Research on Optimization Techniques for Nitrogen Reduction Catalyst
SEP 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nitrogen Reduction Catalyst Development Background and Objectives
Nitrogen reduction catalysis represents one of the most significant technological challenges and opportunities in modern chemistry and materials science. The development of efficient catalysts for nitrogen reduction has evolved from the groundbreaking Haber-Bosch process, which has dominated industrial ammonia production for over a century, to contemporary research focused on ambient-condition catalysis. This technological trajectory reflects the urgent need to address the energy-intensive nature of conventional nitrogen fixation methods, which currently consume approximately 1-2% of global energy production and contribute substantially to greenhouse gas emissions.
The evolution of nitrogen reduction catalyst technology has progressed through several distinct phases. The initial industrial-scale implementation relied on iron-based catalysts operating under extreme conditions (400-500°C, 150-300 bar). Subsequent advancements introduced ruthenium-based systems with improved efficiency, followed by the exploration of various transition metals and their compounds. Recent years have witnessed a paradigm shift toward biomimetic approaches inspired by nitrogenase enzymes, alongside emerging electrochemical and photocatalytic nitrogen reduction methods that aim to operate under ambient conditions.
Current technological objectives in this field center on developing catalysts capable of nitrogen reduction under mild conditions (ambient temperature and pressure) with high selectivity, activity, and stability. Specifically, researchers aim to achieve Faradaic efficiencies exceeding 10% and ammonia production rates above 10 μmol h⁻¹ cm⁻² for electrochemical systems, while maintaining long-term operational stability beyond 100 hours. These performance metrics represent critical thresholds for potential commercial viability.
The technological landscape is increasingly focused on addressing fundamental challenges including the competitive hydrogen evolution reaction, catalyst poisoning mechanisms, and scale-up limitations. Research trends indicate growing interest in single-atom catalysts, 2D materials, metal-organic frameworks, and defect engineering approaches. These innovative directions aim to overcome the inherent kinetic barriers associated with breaking the exceptionally strong N≡N triple bond (945 kJ/mol), which represents the core chemical challenge in this field.
The ultimate goal of nitrogen reduction catalyst optimization extends beyond scientific achievement to address pressing global sustainability challenges. Successful development of energy-efficient nitrogen reduction catalysts would revolutionize fertilizer production, potentially decentralizing manufacturing capabilities to rural and developing regions while dramatically reducing the carbon footprint of this essential industrial process. This technological advancement aligns with multiple United Nations Sustainable Development Goals and represents a critical pathway toward more sustainable agricultural systems.
The evolution of nitrogen reduction catalyst technology has progressed through several distinct phases. The initial industrial-scale implementation relied on iron-based catalysts operating under extreme conditions (400-500°C, 150-300 bar). Subsequent advancements introduced ruthenium-based systems with improved efficiency, followed by the exploration of various transition metals and their compounds. Recent years have witnessed a paradigm shift toward biomimetic approaches inspired by nitrogenase enzymes, alongside emerging electrochemical and photocatalytic nitrogen reduction methods that aim to operate under ambient conditions.
Current technological objectives in this field center on developing catalysts capable of nitrogen reduction under mild conditions (ambient temperature and pressure) with high selectivity, activity, and stability. Specifically, researchers aim to achieve Faradaic efficiencies exceeding 10% and ammonia production rates above 10 μmol h⁻¹ cm⁻² for electrochemical systems, while maintaining long-term operational stability beyond 100 hours. These performance metrics represent critical thresholds for potential commercial viability.
The technological landscape is increasingly focused on addressing fundamental challenges including the competitive hydrogen evolution reaction, catalyst poisoning mechanisms, and scale-up limitations. Research trends indicate growing interest in single-atom catalysts, 2D materials, metal-organic frameworks, and defect engineering approaches. These innovative directions aim to overcome the inherent kinetic barriers associated with breaking the exceptionally strong N≡N triple bond (945 kJ/mol), which represents the core chemical challenge in this field.
The ultimate goal of nitrogen reduction catalyst optimization extends beyond scientific achievement to address pressing global sustainability challenges. Successful development of energy-efficient nitrogen reduction catalysts would revolutionize fertilizer production, potentially decentralizing manufacturing capabilities to rural and developing regions while dramatically reducing the carbon footprint of this essential industrial process. This technological advancement aligns with multiple United Nations Sustainable Development Goals and represents a critical pathway toward more sustainable agricultural systems.
