Artifacts And How To Avoid Them In Cryo-TEM Experiments
AUG 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cryo-TEM Artifacts Background and Research Objectives
Cryo-Transmission Electron Microscopy (Cryo-TEM) has revolutionized structural biology by enabling the visualization of biological specimens in their native state at near-atomic resolution. Since its development in the 1980s by Jacques Dubochet and colleagues, this technique has evolved from a specialized method to an essential tool in modern biological research. The fundamental principle involves rapid freezing of samples in vitreous ice, preserving their structure without the need for chemical fixatives or stains that might introduce artifacts.
The evolution of Cryo-TEM has been marked by significant technological advancements, including the development of direct electron detectors, automated data collection systems, and sophisticated image processing algorithms. These innovations have collectively pushed resolution boundaries from nanometer to near-atomic levels, enabling researchers to visualize molecular structures with unprecedented clarity.
Despite these advances, artifacts remain a persistent challenge in Cryo-TEM experiments. These unwanted features can arise at various stages of the workflow, from sample preparation to image acquisition and processing. Common artifacts include ice contamination, beam-induced damage, preferential orientation of particles, and charging effects. These artifacts not only compromise image quality but can also lead to misinterpretation of biological structures and functions.
The economic and scientific impact of addressing these artifacts is substantial. High-resolution structural information obtained through Cryo-TEM drives drug discovery, vaccine development, and fundamental understanding of cellular processes. However, artifacts can significantly delay research progress and increase costs through wasted resources and repeated experiments.
Recent technological trends in the field focus on automated artifact detection using machine learning algorithms, development of more stable sample supports, and improved vitrification techniques. These innovations aim to minimize human intervention and increase reproducibility in Cryo-TEM experiments.
The primary objective of this research is to comprehensively catalog the various artifacts encountered in Cryo-TEM experiments, understand their underlying physical and chemical causes, and develop systematic approaches to mitigate or eliminate them. By addressing these challenges, we aim to enhance the reliability and efficiency of Cryo-TEM as a research tool.
Secondary objectives include developing standardized protocols for artifact identification, creating educational resources for training new microscopists, and establishing quantitative metrics for assessing image quality. These efforts will contribute to the broader goal of making high-quality structural information more accessible to the scientific community and accelerating discoveries in structural biology and related fields.
The evolution of Cryo-TEM has been marked by significant technological advancements, including the development of direct electron detectors, automated data collection systems, and sophisticated image processing algorithms. These innovations have collectively pushed resolution boundaries from nanometer to near-atomic levels, enabling researchers to visualize molecular structures with unprecedented clarity.
Despite these advances, artifacts remain a persistent challenge in Cryo-TEM experiments. These unwanted features can arise at various stages of the workflow, from sample preparation to image acquisition and processing. Common artifacts include ice contamination, beam-induced damage, preferential orientation of particles, and charging effects. These artifacts not only compromise image quality but can also lead to misinterpretation of biological structures and functions.
The economic and scientific impact of addressing these artifacts is substantial. High-resolution structural information obtained through Cryo-TEM drives drug discovery, vaccine development, and fundamental understanding of cellular processes. However, artifacts can significantly delay research progress and increase costs through wasted resources and repeated experiments.
Recent technological trends in the field focus on automated artifact detection using machine learning algorithms, development of more stable sample supports, and improved vitrification techniques. These innovations aim to minimize human intervention and increase reproducibility in Cryo-TEM experiments.
The primary objective of this research is to comprehensively catalog the various artifacts encountered in Cryo-TEM experiments, understand their underlying physical and chemical causes, and develop systematic approaches to mitigate or eliminate them. By addressing these challenges, we aim to enhance the reliability and efficiency of Cryo-TEM as a research tool.
Secondary objectives include developing standardized protocols for artifact identification, creating educational resources for training new microscopists, and establishing quantitative metrics for assessing image quality. These efforts will contribute to the broader goal of making high-quality structural information more accessible to the scientific community and accelerating discoveries in structural biology and related fields.
Market Demand for High-Quality Cryo-TEM Imaging
The global market for cryo-electron microscopy (cryo-EM) technologies has experienced substantial growth in recent years, with the demand for high-quality cryo-TEM imaging solutions increasing at a remarkable pace. This growth is primarily driven by expanding applications in structural biology, drug discovery, and materials science, where artifact-free imaging is crucial for accurate analysis and interpretation.
Research institutions and pharmaceutical companies are increasingly investing in cryo-TEM technologies to accelerate their R&D processes. The ability to visualize biological macromolecules at near-atomic resolution without artifacts has become a competitive advantage in drug development, reducing time-to-market and improving success rates in clinical trials.
Market analysis indicates that the global cryo-EM market was valued at approximately $650 million in 2021 and is projected to reach $1.4 billion by 2028, growing at a CAGR of 11.5%. The segment specifically focused on artifact reduction technologies and quality control solutions is expanding even faster, highlighting the critical importance of clean, interpretable data.
Academic research facilities represent the largest market segment, accounting for roughly 45% of the total market share. However, pharmaceutical and biotechnology companies are rapidly increasing their adoption rates, with an estimated annual growth of 15-18% in this sector alone.
