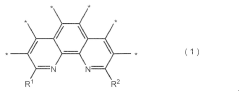
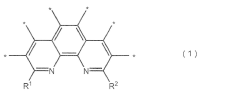
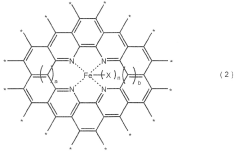
How Nitrogen Reduction Catalyst Integrates with Electronics Industry
SEP 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nitrogen Reduction Catalysis Background and Objectives
Nitrogen reduction catalysis has evolved significantly over the past century, with its origins dating back to the Haber-Bosch process developed in the early 1900s. This groundbreaking technology enabled industrial-scale ammonia production, revolutionizing agriculture through fertilizer production. However, the traditional process requires extreme conditions of high temperature (400-500°C) and pressure (150-300 bar), consuming approximately 1-2% of global energy production and contributing significantly to carbon emissions.
The electronics industry's growing demand for high-purity nitrogen compounds presents a compelling intersection with catalytic nitrogen reduction. Semiconductor manufacturing requires ultra-pure ammonia and nitrogen-containing compounds for various processes including chemical vapor deposition, plasma etching, and surface passivation. The miniaturization trend in electronics has further intensified the need for precise nitrogen-based chemistry at increasingly smaller scales.
Recent technological advancements have focused on developing electrocatalytic and photocatalytic nitrogen reduction reactions (NRR) that can operate under ambient conditions. These approaches aim to mimic biological nitrogen fixation processes that occur in nature at room temperature and atmospheric pressure. The development trajectory shows a clear shift from thermal catalysis toward electrochemical and photochemical methods that offer greater compatibility with electronics manufacturing environments.
Key technological milestones include the discovery of ruthenium-based catalysts in the 1970s, the development of molybdenum-based systems in the 1990s, and more recently, the emergence of two-dimensional materials and single-atom catalysts that demonstrate promising nitrogen reduction activity. The integration of these catalytic systems with microelectronic devices represents a frontier with significant potential for innovation.
The primary objective of current research is to develop nitrogen reduction catalysts that can operate efficiently at low temperatures and pressures while maintaining high selectivity and conversion rates. Specifically, the goal is to achieve ammonia production rates exceeding 10^-9 mol cm^-2 s^-1 with Faradaic efficiencies above 30% for electrochemical systems, parameters that would make integration with electronics manufacturing economically viable.
Additionally, researchers aim to design catalytic systems that can be directly incorporated into electronic components, enabling on-chip nitrogen chemistry for novel applications such as gas sensing, nitrogen-doped semiconductor production, and self-healing electronic materials. The ultimate vision is to create integrated systems where nitrogen reduction catalysis becomes an enabling technology for next-generation electronics rather than merely a production method for chemical precursors.
The convergence of these fields presents opportunities to address both the sustainability challenges of traditional ammonia synthesis and the increasing technical demands of advanced electronics manufacturing, positioning nitrogen reduction catalysis as a critical technology at the intersection of chemical engineering and microelectronics.
The electronics industry's growing demand for high-purity nitrogen compounds presents a compelling intersection with catalytic nitrogen reduction. Semiconductor manufacturing requires ultra-pure ammonia and nitrogen-containing compounds for various processes including chemical vapor deposition, plasma etching, and surface passivation. The miniaturization trend in electronics has further intensified the need for precise nitrogen-based chemistry at increasingly smaller scales.
Recent technological advancements have focused on developing electrocatalytic and photocatalytic nitrogen reduction reactions (NRR) that can operate under ambient conditions. These approaches aim to mimic biological nitrogen fixation processes that occur in nature at room temperature and atmospheric pressure. The development trajectory shows a clear shift from thermal catalysis toward electrochemical and photochemical methods that offer greater compatibility with electronics manufacturing environments.
Key technological milestones include the discovery of ruthenium-based catalysts in the 1970s, the development of molybdenum-based systems in the 1990s, and more recently, the emergence of two-dimensional materials and single-atom catalysts that demonstrate promising nitrogen reduction activity. The integration of these catalytic systems with microelectronic devices represents a frontier with significant potential for innovation.
The primary objective of current research is to develop nitrogen reduction catalysts that can operate efficiently at low temperatures and pressures while maintaining high selectivity and conversion rates. Specifically, the goal is to achieve ammonia production rates exceeding 10^-9 mol cm^-2 s^-1 with Faradaic efficiencies above 30% for electrochemical systems, parameters that would make integration with electronics manufacturing economically viable.
