, the integration of catalysts with novel support materials to improve stability and conductivity, and the exploration of bio-inspired catalysts mimicking natural hydrogen-producing enzymes. These innovations are expected to expand the market and potentially disrupt current pricing structures.
The competitive landscape of the HER catalyst market is characterized by a mix of established chemical companies, specialized catalyst manufacturers, and innovative startups. Collaborations between academic institutions and industry players are common, fostering rapid technology transfer and commercialization of new catalyst designs. As benchmarking methods for catalyst activity become more standardized, competition is likely to intensify, driving further innovation and cost reduction in the sector.
Current Challenges in HER Catalyst Evaluation
Despite significant advancements in hydrogen evolution reaction (HER) catalysis, several challenges persist in accurately evaluating catalyst activity. One of the primary issues is the lack of standardized testing protocols, leading to inconsistencies in reported performance metrics across different research groups. This makes it difficult to compare catalysts and assess their true potential for practical applications.
The complexity of the HER process itself poses another challenge. Factors such as electrode preparation, electrolyte composition, and measurement conditions can significantly influence catalyst performance. Variations in these parameters often result in discrepancies between laboratory-scale experiments and real-world applications, hindering the translation of promising catalysts to industrial settings.
Furthermore, the stability and durability of HER catalysts remain critical concerns. Many studies focus on initial activity measurements but fail to adequately address long-term performance under realistic operating conditions. This gap in understanding limits the ability to predict catalyst lifetimes and assess their economic viability for large-scale hydrogen production.
Another challenge lies in accurately determining the active sites and surface area of catalysts. Traditional methods like BET analysis may not always provide an accurate representation of the electrochemically active surface area, leading to potential overestimation or underestimation of intrinsic catalytic activity. This issue is particularly pronounced for nanostructured materials with complex morphologies.
The influence of mass transport limitations on catalyst performance is often overlooked or inadequately addressed in many studies. Diffusion of reactants and products can significantly affect the observed activity, especially at high current densities. Failure to account for these effects can lead to misinterpretation of catalyst performance and hinder the development of optimized electrode designs.
Additionally, the lack of in-situ and operando characterization techniques limits our understanding of catalyst behavior under reaction conditions. Many studies rely on ex-situ measurements, which may not accurately reflect the dynamic changes occurring at the electrode-electrolyte interface during HER. This knowledge gap impedes the rational design of more efficient catalysts based on structure-activity relationships.
Lastly, the challenge of scaling up promising catalysts from laboratory demonstrations to industrial-scale applications remains a significant hurdle. Factors such as catalyst loading, electrode geometry, and system integration must be carefully considered to maintain high performance at larger scales. Addressing these scaling challenges is crucial for the widespread adoption of HER catalysts in practical hydrogen production systems.
The complexity of the HER process itself poses another challenge. Factors such as electrode preparation, electrolyte composition, and measurement conditions can significantly influence catalyst performance. Variations in these parameters often result in discrepancies between laboratory-scale experiments and real-world applications, hindering the translation of promising catalysts to industrial settings.
Furthermore, the stability and durability of HER catalysts remain critical concerns. Many studies focus on initial activity measurements but fail to adequately address long-term performance under realistic operating conditions. This gap in understanding limits the ability to predict catalyst lifetimes and assess their economic viability for large-scale hydrogen production.
Another challenge lies in accurately determining the active sites and surface area of catalysts. Traditional methods like BET analysis may not always provide an accurate representation of the electrochemically active surface area, leading to potential overestimation or underestimation of intrinsic catalytic activity. This issue is particularly pronounced for nanostructured materials with complex morphologies.
The influence of mass transport limitations on catalyst performance is often overlooked or inadequately addressed in many studies. Diffusion of reactants and products can significantly affect the observed activity, especially at high current densities. Failure to account for these effects can lead to misinterpretation of catalyst performance and hinder the development of optimized electrode designs.
Additionally, the lack of in-situ and operando characterization techniques limits our understanding of catalyst behavior under reaction conditions. Many studies rely on ex-situ measurements, which may not accurately reflect the dynamic changes occurring at the electrode-electrolyte interface during HER. This knowledge gap impedes the rational design of more efficient catalysts based on structure-activity relationships.
Lastly, the challenge of scaling up promising catalysts from laboratory demonstrations to industrial-scale applications remains a significant hurdle. Factors such as catalyst loading, electrode geometry, and system integration must be carefully considered to maintain high performance at larger scales. Addressing these scaling challenges is crucial for the widespread adoption of HER catalysts in practical hydrogen production systems.