Geographically, North America dominates the market with approximately 40% share, followed by Europe at 30% and Asia-Pacific at 25%. Emerging economies in the Asia-Pacific region, particularly China and India, are showing the fastest growth rates as they establish advanced research infrastructure.
The demand for automated artifact detection and correction software has emerged as a particularly strong market trend. End-users are willing to pay premium prices for integrated solutions that can identify and mitigate common artifacts in real-time during experiments, thereby saving valuable research time and expensive reagents.
Training and education services related to artifact-free cryo-TEM imaging techniques have also seen increased demand, with specialized workshops and certification programs experiencing 25-30% annual growth. This reflects the technical complexity of achieving artifact-free results and the high value placed on proper sample preparation and imaging techniques.
Industry surveys indicate that over 70% of cryo-TEM users cite artifact reduction as their primary concern when selecting equipment and protocols, emphasizing the market's sensitivity to image quality issues. This has prompted manufacturers to focus their R&D efforts on developing next-generation solutions specifically designed to address common artifacts such as ice contamination, beam-induced damage, and charging effects.
Research institutions and pharmaceutical companies are increasingly investing in cryo-TEM technologies to accelerate their R&D processes. The ability to visualize biological macromolecules at near-atomic resolution without artifacts has become a competitive advantage in drug development, reducing time-to-market and improving success rates in clinical trials.
Market analysis indicates that the global cryo-EM market was valued at approximately $650 million in 2021 and is projected to reach $1.4 billion by 2028, growing at a CAGR of 11.5%. The segment specifically focused on artifact reduction technologies and quality control solutions is expanding even faster, highlighting the critical importance of clean, interpretable data.
Academic research facilities represent the largest market segment, accounting for roughly 45% of the total market share. However, pharmaceutical and biotechnology companies are rapidly increasing their adoption rates, with an estimated annual growth of 15-18% in this sector alone.
Geographically, North America dominates the market with approximately 40% share, followed by Europe at 30% and Asia-Pacific at 25%. Emerging economies in the Asia-Pacific region, particularly China and India, are showing the fastest growth rates as they establish advanced research infrastructure.
The demand for automated artifact detection and correction software has emerged as a particularly strong market trend. End-users are willing to pay premium prices for integrated solutions that can identify and mitigate common artifacts in real-time during experiments, thereby saving valuable research time and expensive reagents.
Training and education services related to artifact-free cryo-TEM imaging techniques have also seen increased demand, with specialized workshops and certification programs experiencing 25-30% annual growth. This reflects the technical complexity of achieving artifact-free results and the high value placed on proper sample preparation and imaging techniques.
Industry surveys indicate that over 70% of cryo-TEM users cite artifact reduction as their primary concern when selecting equipment and protocols, emphasizing the market's sensitivity to image quality issues. This has prompted manufacturers to focus their R&D efforts on developing next-generation solutions specifically designed to address common artifacts such as ice contamination, beam-induced damage, and charging effects.
Current Challenges in Cryo-TEM Artifact Mitigation
Despite significant advancements in cryo-transmission electron microscopy (cryo-TEM), researchers continue to face numerous challenges in artifact mitigation. The vitrification process, while essential for preserving biological samples in their native state, introduces various artifacts that can compromise data interpretation. Ice contamination remains a persistent issue, with crystalline ice formation distorting the specimen structure and reducing image contrast. Even with modern vitrification techniques, achieving consistent ice thickness across samples proves difficult, leading to uneven imaging quality.
Beam-induced damage presents another significant challenge, as electron beam interaction with biological specimens causes radiation damage, resulting in structural alterations and molecular degradation. Current dose-limitation strategies often sacrifice image resolution and signal-to-noise ratio, creating a difficult trade-off between damage mitigation and data quality.
Sample preparation inconsistencies continue to plague cryo-TEM experiments. Variations in blotting conditions, grid types, and sample concentration can introduce artifacts that are difficult to distinguish from genuine structural features. The development of automated sample preparation systems has improved reproducibility but has not eliminated these issues entirely.
Charging effects represent another persistent challenge, particularly with non-conductive biological samples. The accumulation of electrical charge during imaging can cause specimen movement, contrast variations, and image distortions. While carbon films and conductive coatings help mitigate these effects, they often introduce additional background noise and potential artifacts.
Motion correction algorithms have significantly improved image quality, but specimen movement during data acquisition remains problematic. Beam-induced motion, mechanical instabilities, and thermal drift can blur images and reduce resolution. Current computational approaches can compensate for some movement but cannot fully eliminate all motion-related artifacts.
Contrast enhancement techniques like phase plates and energy filters improve visualization of biological structures but can introduce their own artifacts, including phase shifts, charging effects, and image distortions. Researchers must carefully optimize these tools to balance improved contrast against potential artifacts.
The interpretation of cryo-TEM data presents additional challenges, as distinguishing between genuine structural features and preparation artifacts requires significant expertise. Machine learning approaches show promise in automated artifact detection but remain limited by the availability of training data and the diversity of artifact types.
