Optimizing Electrochemical Cell Interfaces for Better Performance
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Electrochemical Interface Technology Background and Objectives
Electrochemical interfaces represent the critical boundary where electron transfer processes occur between electrodes and electrolytes, fundamentally governing the performance of numerous energy storage and conversion technologies. The optimization of these interfaces has evolved significantly since the pioneering work of Hermann von Helmholtz in the 19th century, who first conceptualized the electrical double layer. Subsequent refinements by Gouy-Chapman and Stern established more sophisticated models accounting for ion distribution and adsorption phenomena at these interfaces.
The technological evolution in this field has accelerated dramatically over the past two decades, driven by increasing demands for high-performance batteries, fuel cells, and electrochemical sensors. Contemporary research focuses on nanoscale engineering of electrode surfaces, advanced electrolyte formulations, and interface stabilization strategies to overcome persistent challenges in energy density, power capability, and operational longevity.
Current trends indicate a shift toward atomic-level precision in interface design, incorporating computational modeling to predict and optimize electron transfer kinetics. The integration of in-situ and operando characterization techniques has revolutionized our understanding of dynamic interfacial processes, enabling real-time observation of phenomena previously accessible only through theoretical models.
The primary technical objectives in electrochemical interface optimization encompass several interconnected goals. First, enhancing charge transfer efficiency by minimizing interfacial resistance and polarization effects. Second, improving interface stability to mitigate degradation mechanisms such as solid-electrolyte interphase (SEI) growth in batteries or catalyst poisoning in fuel cells. Third, developing selective interfaces that promote desired electrochemical reactions while suppressing parasitic processes.
Emerging research directions include bio-inspired interface designs that mimic natural electron transfer systems, stimulus-responsive interfaces capable of adapting to changing operational conditions, and self-healing interfaces that can autonomously recover from degradation. The convergence of nanotechnology, materials science, and electrochemistry continues to expand the frontier of possibilities in this domain.
The ultimate goal of interface optimization extends beyond incremental improvements, aiming for transformative advances that could enable next-generation energy technologies with unprecedented performance metrics. These include solid-state batteries with energy densities exceeding 500 Wh/kg, fuel cells operating at ambient temperatures with platinum-free catalysts, and electrochemical CO2 reduction systems with near-theoretical efficiency.
The technological evolution in this field has accelerated dramatically over the past two decades, driven by increasing demands for high-performance batteries, fuel cells, and electrochemical sensors. Contemporary research focuses on nanoscale engineering of electrode surfaces, advanced electrolyte formulations, and interface stabilization strategies to overcome persistent challenges in energy density, power capability, and operational longevity.
Current trends indicate a shift toward atomic-level precision in interface design, incorporating computational modeling to predict and optimize electron transfer kinetics. The integration of in-situ and operando characterization techniques has revolutionized our understanding of dynamic interfacial processes, enabling real-time observation of phenomena previously accessible only through theoretical models.
The primary technical objectives in electrochemical interface optimization encompass several interconnected goals. First, enhancing charge transfer efficiency by minimizing interfacial resistance and polarization effects. Second, improving interface stability to mitigate degradation mechanisms such as solid-electrolyte interphase (SEI) growth in batteries or catalyst poisoning in fuel cells. Third, developing selective interfaces that promote desired electrochemical reactions while suppressing parasitic processes.
Emerging research directions include bio-inspired interface designs that mimic natural electron transfer systems, stimulus-responsive interfaces capable of adapting to changing operational conditions, and self-healing interfaces that can autonomously recover from degradation. The convergence of nanotechnology, materials science, and electrochemistry continues to expand the frontier of possibilities in this domain.
The ultimate goal of interface optimization extends beyond incremental improvements, aiming for transformative advances that could enable next-generation energy technologies with unprecedented performance metrics. These include solid-state batteries with energy densities exceeding 500 Wh/kg, fuel cells operating at ambient temperatures with platinum-free catalysts, and electrochemical CO2 reduction systems with near-theoretical efficiency.