Established HER Catalyst Benchmarking Protocols
01 Catalyst composition and preparation
Various methods and compositions for preparing catalysts with enhanced activity are described. These include specific formulations of catalyst components, novel synthesis techniques, and treatments to improve catalyst performance in different chemical processes.- Catalyst composition and preparation: This category focuses on the development and preparation of catalysts with improved activity. It includes methods for synthesizing catalysts with specific compositions, structures, and properties to enhance their performance in various chemical reactions. The preparation techniques may involve novel synthesis routes, the use of specific precursors, or the incorporation of promoters to boost catalyst activity.
- Catalyst support and surface modification: This area deals with the optimization of catalyst supports and surface modifications to enhance catalyst activity. It includes techniques for creating high surface area supports, modifying support materials to improve catalyst dispersion, and developing methods to control the porosity and surface chemistry of the catalyst. These approaches aim to maximize the active site accessibility and stability of the catalyst.
- Catalyst regeneration and activity maintenance: This category covers methods for maintaining and regenerating catalyst activity over time. It includes techniques for preventing catalyst deactivation, removing deposited contaminants, and restoring the original activity of spent catalysts. These approaches may involve in-situ or ex-situ regeneration processes, as well as the development of catalysts with improved resistance to deactivation.
- Catalyst testing and characterization: This area focuses on methods and apparatus for testing and characterizing catalyst activity. It includes the development of standardized testing protocols, high-throughput screening techniques, and advanced analytical methods for evaluating catalyst performance. These approaches aim to provide accurate and reliable data on catalyst activity, selectivity, and stability under various reaction conditions.
- Catalyst application in specific processes: This category deals with the application of catalysts in specific industrial processes to improve efficiency and yield. It includes the development of tailored catalysts for particular reactions, such as polymerization, hydrogenation, or oxidation processes. The focus is on optimizing catalyst activity for specific applications, considering factors such as reaction conditions, feedstock composition, and desired product specifications.
02 Catalyst support materials
The choice and modification of catalyst support materials play a crucial role in determining catalyst activity. Different support materials and their modifications are explored to enhance catalyst performance, stability, and selectivity in various reactions.Expand Specific Solutions03 Catalyst regeneration and reactivation
Techniques for regenerating and reactivating catalysts to maintain or restore their activity are discussed. These methods aim to extend catalyst life and improve overall process efficiency in industrial applications.Expand Specific Solutions04 Catalyst testing and characterization
Various methods for testing and characterizing catalyst activity are presented. These include novel analytical techniques, in-situ measurements, and performance evaluation protocols to assess and optimize catalyst efficiency in different reaction conditions.Expand Specific Solutions05 Catalyst modifications for specific applications
Tailoring catalysts for specific industrial applications to enhance their activity and selectivity is explored. This includes modifications for petrochemical processes, environmental catalysis, and fine chemical synthesis, among others.Expand Specific Solutions
Key Players in HER Catalyst Research and Development
The hydrogen evolution reaction (HER) catalyst benchmarking landscape is characterized by a highly competitive and rapidly evolving field. The market is in a growth phase, driven by increasing demand for clean energy solutions and hydrogen production technologies. While the market size is expanding, it remains relatively niche compared to broader energy sectors. Technologically, HER catalysts are advancing quickly, with research institutions and companies like The Regents of the University of California, Fudan University, and Max Planck Gesellschaft leading innovation. These organizations are developing novel materials and methods to improve catalyst efficiency and durability. The involvement of diverse players, from academic institutions to industrial giants like China Petroleum & Chemical Corp., indicates a maturing field with potential for significant breakthroughs in the near future.
The Regents of the University of California
Technical Solution: The University of California system has developed a comprehensive approach to benchmark HER catalyst activity. Their methodology includes the use of standardized protocols for catalyst synthesis and electrochemical testing to ensure reproducibility across different laboratories[13]. They employ advanced electrochemical techniques, such as rotating ring-disk electrode (RRDE) measurements, to simultaneously monitor HER activity and detect potential side reactions[14]. The university has also pioneered the use of operando spectroscopic techniques, including surface-enhanced Raman spectroscopy (SERS), to study catalyst surface chemistry during HER[15]. Additionally, they have developed machine learning algorithms to analyze large datasets of catalyst properties and performance metrics, enabling the prediction of promising new catalyst materials[16].
Strengths: Standardized protocols for improved reproducibility. Integration of machine learning for data analysis and prediction. Weaknesses: Potential challenges in coordinating research efforts across multiple campuses.