Cross-validation with complementary techniques such as X-ray crystallography, NMR spectroscopy, and computational modeling has become essential for confirming structural interpretations and identifying potential artifacts, though integrating data from multiple methodologies introduces its own complexities.
Beam-induced damage presents another significant challenge, as electron beam interaction with biological specimens causes radiation damage, resulting in structural alterations and molecular degradation. Current dose-limitation strategies often sacrifice image resolution and signal-to-noise ratio, creating a difficult trade-off between damage mitigation and data quality.
Sample preparation inconsistencies continue to plague cryo-TEM experiments. Variations in blotting conditions, grid types, and sample concentration can introduce artifacts that are difficult to distinguish from genuine structural features. The development of automated sample preparation systems has improved reproducibility but has not eliminated these issues entirely.
Charging effects represent another persistent challenge, particularly with non-conductive biological samples. The accumulation of electrical charge during imaging can cause specimen movement, contrast variations, and image distortions. While carbon films and conductive coatings help mitigate these effects, they often introduce additional background noise and potential artifacts.
Motion correction algorithms have significantly improved image quality, but specimen movement during data acquisition remains problematic. Beam-induced motion, mechanical instabilities, and thermal drift can blur images and reduce resolution. Current computational approaches can compensate for some movement but cannot fully eliminate all motion-related artifacts.
