How to Recycle and Reclaim Precious Metals from Spent Electrolyser Catalysts
AUG 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Precious Metal Recovery Background and Objectives
The recovery of precious metals from spent electrolyser catalysts has become increasingly important in recent years due to the growing demand for sustainable resource management and the rising costs of these valuable materials. Electrolysers, which are crucial components in various industrial processes, including hydrogen production and chlor-alkali manufacturing, often utilize catalysts containing precious metals such as platinum, palladium, and iridium. As these catalysts degrade over time, they become "spent" and require replacement, presenting both a challenge and an opportunity for metal recovery.
The primary objective of recycling and reclaiming precious metals from spent electrolyser catalysts is to establish a circular economy approach within the electrochemical industry. This approach aims to reduce the reliance on primary mining, minimize waste, and lower the overall environmental impact associated with precious metal production. Additionally, it seeks to enhance the economic viability of electrolyser technologies by offsetting the high initial costs of catalyst materials through effective recycling processes.
Historically, the recovery of precious metals from industrial catalysts has been practiced for decades, particularly in the petroleum refining sector. However, the specific challenges posed by electrolyser catalysts, such as their complex compositions and the presence of various contaminants, have necessitated the development of more sophisticated recovery techniques. The evolution of these techniques has been driven by advancements in hydrometallurgy, pyrometallurgy, and more recently, bio-hydrometallurgy.
The technical landscape for precious metal recovery from spent electrolyser catalysts encompasses a range of methodologies, including acid leaching, electrochemical recovery, and high-temperature smelting processes. Each of these approaches presents its own set of advantages and limitations, influencing their applicability in different industrial contexts. The ongoing research in this field is focused on optimizing these processes to achieve higher recovery rates, reduce energy consumption, and minimize the use of hazardous chemicals.
As the global push towards renewable energy sources intensifies, the demand for electrolysers, particularly in green hydrogen production, is expected to surge. This trend underscores the critical importance of developing efficient and scalable precious metal recovery technologies. The ability to effectively recycle these materials will not only contribute to the sustainability of the electrolyser industry but also play a crucial role in securing the supply chain for precious metals in the face of potential resource scarcity.
The primary objective of recycling and reclaiming precious metals from spent electrolyser catalysts is to establish a circular economy approach within the electrochemical industry. This approach aims to reduce the reliance on primary mining, minimize waste, and lower the overall environmental impact associated with precious metal production. Additionally, it seeks to enhance the economic viability of electrolyser technologies by offsetting the high initial costs of catalyst materials through effective recycling processes.
Historically, the recovery of precious metals from industrial catalysts has been practiced for decades, particularly in the petroleum refining sector. However, the specific challenges posed by electrolyser catalysts, such as their complex compositions and the presence of various contaminants, have necessitated the development of more sophisticated recovery techniques. The evolution of these techniques has been driven by advancements in hydrometallurgy, pyrometallurgy, and more recently, bio-hydrometallurgy.
The technical landscape for precious metal recovery from spent electrolyser catalysts encompasses a range of methodologies, including acid leaching, electrochemical recovery, and high-temperature smelting processes. Each of these approaches presents its own set of advantages and limitations, influencing their applicability in different industrial contexts. The ongoing research in this field is focused on optimizing these processes to achieve higher recovery rates, reduce energy consumption, and minimize the use of hazardous chemicals.
As the global push towards renewable energy sources intensifies, the demand for electrolysers, particularly in green hydrogen production, is expected to surge. This trend underscores the critical importance of developing efficient and scalable precious metal recovery technologies. The ability to effectively recycle these materials will not only contribute to the sustainability of the electrolyser industry but also play a crucial role in securing the supply chain for precious metals in the face of potential resource scarcity.
