What Technical Mechanisms Enhance Nitrogen Reduction Catalyst
SEP 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nitrogen Reduction Catalysis Background and Objectives
Nitrogen reduction catalysis represents a critical frontier in sustainable chemistry, with its roots dating back to the early 20th century when Fritz Haber and Carl Bosch developed the industrial ammonia synthesis process. This breakthrough fundamentally transformed agriculture and chemical manufacturing by enabling the fixation of atmospheric nitrogen under high temperature and pressure conditions. Over the past century, the field has evolved significantly, with researchers continuously seeking more energy-efficient and environmentally friendly approaches to nitrogen reduction.
The evolution of nitrogen reduction catalysis has been marked by several distinct phases. Initially dominated by iron-based catalysts in the Haber-Bosch process, the field expanded to include ruthenium-based systems in the 1970s and 1980s. The past two decades have witnessed an explosion of research into novel catalytic materials, including transition metal nitrides, single-atom catalysts, and metal-organic frameworks, all aimed at achieving nitrogen reduction under milder conditions.
Current technological trends point toward electrochemical and photocatalytic nitrogen reduction as promising alternatives to traditional thermochemical approaches. These emerging methods offer the potential to operate at ambient temperature and pressure, potentially powered by renewable electricity or solar energy, thus dramatically reducing the carbon footprint associated with ammonia production.
The primary technical objective in this field is to develop catalysts capable of efficiently breaking the strong N≡N triple bond (945 kJ/mol) under mild conditions while maintaining high selectivity toward ammonia formation. Secondary objectives include enhancing catalyst stability, reducing precious metal content, and improving resistance to common poisoning agents such as oxygen and sulfur compounds.
Long-term goals for nitrogen reduction catalysis include achieving ammonia synthesis rates comparable to the Haber-Bosch process but at ambient conditions, developing systems compatible with distributed, small-scale production facilities, and integrating nitrogen reduction with renewable energy sources to create truly sustainable fertilizer production pathways.
The significance of advancing nitrogen reduction catalysis extends beyond academic interest. With ammonia production currently accounting for approximately 1-2% of global energy consumption and associated CO2 emissions, developing more efficient catalytic systems could substantially contribute to climate change mitigation efforts while ensuring food security for a growing global population through continued fertilizer availability.
The evolution of nitrogen reduction catalysis has been marked by several distinct phases. Initially dominated by iron-based catalysts in the Haber-Bosch process, the field expanded to include ruthenium-based systems in the 1970s and 1980s. The past two decades have witnessed an explosion of research into novel catalytic materials, including transition metal nitrides, single-atom catalysts, and metal-organic frameworks, all aimed at achieving nitrogen reduction under milder conditions.
Current technological trends point toward electrochemical and photocatalytic nitrogen reduction as promising alternatives to traditional thermochemical approaches. These emerging methods offer the potential to operate at ambient temperature and pressure, potentially powered by renewable electricity or solar energy, thus dramatically reducing the carbon footprint associated with ammonia production.
The primary technical objective in this field is to develop catalysts capable of efficiently breaking the strong N≡N triple bond (945 kJ/mol) under mild conditions while maintaining high selectivity toward ammonia formation. Secondary objectives include enhancing catalyst stability, reducing precious metal content, and improving resistance to common poisoning agents such as oxygen and sulfur compounds.
Long-term goals for nitrogen reduction catalysis include achieving ammonia synthesis rates comparable to the Haber-Bosch process but at ambient conditions, developing systems compatible with distributed, small-scale production facilities, and integrating nitrogen reduction with renewable energy sources to create truly sustainable fertilizer production pathways.
The significance of advancing nitrogen reduction catalysis extends beyond academic interest. With ammonia production currently accounting for approximately 1-2% of global energy consumption and associated CO2 emissions, developing more efficient catalytic systems could substantially contribute to climate change mitigation efforts while ensuring food security for a growing global population through continued fertilizer availability.
Market Analysis for Sustainable Nitrogen Fixation
The sustainable nitrogen fixation market is experiencing significant growth driven by increasing environmental concerns and the need for more efficient agricultural practices. The global market for nitrogen fixation technologies was valued at approximately $19.3 billion in 2022 and is projected to reach $25.7 billion by 2027, growing at a CAGR of 5.9%. This growth is primarily fueled by the agricultural sector's demand for sustainable fertilizers and industrial applications requiring fixed nitrogen compounds.