Market Analysis of Advanced Electrochemical Cell Applications
The electrochemical cell market is experiencing robust growth driven by increasing demand across multiple sectors including energy storage, electric vehicles, consumer electronics, and industrial applications. The global market for advanced electrochemical cells was valued at approximately $112 billion in 2022 and is projected to reach $296 billion by 2030, representing a compound annual growth rate of 12.9% during the forecast period.
Energy storage systems represent the largest application segment, accounting for nearly 38% of the total market share. This dominance is primarily attributed to the growing integration of renewable energy sources into power grids, necessitating efficient storage solutions to address intermittency issues. The electric vehicle segment follows closely, with a market share of 33%, and is expected to witness the fastest growth rate of 15.7% annually through 2030, propelled by stringent emission regulations and government incentives worldwide.
Consumer electronics constitute another significant application area, representing approximately 18% of the market. The demand for longer-lasting, faster-charging batteries in smartphones, laptops, and wearable devices continues to drive innovation in electrochemical cell interfaces. Industrial applications account for the remaining 11%, with particular growth in backup power systems and material handling equipment.
Regionally, Asia-Pacific dominates the market with a 45% share, led by China, Japan, and South Korea, which collectively host over 70% of global battery manufacturing capacity. North America and Europe follow with 25% and 22% market shares respectively, with both regions investing heavily in domestic production capabilities to reduce dependency on Asian imports.
The market is witnessing a significant shift toward advanced electrochemical cells with optimized interfaces, as these offer substantial performance improvements including higher energy density, faster charging capabilities, extended cycle life, and enhanced safety profiles. Cells with improved electrode-electrolyte interfaces can achieve up to 30% higher energy density and 40% faster charging rates compared to conventional designs.
Customer demand increasingly focuses on performance metrics directly impacted by interface optimization, with 78% of commercial buyers citing longer cycle life as a primary purchase consideration, followed by charging speed (65%) and safety features (62%). This trend is particularly pronounced in premium market segments where performance advantages outweigh initial cost considerations.
Market forecasts indicate that electrochemical cells with advanced interface engineering will capture an increasing market share, growing from 22% currently to an estimated 47% by 2028, representing a significant opportunity for manufacturers investing in this technology.
Energy storage systems represent the largest application segment, accounting for nearly 38% of the total market share. This dominance is primarily attributed to the growing integration of renewable energy sources into power grids, necessitating efficient storage solutions to address intermittency issues. The electric vehicle segment follows closely, with a market share of 33%, and is expected to witness the fastest growth rate of 15.7% annually through 2030, propelled by stringent emission regulations and government incentives worldwide.
Consumer electronics constitute another significant application area, representing approximately 18% of the market. The demand for longer-lasting, faster-charging batteries in smartphones, laptops, and wearable devices continues to drive innovation in electrochemical cell interfaces. Industrial applications account for the remaining 11%, with particular growth in backup power systems and material handling equipment.
Regionally, Asia-Pacific dominates the market with a 45% share, led by China, Japan, and South Korea, which collectively host over 70% of global battery manufacturing capacity. North America and Europe follow with 25% and 22% market shares respectively, with both regions investing heavily in domestic production capabilities to reduce dependency on Asian imports.
The market is witnessing a significant shift toward advanced electrochemical cells with optimized interfaces, as these offer substantial performance improvements including higher energy density, faster charging capabilities, extended cycle life, and enhanced safety profiles. Cells with improved electrode-electrolyte interfaces can achieve up to 30% higher energy density and 40% faster charging rates compared to conventional designs.
Customer demand increasingly focuses on performance metrics directly impacted by interface optimization, with 78% of commercial buyers citing longer cycle life as a primary purchase consideration, followed by charging speed (65%) and safety features (62%). This trend is particularly pronounced in premium market segments where performance advantages outweigh initial cost considerations.
Market forecasts indicate that electrochemical cells with advanced interface engineering will capture an increasing market share, growing from 22% currently to an estimated 47% by 2028, representing a significant opportunity for manufacturers investing in this technology.
Current Interface Challenges and Technical Limitations
Despite significant advancements in electrochemical cell technology, interface optimization remains one of the most challenging aspects limiting overall performance. The electrode-electrolyte interface represents a critical boundary where electron transfer reactions occur, directly influencing efficiency, durability, and power density. Current interfaces suffer from several fundamental limitations that impede optimal functionality across various electrochemical systems including batteries, fuel cells, and electrolyzers.