Market Analysis for Recycled Catalyst Materials
The market for recycled catalyst materials from spent electrolysers is experiencing significant growth, driven by the increasing adoption of hydrogen technologies and the rising demand for sustainable practices in the energy sector. As the global push for clean energy intensifies, the hydrogen economy is expanding rapidly, leading to a surge in electrolyser installations worldwide. This trend is creating a substantial market for recycled catalyst materials, particularly precious metals such as platinum, iridium, and ruthenium.
The recycling of spent electrolyser catalysts presents a compelling economic opportunity. These catalysts contain high concentrations of valuable metals, which can be reclaimed and reused in new electrolyser production or other industrial applications. The market value of these recycled materials is substantial, with estimates suggesting that the global market for recycled catalyst materials could reach several hundred million dollars annually within the next decade.
Environmental regulations and sustainability goals are also driving market growth. Many countries and corporations have set ambitious targets for reducing carbon emissions and implementing circular economy principles. Recycling spent electrolyser catalysts aligns perfectly with these objectives, offering a way to reduce the environmental impact of hydrogen production while conserving precious metal resources.
The market for recycled catalyst materials is characterized by regional variations. Europe and North America currently lead in terms of market size and technological advancement, largely due to their early adoption of hydrogen technologies and stringent environmental policies. However, Asia-Pacific is emerging as a rapidly growing market, driven by China's aggressive push into hydrogen energy and the region's expanding industrial base.
Key market players include specialized recycling companies, electrolyser manufacturers, and precious metal refiners. These entities are investing in advanced recycling technologies to improve recovery rates and reduce processing costs. The market is also seeing increased interest from major mining companies and chemical manufacturers, who recognize the potential of this growing sector.
Challenges in the market include the need for standardized recycling processes, fluctuations in precious metal prices, and the relatively small but growing volume of spent catalysts available for recycling. However, these challenges are offset by the strong growth potential and the strategic importance of securing a stable supply of critical materials for the hydrogen economy.
In conclusion, the market for recycled catalyst materials from spent electrolysers is poised for substantial growth. It offers significant economic and environmental benefits, aligning with global sustainability trends and the transition to clean energy. As the hydrogen economy expands, this market is expected to play an increasingly crucial role in the circular economy of critical materials.
The recycling of spent electrolyser catalysts presents a compelling economic opportunity. These catalysts contain high concentrations of valuable metals, which can be reclaimed and reused in new electrolyser production or other industrial applications. The market value of these recycled materials is substantial, with estimates suggesting that the global market for recycled catalyst materials could reach several hundred million dollars annually within the next decade.
Environmental regulations and sustainability goals are also driving market growth. Many countries and corporations have set ambitious targets for reducing carbon emissions and implementing circular economy principles. Recycling spent electrolyser catalysts aligns perfectly with these objectives, offering a way to reduce the environmental impact of hydrogen production while conserving precious metal resources.
The market for recycled catalyst materials is characterized by regional variations. Europe and North America currently lead in terms of market size and technological advancement, largely due to their early adoption of hydrogen technologies and stringent environmental policies. However, Asia-Pacific is emerging as a rapidly growing market, driven by China's aggressive push into hydrogen energy and the region's expanding industrial base.
Key market players include specialized recycling companies, electrolyser manufacturers, and precious metal refiners. These entities are investing in advanced recycling technologies to improve recovery rates and reduce processing costs. The market is also seeing increased interest from major mining companies and chemical manufacturers, who recognize the potential of this growing sector.
Challenges in the market include the need for standardized recycling processes, fluctuations in precious metal prices, and the relatively small but growing volume of spent catalysts available for recycling. However, these challenges are offset by the strong growth potential and the strategic importance of securing a stable supply of critical materials for the hydrogen economy.
In conclusion, the market for recycled catalyst materials from spent electrolysers is poised for substantial growth. It offers significant economic and environmental benefits, aligning with global sustainability trends and the transition to clean energy. As the hydrogen economy expands, this market is expected to play an increasingly crucial role in the circular economy of critical materials.