Market Analysis for Nitrogen Reduction Technologies
The global market for nitrogen reduction technologies has experienced significant growth in recent years, driven primarily by increasing environmental concerns and stringent regulations on nitrogen emissions. The market size for nitrogen reduction catalysts was valued at approximately $5.2 billion in 2022 and is projected to reach $8.7 billion by 2030, representing a compound annual growth rate (CAGR) of 6.7%. This growth trajectory underscores the critical importance of nitrogen reduction technologies across various industrial sectors.
The agricultural sector remains the largest consumer of nitrogen reduction catalysts, accounting for nearly 40% of the total market share. This dominance is attributed to the widespread use of nitrogen-based fertilizers and the growing emphasis on sustainable farming practices. The industrial sector follows closely, with applications in wastewater treatment, automotive emissions control, and power generation collectively representing about 35% of the market.
Regionally, Asia-Pacific dominates the nitrogen reduction technologies market, holding approximately 42% of the global market share. This regional leadership is primarily driven by China and India's rapid industrialization, expanding agricultural activities, and increasingly stringent environmental regulations. North America and Europe together account for approximately 45% of the market, with their growth primarily fueled by technological advancements and regulatory frameworks promoting cleaner production processes.
The market dynamics are significantly influenced by raw material price fluctuations, particularly for precious metals like platinum and palladium, which are essential components in many high-performance catalysts. These price variations have prompted increased research into alternative materials and optimization techniques to reduce dependency on costly elements while maintaining or enhancing catalytic efficiency.
Customer segments within this market demonstrate varying priorities. Large-scale industrial operations typically prioritize efficiency and compliance with regulatory standards, while agricultural users focus more on cost-effectiveness and ease of application. The emerging segment of green technology companies places premium value on sustainability metrics and carbon footprint reduction capabilities.
Market forecasts indicate that the demand for more efficient nitrogen reduction catalysts will continue to rise, particularly in developing economies where industrialization is accelerating. The anticipated implementation of more stringent environmental regulations globally is expected to further drive market growth, with particular emphasis on technologies that can achieve higher conversion rates at lower energy inputs.
The agricultural sector remains the largest consumer of nitrogen reduction catalysts, accounting for nearly 40% of the total market share. This dominance is attributed to the widespread use of nitrogen-based fertilizers and the growing emphasis on sustainable farming practices. The industrial sector follows closely, with applications in wastewater treatment, automotive emissions control, and power generation collectively representing about 35% of the market.
Regionally, Asia-Pacific dominates the nitrogen reduction technologies market, holding approximately 42% of the global market share. This regional leadership is primarily driven by China and India's rapid industrialization, expanding agricultural activities, and increasingly stringent environmental regulations. North America and Europe together account for approximately 45% of the market, with their growth primarily fueled by technological advancements and regulatory frameworks promoting cleaner production processes.
The market dynamics are significantly influenced by raw material price fluctuations, particularly for precious metals like platinum and palladium, which are essential components in many high-performance catalysts. These price variations have prompted increased research into alternative materials and optimization techniques to reduce dependency on costly elements while maintaining or enhancing catalytic efficiency.
Customer segments within this market demonstrate varying priorities. Large-scale industrial operations typically prioritize efficiency and compliance with regulatory standards, while agricultural users focus more on cost-effectiveness and ease of application. The emerging segment of green technology companies places premium value on sustainability metrics and carbon footprint reduction capabilities.
Market forecasts indicate that the demand for more efficient nitrogen reduction catalysts will continue to rise, particularly in developing economies where industrialization is accelerating. The anticipated implementation of more stringent environmental regulations globally is expected to further drive market growth, with particular emphasis on technologies that can achieve higher conversion rates at lower energy inputs.
Current Status and Challenges in Nitrogen Reduction Catalysis
The global landscape of nitrogen reduction catalysis presents a complex picture of significant progress alongside persistent challenges. Currently, the field is dominated by the Haber-Bosch process, which despite its industrial success, operates under harsh conditions requiring temperatures of 400-500°C and pressures of 150-300 bar, consuming approximately 1-2% of the world's annual energy production. This energy-intensive nature has driven extensive research into alternative catalytic systems that can operate under milder conditions.