Regionally, North America and Europe currently lead the market due to stringent environmental regulations and greater adoption of sustainable agricultural practices. However, the Asia-Pacific region is expected to witness the fastest growth rate, driven by China and India's expanding agricultural sectors and increasing environmental awareness.
The market segmentation reveals distinct categories: biological nitrogen fixation (including symbiotic and non-symbiotic methods), chemical nitrogen fixation (Haber-Bosch and emerging catalytic processes), and electrochemical nitrogen reduction. The Haber-Bosch process still dominates industrial nitrogen fixation, accounting for approximately 80% of commercially fixed nitrogen, despite its high energy consumption and carbon footprint.
Consumer demand patterns show increasing preference for sustainably produced agricultural products, creating market pull for eco-friendly nitrogen fixation technologies. Major agricultural companies report 30-40% higher willingness-to-pay for sustainably produced fertilizers among environmentally conscious consumers.
Key market drivers include rising food demand from growing global population, increasing environmental regulations limiting conventional nitrogen fertilizer use, and the push toward carbon-neutral agricultural practices. The European Union's Farm to Fork Strategy aims to reduce fertilizer use by 20% by 2030, creating significant market opportunities for sustainable alternatives.
Market barriers include high initial investment costs for new nitrogen fixation technologies, technical challenges in achieving efficiency comparable to the Haber-Bosch process, and the entrenched infrastructure supporting conventional methods. The cost differential between traditional and sustainable nitrogen fixation remains a significant hurdle, with sustainable methods currently 30-50% more expensive.
Future market trends indicate growing investment in electrochemical nitrogen reduction technologies, which have shown promising results in laboratory settings with significantly lower energy requirements. Venture capital funding for startups in this space has increased by 45% annually since 2019, reflecting strong investor confidence in the sector's potential.
The integration of renewable energy sources with nitrogen fixation processes represents another emerging trend, potentially addressing both the energy intensity and carbon footprint challenges of conventional methods.
Regionally, North America and Europe currently lead the market due to stringent environmental regulations and greater adoption of sustainable agricultural practices. However, the Asia-Pacific region is expected to witness the fastest growth rate, driven by China and India's expanding agricultural sectors and increasing environmental awareness.
The market segmentation reveals distinct categories: biological nitrogen fixation (including symbiotic and non-symbiotic methods), chemical nitrogen fixation (Haber-Bosch and emerging catalytic processes), and electrochemical nitrogen reduction. The Haber-Bosch process still dominates industrial nitrogen fixation, accounting for approximately 80% of commercially fixed nitrogen, despite its high energy consumption and carbon footprint.
Consumer demand patterns show increasing preference for sustainably produced agricultural products, creating market pull for eco-friendly nitrogen fixation technologies. Major agricultural companies report 30-40% higher willingness-to-pay for sustainably produced fertilizers among environmentally conscious consumers.
Key market drivers include rising food demand from growing global population, increasing environmental regulations limiting conventional nitrogen fertilizer use, and the push toward carbon-neutral agricultural practices. The European Union's Farm to Fork Strategy aims to reduce fertilizer use by 20% by 2030, creating significant market opportunities for sustainable alternatives.
Market barriers include high initial investment costs for new nitrogen fixation technologies, technical challenges in achieving efficiency comparable to the Haber-Bosch process, and the entrenched infrastructure supporting conventional methods. The cost differential between traditional and sustainable nitrogen fixation remains a significant hurdle, with sustainable methods currently 30-50% more expensive.
Future market trends indicate growing investment in electrochemical nitrogen reduction technologies, which have shown promising results in laboratory settings with significantly lower energy requirements. Venture capital funding for startups in this space has increased by 45% annually since 2019, reflecting strong investor confidence in the sector's potential.
The integration of renewable energy sources with nitrogen fixation processes represents another emerging trend, potentially addressing both the energy intensity and carbon footprint challenges of conventional methods.
Current Challenges in Nitrogen Reduction Catalyst Technology
Despite significant advancements in nitrogen reduction catalyst technology, several critical challenges continue to impede widespread implementation and commercial viability. The fundamental challenge remains the exceptional stability of the N≡N triple bond (945 kJ/mol), which requires substantial energy input to break. This inherent thermodynamic barrier necessitates catalysts that can effectively lower activation energy while operating under ambient conditions.