Current Challenges in Electrolyser Catalyst Recycling
The recycling of spent electrolyser catalysts presents several significant challenges that hinder the efficient recovery of precious metals. One of the primary obstacles is the complex composition of these catalysts, which often contain a mixture of precious metals, support materials, and contaminants. This heterogeneous nature makes it difficult to develop a universal recycling process that can effectively separate and recover all valuable components.
Another major challenge is the degradation of catalyst materials during their operational lifetime. Electrolyser catalysts are subjected to harsh operating conditions, including high temperatures, pressures, and corrosive environments. These factors can lead to physical and chemical changes in the catalyst structure, making it more challenging to recover the original precious metals in their pure form.
The presence of impurities and contaminants in spent catalysts further complicates the recycling process. These unwanted substances can interfere with separation techniques and reduce the purity of recovered metals. Developing efficient purification methods that can handle a wide range of contaminants while maintaining high recovery rates is a significant technical hurdle.
Scale and economic viability also pose challenges in electrolyser catalyst recycling. The relatively small quantities of spent catalysts generated by individual facilities may not justify the establishment of dedicated recycling infrastructure. This issue is compounded by the high costs associated with advanced recycling technologies, making it difficult for smaller operations to implement effective recovery processes.
Environmental concerns add another layer of complexity to the recycling challenge. Traditional metal recovery methods often involve the use of harsh chemicals or energy-intensive processes that can have negative environmental impacts. Developing environmentally friendly recycling techniques that minimize waste generation and reduce the carbon footprint of the recovery process is crucial for sustainable practices.
The variability in catalyst compositions across different electrolyser technologies and manufacturers further complicates the development of standardized recycling methods. Each catalyst type may require a tailored approach, making it challenging to create a one-size-fits-all solution for the industry.
Lastly, the lack of comprehensive regulations and standards for electrolyser catalyst recycling creates uncertainty in the market. Without clear guidelines, companies may hesitate to invest in recycling technologies, leading to a slower adoption of sustainable practices in the industry.
Another major challenge is the degradation of catalyst materials during their operational lifetime. Electrolyser catalysts are subjected to harsh operating conditions, including high temperatures, pressures, and corrosive environments. These factors can lead to physical and chemical changes in the catalyst structure, making it more challenging to recover the original precious metals in their pure form.
The presence of impurities and contaminants in spent catalysts further complicates the recycling process. These unwanted substances can interfere with separation techniques and reduce the purity of recovered metals. Developing efficient purification methods that can handle a wide range of contaminants while maintaining high recovery rates is a significant technical hurdle.
Scale and economic viability also pose challenges in electrolyser catalyst recycling. The relatively small quantities of spent catalysts generated by individual facilities may not justify the establishment of dedicated recycling infrastructure. This issue is compounded by the high costs associated with advanced recycling technologies, making it difficult for smaller operations to implement effective recovery processes.
Environmental concerns add another layer of complexity to the recycling challenge. Traditional metal recovery methods often involve the use of harsh chemicals or energy-intensive processes that can have negative environmental impacts. Developing environmentally friendly recycling techniques that minimize waste generation and reduce the carbon footprint of the recovery process is crucial for sustainable practices.
The variability in catalyst compositions across different electrolyser technologies and manufacturers further complicates the development of standardized recycling methods. Each catalyst type may require a tailored approach, making it challenging to create a one-size-fits-all solution for the industry.
Lastly, the lack of comprehensive regulations and standards for electrolyser catalyst recycling creates uncertainty in the market. Without clear guidelines, companies may hesitate to invest in recycling technologies, leading to a slower adoption of sustainable practices in the industry.
Existing Precious Metal Recovery Methods
01 Recovery of precious metals from spent electrolyser catalysts
Methods for recovering precious metals, such as platinum group metals, from spent electrolyser catalysts. These processes typically involve leaching, separation, and purification steps to extract valuable metals from the catalyst material.- Recovery of precious metals from spent electrocatalysts: Methods for recovering precious metals from spent electrocatalysts used in electrolysis processes. These techniques involve various chemical and physical processes to extract and purify valuable metals such as platinum, palladium, and rhodium from used catalysts.