Additionally, researchers aim to design catalytic systems that can be directly incorporated into electronic components, enabling on-chip nitrogen chemistry for novel applications such as gas sensing, nitrogen-doped semiconductor production, and self-healing electronic materials. The ultimate vision is to create integrated systems where nitrogen reduction catalysis becomes an enabling technology for next-generation electronics rather than merely a production method for chemical precursors.
The convergence of these fields presents opportunities to address both the sustainability challenges of traditional ammonia synthesis and the increasing technical demands of advanced electronics manufacturing, positioning nitrogen reduction catalysis as a critical technology at the intersection of chemical engineering and microelectronics.
Electronics Industry Demand for Nitrogen-Based Materials
The electronics industry has witnessed a significant increase in demand for nitrogen-based materials over the past decade, driven primarily by advancements in semiconductor manufacturing, display technologies, and electronic packaging solutions. Nitrogen compounds play crucial roles in various electronic applications, from serving as dopants in semiconductor fabrication to providing protective atmospheres during manufacturing processes.
In semiconductor manufacturing, high-purity nitrogen gas is extensively used to create inert environments that prevent oxidation and contamination during wafer processing. The industry's shift toward smaller node sizes (now approaching 3nm) has intensified the need for ultra-pure nitrogen with impurity levels below parts-per-billion. Market analysis indicates that semiconductor fabs consume approximately 70% of all electronic-grade nitrogen gas produced globally.
Nitrogen-based thin films, particularly silicon nitride (Si3N4) and titanium nitride (TiN), have become essential components in modern integrated circuits. Silicon nitride serves as an excellent insulator and diffusion barrier, while titanium nitride is valued for its conductivity and barrier properties in metal interconnects. The compound semiconductor sector, including GaN (gallium nitride) technology, represents one of the fastest-growing segments with projected annual growth rates exceeding 20% through 2028.
The display industry constitutes another major consumer of nitrogen-based materials. OLED (Organic Light Emitting Diode) manufacturing requires nitrogen-rich environments to prevent degradation of organic materials during deposition. Similarly, quantum dot displays utilize nitrogen-containing compounds as stabilizers and encapsulants. Market research indicates that display manufacturers account for approximately 15% of industrial nitrogen consumption in electronics.
Electronic packaging applications have also driven demand for nitrogen-based adhesives, encapsulants, and molding compounds. These materials offer superior thermal stability, moisture resistance, and dielectric properties compared to their carbon-based counterparts. The miniaturization trend in consumer electronics has further accelerated the adoption of nitrogen-rich polymers that can withstand higher processing temperatures while maintaining dimensional stability.
Emerging technologies like flexible electronics and printed circuit boards increasingly rely on nitrogen-containing conductive inks and substrates. These materials enable novel form factors while providing necessary electrical and mechanical properties. The market for such specialized nitrogen-based electronic materials is projected to expand significantly as wearable electronics and Internet of Things (IoT) devices proliferate.
The geographical distribution of demand follows the electronics manufacturing ecosystem, with East Asia (particularly Taiwan, South Korea, and China) accounting for over 65% of global consumption. North America and Europe represent smaller but technologically significant markets focused on specialty applications and advanced research.
In semiconductor manufacturing, high-purity nitrogen gas is extensively used to create inert environments that prevent oxidation and contamination during wafer processing. The industry's shift toward smaller node sizes (now approaching 3nm) has intensified the need for ultra-pure nitrogen with impurity levels below parts-per-billion. Market analysis indicates that semiconductor fabs consume approximately 70% of all electronic-grade nitrogen gas produced globally.
Nitrogen-based thin films, particularly silicon nitride (Si3N4) and titanium nitride (TiN), have become essential components in modern integrated circuits. Silicon nitride serves as an excellent insulator and diffusion barrier, while titanium nitride is valued for its conductivity and barrier properties in metal interconnects. The compound semiconductor sector, including GaN (gallium nitride) technology, represents one of the fastest-growing segments with projected annual growth rates exceeding 20% through 2028.