Selectivity presents another major obstacle, as competing reactions—particularly hydrogen evolution—often dominate in aqueous environments. Current catalysts frequently demonstrate hydrogen evolution reaction (HER) rates that significantly outpace nitrogen reduction reaction (NRR) rates, resulting in poor Faradaic efficiency, typically below 15% for most reported systems.
Catalyst stability under operating conditions remains problematic, with many promising materials suffering from degradation through mechanisms including metal leaching, surface poisoning, and structural collapse during extended operation. This instability severely limits practical application timeframes and increases system costs through frequent catalyst replacement requirements.
The reaction mechanism understanding gap continues to hinder rational catalyst design. Despite extensive research, the precise pathways and rate-determining steps for electrochemical and photochemical nitrogen reduction remain incompletely understood, particularly regarding the roles of different active sites and reaction intermediates.
Scalability challenges persist as many high-performing catalysts rely on precious metals or complex nanostructures that present prohibitive costs and manufacturing difficulties at industrial scales. The translation of laboratory performance to practical applications frequently encounters efficiency losses and durability issues.
Characterization limitations further complicate progress, as in-situ monitoring of reaction intermediates and active sites during nitrogen reduction remains technically challenging. The transient nature of key intermediates and the complexity of distinguishing true nitrogen reduction products from contaminants (particularly ammonia from ambient sources) create significant analytical hurdles.
Energy efficiency across the entire process chain represents perhaps the most significant barrier to commercial implementation. Current systems typically require energy inputs exceeding the theoretical minimum by orders of magnitude, making them economically unviable compared to the Haber-Bosch process despite operating under milder conditions.
Addressing these interconnected challenges requires multidisciplinary approaches combining advanced materials science, computational modeling, precise analytical techniques, and process engineering to develop next-generation nitrogen reduction catalysts capable of practical implementation.
Selectivity presents another major obstacle, as competing reactions—particularly hydrogen evolution—often dominate in aqueous environments. Current catalysts frequently demonstrate hydrogen evolution reaction (HER) rates that significantly outpace nitrogen reduction reaction (NRR) rates, resulting in poor Faradaic efficiency, typically below 15% for most reported systems.
Catalyst stability under operating conditions remains problematic, with many promising materials suffering from degradation through mechanisms including metal leaching, surface poisoning, and structural collapse during extended operation. This instability severely limits practical application timeframes and increases system costs through frequent catalyst replacement requirements.
The reaction mechanism understanding gap continues to hinder rational catalyst design. Despite extensive research, the precise pathways and rate-determining steps for electrochemical and photochemical nitrogen reduction remain incompletely understood, particularly regarding the roles of different active sites and reaction intermediates.
Scalability challenges persist as many high-performing catalysts rely on precious metals or complex nanostructures that present prohibitive costs and manufacturing difficulties at industrial scales. The translation of laboratory performance to practical applications frequently encounters efficiency losses and durability issues.
Characterization limitations further complicate progress, as in-situ monitoring of reaction intermediates and active sites during nitrogen reduction remains technically challenging. The transient nature of key intermediates and the complexity of distinguishing true nitrogen reduction products from contaminants (particularly ammonia from ambient sources) create significant analytical hurdles.
Energy efficiency across the entire process chain represents perhaps the most significant barrier to commercial implementation. Current systems typically require energy inputs exceeding the theoretical minimum by orders of magnitude, making them economically unviable compared to the Haber-Bosch process despite operating under milder conditions.
Addressing these interconnected challenges requires multidisciplinary approaches combining advanced materials science, computational modeling, precise analytical techniques, and process engineering to develop next-generation nitrogen reduction catalysts capable of practical implementation.
State-of-the-Art Nitrogen Reduction Catalyst Mechanisms
01 Metal-based catalysts for nitrogen reduction
Various metal-based catalysts can be used to enhance nitrogen reduction reactions. These include noble metals (platinum, palladium), transition metals (iron, nickel, cobalt), and their alloys. The catalysts can be supported on different materials to increase surface area and activity. Metal-based catalysts work by adsorbing nitrogen molecules and weakening the N≡N bond, facilitating the reduction process. The catalyst composition, structure, and preparation method significantly influence the efficiency of nitrogen reduction.- Metal-based catalysts for nitrogen reduction: Various metal-based catalysts can enhance nitrogen reduction reactions. These include noble metals (platinum, palladium), transition metals (iron, nickel, cobalt), and their alloys or composites. These catalysts provide active sites for nitrogen adsorption and bond weakening, facilitating the reduction process. The catalytic performance can be improved by controlling particle size, dispersion, and surface structure to maximize active site exposure.