Contact resistance at interfaces constitutes a major performance bottleneck, causing significant voltage drops and energy losses. This resistance stems from imperfect physical connections between components and the formation of passivation layers that, while sometimes necessary for stability, often impede ion transport. In lithium-ion batteries, for instance, the solid-electrolyte interphase (SEI) layer simultaneously protects the electrode from further degradation while introducing additional resistance.
Interface degradation mechanisms present another critical challenge. Chemical and electrochemical side reactions at interfaces lead to capacity fading, increased impedance, and shortened device lifetimes. These reactions are particularly problematic at high voltages, high temperatures, or during fast charging conditions, where interface stability is severely compromised.
Mass transport limitations across interfaces significantly restrict reaction kinetics and rate capabilities. Concentration polarization occurs when reactants cannot reach or products cannot leave the interface rapidly enough, creating concentration gradients that reduce cell potential and efficiency. This becomes especially problematic at high current densities required for high-power applications.
Heterogeneity in interface properties presents additional complications. Variations in surface morphology, composition, and reactivity lead to non-uniform current distribution, localized heating, and accelerated degradation at "hot spots." These heterogeneities are difficult to characterize and control during manufacturing processes, resulting in performance inconsistencies between nominally identical cells.
The dynamic nature of interfaces during operation further complicates optimization efforts. Interfaces evolve continuously through processes like dendrite formation, electrode pulverization, and phase transformations. These changes are often irreversible and lead to progressive performance decline that is difficult to mitigate through static design approaches.
Current characterization techniques also present limitations. In-situ and operando methods for studying buried interfaces under realistic operating conditions remain limited in spatial and temporal resolution. This knowledge gap hampers the development of predictive models that could guide interface engineering efforts and accelerate innovation cycles.
Contact resistance at interfaces constitutes a major performance bottleneck, causing significant voltage drops and energy losses. This resistance stems from imperfect physical connections between components and the formation of passivation layers that, while sometimes necessary for stability, often impede ion transport. In lithium-ion batteries, for instance, the solid-electrolyte interphase (SEI) layer simultaneously protects the electrode from further degradation while introducing additional resistance.
Interface degradation mechanisms present another critical challenge. Chemical and electrochemical side reactions at interfaces lead to capacity fading, increased impedance, and shortened device lifetimes. These reactions are particularly problematic at high voltages, high temperatures, or during fast charging conditions, where interface stability is severely compromised.
Mass transport limitations across interfaces significantly restrict reaction kinetics and rate capabilities. Concentration polarization occurs when reactants cannot reach or products cannot leave the interface rapidly enough, creating concentration gradients that reduce cell potential and efficiency. This becomes especially problematic at high current densities required for high-power applications.
Heterogeneity in interface properties presents additional complications. Variations in surface morphology, composition, and reactivity lead to non-uniform current distribution, localized heating, and accelerated degradation at "hot spots." These heterogeneities are difficult to characterize and control during manufacturing processes, resulting in performance inconsistencies between nominally identical cells.
The dynamic nature of interfaces during operation further complicates optimization efforts. Interfaces evolve continuously through processes like dendrite formation, electrode pulverization, and phase transformations. These changes are often irreversible and lead to progressive performance decline that is difficult to mitigate through static design approaches.
Current characterization techniques also present limitations. In-situ and operando methods for studying buried interfaces under realistic operating conditions remain limited in spatial and temporal resolution. This knowledge gap hampers the development of predictive models that could guide interface engineering efforts and accelerate innovation cycles.
State-of-the-Art Interface Optimization Approaches
01 Interface modification for improved electrochemical performance
Modification of electrochemical cell interfaces can significantly enhance performance by reducing interfacial resistance and improving charge transfer. Various techniques include surface treatments, coatings, and the introduction of functional layers that optimize the interaction between electrodes and electrolytes. These modifications can lead to better stability, increased power density, and extended cycle life of electrochemical cells.- Electrode-electrolyte interface optimization: The interface between electrodes and electrolytes is critical for electrochemical cell performance. Optimization techniques include surface modifications, specialized coatings, and nanostructured designs that enhance ion transport and reduce interfacial resistance. These improvements lead to better charge transfer kinetics, increased energy density, and extended cycle life of electrochemical cells.