Contrast enhancement techniques like phase plates and energy filters improve visualization of biological structures but can introduce their own artifacts, including phase shifts, charging effects, and image distortions. Researchers must carefully optimize these tools to balance improved contrast against potential artifacts.
The interpretation of cryo-TEM data presents additional challenges, as distinguishing between genuine structural features and preparation artifacts requires significant expertise. Machine learning approaches show promise in automated artifact detection but remain limited by the availability of training data and the diversity of artifact types.
Cross-validation with complementary techniques such as X-ray crystallography, NMR spectroscopy, and computational modeling has become essential for confirming structural interpretations and identifying potential artifacts, though integrating data from multiple methodologies introduces its own complexities.
Established Protocols for Artifact Prevention
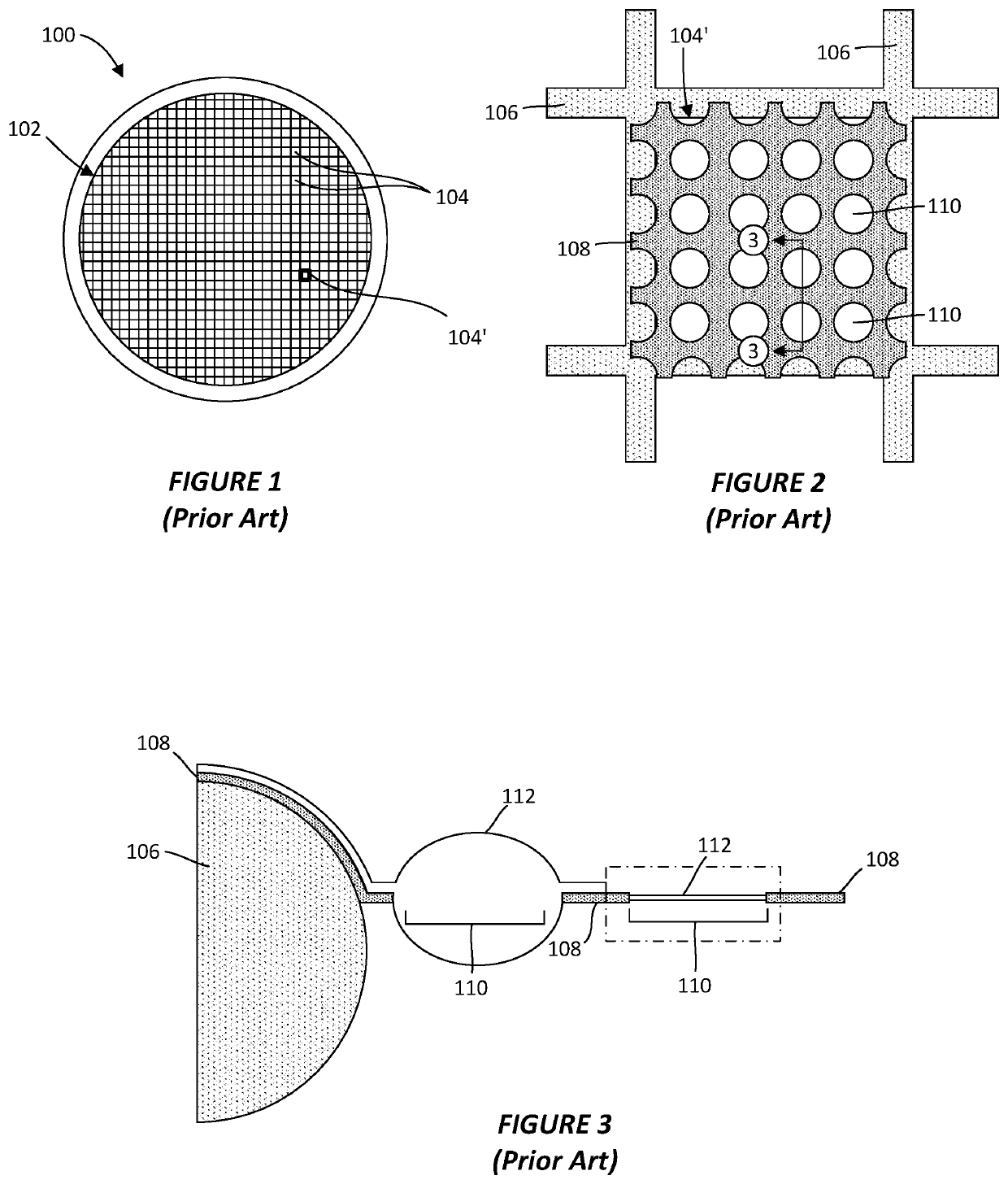
01 Ice crystal formation artifacts in cryo-TEM
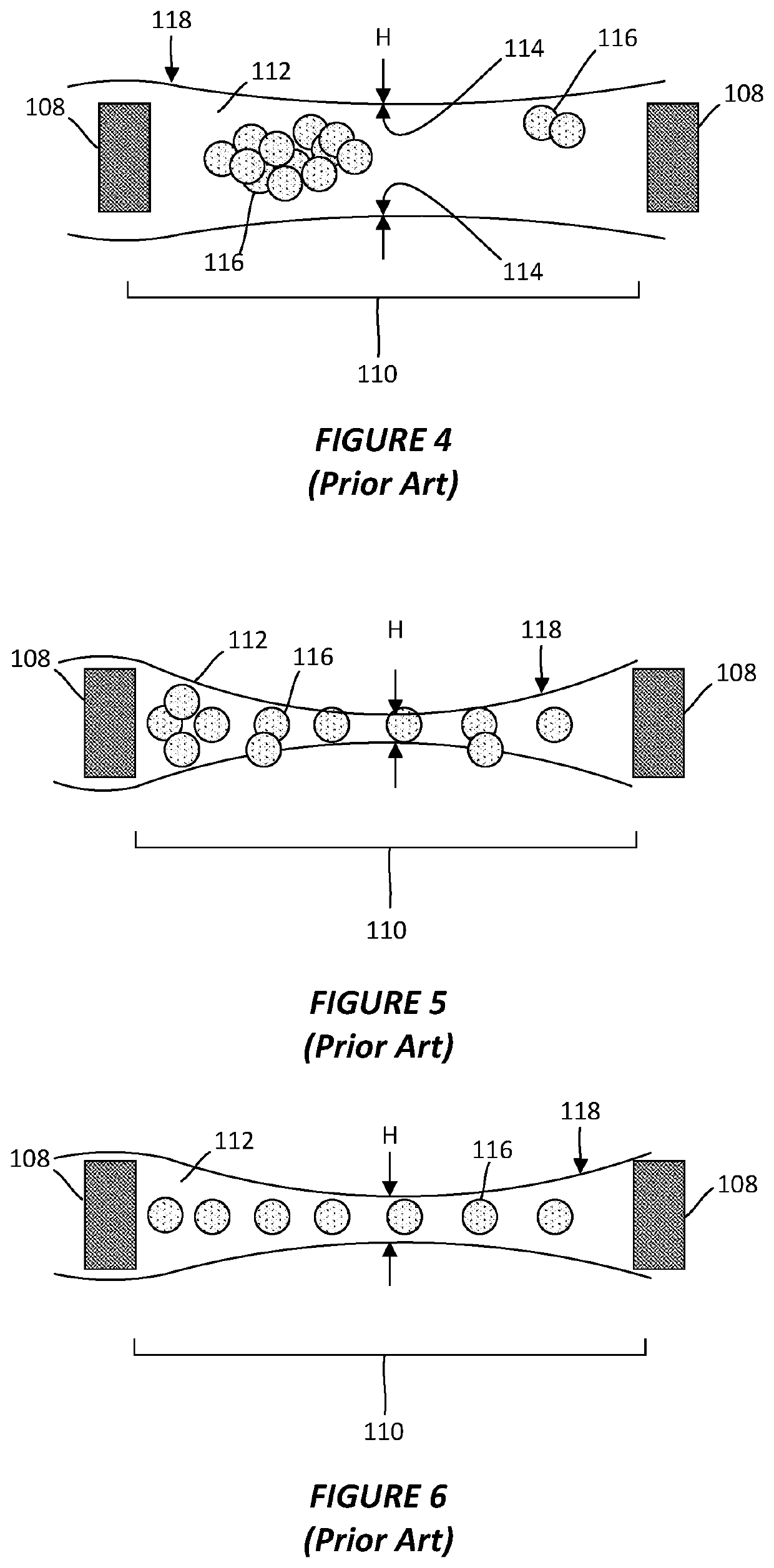
Ice crystal formation is a common artifact in cryo-TEM that occurs during sample preparation when the vitrification process is not rapid enough. These crystals can damage the specimen structure and create visual artifacts in the resulting images. Techniques to minimize ice crystal formation include optimized rapid freezing protocols, use of cryoprotectants, and controlled temperature environments during sample preparation and imaging.- Ice crystal formation artifacts in cryo-TEM: Ice crystal formation is a common artifact in cryo-TEM that occurs during sample preparation when the vitrification process is not rapid enough. These crystals can damage the specimen structure and create visual artifacts in the resulting images. Techniques to minimize ice crystal formation include optimized rapid freezing protocols, use of cryoprotectants, and controlled temperature environments during sample preparation and imaging.