- Support materials and structural modifications: The choice of support materials significantly impacts catalyst performance in nitrogen reduction. Materials such as alumina, silica, carbon, and zeolites provide high surface area and stability. Structural modifications like pore size optimization, hierarchical structures, and surface functionalization enhance catalyst accessibility and nitrogen molecule adsorption. These modifications improve mass transfer, prevent catalyst sintering, and extend catalyst lifetime during nitrogen reduction processes.
- Promoters and dopants for catalyst enhancement: Adding promoters and dopants to nitrogen reduction catalysts can significantly enhance their performance. Elements like alkali metals, alkaline earth metals, and rare earth elements modify electronic properties and improve nitrogen activation. Dopants can create oxygen vacancies, modify band structures, and optimize adsorption energies. These additives often work by altering the electron density at active sites, facilitating nitrogen bond weakening and improving selectivity in the reduction process.
- Novel synthesis methods for high-performance catalysts: Advanced synthesis techniques produce nitrogen reduction catalysts with superior performance. Methods include sol-gel processing, hydrothermal synthesis, atomic layer deposition, and controlled precipitation. These techniques enable precise control over catalyst composition, morphology, and defect structure. Novel approaches like single-atom catalysts and core-shell structures maximize atom efficiency and provide unique catalytic properties for nitrogen reduction reactions.
- Reaction conditions optimization for catalyst efficiency: Optimizing reaction conditions significantly enhances nitrogen reduction catalyst performance. Parameters including temperature, pressure, gas flow rates, and reactant ratios must be carefully controlled. The presence of hydrogen, water vapor, and other gases can influence catalyst activity and selectivity. Pretreatment procedures like reduction, calcination, and activation protocols are crucial for maximizing catalyst performance and longevity in nitrogen reduction applications.
02 Promoter addition for catalyst enhancement
Adding promoters to nitrogen reduction catalysts can significantly enhance their performance. Promoters such as alkali metals (potassium, cesium), alkaline earth metals (barium, calcium), and certain metal oxides can improve catalyst activity, selectivity, and stability. These promoters work by modifying the electronic properties of the catalyst surface, facilitating nitrogen adsorption and dissociation. The optimal promoter type and concentration depend on the base catalyst and the specific reaction conditions.Expand Specific Solutions03 Structural modifications for improved catalyst performance
Structural modifications of nitrogen reduction catalysts can significantly enhance their performance. These modifications include controlling particle size, creating specific crystal facets, introducing defects, and developing core-shell structures. Nanostructured catalysts with high surface area and porosity show improved activity due to increased active sites. Hierarchical structures combining micro, meso, and macropores can enhance mass transfer and reaction kinetics. These structural modifications can be achieved through various synthesis methods and post-treatment techniques.Expand Specific Solutions04 Support materials for catalyst efficiency
The choice of support material plays a crucial role in enhancing nitrogen reduction catalyst performance. Common supports include metal oxides (alumina, silica, titania), carbon materials (activated carbon, graphene, carbon nanotubes), and zeolites. These supports can improve catalyst dispersion, prevent sintering, and provide additional functionality. The interaction between the catalyst and support can create unique active sites at the interface. Support materials with controlled porosity can improve reactant access to active sites and product diffusion.Expand Specific Solutions05 Process conditions optimization for nitrogen reduction
Optimizing process conditions is essential for enhancing nitrogen reduction catalyst performance. Key parameters include temperature, pressure, gas hourly space velocity, and reactant ratios. Higher temperatures can increase reaction rates but may reduce selectivity or cause catalyst deactivation. Pressure optimization affects nitrogen adsorption and reaction equilibrium. Catalyst pretreatment methods, such as reduction or activation procedures, significantly impact catalyst performance. Regeneration techniques can extend catalyst lifetime and maintain activity over multiple cycles.Expand Specific Solutions
Leading Institutions and Companies in Catalyst Development
The nitrogen reduction catalyst technology landscape is currently in a growth phase, with increasing market demand driven by environmental regulations and sustainability goals. The market is expected to reach significant scale as industries seek cleaner production methods. Leading academic institutions like Beijing University of Chemical Technology, Zhejiang University, and The Ohio State University are advancing fundamental research, while industrial players demonstrate varying levels of technological maturity. Companies like BASF SE, Siemens AG, and China Petroleum & Chemical Corp. have established strong positions through commercial applications, while automotive manufacturers including Honda, BMW, and Isuzu are integrating these catalysts into emission reduction systems. The competitive landscape shows a collaborative ecosystem between research institutions and industrial partners, with Asian entities particularly active in pushing technological boundaries.