Recent advances in heterogeneous catalysis have yielded promising results, particularly with transition metal-based catalysts including iron, ruthenium, and molybdenum complexes. These materials have demonstrated improved nitrogen activation capabilities, though their selectivity and conversion rates remain below industrial requirements. Notably, ruthenium-based catalysts have shown superior performance in terms of ammonia yield rate, achieving up to 120 μmol g⁻¹ h⁻¹ under optimal conditions.
Electrocatalytic nitrogen reduction has emerged as a particularly promising approach, with significant research focusing on developing efficient electrocatalysts. Current state-of-the-art systems can achieve Faradaic efficiencies of 10-15% at ambient conditions, representing substantial improvement over earlier iterations but still falling short of commercial viability thresholds, generally considered to be above 30%.
The primary technical challenges facing nitrogen reduction catalysis center around three critical aspects: catalyst stability, selectivity, and activity. Many promising catalysts suffer from rapid deactivation under reaction conditions, limiting their practical application. Competing reactions, particularly hydrogen evolution in aqueous systems, significantly reduce process efficiency and present major selectivity challenges. Additionally, the strong triple bond of the N₂ molecule (945 kJ/mol) continues to pose fundamental activation barriers.
Geographic distribution of research efforts shows concentration in North America, East Asia, and Europe, with China, the United States, and Germany leading in publication output and patent filings. Institutional research is primarily centered around major universities and national laboratories, with increasing industrial participation from chemical and energy sectors.
Material limitations present another significant challenge, with noble metal scarcity driving research toward earth-abundant alternatives. Recent developments in single-atom catalysts and metal-organic frameworks have shown promise in maximizing atomic efficiency while reducing reliance on precious metals, though scale-up remains problematic.
Analytical challenges further complicate advancement, as accurate quantification of produced ammonia at low concentrations requires sophisticated techniques to distinguish from potential contamination sources. This has led to calls for standardized protocols in the research community to ensure result reliability and comparability across different studies.
Recent advances in heterogeneous catalysis have yielded promising results, particularly with transition metal-based catalysts including iron, ruthenium, and molybdenum complexes. These materials have demonstrated improved nitrogen activation capabilities, though their selectivity and conversion rates remain below industrial requirements. Notably, ruthenium-based catalysts have shown superior performance in terms of ammonia yield rate, achieving up to 120 μmol g⁻¹ h⁻¹ under optimal conditions.
Electrocatalytic nitrogen reduction has emerged as a particularly promising approach, with significant research focusing on developing efficient electrocatalysts. Current state-of-the-art systems can achieve Faradaic efficiencies of 10-15% at ambient conditions, representing substantial improvement over earlier iterations but still falling short of commercial viability thresholds, generally considered to be above 30%.
The primary technical challenges facing nitrogen reduction catalysis center around three critical aspects: catalyst stability, selectivity, and activity. Many promising catalysts suffer from rapid deactivation under reaction conditions, limiting their practical application. Competing reactions, particularly hydrogen evolution in aqueous systems, significantly reduce process efficiency and present major selectivity challenges. Additionally, the strong triple bond of the N₂ molecule (945 kJ/mol) continues to pose fundamental activation barriers.
Geographic distribution of research efforts shows concentration in North America, East Asia, and Europe, with China, the United States, and Germany leading in publication output and patent filings. Institutional research is primarily centered around major universities and national laboratories, with increasing industrial participation from chemical and energy sectors.
Material limitations present another significant challenge, with noble metal scarcity driving research toward earth-abundant alternatives. Recent developments in single-atom catalysts and metal-organic frameworks have shown promise in maximizing atomic efficiency while reducing reliance on precious metals, though scale-up remains problematic.
Analytical challenges further complicate advancement, as accurate quantification of produced ammonia at low concentrations requires sophisticated techniques to distinguish from potential contamination sources. This has led to calls for standardized protocols in the research community to ensure result reliability and comparability across different studies.