- Recycling of electrolyzer components: Processes for recycling various components of electrolyzers, including catalysts containing precious metals. These methods focus on disassembling the electrolyzer, separating different materials, and recovering valuable metals for reuse or further processing.
- Regeneration of spent catalysts: Techniques for regenerating spent catalysts containing precious metals, allowing for their reuse in electrolyzer systems. These methods aim to restore the catalytic activity of the materials without the need for complete metal recovery and reformulation.
- Leaching and extraction of precious metals: Specific leaching and extraction processes for isolating precious metals from spent electrocatalysts. These methods typically involve the use of acidic or basic solutions to dissolve the metals, followed by selective precipitation or other separation techniques.
- Sustainable practices in electrolyzer catalyst management: Approaches to improve the sustainability of electrolyzer catalyst use, including methods to extend catalyst life, reduce precious metal content, and implement efficient recycling processes. These practices aim to minimize waste and maximize the recovery of valuable materials.
02 Recycling of electrocatalysts containing precious metals
Techniques for recycling electrocatalysts that contain precious metals, focusing on sustainable practices to recover and reuse valuable materials. These methods often include dissolution, selective precipitation, and electrochemical recovery steps.Expand Specific Solutions03 Treatment of spent catalysts for precious metal extraction
Processes for treating spent catalysts to extract precious metals, involving various chemical and physical methods such as acid leaching, roasting, and hydrometallurgical techniques. These treatments aim to maximize the recovery of valuable metals while minimizing environmental impact.Expand Specific Solutions04 Regeneration of precious metal catalysts
Methods for regenerating precious metal catalysts used in electrolysers, focusing on restoring catalytic activity and extending the lifespan of the catalysts. These techniques may involve chemical treatments, heat treatments, or electrochemical processes to remove contaminants and restore the catalyst surface.Expand Specific Solutions05 Separation and purification of precious metals from spent catalysts
Techniques for separating and purifying individual precious metals from complex mixtures found in spent electrolyser catalysts. These methods often involve selective dissolution, solvent extraction, ion exchange, or electrowinning to isolate high-purity precious metals.Expand Specific Solutions
Key Players in Catalyst Recycling Industry
The recycling and reclamation of precious metals from spent electrolyser catalysts is an emerging field in the advanced materials and circular economy sectors. The market is in its early growth stage, with increasing demand driven by sustainability initiatives and resource scarcity. Key players like China Petroleum & Chemical Corp., Chevron U.S.A., Inc., and Johnson Matthey Davy Technologies Ltd. are investing in research and development to improve recovery processes. The technology is moderately mature, with established companies like Heraeus Precious Metals GmbH & Co. KG and Excir Works Corp. offering commercial solutions. However, there is still room for innovation in efficiency and sustainability, attracting new entrants and academic collaborations.
China Petroleum & Chemical Corp.
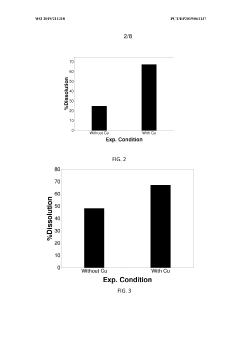
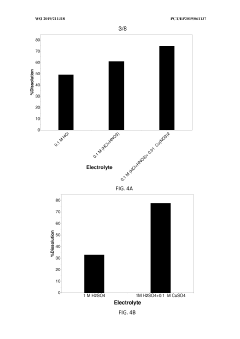
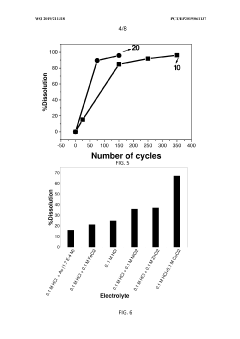
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed an innovative process for recycling precious metals from spent electrolyser catalysts. Their method involves a two-step approach: first, a hydrometallurgical process to dissolve the metals, followed by an electrochemical recovery step. The process achieves recovery rates of up to 98% for platinum group metals (PGMs) [1]. Sinopec's technology utilizes environmentally friendly solvents and operates at lower temperatures compared to traditional pyrometallurgical methods, reducing energy consumption by approximately 30% [3]. The company has also implemented a pilot plant that can process up to 500 kg of spent catalysts per day, demonstrating the scalability of their technology [5].