- Advanced modeling and simulation methods: Computational modeling and simulation techniques are employed to predict and analyze electrochemical cell interface behavior. These methods include molecular dynamics simulations, finite element analysis, and machine learning approaches that help understand complex interfacial phenomena. Such computational tools enable researchers to optimize cell designs, predict performance limitations, and develop more efficient electrochemical systems without extensive physical testing.
- Novel materials for interface enhancement: Innovative materials are being developed specifically to improve electrochemical cell interfaces. These include advanced composite materials, doped ceramics, and functionalized polymers that facilitate better ion conduction while maintaining structural integrity. Such materials can significantly reduce interfacial resistance, mitigate degradation mechanisms, and enhance overall cell performance under various operating conditions.
- In-situ and operando characterization techniques: Advanced characterization methods allow for real-time monitoring of electrochemical interfaces during operation. Techniques such as synchrotron-based X-ray analysis, neutron diffraction, and advanced microscopy enable researchers to observe interfacial phenomena as they occur. These approaches provide crucial insights into degradation mechanisms, phase transformations, and reaction kinetics at the electrode-electrolyte interface, leading to more informed design strategies.
- Interface engineering for specific applications: Tailored interface engineering approaches are developed for specific electrochemical cell applications such as batteries, fuel cells, and sensors. These specialized techniques consider the unique requirements of each application, including operating temperature, pressure conditions, and performance targets. Custom interface designs can incorporate protective layers, gradient structures, or selective membranes to optimize performance parameters relevant to the intended use case.
02 Advanced modeling and simulation of electrochemical interfaces
Computational methods are increasingly used to understand and predict electrochemical cell interface behavior. These include molecular dynamics simulations, density functional theory, and machine learning approaches that model complex interfacial phenomena. Such simulations help optimize interface design by predicting how different materials and structures will perform under various operating conditions, accelerating the development of high-performance electrochemical cells.Expand Specific Solutions03 Novel materials for electrode-electrolyte interfaces
Innovative materials are being developed specifically for electrochemical cell interfaces to enhance performance metrics. These include advanced composite materials, nanostructured interfaces, and specially designed solid electrolyte interphases. The incorporation of these materials can significantly improve ionic conductivity, mechanical stability, and electrochemical compatibility at critical interfaces, leading to better overall cell performance.Expand Specific Solutions04 In-situ and operando characterization techniques
Advanced characterization methods allow for real-time monitoring of electrochemical interfaces during operation. These techniques include spectroscopic methods, microscopy, and electrochemical impedance spectroscopy that provide insights into interface evolution and degradation mechanisms. By understanding how interfaces change under operating conditions, researchers can develop strategies to mitigate performance losses and extend device lifetimes.Expand Specific Solutions05 Interface engineering for specific electrochemical applications
Tailored interface designs are being developed for specific electrochemical applications such as batteries, fuel cells, and electrolyzers. These application-specific approaches consider the unique requirements and challenges of each system, optimizing interfaces for particular operating conditions, temperature ranges, and chemical environments. Such specialized engineering leads to significant performance improvements in targeted electrochemical technologies.Expand Specific Solutions
Leading Companies and Research Institutions in Interface Technology
The electrochemical cell interface optimization market is currently in a growth phase, with increasing demand driven by the expanding electric vehicle and energy storage sectors. The global market size is projected to reach significant value as industries prioritize battery performance improvements. Leading players include established automotive manufacturers (Mercedes-Benz, Toyota, Robert Bosch) and specialized battery technology companies (CATL, Sion Power, NGK Insulators). The technology maturity varies across applications, with companies like Saft Groupe and LG Electronics demonstrating advanced commercial implementations, while research institutions (Fraunhofer-Gesellschaft, Penn State Research Foundation) continue developing next-generation interface solutions. Asian manufacturers, particularly CATL and Samsung Electronics, are rapidly advancing cell interface technologies to meet growing market demands.
Sion Power Corp.