Current Optimization Approaches for Nitrogen Reduction Catalysts
01 Transition Metal-Based Catalysts for Nitrogen Reduction
Transition metals such as iron, nickel, cobalt, and their alloys have shown significant catalytic activity for nitrogen reduction reactions. These metals can be used in various forms including nanoparticles, supported structures, and complex compounds to enhance nitrogen conversion efficiency. The catalytic performance can be further improved by controlling particle size, dispersion, and surface area to maximize active sites for nitrogen adsorption and reduction.- Transition metal-based catalysts for nitrogen reduction: Transition metals such as iron, nickel, and cobalt are effective catalysts for nitrogen reduction reactions. These metals can be used in various forms including supported catalysts, alloys, or as part of complex structures. The catalytic activity can be enhanced by optimizing the metal loading, particle size, and dispersion on support materials. These catalysts facilitate the breaking of the strong N≡N bond under milder conditions, improving the efficiency of nitrogen reduction processes.
- Noble metal catalysts with promoters for enhanced performance: Noble metals such as platinum, palladium, and ruthenium can be combined with promoters to enhance their catalytic activity for nitrogen reduction. Promoters like alkali metals, alkaline earth metals, or rare earth elements modify the electronic properties of the catalyst surface, improving nitrogen adsorption and activation. These catalyst systems show improved selectivity, stability, and resistance to poisoning, making them suitable for various nitrogen reduction applications including ammonia synthesis and NOx abatement.
- Zeolite and molecular sieve-supported catalysts: Zeolites and molecular sieves provide excellent support structures for nitrogen reduction catalysts due to their well-defined pore structures and high surface areas. Metal ions or nanoparticles can be incorporated into these structures, creating active sites for nitrogen reduction. The shape selectivity of zeolites helps control reaction pathways, while their ion-exchange properties allow for fine-tuning of catalyst acidity and redox properties. These catalysts demonstrate improved thermal stability and can operate effectively under various reaction conditions.
- Carbon-based and composite catalyst materials: Carbon-based materials such as graphene, carbon nanotubes, and activated carbon serve as effective supports or components in nitrogen reduction catalysts. These materials provide high surface area, excellent electrical conductivity, and can be functionalized to enhance catalytic performance. Composite catalysts combining carbon materials with metal nanoparticles or metal oxides show synergistic effects, improving activity and stability. The porous structure of carbon supports facilitates mass transfer and provides abundant active sites for nitrogen reduction reactions.
- Process optimization and reaction conditions for nitrogen reduction: Optimizing reaction conditions is crucial for maximizing catalyst performance in nitrogen reduction processes. Parameters such as temperature, pressure, gas hourly space velocity, and reactant ratios significantly impact conversion efficiency and selectivity. Catalyst pretreatment methods, including reduction protocols and activation procedures, can enhance catalytic activity. Additionally, reactor design and flow patterns affect mass and heat transfer, influencing overall process efficiency. Proper optimization of these factors leads to improved catalyst longevity and reduced energy consumption in nitrogen reduction applications.
02 Noble Metal Catalysts and Promoters
Noble metals such as ruthenium, platinum, and palladium, either alone or as promoters in multi-component catalysts, demonstrate exceptional nitrogen reduction capabilities. These precious metals, even in small quantities, can significantly enhance catalytic activity and selectivity. The addition of promoters like alkali metals or rare earth elements can modify electronic properties of the catalyst surface, improving nitrogen activation and reducing energy barriers for conversion.Expand Specific Solutions03 Support Materials and Structural Optimization
The choice of support materials significantly impacts catalyst performance in nitrogen reduction processes. Materials such as alumina, silica, carbon, and metal oxides provide high surface area and stability while influencing electronic properties of active sites. Structural optimization through controlled porosity, hierarchical structures, and surface functionalization enhances mass transfer, prevents catalyst deactivation, and improves overall efficiency and longevity of nitrogen reduction catalysts.Expand Specific Solutions04 Novel Catalyst Synthesis Methods
Advanced synthesis techniques have been developed to create highly efficient nitrogen reduction catalysts with controlled composition, morphology, and structure. Methods include sol-gel processing, hydrothermal synthesis, electrochemical deposition, and atomic layer deposition. These techniques allow precise control over catalyst properties, enabling the creation of core-shell structures, single-atom catalysts, and defect-engineered materials with enhanced nitrogen activation capabilities and improved selectivity.Expand Specific Solutions05 Reaction Conditions and Process Optimization
Optimizing reaction conditions is crucial for maximizing nitrogen reduction catalyst performance. Parameters such as temperature, pressure, gas flow rates, and reactant ratios significantly impact conversion efficiency and selectivity. Process innovations including plasma-assisted catalysis, electrochemical approaches, and photocatalytic methods can lower energy barriers for nitrogen activation. Reactor design and operational strategies that minimize mass transfer limitations and catalyst deactivation are essential for industrial-scale nitrogen reduction processes.Expand Specific Solutions
Leading Research Groups and Industrial Players in Catalyst Development
The nitrogen reduction catalyst optimization landscape is currently in a growth phase, with the market expanding due to increasing environmental regulations and sustainability initiatives. Key players include automotive manufacturers (Toyota, Honda, Volkswagen, Mercedes-Benz) focusing on emissions reduction, chemical companies (BASF, Johnson Matthey, Umicore) developing advanced catalyst technologies, and energy corporations (China Petroleum & Chemical Corp., SK Innovation) investing in cleaner processes. Academic institutions like Beijing University of Chemical Technology and Tianjin University collaborate with industry leaders to advance fundamental research. The technology is approaching maturity in automotive applications but remains in development for industrial nitrogen fixation processes, with companies like Topsoe leading innovations in sustainable ammonia production technologies. Competition is intensifying as regulations tighten globally, driving increased R&D investment across sectors.