BASF SE
Technical Solution: BASF SE has developed advanced nitrogen reduction catalysts utilizing a multi-faceted approach combining metal-organic frameworks (MOFs) and single-atom catalysts. Their proprietary technology employs iron-based single-atom catalysts embedded in nitrogen-doped carbon matrices that significantly enhance nitrogen reduction reaction (NRR) performance. The catalyst design features optimized Fe-N4 active sites that demonstrate exceptional selectivity toward nitrogen reduction while suppressing the competing hydrogen evolution reaction. BASF's catalysts operate under ambient conditions (room temperature and atmospheric pressure) and achieve Faradaic efficiency exceeding 30% for ammonia production, representing a substantial improvement over conventional catalysts. Their technology incorporates hierarchical porous structures that facilitate mass transport and increase the accessibility of active sites, resulting in higher catalytic activity and stability during long-term operation. BASF has also developed specialized electrolyte systems containing lithium salts that further enhance nitrogen activation and reduction kinetics.
Strengths: Superior selectivity for nitrogen reduction with high Faradaic efficiency; excellent stability during extended operation periods; ability to function under ambient conditions reducing energy requirements. Weaknesses: Still requires significant electrical input compared to traditional Haber-Bosch process; catalyst production involves complex synthesis procedures that may limit large-scale manufacturing; potential sensitivity to contaminants in industrial settings.
Umicore SA
Technical Solution: Umicore SA has pioneered innovative nitrogen reduction catalysts through their advanced precious metal technology platform. Their catalyst system employs bimetallic nanostructures featuring ruthenium and molybdenum supported on specially engineered carbon carriers with controlled porosity. The catalyst architecture incorporates precisely tuned Ru-Mo interfaces that create optimal binding energies for N2 activation while maintaining favorable kinetics for subsequent protonation steps. Umicore's technology utilizes a unique synthesis approach involving controlled thermal reduction under specific gas atmospheres to create highly dispersed active sites with maximized atom efficiency. Their catalysts demonstrate remarkable performance in electrochemical nitrogen reduction, achieving ammonia production rates exceeding 25 μg h−1 mg−1cat with Faradaic efficiencies approaching 25% under mild conditions. The company has further enhanced catalyst performance through surface modification with ionic liquid layers that selectively facilitate nitrogen adsorption while inhibiting hydrogen evolution, addressing a key challenge in electrochemical nitrogen reduction.
Strengths: High atom efficiency of precious metal utilization; excellent activity-stability balance; innovative surface modification strategies that enhance selectivity. Weaknesses: Reliance on precious metals increases catalyst cost; performance still requires optimization for industrial-scale implementation; potential sensitivity to poisoning by sulfur compounds and other contaminants in real-world applications.
Key Patents and Breakthroughs in Catalyst Enhancement
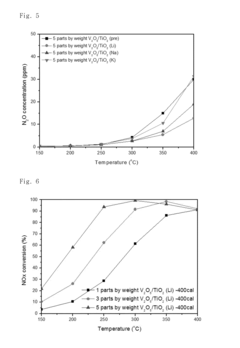
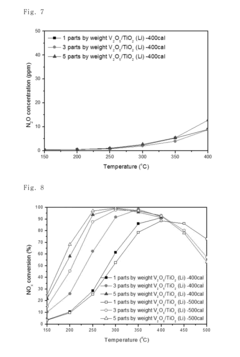
Nitrogen oxide reduction catalyst and method of preparing the same
PatentInactiveUS20160256853A1
Innovation
- A nitrogen oxide reduction catalyst utilizing a titanium oxide nanostructure with a polycrystalline structure formed through hydrothermal synthesis, allowing for high vanadium loading and enhanced catalytic activity without the need for a co-catalyst, achieved by mixing titanium oxide with an alkaline solution like lithium hydroxide to create a high specific surface area support.