- Beam damage artifacts in cryo-TEM imaging: Electron beam damage is a significant source of artifacts in cryo-TEM, where the high-energy electrons can cause structural changes in the vitrified sample. This includes radiolysis, bubble formation, and molecular degradation that can alter the specimen's native structure. Low-dose imaging techniques, beam-blanking strategies, and advanced detection systems are employed to minimize these artifacts while maintaining sufficient image quality for analysis.
- Sample preparation artifacts in cryo-TEM: Sample preparation for cryo-TEM can introduce various artifacts including concentration gradients, aggregation, and adsorption effects at air-water interfaces. These artifacts can misrepresent the native state of the specimen. Advanced preparation methods such as controlled blotting parameters, specialized grid treatments, and automated vitrification systems help reduce these artifacts and improve the reliability of structural observations.
- Image processing and reconstruction artifacts: Computational processing of cryo-TEM data can introduce artifacts during image alignment, averaging, and 3D reconstruction. These include missing wedge artifacts, alignment errors, and classification biases that can lead to misinterpretation of molecular structures. Advanced algorithms, validation protocols, and resolution estimation methods are developed to identify and mitigate these computational artifacts in the final reconstructions.
- Contrast and resolution artifacts in cryo-TEM: Contrast and resolution limitations in cryo-TEM can create artifacts that obscure structural details. These include phase contrast variations, defocus-related artifacts, and Thon rings that affect image interpretation. Technological advancements such as phase plates, direct electron detectors, and aberration correctors are implemented to enhance contrast while minimizing these artifacts, enabling more accurate visualization of biological and material specimens at near-atomic resolution.
02 Beam damage artifacts in cryo-TEM imaging
Electron beam damage is a significant challenge in cryo-TEM, where the high-energy electrons can cause structural changes in the frozen-hydrated specimens. This results in artifacts such as bubbling, specimen movement, and loss of high-resolution information. Methods to reduce beam damage include low-dose imaging techniques, beam-stable sample supports, and advanced image processing algorithms that can compensate for beam-induced specimen changes.Expand Specific Solutions03 Sample preparation artifacts in cryo-TEM
Sample preparation for cryo-TEM can introduce various artifacts including concentration gradients, aggregation, and adsorption effects at air-water interfaces. These artifacts can significantly alter the observed structures and lead to misinterpretation of results. Advanced preparation techniques such as controlled blotting parameters, specialized grid treatments, and environmental control during sample application help minimize these preparation-related artifacts.Expand Specific Solutions04 Image processing and reconstruction artifacts
Computational processing of cryo-TEM data can introduce artifacts during 3D reconstruction, particle picking, and contrast enhancement. These include missing wedge artifacts, preferential orientation issues, and artificial features from excessive filtering or classification. Advanced algorithms and validation protocols are being developed to identify and minimize these computational artifacts, ensuring more reliable structural interpretations from cryo-TEM data.Expand Specific Solutions05 Contamination and environmental artifacts
Environmental factors can introduce contamination artifacts in cryo-TEM, including ice contamination from atmospheric water vapor, carbon contamination from the microscope vacuum system, and particulate contaminants. These artifacts can obscure structural details and interfere with image interpretation. Specialized equipment such as anti-contamination devices, improved vacuum systems, and controlled transfer environments help minimize these environmental artifacts during cryo-TEM imaging.Expand Specific Solutions
Leading Manufacturers and Research Groups in Cryo-TEM
Cryo-TEM artifact avoidance represents a critical challenge in the evolving field of cryogenic electron microscopy, currently transitioning from early adoption to mainstream implementation. The market is experiencing robust growth, projected to reach significant scale as structural biology applications expand. Technologically, the field shows varying maturity levels, with companies like FEI Co. (now part of Thermo Fisher) leading in instrumentation development, while United Imaging Healthcare, Siemens Healthineers, and Hitachi contribute significant innovations in imaging technologies. Specialized players such as NanoSoft and Simple Origin focus on cryo-specific accessories and sample preparation solutions. Academic institutions including Max Planck Society and University of Washington collaborate with industry partners to address fundamental challenges in ice contamination, beam-induced damage, and specimen preparation artifacts that currently limit widespread adoption.