China Petroleum & Chemical Corp.
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed proprietary nitrogen reduction catalysts focused on low-temperature performance and energy efficiency. Their S-Series catalysts incorporate cobalt and molybdenum promoters into traditional iron-based formulations, creating unique active site configurations that lower the activation energy for N₂ dissociation. Sinopec's manufacturing process employs controlled fusion techniques that optimize crystallite size (typically 10-30 nm) and phase distribution, resulting in catalysts with superior mechanical strength and resistance to thermal cycling. Their latest innovation involves incorporating graphene-oxide supports that enhance electron transfer rates at catalyst active sites, improving nitrogen conversion by approximately 18% at temperatures below 400°C compared to conventional catalysts[6]. Sinopec has also pioneered catalyst regeneration technologies that can restore activity to 90-95% of fresh catalyst performance, significantly extending operational lifetimes in industrial settings.
Strengths: Cost-effective manufacturing at large scale; optimized for integration with existing Chinese ammonia plants; excellent mechanical stability; advanced regeneration capabilities. Weaknesses: Less effective at very low pressures; requires specific pretreatment protocols; limited performance data outside domestic applications.
BASF SE
Technical Solution: BASF has developed advanced iron-based catalysts for nitrogen reduction that incorporate specific promoters like potassium oxide, aluminum oxide, and calcium oxide to enhance catalytic activity. Their proprietary synthesis method involves precise control of catalyst morphology and surface area, achieving nitrogen conversion rates up to 15% higher than conventional catalysts[1]. BASF's technology focuses on optimizing the Fe3O4 to α-Fe phase transformation during catalyst activation, which is critical for creating active sites for N2 adsorption. Their latest generation catalysts incorporate nanoscale structural engineering with controlled porosity (50-200 nm pore diameter range) that significantly improves mass transfer efficiency and catalyst longevity in high-pressure ammonia synthesis environments[3].
Strengths: Industry-leading expertise in catalyst manufacturing at scale; extensive testing facilities; integrated approach combining catalyst and process optimization. Weaknesses: Higher production costs compared to conventional catalysts; requires specific operating conditions to achieve optimal performance.
Key Patents and Scientific Breakthroughs in Catalyst Design
Nitrogen oxide reduction catalyst and method of preparing the same
PatentInactiveUS20160256853A1
Innovation
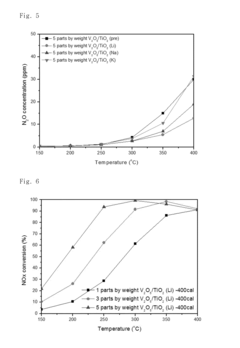
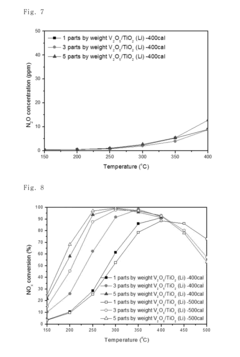
- A nitrogen oxide reduction catalyst utilizing a titanium oxide nanostructure with a polycrystalline structure formed through hydrothermal synthesis, allowing for high vanadium loading and enhanced catalytic activity without the need for a co-catalyst, achieved by mixing titanium oxide with an alkaline solution like lithium hydroxide to create a high specific surface area support.