Research & Business Foundation Sungkyunkwan University
Technical Solution: Sungkyunkwan University has developed a systematic approach to benchmark HER catalyst activity. Their methodology includes the use of advanced electrochemical techniques, such as electrochemical quartz crystal microbalance (EQCM), to monitor catalyst mass changes during HER[17]. They have implemented in-situ Raman spectroscopy to study the formation and breakdown of reaction intermediates on catalyst surfaces[18]. The university has also developed novel nanostructured electrode designs to enhance catalyst performance and stability[19]. Additionally, they utilize advanced computational methods, including machine learning-assisted DFT calculations, to predict catalyst activity and guide experimental design[20].
Strengths: Integration of advanced in-situ spectroscopic techniques. Novel electrode designs for enhanced performance. Weaknesses: Potential focus on nanostructured materials may limit applicability to some catalyst systems.
Critical Innovations in HER Activity Measurement
Hydrogen evolution reaction catalyst
PatentActiveUS12297548B2
Innovation
- Development of HER catalysts comprising interspersed metal catalyst species (such as Ni or Co) with vanadium species, which enhances catalytic activity and prevents passivation, and can operate effectively in both alkaline and neutral solutions.
Catalysis of hydrogen evolution reaction using ruthenium ion complexed carbon nitride materials
PatentWO2019070844A1
Innovation
- Incorporation of ruthenium ions into carbon nitride nanosheets, with the addition of graphene oxide, forming a composite that enhances the catalytic activity and stability of the HER process, achieving performance comparable to platinum catalysts with significantly less metal usage.
Standardization Efforts in HER Catalyst Benchmarking
Efforts to standardize benchmarking methods for hydrogen evolution reaction (HER) catalysts have gained significant momentum in recent years. The scientific community recognizes the critical need for consistent and reliable protocols to evaluate catalyst performance accurately. These standardization initiatives aim to address the challenges of comparing results across different research groups and experimental setups.
One of the primary focuses of standardization efforts is the establishment of uniform testing conditions. This includes defining standard electrolyte compositions, pH levels, and temperature ranges for HER catalyst evaluation. By adopting these standardized conditions, researchers can ensure that performance metrics are directly comparable across different studies.
Another key aspect of standardization is the development of agreed-upon performance metrics. While current density at a specific overpotential is commonly used, there is a push to incorporate additional parameters such as turnover frequency, mass activity, and long-term stability measurements. These comprehensive metrics provide a more holistic view of catalyst performance and durability.
Electrode preparation techniques have also been a subject of standardization efforts. Guidelines for catalyst loading, substrate selection, and ink formulation are being established to minimize variations in catalyst film quality and thickness. This standardization helps to isolate the intrinsic catalytic activity from effects related to electrode fabrication.
Reporting standards are another crucial area of focus. Standardization initiatives are working to define a minimum set of data that should be reported in publications, including detailed experimental procedures, raw data, and statistical analysis. This approach enhances reproducibility and facilitates meta-analyses of catalyst performance across multiple studies.
Interlaboratory studies have played a vital role in validating standardized protocols. These collaborative efforts involve multiple research groups performing identical experiments on benchmark catalysts, allowing for the assessment of protocol robustness and inter-laboratory variability. The results of these studies inform further refinements to standardization guidelines.
As standardization efforts progress, there is an increasing emphasis on developing reference materials and calibration standards for HER catalyst benchmarking. These materials serve as controls to validate experimental setups and ensure consistency across different laboratories. The availability of such standards is expected to significantly improve the reliability and comparability of HER catalyst performance data.
One of the primary focuses of standardization efforts is the establishment of uniform testing conditions. This includes defining standard electrolyte compositions, pH levels, and temperature ranges for HER catalyst evaluation. By adopting these standardized conditions, researchers can ensure that performance metrics are directly comparable across different studies.
Another key aspect of standardization is the development of agreed-upon performance metrics. While current density at a specific overpotential is commonly used, there is a push to incorporate additional parameters such as turnover frequency, mass activity, and long-term stability measurements. These comprehensive metrics provide a more holistic view of catalyst performance and durability.
Electrode preparation techniques have also been a subject of standardization efforts. Guidelines for catalyst loading, substrate selection, and ink formulation are being established to minimize variations in catalyst film quality and thickness. This standardization helps to isolate the intrinsic catalytic activity from effects related to electrode fabrication.
Reporting standards are another crucial area of focus. Standardization initiatives are working to define a minimum set of data that should be reported in publications, including detailed experimental procedures, raw data, and statistical analysis. This approach enhances reproducibility and facilitates meta-analyses of catalyst performance across multiple studies.