Siemens Healthineers AG
Technical Solution: Siemens Healthineers has developed advanced artifact reduction technologies for their Cryo-TEM systems focused on medical and diagnostic applications. Their proprietary Direct Electron Detection system incorporates radiation-hardened sensors that minimize counting artifacts while enabling extremely low-dose imaging protocols. Siemens' automated specimen handling system maintains samples in a contamination-free environment throughout the preparation and transfer process, eliminating ice contamination artifacts. Their TrueAlign technology provides automated alignment procedures that correct for optical aberrations and astigmatism that can create artificial structures in images. Siemens has implemented sophisticated drift correction algorithms that compensate for both mechanical and thermal instabilities during long exposure times. Their systems feature specialized energy filters that remove inelastically scattered electrons, significantly improving signal-to-noise ratio and reducing chromatic aberration artifacts. Additionally, Siemens' image processing suite includes AI-powered artifact recognition and correction tools that can identify and remove common artifacts during post-processing.
Strengths: Exceptional integration with medical imaging workflows; advanced automation reduces operator-dependent artifacts; sophisticated software tools for artifact identification and correction. Weaknesses: Systems optimized for medical applications may lack flexibility for certain research protocols; premium pricing structure; proprietary software ecosystems can limit integration with third-party tools.
FEI Co.
Technical Solution: FEI Co. (now part of Thermo Fisher Scientific) has developed comprehensive artifact mitigation solutions for Cryo-TEM through their Vitrobot sample preparation system. This automated vitrification device precisely controls blotting parameters, humidity, and temperature to ensure optimal ice thickness and prevent crystallization artifacts. Their Direct Electron Detectors employ electron counting technology that significantly reduces beam-induced motion artifacts by capturing multiple frames per second and applying sophisticated motion correction algorithms. FEI's automated low-dose imaging workflows minimize beam damage artifacts by limiting electron exposure while maintaining image quality. Their phase plate technology enhances contrast without increasing electron dose, reducing beam-induced artifacts in biological samples. Additionally, FEI has pioneered energy filtering systems that remove inelastically scattered electrons, dramatically improving signal-to-noise ratio and reducing chromatic aberration artifacts in Cryo-TEM images.
Strengths: Industry-leading sample preparation automation reduces user-dependent artifacts; integrated workflow solutions address multiple artifact sources simultaneously; high-sensitivity detectors enable lower dose imaging with reduced beam damage. Weaknesses: Premium equipment comes with high acquisition and maintenance costs; complex systems require significant user training; some solutions may be overengineered for basic research applications.
Critical Analysis of Artifact Reduction Technologies
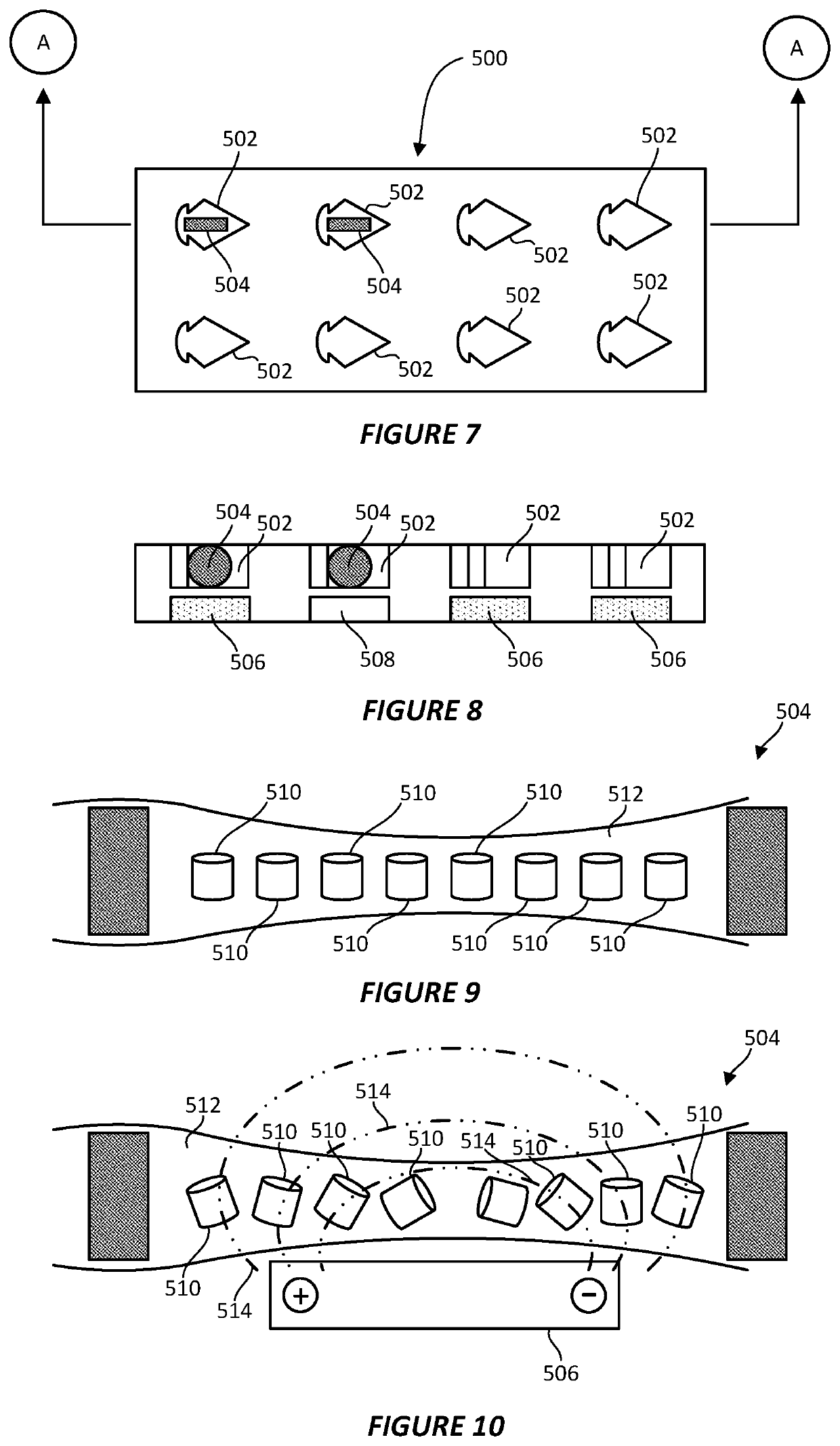
Cryogenic transmission electron microscopy sample preparation
PatentPendingEP4105632A1
Innovation
- A blotless technique involving a hydrophilic porous material that is loaded with a fluidic sample and brought into contact with a hydrophilic TEM substrate, allowing sample transfer and vitrification without excess volume removal, utilizing a synchronized motion to plunge the substrate into a cryogen for efficient sample preparation.
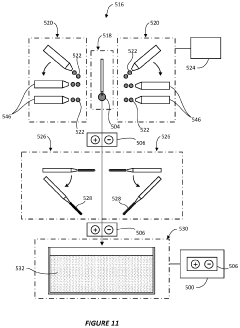
System and method for preparing CRYO-em grids
PatentActiveUS20200363301A1