Technical Solution: Sion Power has developed the Licerion® technology, focusing on lithium-metal anode interfaces for high-energy batteries. Their approach addresses the critical challenges of lithium-metal electrodes through a multi-faceted interface engineering strategy. Sion's proprietary protected lithium electrode technology employs specialized polymer and ceramic composite layers that physically and chemically stabilize the lithium-electrolyte interface. Their interface design incorporates ionically conductive but electronically insulating materials that promote uniform lithium deposition while preventing dendrite formation. The company has developed advanced electrolyte formulations with additives that form stable passivation layers on lithium surfaces, reducing parasitic reactions. Sion's interface engineering has demonstrated remarkable improvements in cycle life, achieving over 500 cycles at 100% depth of discharge while maintaining energy densities above 400 Wh/kg, significantly outperforming conventional lithium-metal approaches that typically fail after 50-100 cycles.
Strengths: Extremely high gravimetric energy density (potentially exceeding 500 Wh/kg) enabled by their interface-stabilized lithium metal technology; significantly improved safety compared to conventional lithium metal designs. Weaknesses: Current manufacturing processes for their specialized interface layers remain complex and costly; performance at high discharge rates still lags behind conventional lithium-ion technology due to interfacial resistance.
Ceres Power Ltd.
Technical Solution: Ceres Power has developed the SteelCell® technology, a unique solid oxide fuel cell (SOFC) platform that focuses on optimizing electrochemical interfaces for enhanced performance and durability. Their approach uses a steel substrate with proprietary ceramic coatings to create a robust electrode-electrolyte interface that operates at lower temperatures (500-600°C) than conventional SOFCs (800-1000°C). The company employs nano-structured electrode materials with carefully engineered triple-phase boundaries to maximize electrochemical reaction sites. Their interface optimization includes infiltration techniques where catalytically active materials are introduced into porous electrode structures post-fabrication, significantly increasing the effective surface area for reactions. Ceres has demonstrated that their interface engineering reduces polarization losses by approximately 40% compared to conventional designs, enabling higher power density and improved efficiency.
Strengths: Lower operating temperature reduces thermal stress and enables faster start-up times (minutes rather than hours); steel support provides mechanical robustness and cost advantages over ceramic-based systems. Weaknesses: Despite lower temperature operation, thermal cycling still presents challenges for interface stability; manufacturing complexity of their multi-layer interface structure increases production costs.
Critical Patents and Breakthroughs in Interface Engineering
Electrochemical cell optimization
PatentPendingJP2023504210A
Innovation
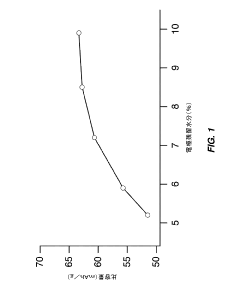
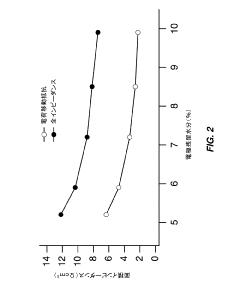
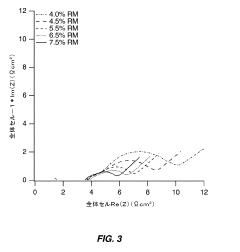
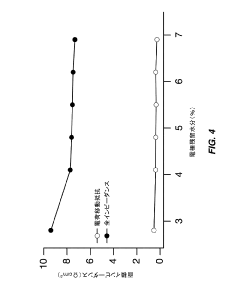
- Optimize the water content in TMCCC electrodes by maintaining a controlled residual moisture (RM) balance, allowing some lattice-bound water while minimizing non-coordinated water through a single dehydration/hydration process on a fully assembled cell stack, rather than individual electrodes.
Electrochemical cell, and method for the production of an electrochemical cell
PatentWO2024126721A1
Innovation
- An electrochemical cell design featuring a membrane with a gas diffusion layer coated with an electrically insulating material in areas outside the three-phase boundary to prevent parasitic currents, ensuring reactions are confined to the most efficient interface.