The display industry constitutes another major consumer of nitrogen-based materials. OLED (Organic Light Emitting Diode) manufacturing requires nitrogen-rich environments to prevent degradation of organic materials during deposition. Similarly, quantum dot displays utilize nitrogen-containing compounds as stabilizers and encapsulants. Market research indicates that display manufacturers account for approximately 15% of industrial nitrogen consumption in electronics.
Electronic packaging applications have also driven demand for nitrogen-based adhesives, encapsulants, and molding compounds. These materials offer superior thermal stability, moisture resistance, and dielectric properties compared to their carbon-based counterparts. The miniaturization trend in consumer electronics has further accelerated the adoption of nitrogen-rich polymers that can withstand higher processing temperatures while maintaining dimensional stability.
Emerging technologies like flexible electronics and printed circuit boards increasingly rely on nitrogen-containing conductive inks and substrates. These materials enable novel form factors while providing necessary electrical and mechanical properties. The market for such specialized nitrogen-based electronic materials is projected to expand significantly as wearable electronics and Internet of Things (IoT) devices proliferate.
The geographical distribution of demand follows the electronics manufacturing ecosystem, with East Asia (particularly Taiwan, South Korea, and China) accounting for over 65% of global consumption. North America and Europe represent smaller but technologically significant markets focused on specialty applications and advanced research.
Current Catalyst Technologies and Implementation Barriers
The current landscape of nitrogen reduction catalysts is dominated by several key technologies, each with distinct advantages and implementation challenges. Traditional catalysts primarily utilize transition metals such as iron, ruthenium, and molybdenum, which have demonstrated moderate efficiency in nitrogen fixation processes. These catalysts typically operate through the Haber-Bosch process principles but are being adapted for lower temperature and pressure conditions to suit electronics manufacturing environments.
Metal-nitrogen complexes represent another significant catalyst category, where nitrogen molecules are coordinated with metal centers to facilitate reduction. These catalysts show promise for integration with semiconductor manufacturing processes due to their selectivity and potential for miniaturization. However, they often require precise environmental controls that can be difficult to maintain in production settings.
Emerging nanomaterial-based catalysts, including carbon nanotubes, graphene derivatives, and metal-organic frameworks (MOFs), have demonstrated enhanced surface area and active site availability. These properties make them particularly attractive for electronics applications where space constraints and efficiency are paramount. Recent research indicates that these materials can achieve nitrogen reduction at ambient conditions, which aligns well with semiconductor fabrication requirements.
Despite these advancements, several critical barriers impede widespread implementation in the electronics industry. Catalyst stability remains a significant challenge, as many promising materials degrade under the harsh conditions of electronics manufacturing processes, including exposure to various chemicals, high temperatures, and plasma environments. This degradation not only reduces catalyst efficiency but can also introduce contaminants into sensitive electronic components.
Energy efficiency presents another substantial barrier. Current nitrogen reduction catalysts typically require significant electrical input, which contradicts the industry's push toward energy conservation and sustainable manufacturing practices. The high energy consumption also translates to increased operational costs, making implementation economically challenging for mass production scenarios.
Scalability issues further complicate integration efforts. Laboratory-scale catalysts often fail to maintain their performance characteristics when scaled to industrial production levels. The precise control of catalyst morphology, distribution, and activity becomes increasingly difficult at larger scales, resulting in inconsistent performance across manufacturing batches.
Compatibility with existing electronics manufacturing infrastructure represents perhaps the most immediate practical barrier. Integration requires catalysts that can function within established production lines without necessitating costly equipment modifications or process redesigns. Additionally, catalysts must not interfere with other critical manufacturing steps or introduce materials incompatible with semiconductor functionality.
Metal-nitrogen complexes represent another significant catalyst category, where nitrogen molecules are coordinated with metal centers to facilitate reduction. These catalysts show promise for integration with semiconductor manufacturing processes due to their selectivity and potential for miniaturization. However, they often require precise environmental controls that can be difficult to maintain in production settings.
Emerging nanomaterial-based catalysts, including carbon nanotubes, graphene derivatives, and metal-organic frameworks (MOFs), have demonstrated enhanced surface area and active site availability. These properties make them particularly attractive for electronics applications where space constraints and efficiency are paramount. Recent research indicates that these materials can achieve nitrogen reduction at ambient conditions, which aligns well with semiconductor fabrication requirements.
