Research on Solid sorbents for CO2 capture material properties and performance
SEP 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
CO2 Capture Sorbent Evolution and Objectives
Carbon dioxide capture technology has evolved significantly over the past decades, driven by the urgent need to mitigate climate change impacts. The journey began with conventional absorption methods using liquid amines in the 1930s, primarily for natural gas sweetening rather than climate concerns. By the 1990s, as global warming awareness increased, research into CO2 capture technologies accelerated, with solid sorbents emerging as promising alternatives to liquid systems due to their potential energy efficiency advantages.
The evolution of solid sorbents for CO2 capture has progressed through several distinct phases. First-generation materials focused on activated carbons and zeolites, which offered moderate CO2 selectivity but suffered from moisture sensitivity and capacity limitations. The second generation, emerging in the early 2000s, introduced metal-organic frameworks (MOFs) and functionalized porous silicas, which demonstrated significantly improved CO2 adsorption capacities and selectivity under controlled conditions.
Current third-generation materials represent a convergence of materials science and chemical engineering, with hybrid sorbents combining the advantages of multiple material classes. These include amine-functionalized MOFs, hierarchical porous structures, and composite materials designed to overcome the traditional capacity-kinetics trade-off that has limited practical applications.
The primary technical objectives in solid sorbent development focus on several critical performance parameters. First, achieving high CO2 selectivity (>100) in the presence of other flue gas components, particularly water vapor, remains essential for practical deployment. Second, rapid adsorption-desorption kinetics is crucial for process economics, with target adsorption times under 60 seconds for viable industrial implementation.
Energy efficiency represents another critical objective, with researchers aiming to reduce regeneration energy requirements below 2 GJ/ton CO2, significantly lower than the 3.5-4 GJ/ton typical of conventional amine scrubbing. Mechanical and chemical stability under repeated cycling (>10,000 cycles) and resistance to contaminants like SOx and NOx constitute additional key targets.
Looking forward, the field is trending toward multifunctional materials that can simultaneously capture CO2 and convert it into value-added products, potentially transforming carbon capture from a cost center to a revenue generator. Computational materials design and high-throughput screening methodologies are accelerating discovery in this space, with machine learning approaches helping to identify promising candidate materials from vast theoretical possibilities.
The ultimate objective remains developing economically viable solid sorbents that can be deployed at scale across various industrial sectors, from power generation to cement and steel production, enabling meaningful reductions in global CO2 emissions while maintaining industrial productivity and economic growth.
The evolution of solid sorbents for CO2 capture has progressed through several distinct phases. First-generation materials focused on activated carbons and zeolites, which offered moderate CO2 selectivity but suffered from moisture sensitivity and capacity limitations. The second generation, emerging in the early 2000s, introduced metal-organic frameworks (MOFs) and functionalized porous silicas, which demonstrated significantly improved CO2 adsorption capacities and selectivity under controlled conditions.
Current third-generation materials represent a convergence of materials science and chemical engineering, with hybrid sorbents combining the advantages of multiple material classes. These include amine-functionalized MOFs, hierarchical porous structures, and composite materials designed to overcome the traditional capacity-kinetics trade-off that has limited practical applications.
The primary technical objectives in solid sorbent development focus on several critical performance parameters. First, achieving high CO2 selectivity (>100) in the presence of other flue gas components, particularly water vapor, remains essential for practical deployment. Second, rapid adsorption-desorption kinetics is crucial for process economics, with target adsorption times under 60 seconds for viable industrial implementation.
Energy efficiency represents another critical objective, with researchers aiming to reduce regeneration energy requirements below 2 GJ/ton CO2, significantly lower than the 3.5-4 GJ/ton typical of conventional amine scrubbing. Mechanical and chemical stability under repeated cycling (>10,000 cycles) and resistance to contaminants like SOx and NOx constitute additional key targets.
Looking forward, the field is trending toward multifunctional materials that can simultaneously capture CO2 and convert it into value-added products, potentially transforming carbon capture from a cost center to a revenue generator. Computational materials design and high-throughput screening methodologies are accelerating discovery in this space, with machine learning approaches helping to identify promising candidate materials from vast theoretical possibilities.
The ultimate objective remains developing economically viable solid sorbents that can be deployed at scale across various industrial sectors, from power generation to cement and steel production, enabling meaningful reductions in global CO2 emissions while maintaining industrial productivity and economic growth.