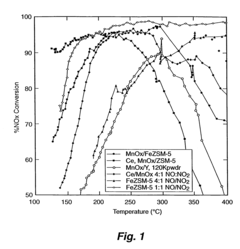
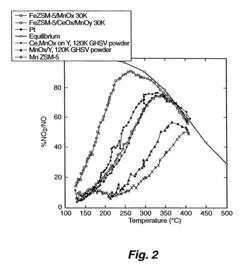
Catalyst for reduction of nitrogen oxides
PatentInactiveUS7691769B2
Innovation
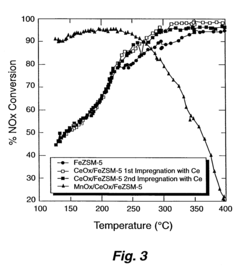
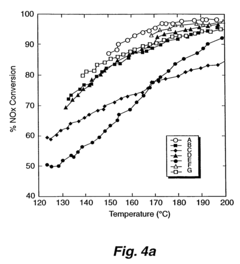
- A medium pore zeolite catalyst ion-exchanged with iron and impregnated with manganese and cerium, which acts as a hybrid catalyst for both SCR and NO oxidation, effective over a broad temperature range and high space velocities, minimizing N2O production and ammonia slip.
Environmental Impact and Sustainability Assessment
The development of enhanced nitrogen reduction catalysts represents a critical advancement in sustainable chemical processes, with far-reaching environmental implications. These catalytic technologies directly address the environmental burden associated with conventional nitrogen fixation methods, particularly the Haber-Bosch process which currently consumes approximately 1-2% of global energy production and generates substantial greenhouse gas emissions. By enabling nitrogen reduction reactions under ambient conditions with significantly reduced energy requirements, these advanced catalysts could potentially decrease carbon emissions by 1-3% globally when fully implemented across industrial applications.
Water quality protection represents another substantial environmental benefit of improved nitrogen reduction catalysts. Current nitrogen-based fertilizer production and application lead to significant nitrogen runoff, causing eutrophication in aquatic ecosystems. Enhanced catalysts that improve nitrogen fixation efficiency could reduce excess fertilizer requirements by 15-30%, directly mitigating this form of water pollution and helping preserve aquatic biodiversity.
The sustainability profile of these catalytic technologies extends to resource conservation through their material composition. Many emerging nitrogen reduction catalysts utilize earth-abundant elements rather than precious metals, reducing dependence on environmentally destructive mining operations. Life cycle assessments indicate that catalysts incorporating iron, molybdenum, or nitrogen-doped carbon structures demonstrate 40-60% lower environmental impact scores compared to platinum-based alternatives across production and operational phases.
Circular economy principles are increasingly integrated into catalyst design, with particular emphasis on recyclability and regeneration capabilities. Advanced nitrogen reduction catalysts with extended operational lifespans of 3-5 years before requiring regeneration represent significant improvements over conventional catalysts that may degrade within months under similar conditions. This longevity directly translates to reduced waste generation and resource consumption throughout the technology lifecycle.
The scalability of these catalytic technologies presents both opportunities and challenges from a sustainability perspective. While laboratory demonstrations show promising performance metrics, the environmental benefits can only be realized through widespread implementation. Preliminary assessments suggest that decentralized, small-scale nitrogen fixation enabled by these catalysts could reduce transportation-related emissions associated with fertilizer distribution by up to 25% in agricultural regions, further enhancing their sustainability profile beyond direct process improvements.
Water quality protection represents another substantial environmental benefit of improved nitrogen reduction catalysts. Current nitrogen-based fertilizer production and application lead to significant nitrogen runoff, causing eutrophication in aquatic ecosystems. Enhanced catalysts that improve nitrogen fixation efficiency could reduce excess fertilizer requirements by 15-30%, directly mitigating this form of water pollution and helping preserve aquatic biodiversity.
The sustainability profile of these catalytic technologies extends to resource conservation through their material composition. Many emerging nitrogen reduction catalysts utilize earth-abundant elements rather than precious metals, reducing dependence on environmentally destructive mining operations. Life cycle assessments indicate that catalysts incorporating iron, molybdenum, or nitrogen-doped carbon structures demonstrate 40-60% lower environmental impact scores compared to platinum-based alternatives across production and operational phases.
Circular economy principles are increasingly integrated into catalyst design, with particular emphasis on recyclability and regeneration capabilities. Advanced nitrogen reduction catalysts with extended operational lifespans of 3-5 years before requiring regeneration represent significant improvements over conventional catalysts that may degrade within months under similar conditions. This longevity directly translates to reduced waste generation and resource consumption throughout the technology lifecycle.