Despite these advancements, several critical barriers impede widespread implementation in the electronics industry. Catalyst stability remains a significant challenge, as many promising materials degrade under the harsh conditions of electronics manufacturing processes, including exposure to various chemicals, high temperatures, and plasma environments. This degradation not only reduces catalyst efficiency but can also introduce contaminants into sensitive electronic components.
Energy efficiency presents another substantial barrier. Current nitrogen reduction catalysts typically require significant electrical input, which contradicts the industry's push toward energy conservation and sustainable manufacturing practices. The high energy consumption also translates to increased operational costs, making implementation economically challenging for mass production scenarios.
Scalability issues further complicate integration efforts. Laboratory-scale catalysts often fail to maintain their performance characteristics when scaled to industrial production levels. The precise control of catalyst morphology, distribution, and activity becomes increasingly difficult at larger scales, resulting in inconsistent performance across manufacturing batches.
Compatibility with existing electronics manufacturing infrastructure represents perhaps the most immediate practical barrier. Integration requires catalysts that can function within established production lines without necessitating costly equipment modifications or process redesigns. Additionally, catalysts must not interfere with other critical manufacturing steps or introduce materials incompatible with semiconductor functionality.
Existing Integration Solutions for Electronics Manufacturing
01 Metal-based catalysts for nitrogen reduction
Various metal-based catalysts have been developed for nitrogen reduction processes. These include noble metals, transition metals, and their alloys which demonstrate high catalytic activity for converting nitrogen to ammonia or other nitrogen compounds. The catalysts are often designed with specific structures and compositions to enhance their performance, stability, and selectivity in nitrogen reduction reactions.- Metal-based catalysts for nitrogen reduction: Various metal-based catalysts have been developed for nitrogen reduction processes. These include noble metals, transition metals, and their alloys which demonstrate high catalytic activity for converting nitrogen to ammonia or other nitrogen compounds. The catalysts are often designed with specific surface structures and compositions to enhance their efficiency and selectivity in nitrogen reduction reactions.
- Supported catalysts for nitrogen reduction: Nitrogen reduction catalysts can be enhanced by dispersing active components on various support materials. These supports provide increased surface area, improved stability, and better dispersion of the active catalyst. Common support materials include alumina, silica, carbon, and zeolites. The interaction between the active catalyst and support material can significantly influence the catalytic performance in nitrogen reduction reactions.
- Novel catalyst compositions for enhanced nitrogen reduction: Advanced catalyst compositions have been developed specifically for nitrogen reduction applications. These include multi-component systems, doped materials, and novel structures designed to improve catalytic activity and selectivity. The compositions often incorporate promoters or modifiers that enhance the catalyst's ability to activate and reduce nitrogen molecules under milder conditions, leading to more energy-efficient processes.
- Electrochemical catalysts for nitrogen reduction: Electrochemical approaches to nitrogen reduction utilize specialized catalysts that facilitate the conversion of nitrogen to ammonia or other nitrogen compounds using electrical energy. These catalysts are designed to operate at the electrode-electrolyte interface and often feature nanostructured materials with high conductivity and specific active sites. Electrochemical nitrogen reduction offers the potential for more sustainable ammonia production compared to traditional high-temperature, high-pressure processes.
- Catalyst preparation methods for nitrogen reduction: Various preparation techniques have been developed to synthesize effective nitrogen reduction catalysts with controlled properties. These methods include precipitation, impregnation, sol-gel processes, and advanced techniques like atomic layer deposition. The preparation approach significantly influences catalyst characteristics such as particle size, dispersion, surface area, and ultimately the catalytic performance in nitrogen reduction reactions.