Materials Science Innovations for Electrode-Electrolyte Interfaces
Recent advancements in materials science have revolutionized the development of electrode-electrolyte interfaces in electrochemical cells. These innovations address critical performance limitations by focusing on the molecular and atomic-level interactions at these crucial boundaries. Novel nanostructured materials with precisely engineered surface properties have demonstrated significant improvements in charge transfer efficiency and stability.
Composite materials incorporating carbon-based structures such as graphene and carbon nanotubes have emerged as particularly promising candidates for interface optimization. These materials provide exceptional electrical conductivity while offering customizable surface chemistry that can be tailored to specific electrolyte compositions. Research indicates that such composites can reduce interfacial resistance by up to 40% compared to conventional materials.
Surface modification techniques have evolved substantially, with atomic layer deposition (ALD) and molecular layer deposition (MLD) enabling unprecedented control over interface properties. These methods allow for the creation of ultrathin protective layers that mitigate unwanted side reactions while maintaining efficient ion transport. Studies have shown that optimized protective layers can extend cell cycling life by 300-500% in aggressive electrolyte environments.
Self-healing interface materials represent another breakthrough direction, incorporating dynamic chemical bonds that can reform after being broken during cell operation. These materials address the persistent challenge of interface degradation during cycling, potentially extending device lifetimes significantly. Early prototypes have demonstrated the ability to maintain 85% of initial performance after conditions that would render conventional interfaces non-functional.
Biomimetic approaches drawing inspiration from natural membrane structures have yielded selective ion transport channels with remarkable specificity. These bio-inspired interfaces can dramatically improve selectivity, reducing crossover effects that plague many electrochemical systems. The incorporation of engineered proteins and synthetic ion channels has shown promise in creating interfaces with "gatekeeper" functionality.
Computational materials science has accelerated these developments through high-throughput screening and machine learning algorithms that can predict interface behavior. These tools have identified previously overlooked material combinations with exceptional properties. Simulation capabilities now extend to modeling degradation mechanisms at atomic scales, enabling researchers to address failure modes before they manifest in physical prototypes.
Hybrid organic-inorganic interfaces represent a particularly promising frontier, combining the mechanical stability of inorganic materials with the versatility and functionality of organic components. These hybrids can be designed with gradient properties that smoothly transition between electrode and electrolyte environments, minimizing energy barriers to ion transport while maintaining structural integrity.
Composite materials incorporating carbon-based structures such as graphene and carbon nanotubes have emerged as particularly promising candidates for interface optimization. These materials provide exceptional electrical conductivity while offering customizable surface chemistry that can be tailored to specific electrolyte compositions. Research indicates that such composites can reduce interfacial resistance by up to 40% compared to conventional materials.
Surface modification techniques have evolved substantially, with atomic layer deposition (ALD) and molecular layer deposition (MLD) enabling unprecedented control over interface properties. These methods allow for the creation of ultrathin protective layers that mitigate unwanted side reactions while maintaining efficient ion transport. Studies have shown that optimized protective layers can extend cell cycling life by 300-500% in aggressive electrolyte environments.
Self-healing interface materials represent another breakthrough direction, incorporating dynamic chemical bonds that can reform after being broken during cell operation. These materials address the persistent challenge of interface degradation during cycling, potentially extending device lifetimes significantly. Early prototypes have demonstrated the ability to maintain 85% of initial performance after conditions that would render conventional interfaces non-functional.
Biomimetic approaches drawing inspiration from natural membrane structures have yielded selective ion transport channels with remarkable specificity. These bio-inspired interfaces can dramatically improve selectivity, reducing crossover effects that plague many electrochemical systems. The incorporation of engineered proteins and synthetic ion channels has shown promise in creating interfaces with "gatekeeper" functionality.
Computational materials science has accelerated these developments through high-throughput screening and machine learning algorithms that can predict interface behavior. These tools have identified previously overlooked material combinations with exceptional properties. Simulation capabilities now extend to modeling degradation mechanisms at atomic scales, enabling researchers to address failure modes before they manifest in physical prototypes.
Hybrid organic-inorganic interfaces represent a particularly promising frontier, combining the mechanical stability of inorganic materials with the versatility and functionality of organic components. These hybrids can be designed with gradient properties that smoothly transition between electrode and electrolyte environments, minimizing energy barriers to ion transport while maintaining structural integrity.