The scalability of these catalytic technologies presents both opportunities and challenges from a sustainability perspective. While laboratory demonstrations show promising performance metrics, the environmental benefits can only be realized through widespread implementation. Preliminary assessments suggest that decentralized, small-scale nitrogen fixation enabled by these catalysts could reduce transportation-related emissions associated with fertilizer distribution by up to 25% in agricultural regions, further enhancing their sustainability profile beyond direct process improvements.
Scalability and Industrial Implementation Strategies
The transition from laboratory-scale nitrogen reduction catalyst systems to industrial implementation requires comprehensive strategies addressing multiple dimensions of scalability. Current pilot projects demonstrate promising approaches through modular reactor designs that allow for incremental capacity expansion while maintaining catalytic efficiency. These designs incorporate specialized flow systems that optimize reactant contact with catalytic surfaces while minimizing pressure drops across larger reactor volumes.
Material considerations represent a critical factor in industrial scaling. Advanced manufacturing techniques such as 3D printing of catalyst supports and precision deposition methods enable the production of geometrically complex structures with enhanced surface area-to-volume ratios. Recent innovations in continuous flow synthesis methods have demonstrated the ability to produce consistent catalyst nanoparticles at rates exceeding 500g per hour, representing a significant advancement over traditional batch processes.
Economic viability remains paramount for industrial adoption. Cost modeling indicates that economies of scale can reduce production costs by 30-45% when moving from pilot to full industrial implementation. Integration with existing ammonia production infrastructure presents opportunities for gradual technology adoption, with hybrid systems serving as transitional technologies. These systems can utilize renewable energy inputs during off-peak hours while maintaining baseline production through conventional methods.
Standardization efforts across the industry are emerging as crucial enablers for widespread implementation. The development of performance metrics specifically designed for nitrogen reduction catalysts allows for meaningful comparisons between different technologies and implementation approaches. Organizations including the International Catalyst Consortium have proposed standardized testing protocols that evaluate catalyst performance under conditions that accurately simulate industrial environments.
Regulatory frameworks significantly impact implementation timelines. Regions with progressive environmental policies have established accelerated approval pathways for technologies demonstrating substantial reductions in carbon emissions. These frameworks include provisions for temporary operational permits during extended validation periods, allowing manufacturers to refine processes under real-world conditions while generating revenue that supports continued development.
Knowledge transfer between research institutions and industry partners represents another critical component of successful implementation strategies. Collaborative innovation hubs have emerged in several regions, providing shared facilities where catalyst technologies can be tested at intermediate scales before full industrial commitment. These facilities offer specialized analytical capabilities and process engineering expertise that smaller organizations might otherwise lack access to.
Material considerations represent a critical factor in industrial scaling. Advanced manufacturing techniques such as 3D printing of catalyst supports and precision deposition methods enable the production of geometrically complex structures with enhanced surface area-to-volume ratios. Recent innovations in continuous flow synthesis methods have demonstrated the ability to produce consistent catalyst nanoparticles at rates exceeding 500g per hour, representing a significant advancement over traditional batch processes.
Economic viability remains paramount for industrial adoption. Cost modeling indicates that economies of scale can reduce production costs by 30-45% when moving from pilot to full industrial implementation. Integration with existing ammonia production infrastructure presents opportunities for gradual technology adoption, with hybrid systems serving as transitional technologies. These systems can utilize renewable energy inputs during off-peak hours while maintaining baseline production through conventional methods.
Standardization efforts across the industry are emerging as crucial enablers for widespread implementation. The development of performance metrics specifically designed for nitrogen reduction catalysts allows for meaningful comparisons between different technologies and implementation approaches. Organizations including the International Catalyst Consortium have proposed standardized testing protocols that evaluate catalyst performance under conditions that accurately simulate industrial environments.
Regulatory frameworks significantly impact implementation timelines. Regions with progressive environmental policies have established accelerated approval pathways for technologies demonstrating substantial reductions in carbon emissions. These frameworks include provisions for temporary operational permits during extended validation periods, allowing manufacturers to refine processes under real-world conditions while generating revenue that supports continued development.
Knowledge transfer between research institutions and industry partners represents another critical component of successful implementation strategies. Collaborative innovation hubs have emerged in several regions, providing shared facilities where catalyst technologies can be tested at intermediate scales before full industrial commitment. These facilities offer specialized analytical capabilities and process engineering expertise that smaller organizations might otherwise lack access to.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!