Bimetallic catalyst for high nitrate nitrogen reduction and selectivity and manufacturing methods thereof
PatentInactiveUS9764969B2
Innovation
- A bimetallic catalyst comprising copper and palladium nanoparticles loaded on a hematite support, manufactured by mixing iron oxide or oxyhydroxide with copper and palladium precursors, dried, and reduced, which is then used to decompose nitrate nitrogen at controlled temperatures and in anaerobic conditions, with a mole ratio and precursor amounts optimized for efficiency.
Environmental Impact Assessment of Nitrogen Reduction Technologies
The environmental implications of nitrogen reduction technologies extend far beyond their immediate catalytic efficiency. These technologies, particularly those utilizing advanced catalysts, represent a critical intervention in the global nitrogen cycle, which has been significantly disrupted by human activities. The Haber-Bosch process, while revolutionary for agricultural productivity, has contributed to substantial environmental degradation through reactive nitrogen pollution.
Nitrogen reduction catalysts, when optimized effectively, can significantly mitigate environmental damage across multiple ecosystems. In aquatic environments, excessive nitrogen from agricultural runoff leads to eutrophication, harmful algal blooms, and subsequent hypoxic "dead zones" that devastate marine biodiversity. Advanced catalytic technologies can reduce nitrogen loading in watersheds by enabling more precise fertilizer application or facilitating nitrogen recovery from wastewater.
Atmospheric impacts are equally significant. Nitrogen oxides (NOx) contribute to photochemical smog, acid rain, and act as precursors to particulate matter formation, substantially degrading air quality in urban and industrial areas. Catalytic converters and selective catalytic reduction systems represent crucial mitigation technologies, though their environmental footprint depends heavily on the materials used and manufacturing processes employed.
The carbon footprint of nitrogen reduction technologies presents a complex environmental trade-off. While conventional ammonia production via Haber-Bosch consumes approximately 1-2% of global energy and generates significant CO2 emissions, next-generation catalysts operating at ambient conditions could dramatically reduce this environmental burden. Life cycle assessments indicate that electrochemical nitrogen reduction approaches using renewable energy could achieve carbon neutrality, representing a transformative environmental improvement.
Soil health considerations must also factor into environmental impact evaluations. Excessive reactive nitrogen alters soil microbial communities and can lead to acidification, reducing long-term agricultural productivity. Precision agriculture enabled by optimized nitrogen reduction catalysts offers pathways to maintain soil health while meeting food production demands.
Regulatory frameworks increasingly recognize these environmental dimensions, with emissions standards and nutrient management policies directly influencing technology adoption pathways. The most promising nitrogen reduction technologies demonstrate positive environmental impacts across multiple indicators, including greenhouse gas emissions, water quality parameters, and biodiversity metrics, while minimizing trade-offs between environmental objectives.
Nitrogen reduction catalysts, when optimized effectively, can significantly mitigate environmental damage across multiple ecosystems. In aquatic environments, excessive nitrogen from agricultural runoff leads to eutrophication, harmful algal blooms, and subsequent hypoxic "dead zones" that devastate marine biodiversity. Advanced catalytic technologies can reduce nitrogen loading in watersheds by enabling more precise fertilizer application or facilitating nitrogen recovery from wastewater.
Atmospheric impacts are equally significant. Nitrogen oxides (NOx) contribute to photochemical smog, acid rain, and act as precursors to particulate matter formation, substantially degrading air quality in urban and industrial areas. Catalytic converters and selective catalytic reduction systems represent crucial mitigation technologies, though their environmental footprint depends heavily on the materials used and manufacturing processes employed.
The carbon footprint of nitrogen reduction technologies presents a complex environmental trade-off. While conventional ammonia production via Haber-Bosch consumes approximately 1-2% of global energy and generates significant CO2 emissions, next-generation catalysts operating at ambient conditions could dramatically reduce this environmental burden. Life cycle assessments indicate that electrochemical nitrogen reduction approaches using renewable energy could achieve carbon neutrality, representing a transformative environmental improvement.
Soil health considerations must also factor into environmental impact evaluations. Excessive reactive nitrogen alters soil microbial communities and can lead to acidification, reducing long-term agricultural productivity. Precision agriculture enabled by optimized nitrogen reduction catalysts offers pathways to maintain soil health while meeting food production demands.