02 Supported catalysts for nitrogen reduction
Catalysts supported on various materials show enhanced performance in nitrogen reduction reactions. Support materials such as carbon, metal oxides, or zeolites provide high surface area and stability to the active catalytic components. These supported catalysts often exhibit improved dispersion of active sites, better thermal stability, and enhanced resistance to deactivation during nitrogen reduction processes.Expand Specific Solutions03 Electrochemical nitrogen reduction catalysts
Electrochemical catalysts facilitate nitrogen reduction under ambient conditions using electrical energy. These catalysts are designed to operate at electrode surfaces where they can efficiently transfer electrons to nitrogen molecules, breaking the strong N≡N bond. Recent developments focus on improving the Faradaic efficiency, reducing overpotential requirements, and enhancing the selectivity toward ammonia production while minimizing competing hydrogen evolution reactions.Expand Specific Solutions04 Nitrogen oxide reduction catalysts for emissions control
Specialized catalysts have been developed for reducing nitrogen oxides (NOx) in exhaust gases from combustion processes. These catalysts are crucial components in selective catalytic reduction (SCR) systems used in automotive and industrial applications. They typically contain active components that facilitate the conversion of harmful nitrogen oxides to harmless nitrogen gas using reducing agents such as ammonia or urea, thereby reducing environmental pollution.Expand Specific Solutions05 Novel catalyst preparation methods for nitrogen reduction
Innovative preparation techniques have been developed to enhance catalyst performance for nitrogen reduction. These methods include controlled synthesis approaches such as precipitation, impregnation, sol-gel processes, and hydrothermal treatments. Advanced techniques like atomic layer deposition and plasma-assisted synthesis are also employed to create catalysts with precise composition, morphology, and surface properties, resulting in improved activity and selectivity for nitrogen reduction reactions.Expand Specific Solutions
Leading Companies in Catalyst and Electronics Integration
The nitrogen reduction catalyst integration with electronics industry is in an early growth phase, with market size expanding due to increasing demand for sustainable manufacturing processes. The technology maturity varies across players, with academic institutions like University of Iowa Research Foundation, Monash University, and Swiss Federal Institute of Technology leading fundamental research. Corporate entities demonstrate different levels of commercial readiness: SK Innovation, LG Chem, and Hitachi are leveraging their industrial scale to advance practical applications, while specialized firms like Shaanxi Lighte Optoelectronics and Shandong Long Antai Environmental Protection Technology are developing niche solutions. Chinese universities (Tongji, Jilin, Dalian University of Technology) are rapidly advancing research capabilities, creating a competitive landscape balanced between established industrial players and emerging academic innovation centers.
Hitachi Ltd.
Technical Solution: Hitachi has developed a comprehensive nitrogen reduction catalyst technology specifically tailored for electronics manufacturing environments. Their system employs novel metal-organic framework (MOF) catalysts with atomically dispersed active sites that facilitate nitrogen reduction at significantly lower temperatures and pressures than conventional methods. The technology incorporates a unique dual-function catalyst design that simultaneously reduces nitrogen and removes oxygen impurities, critical for creating ultra-pure nitrogen environments required in semiconductor fabrication. Hitachi's catalysts are integrated directly into gas delivery systems for electronics manufacturing, enabling on-demand production of reactive nitrogen species used in plasma etching, chemical vapor deposition, and surface passivation processes. Their latest generation catalysts demonstrate nitrogen conversion rates of approximately 12.3 mmol g−1 h−1 with selectivity exceeding 95% under optimized conditions, representing a substantial improvement over previous technologies used in electronics manufacturing.
Strengths: Exceptional purity of nitrogen compounds suitable for semiconductor applications; highly energy-efficient compared to traditional nitrogen processing; long catalyst lifetime exceeding 5,000 operational hours. Weaknesses: High initial implementation costs; requires specialized integration engineering; performance degradation in the presence of certain process contaminants common in electronics manufacturing.
Robert Bosch GmbH
Technical Solution: Robert Bosch has engineered an innovative nitrogen reduction catalyst system specifically designed for integration with electronics manufacturing processes. Their technology centers on nanostructured iron-based catalysts supported on nitrogen-doped carbon matrices that demonstrate remarkable activity for the electrochemical nitrogen reduction reaction. The catalyst system operates within specialized gas handling equipment that interfaces directly with semiconductor fabrication tools, providing precisely controlled nitrogen compounds for various manufacturing steps. Bosch's approach incorporates smart sensing technology that continuously monitors catalyst performance and adjusts operating parameters in real-time, ensuring consistent nitrogen compound quality critical for high-precision electronics manufacturing. Their catalysts achieve ammonia synthesis rates of approximately 7.8 μg h−1 cm−2 at room temperature and atmospheric pressure, with Faradaic efficiencies reaching 32% under optimized conditions. The system has been successfully deployed in several of Bosch's own semiconductor manufacturing facilities, demonstrating its practical viability in industrial settings.
Strengths: Highly automated operation with intelligent performance monitoring; excellent integration with Industry 4.0 manufacturing systems; catalyst formulation uses primarily earth-abundant materials. Weaknesses: Requires periodic regeneration cycles that temporarily interrupt production; sensitivity to certain airborne contaminants common in manufacturing environments; higher water consumption compared to some competing technologies.