Market Analysis for Carbon Capture Technologies
The global carbon capture and storage (CCS) market is experiencing significant growth, driven by increasing environmental regulations and the urgent need to reduce greenhouse gas emissions. As of 2023, the market was valued at approximately $7.5 billion and is projected to reach $15.3 billion by 2030, representing a compound annual growth rate (CAGR) of 10.7%. This growth trajectory is primarily fueled by governmental commitments to achieve net-zero emissions targets and the implementation of carbon pricing mechanisms across various regions.
North America currently dominates the carbon capture market, accounting for roughly 40% of global installations, with the United States leading in terms of operational facilities. Europe follows closely, with the European Union's ambitious climate policies driving substantial investments in carbon capture technologies. The Asia-Pacific region, particularly China and Japan, is emerging as the fastest-growing market, with projected growth rates exceeding 12% annually through 2030.
Within the carbon capture technology landscape, solid sorbent-based systems are gaining significant traction. These technologies currently represent approximately 25% of the carbon capture market but are expected to expand to 35% by 2028. This growth is attributed to their lower energy requirements compared to traditional liquid solvent systems, which typically consume 20-30% less energy for the regeneration process.
The industrial sector remains the largest end-user of carbon capture technologies, accounting for 65% of applications, particularly in cement production, steel manufacturing, and power generation. However, the direct air capture (DAC) segment, though currently small at about 5% market share, is projected to be the fastest-growing application area with an anticipated CAGR of 15.2% through 2030.
Key market drivers include increasingly stringent carbon emission regulations, with over 40 countries now implementing some form of carbon pricing. Additionally, tax incentives such as the 45Q tax credit in the United States, which offers up to $85 per metric ton of CO2 stored, are significantly improving the economic viability of carbon capture projects. The integration of carbon capture with utilization pathways, creating valuable products from captured CO2, is also enhancing market attractiveness.
Market barriers include high capital expenditure requirements, with typical industrial-scale installations costing between $400-800 million, and operational costs ranging from $40-120 per ton of CO2 captured. Technological limitations of current sorbent materials, including degradation issues and selectivity challenges in mixed gas streams, also present significant hurdles to widespread adoption. Infrastructure constraints, particularly the limited availability of CO2 transport and storage networks, further impede market expansion in many regions.
North America currently dominates the carbon capture market, accounting for roughly 40% of global installations, with the United States leading in terms of operational facilities. Europe follows closely, with the European Union's ambitious climate policies driving substantial investments in carbon capture technologies. The Asia-Pacific region, particularly China and Japan, is emerging as the fastest-growing market, with projected growth rates exceeding 12% annually through 2030.
Within the carbon capture technology landscape, solid sorbent-based systems are gaining significant traction. These technologies currently represent approximately 25% of the carbon capture market but are expected to expand to 35% by 2028. This growth is attributed to their lower energy requirements compared to traditional liquid solvent systems, which typically consume 20-30% less energy for the regeneration process.
The industrial sector remains the largest end-user of carbon capture technologies, accounting for 65% of applications, particularly in cement production, steel manufacturing, and power generation. However, the direct air capture (DAC) segment, though currently small at about 5% market share, is projected to be the fastest-growing application area with an anticipated CAGR of 15.2% through 2030.
Key market drivers include increasingly stringent carbon emission regulations, with over 40 countries now implementing some form of carbon pricing. Additionally, tax incentives such as the 45Q tax credit in the United States, which offers up to $85 per metric ton of CO2 stored, are significantly improving the economic viability of carbon capture projects. The integration of carbon capture with utilization pathways, creating valuable products from captured CO2, is also enhancing market attractiveness.
Market barriers include high capital expenditure requirements, with typical industrial-scale installations costing between $400-800 million, and operational costs ranging from $40-120 per ton of CO2 captured. Technological limitations of current sorbent materials, including degradation issues and selectivity challenges in mixed gas streams, also present significant hurdles to widespread adoption. Infrastructure constraints, particularly the limited availability of CO2 transport and storage networks, further impede market expansion in many regions.
Current Solid Sorbents Landscape and Challenges
The global landscape of solid sorbents for CO2 capture is rapidly evolving, with significant advancements in material development across multiple categories. Currently, the field is dominated by several major classes of materials including activated carbons, zeolites, metal-organic frameworks (MOFs), amine-functionalized sorbents, and hydrotalcite-like compounds. Each category presents distinct advantages and limitations that influence their commercial viability and application scope.