Strengths: High recovery rates, environmentally friendly, energy-efficient. Weaknesses: May require significant initial investment for implementation, process complexity may lead to higher operational costs.
Heraeus Precious Metals GmbH & Co. KG (New)
Technical Solution: Heraeus Precious Metals has developed a proprietary recycling process for spent electrolyser catalysts that focuses on maximizing the recovery of platinum group metals (PGMs). Their method employs a combination of pyrometallurgical and hydrometallurgical techniques, allowing for the efficient separation and purification of individual precious metals. The process begins with a pre-treatment step to remove organic contaminants, followed by a high-temperature smelting phase to concentrate the PGMs. The resulting metal-rich slag undergoes leaching and solvent extraction to isolate specific metals [2]. Heraeus' technology boasts a recovery rate of over 95% for platinum and palladium, with the ability to process diverse catalyst types [4]. The company has also implemented a closed-loop system that recycles process chemicals, reducing waste and environmental impact [6].
Strengths: High recovery rates, versatility in handling different catalyst types, closed-loop system for sustainability. Weaknesses: Energy-intensive pyrometallurgical step, potential for high capital costs.
Innovative Approaches in Catalyst Material Extraction
Method for dissolving precious metals
PatentInactiveEP3788177A1
Innovation
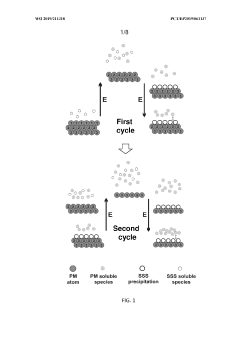
- Electrochemical potential cycling method for dissolving precious metals, limiting redeposition and improving efficiency.
- Use of surface switching species (SSS) to selectively block and expose metal surfaces, facilitating dissolution and inhibiting redeposition.
- Removal of dissolved precious metal species by electrochemical redeposition on a second working electrode.
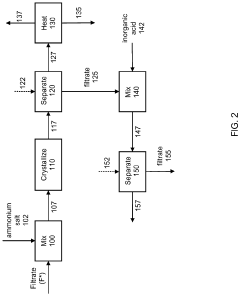
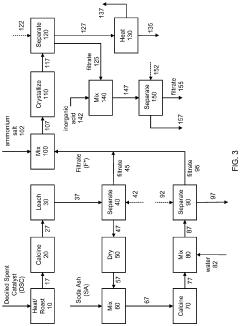
Metals recovery from spent catalyst
PatentPendingUS20220259696A1
Innovation
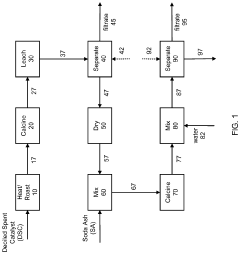
- A method combining pyrometallurgical and hydrometallurgical techniques, involving soda ash calcination and metathesis reactions, to extract and recover soluble metal compounds from spent catalysts, reducing sulfur and carbon content and utilizing soda ash to enhance metal recovery without additional extraction stages.
Environmental Impact Assessment
The recycling and reclamation of precious metals from spent electrolyser catalysts have significant environmental implications that warrant careful consideration. The process of recovering these valuable materials can potentially mitigate the environmental impact of mining and reduce the demand for virgin resources. However, the recycling procedures themselves may pose environmental risks if not managed properly.