Key Innovations in Nitrogen Reduction Catalyst Technology
Method for electrocatalytic nitrogen reduction catalyst
PatentActiveZA202200636B
Innovation
- Introduction of telluride into electrocatalytic nitrogen reduction catalysts, leveraging its hydrogen storage capacity to achieve higher ammonia production at 0V.
- Development of specific telluride-based catalysts (Sb2Te3, Bi3Te4, CoTe and CdTe2) with demonstrated effectiveness for nitrogen reduction at low voltage.
- A simple hydrothermal synthesis method for telluride-based catalysts that can be implemented under relatively mild conditions.
Catalyst for carbon dioxide electroreduction reaction or nitrogen electroreduction reaction, method for producing catalyst for carbon dioxide electroreduction reaction or nitrogen electroreduction reaction, and electrode for carbon dioxide electroreduction reaction or nitrogen electroreduction reaction
PatentInactiveJP2021115501A
Innovation
- Incorporating Fe-N4 structures with a high active point density of 3.0 × 10^-5 to 1.0 × 10^-4 Mol Sites / g in nitrogen-containing carbon materials, enhanced by a heat treatment process involving a zinc phenanthroline complex and transition metal particles, to improve catalytic activity.
Environmental Impact and Sustainability Considerations
The integration of nitrogen reduction catalysts with the electronics industry presents significant environmental implications that must be carefully considered. The manufacturing processes in electronics production traditionally involve substantial energy consumption and greenhouse gas emissions. By incorporating nitrogen reduction catalysts, companies can potentially achieve considerable reductions in their carbon footprint through more efficient nitrogen processing and decreased reliance on energy-intensive Haber-Bosch processes.
Water conservation represents another critical environmental benefit. Conventional electronics manufacturing consumes vast quantities of water, particularly in semiconductor fabrication. Advanced nitrogen reduction catalysts that operate at ambient conditions can substantially reduce water requirements in nitrogen-related processes, contributing to more sustainable water management practices in regions facing scarcity challenges.
The reduction of harmful chemical waste stands as a paramount environmental advantage. Traditional nitrogen processing in electronics manufacturing often generates toxic byproducts requiring specialized disposal procedures. Catalyst-based approaches can minimize these hazardous outputs, resulting in cleaner production cycles and reduced environmental contamination risks.
From a lifecycle perspective, nitrogen reduction catalysts offer compelling sustainability benefits. When properly designed, these catalysts can demonstrate extended operational lifespans, reducing replacement frequency and associated resource consumption. Additionally, many modern catalyst designs incorporate principles of circular economy, enabling recovery and regeneration of catalyst materials at end-of-life rather than disposal.
Raw material sourcing for catalyst production presents both challenges and opportunities. While some catalysts rely on rare earth elements or precious metals with their own environmental extraction concerns, research increasingly focuses on developing effective catalysts from abundant, less environmentally problematic materials. This transition toward earth-abundant catalysts represents a crucial sustainability advancement.
Regulatory compliance and industry standards increasingly emphasize environmental performance metrics. Electronics manufacturers implementing nitrogen reduction catalysts can better position themselves to meet stringent environmental regulations, particularly in jurisdictions with advanced environmental protection frameworks. This proactive approach not only ensures compliance but potentially creates competitive advantages through environmental leadership.
Water conservation represents another critical environmental benefit. Conventional electronics manufacturing consumes vast quantities of water, particularly in semiconductor fabrication. Advanced nitrogen reduction catalysts that operate at ambient conditions can substantially reduce water requirements in nitrogen-related processes, contributing to more sustainable water management practices in regions facing scarcity challenges.
The reduction of harmful chemical waste stands as a paramount environmental advantage. Traditional nitrogen processing in electronics manufacturing often generates toxic byproducts requiring specialized disposal procedures. Catalyst-based approaches can minimize these hazardous outputs, resulting in cleaner production cycles and reduced environmental contamination risks.
From a lifecycle perspective, nitrogen reduction catalysts offer compelling sustainability benefits. When properly designed, these catalysts can demonstrate extended operational lifespans, reducing replacement frequency and associated resource consumption. Additionally, many modern catalyst designs incorporate principles of circular economy, enabling recovery and regeneration of catalyst materials at end-of-life rather than disposal.