Activated carbons remain among the most widely deployed sorbents due to their relatively low production costs and established manufacturing infrastructure. However, they typically exhibit moderate CO2 selectivity and capacity compared to newer materials, particularly at low partial pressures relevant to post-combustion capture scenarios. Their performance is heavily dependent on pore structure optimization and surface chemistry modifications.
Zeolites, particularly those with high aluminum content such as 13X and 5A, demonstrate excellent CO2 adsorption capacities at ambient temperatures but suffer from severe performance degradation in humid conditions. This moisture sensitivity represents a significant challenge for real-world applications where flue gases typically contain 5-15% water vapor.
Metal-organic frameworks have emerged as promising candidates with record-breaking surface areas (exceeding 7000 m²/g) and highly tunable pore structures. Notable examples include Mg-MOF-74 and HKUST-1, which exhibit exceptional CO2 uptake capacities. However, challenges persist regarding their hydrothermal stability, manufacturing scalability, and production costs that currently limit widespread industrial adoption.
Amine-functionalized sorbents, including amine-grafted silicas and porous polymers, leverage the selective chemistry between amines and CO2 to achieve high selectivity even at low CO2 concentrations. While these materials show promising performance in humid conditions, they face challenges related to amine leaching, oxidative degradation, and energy-intensive regeneration requirements.
Geographically, research leadership in solid sorbent development shows distinct patterns. North America and Europe maintain strong positions in fundamental research and pilot-scale demonstrations, while East Asian countries, particularly China, are rapidly accelerating both research output and commercial implementation efforts. This shifting landscape is creating new competitive dynamics in intellectual property development and commercialization pathways.
The primary technical challenges currently facing the field include: (1) balancing high CO2 selectivity with reasonable regeneration energy requirements; (2) maintaining performance stability over thousands of adsorption-desorption cycles; (3) developing materials with sufficient mechanical strength for industrial implementation; (4) addressing manufacturing scalability and cost barriers; and (5) optimizing system integration to minimize parasitic energy losses in full-scale capture operations.
Activated carbons remain among the most widely deployed sorbents due to their relatively low production costs and established manufacturing infrastructure. However, they typically exhibit moderate CO2 selectivity and capacity compared to newer materials, particularly at low partial pressures relevant to post-combustion capture scenarios. Their performance is heavily dependent on pore structure optimization and surface chemistry modifications.
Zeolites, particularly those with high aluminum content such as 13X and 5A, demonstrate excellent CO2 adsorption capacities at ambient temperatures but suffer from severe performance degradation in humid conditions. This moisture sensitivity represents a significant challenge for real-world applications where flue gases typically contain 5-15% water vapor.
Metal-organic frameworks have emerged as promising candidates with record-breaking surface areas (exceeding 7000 m²/g) and highly tunable pore structures. Notable examples include Mg-MOF-74 and HKUST-1, which exhibit exceptional CO2 uptake capacities. However, challenges persist regarding their hydrothermal stability, manufacturing scalability, and production costs that currently limit widespread industrial adoption.
Amine-functionalized sorbents, including amine-grafted silicas and porous polymers, leverage the selective chemistry between amines and CO2 to achieve high selectivity even at low CO2 concentrations. While these materials show promising performance in humid conditions, they face challenges related to amine leaching, oxidative degradation, and energy-intensive regeneration requirements.
Geographically, research leadership in solid sorbent development shows distinct patterns. North America and Europe maintain strong positions in fundamental research and pilot-scale demonstrations, while East Asian countries, particularly China, are rapidly accelerating both research output and commercial implementation efforts. This shifting landscape is creating new competitive dynamics in intellectual property development and commercialization pathways.
The primary technical challenges currently facing the field include: (1) balancing high CO2 selectivity with reasonable regeneration energy requirements; (2) maintaining performance stability over thousands of adsorption-desorption cycles; (3) developing materials with sufficient mechanical strength for industrial implementation; (4) addressing manufacturing scalability and cost barriers; and (5) optimizing system integration to minimize parasitic energy losses in full-scale capture operations.