One of the primary environmental benefits of recycling spent electrolyser catalysts is the conservation of natural resources. By reclaiming precious metals such as platinum, palladium, and rhodium, the need for extensive mining operations is reduced. This, in turn, helps to preserve ecosystems, minimize habitat destruction, and reduce the carbon footprint associated with raw material extraction. Additionally, recycling these catalysts contributes to the circular economy, promoting sustainable resource management and reducing waste accumulation in landfills.
The recycling process itself, however, requires careful environmental management. Chemical treatments used to extract precious metals from spent catalysts can generate hazardous waste streams if not properly handled. These may include acidic solutions, heavy metal residues, and other potentially toxic byproducts. Proper waste treatment and disposal systems must be implemented to prevent soil and water contamination. Furthermore, the energy consumption associated with recycling processes should be evaluated to ensure that the environmental benefits outweigh the energy costs.
Air quality is another important consideration in the environmental impact assessment of catalyst recycling. Some recovery methods may involve high-temperature processes that could release particulate matter or gaseous emissions. Implementing effective air pollution control measures, such as scrubbers and filters, is crucial to minimize the release of harmful substances into the atmosphere.
Water usage and management are also critical factors in the environmental assessment. Recycling processes often require significant amounts of water for washing and separation stages. Implementing water recycling systems and efficient water treatment technologies can help reduce the overall water footprint of the operation and minimize the discharge of contaminated wastewater.
The transportation of spent catalysts to recycling facilities and the distribution of reclaimed materials should also be factored into the environmental impact assessment. Optimizing logistics to reduce transportation distances and employing low-emission vehicles can help minimize the carbon footprint associated with these activities.
Lastly, the long-term environmental benefits of recycling precious metals from spent electrolyser catalysts should be quantified. This includes assessing the reduction in greenhouse gas emissions, energy savings, and conservation of non-renewable resources achieved through recycling compared to primary production methods. Such analysis can provide valuable insights into the overall environmental sustainability of the recycling process and guide future improvements in recycling technologies and practices.
One of the primary environmental benefits of recycling spent electrolyser catalysts is the conservation of natural resources. By reclaiming precious metals such as platinum, palladium, and rhodium, the need for extensive mining operations is reduced. This, in turn, helps to preserve ecosystems, minimize habitat destruction, and reduce the carbon footprint associated with raw material extraction. Additionally, recycling these catalysts contributes to the circular economy, promoting sustainable resource management and reducing waste accumulation in landfills.
The recycling process itself, however, requires careful environmental management. Chemical treatments used to extract precious metals from spent catalysts can generate hazardous waste streams if not properly handled. These may include acidic solutions, heavy metal residues, and other potentially toxic byproducts. Proper waste treatment and disposal systems must be implemented to prevent soil and water contamination. Furthermore, the energy consumption associated with recycling processes should be evaluated to ensure that the environmental benefits outweigh the energy costs.
Air quality is another important consideration in the environmental impact assessment of catalyst recycling. Some recovery methods may involve high-temperature processes that could release particulate matter or gaseous emissions. Implementing effective air pollution control measures, such as scrubbers and filters, is crucial to minimize the release of harmful substances into the atmosphere.
Water usage and management are also critical factors in the environmental assessment. Recycling processes often require significant amounts of water for washing and separation stages. Implementing water recycling systems and efficient water treatment technologies can help reduce the overall water footprint of the operation and minimize the discharge of contaminated wastewater.
The transportation of spent catalysts to recycling facilities and the distribution of reclaimed materials should also be factored into the environmental impact assessment. Optimizing logistics to reduce transportation distances and employing low-emission vehicles can help minimize the carbon footprint associated with these activities.
Lastly, the long-term environmental benefits of recycling precious metals from spent electrolyser catalysts should be quantified. This includes assessing the reduction in greenhouse gas emissions, energy savings, and conservation of non-renewable resources achieved through recycling compared to primary production methods. Such analysis can provide valuable insights into the overall environmental sustainability of the recycling process and guide future improvements in recycling technologies and practices.