Interlaboratory studies have played a vital role in validating standardized protocols. These collaborative efforts involve multiple research groups performing identical experiments on benchmark catalysts, allowing for the assessment of protocol robustness and inter-laboratory variability. The results of these studies inform further refinements to standardization guidelines.
As standardization efforts progress, there is an increasing emphasis on developing reference materials and calibration standards for HER catalyst benchmarking. These materials serve as controls to validate experimental setups and ensure consistency across different laboratories. The availability of such standards is expected to significantly improve the reliability and comparability of HER catalyst performance data.
Environmental Impact of HER Catalyst Development
The development of catalysts for the Hydrogen Evolution Reaction (HER) has significant environmental implications that extend beyond their immediate application in hydrogen production. As research in this field progresses, it is crucial to consider the broader environmental impact of catalyst development and implementation.
One of the primary environmental benefits of advancing HER catalyst technology is the potential for more efficient and cost-effective hydrogen production. Improved catalysts can lead to reduced energy requirements for hydrogen generation, thereby decreasing the overall carbon footprint associated with this process. This is particularly important as hydrogen is increasingly viewed as a clean energy carrier and a potential replacement for fossil fuels in various applications.
However, the environmental impact of HER catalyst development is not solely positive. The production of catalysts often involves the use of rare and precious metals, such as platinum, iridium, or ruthenium. The mining and processing of these materials can have substantial environmental consequences, including habitat destruction, water pollution, and greenhouse gas emissions. As research continues, there is a growing emphasis on developing catalysts using more abundant and environmentally friendly materials, such as nickel, iron, or cobalt-based compounds.
The lifecycle assessment of HER catalysts is another critical aspect to consider. This includes evaluating the environmental impact of catalyst synthesis, use, and disposal. Researchers are increasingly focusing on developing catalysts with longer lifespans and improved stability to reduce the frequency of replacement and associated waste generation. Additionally, efforts are being made to design catalysts that can be easily recycled or regenerated, further minimizing their environmental footprint.
Water consumption and quality are also important environmental factors in HER catalyst development. While the HER process itself uses water as a feedstock, the production and testing of catalysts may require significant amounts of water and potentially generate wastewater containing trace metals or other contaminants. Addressing these issues through improved synthesis methods and wastewater treatment techniques is an ongoing area of research.
As the field advances, there is a growing recognition of the need for a holistic approach to catalyst development that considers not only performance metrics but also environmental sustainability. This includes exploring bio-inspired catalysts, utilizing green chemistry principles in synthesis, and investigating the potential for integrating HER catalysts with renewable energy sources to create fully sustainable hydrogen production systems.
One of the primary environmental benefits of advancing HER catalyst technology is the potential for more efficient and cost-effective hydrogen production. Improved catalysts can lead to reduced energy requirements for hydrogen generation, thereby decreasing the overall carbon footprint associated with this process. This is particularly important as hydrogen is increasingly viewed as a clean energy carrier and a potential replacement for fossil fuels in various applications.
However, the environmental impact of HER catalyst development is not solely positive. The production of catalysts often involves the use of rare and precious metals, such as platinum, iridium, or ruthenium. The mining and processing of these materials can have substantial environmental consequences, including habitat destruction, water pollution, and greenhouse gas emissions. As research continues, there is a growing emphasis on developing catalysts using more abundant and environmentally friendly materials, such as nickel, iron, or cobalt-based compounds.
The lifecycle assessment of HER catalysts is another critical aspect to consider. This includes evaluating the environmental impact of catalyst synthesis, use, and disposal. Researchers are increasingly focusing on developing catalysts with longer lifespans and improved stability to reduce the frequency of replacement and associated waste generation. Additionally, efforts are being made to design catalysts that can be easily recycled or regenerated, further minimizing their environmental footprint.
Water consumption and quality are also important environmental factors in HER catalyst development. While the HER process itself uses water as a feedstock, the production and testing of catalysts may require significant amounts of water and potentially generate wastewater containing trace metals or other contaminants. Addressing these issues through improved synthesis methods and wastewater treatment techniques is an ongoing area of research.
As the field advances, there is a growing recognition of the need for a holistic approach to catalyst development that considers not only performance metrics but also environmental sustainability. This includes exploring bio-inspired catalysts, utilizing green chemistry principles in synthesis, and investigating the potential for integrating HER catalysts with renewable energy sources to create fully sustainable hydrogen production systems.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!