Existing Solid Sorbent Material Solutions
01 Metal-organic frameworks (MOFs) for CO2 capture
Metal-organic frameworks represent a class of porous materials with high surface area and tunable pore structures that show promising performance for CO2 capture. These crystalline materials consist of metal ions or clusters coordinated with organic ligands, creating a framework with exceptional adsorption capacity. MOFs can be designed with specific functional groups to enhance CO2 selectivity and binding affinity, while maintaining good regeneration properties under various operating conditions.- Metal-organic frameworks (MOFs) for CO2 capture: Metal-organic frameworks are crystalline porous materials composed of metal ions or clusters coordinated with organic ligands. They exhibit exceptional properties for CO2 capture including high surface area, tunable pore size, and chemical functionality. MOFs can be designed with specific binding sites for CO2 molecules, enhancing selectivity and adsorption capacity. Their modular nature allows for customization to optimize CO2 capture performance under various conditions, making them promising candidates for industrial carbon capture applications.
- Amine-functionalized sorbents for enhanced CO2 adsorption: Amine-functionalized materials represent a significant class of solid sorbents for CO2 capture. These materials incorporate amine groups that chemically react with CO2 through carbamate formation, resulting in high selectivity and adsorption capacity. Common supports include silica, activated carbon, and polymeric substrates that are modified with various amine compounds. The performance of these sorbents depends on amine loading, accessibility of binding sites, and thermal stability. They typically operate at lower temperatures compared to traditional aqueous amine scrubbing and can be regenerated with less energy input.
- Zeolites and molecular sieves for selective CO2 adsorption: Zeolites and molecular sieves are aluminosilicate materials with well-defined pore structures that enable molecular sieving effects for selective CO2 capture. Their crystalline framework contains cations that create strong electrostatic fields, enhancing interaction with CO2 molecules. These materials demonstrate good thermal stability and can be regenerated multiple times without significant performance degradation. The CO2 adsorption capacity and selectivity can be tuned by adjusting the silicon-to-aluminum ratio, cation type, and pore dimensions. They are particularly effective for pressure swing adsorption processes in industrial applications.
- Carbon-based sorbents with enhanced surface properties: Carbon-based materials, including activated carbon, carbon nanotubes, and graphene derivatives, serve as effective CO2 sorbents due to their high surface area and pore volume. These materials can be modified through physical or chemical activation processes to enhance their CO2 capture performance. Surface functionalization with nitrogen, oxygen, or sulfur-containing groups improves CO2 binding affinity. The hierarchical pore structure facilitates rapid diffusion and access to adsorption sites. Carbon-based sorbents offer advantages including low cost, high thermal stability, and resistance to moisture, making them suitable for practical carbon capture applications.
- Regeneration methods and cyclic performance of CO2 sorbents: The practical application of solid sorbents for CO2 capture depends significantly on their regeneration capabilities and performance stability over multiple adsorption-desorption cycles. Various regeneration methods include temperature swing, pressure swing, vacuum swing, and combinations thereof. Key performance metrics include working capacity, regeneration energy requirements, and degradation rate over cycles. Material properties that influence regeneration include heat capacity, thermal conductivity, and structural stability. Advanced sorbent designs incorporate features to minimize energy penalties during regeneration while maintaining high CO2 capture efficiency throughout the operational lifetime.
02 Amine-functionalized sorbents
Amine-functionalized materials are widely used as solid sorbents for CO2 capture due to their strong chemical interaction with CO2 molecules. These sorbents typically consist of amines grafted onto porous supports such as silica, activated carbon, or polymeric materials. The amine groups form carbamates or bicarbonates upon reaction with CO2, enabling high selectivity even at low CO2 concentrations. Key performance factors include amine loading, accessibility, stability during multiple adsorption-desorption cycles, and heat management during regeneration.Expand Specific Solutions03 Zeolites and molecular sieves
Zeolites and molecular sieves are crystalline aluminosilicate materials with well-defined pore structures that enable molecular sieving of CO2. These materials offer high thermal stability, mechanical strength, and relatively low cost for CO2 capture applications. Their performance depends on the Si/Al ratio, cation type, pore size distribution, and framework topology. While zeolites generally exhibit good CO2 selectivity over nitrogen, their capacity can be negatively affected by moisture, requiring consideration of water management strategies in practical applications.Expand Specific Solutions04 Carbon-based sorbents