Innovation
- A method and apparatus that utilize an electromagnetic field (EMF) to re-orient proteins and a system with a sample shaping element and cryogenic vitrifying element to deposit, thin, and vitrify samples on cryo-EM grids, allowing for precise control over sample thickness and orientation, and incorporating a storage device to further randomize protein orientations post-vitrification.
Sample Preparation Optimization Strategies
Sample preparation represents the most critical step in achieving high-quality cryo-TEM results while minimizing artifacts. Optimizing this process requires a systematic approach that addresses multiple variables affecting specimen quality. The vitrification process must be carefully controlled to ensure rapid freezing without ice crystal formation, which can severely distort biological structures and create misleading images.
Temperature management during sample handling is paramount. All tools and grids should be maintained at optimal temperatures to prevent premature freezing or warming effects that can introduce structural artifacts. Pre-cooling instruments to appropriate temperatures (typically 4°C for initial handling and near liquid nitrogen temperatures for vitrification steps) significantly reduces thermal gradient-induced damage to delicate specimens.
Buffer composition plays a crucial role in sample stability and image contrast. Phosphate buffers should generally be avoided as they can form electron-dense precipitates during vitrification. Instead, HEPES, Tris, or MES buffers at concentrations between 20-50 mM typically provide better results. The addition of small amounts (1-3%) of trehalose or glycerol can act as cryoprotectants, reducing ice crystal formation without significantly altering biological structures.
Grid selection and treatment significantly impact sample distribution and ice quality. Hydrophilicity adjustments through glow discharge or plasma cleaning optimize the spread of aqueous samples across the grid surface. The duration and intensity of these treatments must be calibrated for specific sample types - typically 30-60 seconds of glow discharge for protein solutions, while lipid-containing samples may require shorter treatments to maintain appropriate hydrophobic/hydrophilic balance.
Blotting parameters represent another critical variable requiring optimization. The blotting force, duration (typically 3-7 seconds), and environmental humidity (optimally 80-95%) must be adjusted based on sample viscosity and concentration. Double-sided blotting generally produces more uniform ice thickness compared to single-sided approaches, though sample-specific testing is essential.
Plunge freezing velocity and depth affect vitrification quality. Modern automated systems maintain consistent plunge speeds of approximately 1-2 m/s, which typically achieves optimal vitrification rates. The liquid ethane or propane used for freezing should be maintained just above its freezing point to maximize cooling efficiency without creating temperature gradients that could induce ice crystal formation.
Post-vitrification handling requires extreme care to prevent warming events that could cause devitrification. Transfer systems with minimal exposure to ambient conditions and proper storage in liquid nitrogen are essential for maintaining sample integrity until imaging can be performed.
Temperature management during sample handling is paramount. All tools and grids should be maintained at optimal temperatures to prevent premature freezing or warming effects that can introduce structural artifacts. Pre-cooling instruments to appropriate temperatures (typically 4°C for initial handling and near liquid nitrogen temperatures for vitrification steps) significantly reduces thermal gradient-induced damage to delicate specimens.