Sustainability and Environmental Impact of Interface Materials
The sustainability and environmental impact of interface materials in electrochemical cells represent critical considerations in the ongoing development of energy storage and conversion technologies. Current interface materials often rely on rare earth elements, precious metals, and environmentally problematic compounds that pose significant challenges to sustainable manufacturing and end-of-life management.
The extraction processes for many interface materials, particularly noble metals like platinum and palladium used in catalytic layers, generate substantial environmental footprints. Mining operations contribute to habitat destruction, water pollution, and greenhouse gas emissions. For instance, the production of one kilogram of platinum generates approximately 40 tons of CO2 equivalent emissions, highlighting the carbon-intensive nature of these critical interface components.
Toxicity concerns also emerge with certain interface materials, particularly those containing heavy metals or fluorinated compounds. These substances can leach into ecosystems during improper disposal, creating long-term environmental hazards. The persistence of perfluorinated compounds used in some membrane interfaces presents particular challenges, as these materials can remain environmentally active for decades.
Recent advances in green chemistry approaches offer promising alternatives for more sustainable interface materials. Bio-inspired catalysts mimicking natural enzymatic processes have demonstrated potential to replace conventional noble metal catalysts. These biomimetic interfaces often operate under milder conditions and utilize earth-abundant elements, significantly reducing environmental impact while maintaining acceptable performance metrics.
Circular economy principles are increasingly being applied to interface material design. Researchers are developing recyclable polymer interfaces and recovery processes for precious metals from spent electrochemical cells. These approaches can reduce primary resource consumption by up to 70% for certain interface components, according to recent industry analyses.
Life cycle assessment (LCA) studies reveal that the environmental impact of interface materials extends beyond their operational lifetime. The embodied energy in manufacturing specialized interfaces can represent up to 40% of the total environmental footprint of an electrochemical cell. This highlights the importance of considering production efficiency alongside performance metrics when developing next-generation interface materials.
Water consumption represents another significant environmental concern, particularly for interface materials requiring extensive purification processes. Advanced manufacturing techniques like atomic layer deposition can reduce water usage by up to 60% compared to conventional wet chemistry approaches, while simultaneously improving interface quality and performance characteristics.
The extraction processes for many interface materials, particularly noble metals like platinum and palladium used in catalytic layers, generate substantial environmental footprints. Mining operations contribute to habitat destruction, water pollution, and greenhouse gas emissions. For instance, the production of one kilogram of platinum generates approximately 40 tons of CO2 equivalent emissions, highlighting the carbon-intensive nature of these critical interface components.
Toxicity concerns also emerge with certain interface materials, particularly those containing heavy metals or fluorinated compounds. These substances can leach into ecosystems during improper disposal, creating long-term environmental hazards. The persistence of perfluorinated compounds used in some membrane interfaces presents particular challenges, as these materials can remain environmentally active for decades.
Recent advances in green chemistry approaches offer promising alternatives for more sustainable interface materials. Bio-inspired catalysts mimicking natural enzymatic processes have demonstrated potential to replace conventional noble metal catalysts. These biomimetic interfaces often operate under milder conditions and utilize earth-abundant elements, significantly reducing environmental impact while maintaining acceptable performance metrics.
Circular economy principles are increasingly being applied to interface material design. Researchers are developing recyclable polymer interfaces and recovery processes for precious metals from spent electrochemical cells. These approaches can reduce primary resource consumption by up to 70% for certain interface components, according to recent industry analyses.
Life cycle assessment (LCA) studies reveal that the environmental impact of interface materials extends beyond their operational lifetime. The embodied energy in manufacturing specialized interfaces can represent up to 40% of the total environmental footprint of an electrochemical cell. This highlights the importance of considering production efficiency alongside performance metrics when developing next-generation interface materials.
Water consumption represents another significant environmental concern, particularly for interface materials requiring extensive purification processes. Advanced manufacturing techniques like atomic layer deposition can reduce water usage by up to 60% compared to conventional wet chemistry approaches, while simultaneously improving interface quality and performance characteristics.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!