Carbon-based materials, including activated carbon, carbon nanotubes, and graphene derivatives, serve as effective CO2 sorbents due to their high surface area and pore volume. These materials can be produced from various precursors including biomass, polymers, or fossil resources. Their CO2 capture performance can be enhanced through surface modification, heteroatom doping (N, S, or O), and pore structure optimization. Carbon-based sorbents typically exhibit good stability, relatively low regeneration energy, and potential for large-scale production at reasonable cost.Expand Specific Solutions05 Alkali metal-based sorbents
Alkali metal-based sorbents, particularly those containing lithium, sodium, or potassium compounds, demonstrate high CO2 capture capacity through carbonate formation mechanisms. These materials include lithium zirconates, lithium silicates, sodium carbonates, and potassium-promoted oxides. They typically operate at elevated temperatures (200-700°C) and are suitable for pre-combustion or post-combustion capture systems. Key performance factors include reaction kinetics, mechanical stability during multiple carbonation-calcination cycles, and resistance to contaminants in flue gas streams.Expand Specific Solutions
Leading Organizations in CO2 Capture Materials Research
The solid sorbent CO2 capture technology market is in a growth phase, with increasing focus on carbon neutrality driving research and commercialization. The global carbon capture market is projected to expand significantly as climate policies tighten worldwide. Technologically, the field shows varying maturity levels across different sorbent types. Leading players include specialized carbon capture companies like Climeworks AG and Carboncapture, Inc., which are commercializing direct air capture technologies, alongside energy giants such as ExxonMobil and Sinopec developing industrial-scale solutions. Research institutions including Tianjin University, West Virginia University, and Shanghai Advanced Research Institute are advancing fundamental materials science, while companies like Susteon are bridging the gap between laboratory research and commercial deployment through innovative sorbent development.
Climeworks AG
Technical Solution: Climeworks has developed a proprietary Direct Air Capture (DAC) technology using solid sorbents to capture CO2 directly from ambient air. Their approach employs a two-step temperature/vacuum swing adsorption process with specialized amine-functionalized filter materials. The solid sorbents are packed into collector units where ambient air flows through, allowing CO2 molecules to chemically bind to the sorbent surface. Once saturated, the collectors are heated to approximately 100°C, releasing concentrated CO2 that can be collected for storage or utilization. Climeworks' technology has been commercially deployed in multiple facilities, including their Orca plant in Iceland which captures 4,000 tons of CO2 annually and their larger Mammoth plant under development with a planned capacity of 36,000 tons per year. Their solid sorbents are designed for thousands of adsorption-desorption cycles, maintaining performance over extended operational periods.
Strengths: Modular, scalable design allows for flexible deployment; integration with geothermal energy in Iceland provides renewable heat source for regeneration; proven commercial implementation with multiple operational plants. Weaknesses: Higher energy requirements and costs compared to point-source capture; current capacity remains limited relative to global emissions; regeneration process requires significant thermal energy input.
China Petroleum & Chemical Corp.
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed advanced solid sorbent technologies for CO2 capture focusing on post-combustion applications at fossil fuel power plants and industrial facilities. Their primary approach utilizes structured amine-modified mesoporous silica materials with optimized pore structures to enhance CO2 diffusion and adsorption kinetics. Sinopec's solid sorbents feature high CO2 selectivity in flue gas environments containing moisture and other contaminants, with adsorption capacities exceeding 3 mmol CO2/g sorbent under typical flue gas conditions. The company has implemented temperature-swing adsorption systems where regeneration occurs at temperatures between 90-120°C, significantly lower than conventional amine scrubbing processes. Sinopec has conducted extensive pilot testing at coal-fired power plants in China, demonstrating sorbent stability over thousands of adsorption-desorption cycles with minimal performance degradation. Their most recent developments include composite materials incorporating heat-conducting elements to improve thermal management during regeneration, reducing energy penalties by approximately 30% compared to first-generation solid sorbents. Sinopec has also explored integration of their capture technology with existing industrial processes to utilize waste heat for sorbent regeneration.
Strengths: Extensive industrial implementation experience at large-scale facilities; sorbent materials optimized for real-world flue gas conditions; strong integration with existing power plant infrastructure; access to significant R&D resources. Weaknesses: Technology primarily focused on point-source emissions rather than direct air capture; potential challenges with sorbent poisoning from flue gas contaminants over extended operation; regeneration still requires substantial thermal energy input.