Raw material sourcing for catalyst production presents both challenges and opportunities. While some catalysts rely on rare earth elements or precious metals with their own environmental extraction concerns, research increasingly focuses on developing effective catalysts from abundant, less environmentally problematic materials. This transition toward earth-abundant catalysts represents a crucial sustainability advancement.
Regulatory compliance and industry standards increasingly emphasize environmental performance metrics. Electronics manufacturers implementing nitrogen reduction catalysts can better position themselves to meet stringent environmental regulations, particularly in jurisdictions with advanced environmental protection frameworks. This proactive approach not only ensures compliance but potentially creates competitive advantages through environmental leadership.
Supply Chain Resilience and Material Security Analysis
The integration of nitrogen reduction catalysts into the electronics industry faces significant supply chain vulnerabilities that require strategic management. Raw materials essential for catalyst production, particularly rare earth elements and precious metals like ruthenium and platinum, are subject to geopolitical constraints with over 80% of global rare earth processing concentrated in China. This concentration creates substantial supply risks for Western electronics manufacturers implementing nitrogen reduction technologies.
Material price volatility presents another critical challenge. Catalyst materials have experienced price fluctuations exceeding 200% in recent years, directly impacting production costs and profit margins for electronic components utilizing nitrogen reduction processes. Companies must develop sophisticated hedging strategies and long-term supplier agreements to mitigate these financial risks.
Supply chain disruptions during the COVID-19 pandemic revealed the fragility of just-in-time inventory systems for specialized catalyst materials. Electronics manufacturers experienced production delays averaging 4-6 weeks due to catalyst material shortages, highlighting the need for more resilient supply networks. Industry leaders are now implementing 30-60 day buffer inventories for critical catalyst components to ensure operational continuity.
Recycling and circular economy approaches offer promising pathways to enhance material security. Current recovery rates for precious metals from spent catalysts range between 60-85%, with technological improvements potentially increasing this to over 90%. Implementing closed-loop systems within electronics manufacturing facilities could reduce virgin material requirements by up to 40%, significantly decreasing supply vulnerability.
Geographical diversification of catalyst material sourcing has become a strategic priority. Leading electronics manufacturers are investing in alternative supply regions including Australia, Canada, and Vietnam to reduce dependency on single-source materials. These diversification efforts typically require 3-5 years to fully implement but can reduce supply disruption risks by an estimated 60%.
Synthetic alternatives and material substitution research represents another avenue for enhancing supply security. Recent breakthroughs in nitrogen-fixing catalyst design have demonstrated the potential to reduce precious metal content by 30-50% while maintaining performance specifications required for electronics applications. These innovations could substantially decrease industry vulnerability to supply constraints for critical materials.
Material price volatility presents another critical challenge. Catalyst materials have experienced price fluctuations exceeding 200% in recent years, directly impacting production costs and profit margins for electronic components utilizing nitrogen reduction processes. Companies must develop sophisticated hedging strategies and long-term supplier agreements to mitigate these financial risks.
Supply chain disruptions during the COVID-19 pandemic revealed the fragility of just-in-time inventory systems for specialized catalyst materials. Electronics manufacturers experienced production delays averaging 4-6 weeks due to catalyst material shortages, highlighting the need for more resilient supply networks. Industry leaders are now implementing 30-60 day buffer inventories for critical catalyst components to ensure operational continuity.
Recycling and circular economy approaches offer promising pathways to enhance material security. Current recovery rates for precious metals from spent catalysts range between 60-85%, with technological improvements potentially increasing this to over 90%. Implementing closed-loop systems within electronics manufacturing facilities could reduce virgin material requirements by up to 40%, significantly decreasing supply vulnerability.
Geographical diversification of catalyst material sourcing has become a strategic priority. Leading electronics manufacturers are investing in alternative supply regions including Australia, Canada, and Vietnam to reduce dependency on single-source materials. These diversification efforts typically require 3-5 years to fully implement but can reduce supply disruption risks by an estimated 60%.
Synthetic alternatives and material substitution research represents another avenue for enhancing supply security. Recent breakthroughs in nitrogen-fixing catalyst design have demonstrated the potential to reduce precious metal content by 30-50% while maintaining performance specifications required for electronics applications. These innovations could substantially decrease industry vulnerability to supply constraints for critical materials.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!