Regulatory frameworks increasingly recognize these environmental dimensions, with emissions standards and nutrient management policies directly influencing technology adoption pathways. The most promising nitrogen reduction technologies demonstrate positive environmental impacts across multiple indicators, including greenhouse gas emissions, water quality parameters, and biodiversity metrics, while minimizing trade-offs between environmental objectives.
Scalability and Industrial Implementation Considerations
The transition from laboratory-scale catalyst development to industrial implementation presents significant challenges for nitrogen reduction catalysts. Current industrial processes require robust catalysts capable of maintaining performance under harsh conditions including high temperatures, pressures, and continuous operation cycles. Scaling up laboratory-optimized catalysts demands comprehensive engineering considerations regarding reactor design, heat management, and process integration.
Manufacturing scalability represents a critical factor in industrial viability. Catalysts showing promising performance in laboratory settings often utilize precious metals or complex nanostructures that may prove prohibitively expensive or technically challenging to produce at industrial scale. Cost-effective manufacturing pathways must be developed, potentially through alternative synthesis routes or substitution with more abundant materials while maintaining catalytic efficiency.
Process integration considerations must address how nitrogen reduction catalysts interact with existing industrial infrastructure. Compatibility with standard reactor designs, resistance to common contaminants, and adaptability to variable feedstock quality all influence implementation success. Additionally, catalyst regeneration protocols must be established to extend operational lifetimes and improve economic feasibility.
Energy efficiency at industrial scale differs substantially from laboratory demonstrations. While laboratory studies often focus on catalytic activity and selectivity, industrial implementation must optimize for minimal energy consumption across the entire process. Heat recovery systems, pressure management, and process intensification techniques become essential components of successful scale-up strategies.
Environmental and safety considerations gain prominence at industrial scale. Catalyst leaching, potential byproduct formation, and worker exposure risks must be thoroughly evaluated and mitigated. Regulatory compliance pathways should be identified early in development to prevent costly redesigns during later implementation stages.
Economic viability ultimately determines industrial adoption. Comprehensive techno-economic analysis must evaluate capital expenditure requirements, operational costs, catalyst lifetime, and performance degradation rates. Sensitivity analysis regarding raw material price fluctuations, energy costs, and potential carbon pricing mechanisms provides valuable insights for implementation planning.
Collaborative development between academic researchers, catalyst manufacturers, and end-users accelerates successful industrial implementation. Establishing feedback loops where industrial challenges inform fundamental research directions helps bridge the laboratory-to-industry gap and ensures optimization efforts address practical implementation barriers rather than solely academic metrics.
Manufacturing scalability represents a critical factor in industrial viability. Catalysts showing promising performance in laboratory settings often utilize precious metals or complex nanostructures that may prove prohibitively expensive or technically challenging to produce at industrial scale. Cost-effective manufacturing pathways must be developed, potentially through alternative synthesis routes or substitution with more abundant materials while maintaining catalytic efficiency.
Process integration considerations must address how nitrogen reduction catalysts interact with existing industrial infrastructure. Compatibility with standard reactor designs, resistance to common contaminants, and adaptability to variable feedstock quality all influence implementation success. Additionally, catalyst regeneration protocols must be established to extend operational lifetimes and improve economic feasibility.
Energy efficiency at industrial scale differs substantially from laboratory demonstrations. While laboratory studies often focus on catalytic activity and selectivity, industrial implementation must optimize for minimal energy consumption across the entire process. Heat recovery systems, pressure management, and process intensification techniques become essential components of successful scale-up strategies.
Environmental and safety considerations gain prominence at industrial scale. Catalyst leaching, potential byproduct formation, and worker exposure risks must be thoroughly evaluated and mitigated. Regulatory compliance pathways should be identified early in development to prevent costly redesigns during later implementation stages.
Economic viability ultimately determines industrial adoption. Comprehensive techno-economic analysis must evaluate capital expenditure requirements, operational costs, catalyst lifetime, and performance degradation rates. Sensitivity analysis regarding raw material price fluctuations, energy costs, and potential carbon pricing mechanisms provides valuable insights for implementation planning.
Collaborative development between academic researchers, catalyst manufacturers, and end-users accelerates successful industrial implementation. Establishing feedback loops where industrial challenges inform fundamental research directions helps bridge the laboratory-to-industry gap and ensures optimization efforts address practical implementation barriers rather than solely academic metrics.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!