Key Advancements in Sorbent Material Properties
Solid sorbents for capturing co 2
PatentWO2023232666A1
Innovation
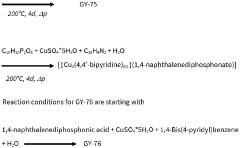
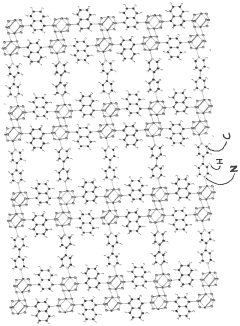
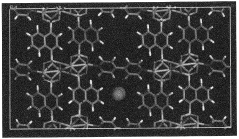
- Development of phosphonate and organoarsonate MOFs with specific molecular formulas, such as [{M2(4,4’-bipyridine)0.5}(l,4-naphthalenediphosphonate)] and [{M2(4,4’-bipyridine)0.5}(l,4-naphthalenediarsonate)], which maintain selectivity and stability under harsh conditions, including high humidity and temperatures up to 360°C, by creating a hydrophobic environment that favors CO2 physisorption over H2O.
Environmental Impact Assessment of Sorbent Materials
The environmental impact of solid sorbents for CO2 capture extends beyond their primary function of reducing greenhouse gas emissions. Life cycle assessment (LCA) studies reveal that while these materials contribute positively to climate change mitigation, their production, use, and disposal create environmental footprints that must be carefully evaluated.
Production of solid sorbents, particularly amine-functionalized materials, often involves energy-intensive processes and potentially hazardous chemicals. Zeolites require high-temperature calcination, while metal-organic frameworks (MOFs) frequently utilize solvents that may pose environmental risks if not properly managed. The extraction of raw materials for these sorbents, such as rare earth elements for certain MOFs, can lead to habitat destruction, water pollution, and soil degradation in mining regions.
During operational phases, solid sorbents demonstrate varying environmental profiles. Materials requiring high regeneration temperatures contribute to indirect CO2 emissions when powered by fossil fuels. Conversely, low-temperature sorbents like polyethylenimine-based materials offer reduced energy penalties but may face durability challenges, necessitating more frequent replacement and consequently increasing material consumption and waste generation.
Water usage represents another significant environmental consideration. Hydrophilic sorbents may compete with CO2 for adsorption sites, reducing capture efficiency in humid conditions while simultaneously depleting water resources during regeneration cycles. This water-energy nexus becomes particularly problematic in water-stressed regions where carbon capture facilities might operate.
End-of-life management of spent sorbents presents additional environmental challenges. Heavy metal content in certain MOFs and zeolites may classify them as hazardous waste, requiring specialized disposal procedures. Research into sorbent recycling remains limited, with few established protocols for material recovery or safe disposal pathways.
Toxicity assessments indicate varying levels of environmental risk. Amine-based sorbents can degrade to form potentially harmful compounds including nitrosamines, while dust from zeolite handling may pose respiratory hazards. Emerging nanoporous materials introduce uncertainties regarding long-term environmental persistence and potential ecosystem impacts.
Comparative analyses between different sorbent classes suggest that materials with longer operational lifespans, lower regeneration energy requirements, and reduced toxic component profiles generally demonstrate superior environmental performance. However, these advantages must be balanced against capture efficiency to ensure net positive environmental outcomes in real-world carbon capture applications.
Production of solid sorbents, particularly amine-functionalized materials, often involves energy-intensive processes and potentially hazardous chemicals. Zeolites require high-temperature calcination, while metal-organic frameworks (MOFs) frequently utilize solvents that may pose environmental risks if not properly managed. The extraction of raw materials for these sorbents, such as rare earth elements for certain MOFs, can lead to habitat destruction, water pollution, and soil degradation in mining regions.
During operational phases, solid sorbents demonstrate varying environmental profiles. Materials requiring high regeneration temperatures contribute to indirect CO2 emissions when powered by fossil fuels. Conversely, low-temperature sorbents like polyethylenimine-based materials offer reduced energy penalties but may face durability challenges, necessitating more frequent replacement and consequently increasing material consumption and waste generation.
Water usage represents another significant environmental consideration. Hydrophilic sorbents may compete with CO2 for adsorption sites, reducing capture efficiency in humid conditions while simultaneously depleting water resources during regeneration cycles. This water-energy nexus becomes particularly problematic in water-stressed regions where carbon capture facilities might operate.
End-of-life management of spent sorbents presents additional environmental challenges. Heavy metal content in certain MOFs and zeolites may classify them as hazardous waste, requiring specialized disposal procedures. Research into sorbent recycling remains limited, with few established protocols for material recovery or safe disposal pathways.