Economic Feasibility of Recycling Processes
The economic feasibility of recycling processes for precious metals from spent electrolyser catalysts is a critical factor in determining the viability of such operations. The cost-effectiveness of these processes depends on several key variables, including the concentration of precious metals in the spent catalysts, the efficiency of the recovery methods, and the market prices of the reclaimed metals.
Initial investment costs for establishing a recycling facility can be substantial, encompassing equipment for crushing, grinding, and chemical processing, as well as environmental control systems. However, these capital expenditures can be offset by the potential long-term returns from recovered metals. The operational costs, including energy consumption, chemical reagents, and labor, must be carefully balanced against the value of the reclaimed materials.
The economic viability is significantly influenced by the precious metal content in the spent catalysts. Higher concentrations of platinum, palladium, or other valuable metals improve the cost-benefit ratio of the recycling process. Advanced analytical techniques are essential for accurately assessing the metal content and optimizing the recovery strategy.
Market dynamics play a crucial role in the economic feasibility. Fluctuations in precious metal prices can dramatically impact the profitability of recycling operations. A robust business model should account for these market variations and potentially include hedging strategies to mitigate risks associated with price volatility.
The scale of operations is another important consideration. Larger-scale recycling facilities can benefit from economies of scale, reducing per-unit processing costs. However, this must be balanced against the availability of spent catalysts and the logistics of collection and transportation.
Regulatory compliance and environmental considerations also factor into the economic equation. While stringent environmental regulations may increase operational costs, they also create opportunities for companies that can demonstrate sustainable and responsible recycling practices.
Technological advancements in recycling processes can significantly enhance economic feasibility. Innovations in hydrometallurgical or pyrometallurgical techniques that improve recovery rates or reduce energy consumption can lead to more cost-effective operations. Continuous research and development in this area are crucial for maintaining competitive advantage.
In conclusion, the economic feasibility of recycling precious metals from spent electrolyser catalysts is a complex interplay of technical, economic, and environmental factors. A thorough cost-benefit analysis, considering all aspects from collection to final metal recovery, is essential for determining the viability of such ventures in the evolving landscape of resource recovery and circular economy principles.
Initial investment costs for establishing a recycling facility can be substantial, encompassing equipment for crushing, grinding, and chemical processing, as well as environmental control systems. However, these capital expenditures can be offset by the potential long-term returns from recovered metals. The operational costs, including energy consumption, chemical reagents, and labor, must be carefully balanced against the value of the reclaimed materials.
The economic viability is significantly influenced by the precious metal content in the spent catalysts. Higher concentrations of platinum, palladium, or other valuable metals improve the cost-benefit ratio of the recycling process. Advanced analytical techniques are essential for accurately assessing the metal content and optimizing the recovery strategy.
Market dynamics play a crucial role in the economic feasibility. Fluctuations in precious metal prices can dramatically impact the profitability of recycling operations. A robust business model should account for these market variations and potentially include hedging strategies to mitigate risks associated with price volatility.
The scale of operations is another important consideration. Larger-scale recycling facilities can benefit from economies of scale, reducing per-unit processing costs. However, this must be balanced against the availability of spent catalysts and the logistics of collection and transportation.
Regulatory compliance and environmental considerations also factor into the economic equation. While stringent environmental regulations may increase operational costs, they also create opportunities for companies that can demonstrate sustainable and responsible recycling practices.
Technological advancements in recycling processes can significantly enhance economic feasibility. Innovations in hydrometallurgical or pyrometallurgical techniques that improve recovery rates or reduce energy consumption can lead to more cost-effective operations. Continuous research and development in this area are crucial for maintaining competitive advantage.
In conclusion, the economic feasibility of recycling precious metals from spent electrolyser catalysts is a complex interplay of technical, economic, and environmental factors. A thorough cost-benefit analysis, considering all aspects from collection to final metal recovery, is essential for determining the viability of such ventures in the evolving landscape of resource recovery and circular economy principles.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!