Buffer composition plays a crucial role in sample stability and image contrast. Phosphate buffers should generally be avoided as they can form electron-dense precipitates during vitrification. Instead, HEPES, Tris, or MES buffers at concentrations between 20-50 mM typically provide better results. The addition of small amounts (1-3%) of trehalose or glycerol can act as cryoprotectants, reducing ice crystal formation without significantly altering biological structures.
Grid selection and treatment significantly impact sample distribution and ice quality. Hydrophilicity adjustments through glow discharge or plasma cleaning optimize the spread of aqueous samples across the grid surface. The duration and intensity of these treatments must be calibrated for specific sample types - typically 30-60 seconds of glow discharge for protein solutions, while lipid-containing samples may require shorter treatments to maintain appropriate hydrophobic/hydrophilic balance.
Blotting parameters represent another critical variable requiring optimization. The blotting force, duration (typically 3-7 seconds), and environmental humidity (optimally 80-95%) must be adjusted based on sample viscosity and concentration. Double-sided blotting generally produces more uniform ice thickness compared to single-sided approaches, though sample-specific testing is essential.
Plunge freezing velocity and depth affect vitrification quality. Modern automated systems maintain consistent plunge speeds of approximately 1-2 m/s, which typically achieves optimal vitrification rates. The liquid ethane or propane used for freezing should be maintained just above its freezing point to maximize cooling efficiency without creating temperature gradients that could induce ice crystal formation.
Post-vitrification handling requires extreme care to prevent warming events that could cause devitrification. Transfer systems with minimal exposure to ambient conditions and proper storage in liquid nitrogen are essential for maintaining sample integrity until imaging can be performed.
Quality Control Standards for Cryo-TEM Data Validation
Establishing robust quality control standards is essential for validating cryo-TEM data and distinguishing genuine structural information from artifacts. These standards must encompass multiple levels of validation, beginning with sample preparation quality assessment. Vitrified specimens should be examined for ice thickness uniformity, absence of crystalline ice, and appropriate particle distribution. Documentation of grid preparation parameters, including blotting conditions and freezing rates, provides critical context for evaluating potential artifacts.
Image acquisition parameters require standardized documentation protocols, including microscope settings (acceleration voltage, beam current, dose rate), detector configurations, and imaging conditions (defocus values, exposure times). These parameters significantly influence image quality and must be recorded to ensure reproducibility and facilitate troubleshooting when artifacts are detected.
Post-processing validation represents another critical quality control checkpoint. All image processing steps should be documented with clear justification for filter applications, contrast adjustments, and any computational corrections. Comparison between raw and processed images must be maintained to verify that processing has not introduced artificial features or eliminated genuine structural elements.
Statistical validation approaches are increasingly important for cryo-TEM data quality assessment. Multiple technical and biological replicates should be analyzed to confirm structural observations. Quantitative metrics such as resolution measurements, signal-to-noise ratios, and particle classification statistics provide objective measures of data quality. Implementation of automated detection algorithms for common artifacts (beam-induced motion, charging effects, ice contamination) can standardize quality assessment across laboratories.
External validation mechanisms further strengthen data integrity. Correlation with complementary structural techniques (X-ray crystallography, NMR, mass spectrometry) provides independent verification of observed structures. Blind testing protocols, where analysts evaluate data without knowledge of experimental conditions, help identify subtle artifacts that might otherwise be overlooked.
Implementation of standardized reporting requirements for publication ensures transparency in the scientific community. These should include detailed methods sections documenting all sample preparation steps, imaging conditions, and processing workflows. Raw data deposition in public repositories enables independent verification and reanalysis, fostering greater confidence in structural conclusions derived from cryo-TEM experiments.
Image acquisition parameters require standardized documentation protocols, including microscope settings (acceleration voltage, beam current, dose rate), detector configurations, and imaging conditions (defocus values, exposure times). These parameters significantly influence image quality and must be recorded to ensure reproducibility and facilitate troubleshooting when artifacts are detected.
Post-processing validation represents another critical quality control checkpoint. All image processing steps should be documented with clear justification for filter applications, contrast adjustments, and any computational corrections. Comparison between raw and processed images must be maintained to verify that processing has not introduced artificial features or eliminated genuine structural elements.
Statistical validation approaches are increasingly important for cryo-TEM data quality assessment. Multiple technical and biological replicates should be analyzed to confirm structural observations. Quantitative metrics such as resolution measurements, signal-to-noise ratios, and particle classification statistics provide objective measures of data quality. Implementation of automated detection algorithms for common artifacts (beam-induced motion, charging effects, ice contamination) can standardize quality assessment across laboratories.
External validation mechanisms further strengthen data integrity. Correlation with complementary structural techniques (X-ray crystallography, NMR, mass spectrometry) provides independent verification of observed structures. Blind testing protocols, where analysts evaluate data without knowledge of experimental conditions, help identify subtle artifacts that might otherwise be overlooked.
Implementation of standardized reporting requirements for publication ensures transparency in the scientific community. These should include detailed methods sections documenting all sample preparation steps, imaging conditions, and processing workflows. Raw data deposition in public repositories enables independent verification and reanalysis, fostering greater confidence in structural conclusions derived from cryo-TEM experiments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!