Toxicity assessments indicate varying levels of environmental risk. Amine-based sorbents can degrade to form potentially harmful compounds including nitrosamines, while dust from zeolite handling may pose respiratory hazards. Emerging nanoporous materials introduce uncertainties regarding long-term environmental persistence and potential ecosystem impacts.
Comparative analyses between different sorbent classes suggest that materials with longer operational lifespans, lower regeneration energy requirements, and reduced toxic component profiles generally demonstrate superior environmental performance. However, these advantages must be balanced against capture efficiency to ensure net positive environmental outcomes in real-world carbon capture applications.
Scalability and Economic Viability Analysis
The scalability of solid sorbents for CO2 capture represents a critical factor in their commercial viability. Current laboratory-scale demonstrations have shown promising results, but significant challenges emerge when considering industrial implementation. The transition from gram-scale to ton-scale production introduces manufacturing complexities, including maintaining consistent material properties, structural integrity, and adsorption performance. Materials such as metal-organic frameworks (MOFs) and amine-functionalized silica demonstrate excellent CO2 selectivity in controlled environments but face stability issues under prolonged industrial operating conditions.
Economic viability analysis reveals that material cost remains a primary barrier to widespread adoption. High-performance sorbents like zeolites and MOFs currently range from $50-200/kg, significantly higher than the $5-15/kg threshold identified for economical carbon capture operations. Production scaling could potentially reduce costs by 30-60%, according to recent techno-economic assessments, but requires substantial initial capital investment. The regeneration energy requirement, typically 2.5-4.0 GJ/ton CO2 for leading solid sorbents, compares favorably to liquid amine systems (3.5-5.0 GJ/ton CO2) but still impacts operational economics considerably.
Life-cycle analysis indicates that solid sorbents generally offer a 15-25% reduction in overall environmental impact compared to conventional amine scrubbing technologies. However, this advantage diminishes if sorbent replacement frequency exceeds projections due to degradation under real-world conditions. Current estimates suggest replacement intervals of 1-3 years for most advanced materials, though field demonstrations have shown variability depending on flue gas composition and operating parameters.
Infrastructure compatibility presents another dimension of scalability challenges. Retrofitting existing power plants and industrial facilities requires custom engineering solutions that can increase implementation costs by 25-40%. Modular designs being developed by companies like Carbon Clean and Svante aim to reduce this premium through standardized capture units, potentially bringing retrofit costs down to $30-50 per ton of CO2 capture capacity.
Market analysis projects that solid sorbent technologies could achieve economic viability in specific high-value applications by 2025-2027, with broader market penetration possible by 2030 if production scaling and material advances continue at current rates. Government incentives, such as carbon pricing mechanisms and tax credits for carbon capture, significantly influence the economic equation, potentially accelerating commercial deployment timelines by 3-5 years in supportive regulatory environments.
Economic viability analysis reveals that material cost remains a primary barrier to widespread adoption. High-performance sorbents like zeolites and MOFs currently range from $50-200/kg, significantly higher than the $5-15/kg threshold identified for economical carbon capture operations. Production scaling could potentially reduce costs by 30-60%, according to recent techno-economic assessments, but requires substantial initial capital investment. The regeneration energy requirement, typically 2.5-4.0 GJ/ton CO2 for leading solid sorbents, compares favorably to liquid amine systems (3.5-5.0 GJ/ton CO2) but still impacts operational economics considerably.
Life-cycle analysis indicates that solid sorbents generally offer a 15-25% reduction in overall environmental impact compared to conventional amine scrubbing technologies. However, this advantage diminishes if sorbent replacement frequency exceeds projections due to degradation under real-world conditions. Current estimates suggest replacement intervals of 1-3 years for most advanced materials, though field demonstrations have shown variability depending on flue gas composition and operating parameters.
Infrastructure compatibility presents another dimension of scalability challenges. Retrofitting existing power plants and industrial facilities requires custom engineering solutions that can increase implementation costs by 25-40%. Modular designs being developed by companies like Carbon Clean and Svante aim to reduce this premium through standardized capture units, potentially bringing retrofit costs down to $30-50 per ton of CO2 capture capacity.
Market analysis projects that solid sorbent technologies could achieve economic viability in specific high-value applications by 2025-2027, with broader market penetration possible by 2030 if production scaling and material advances continue at current rates. Government incentives, such as carbon pricing mechanisms and tax credits for carbon capture, significantly influence the economic equation, potentially accelerating commercial deployment timelines by 3-5 years in supportive regulatory environments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!