Comparative Guide: Cryo-EM Vs. Conventional TEM For Soft Matter
AUG 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cryo-EM and TEM Evolution for Soft Matter Analysis
The evolution of electron microscopy techniques for soft matter analysis represents a significant advancement in materials science and structural biology. Transmission Electron Microscopy (TEM) emerged in the 1930s as a revolutionary tool for visualizing structures beyond the resolution limits of light microscopy. However, conventional TEM faced substantial limitations when examining soft materials due to the high vacuum environment and electron beam damage that often destroyed delicate biological and polymer samples.
The 1980s marked a pivotal turning point with the development of cryo-electron microscopy (cryo-EM), pioneered by Jacques Dubochet and colleagues. This technique involved rapid freezing of samples in vitreous ice, preserving their native structure while minimizing radiation damage. This innovation fundamentally changed how researchers could examine soft matter, enabling visualization of hydrated specimens in near-native states.
Throughout the 1990s, incremental improvements in electron detectors and computational methods gradually enhanced image quality, though resolution remained limited compared to crystallographic techniques. The real breakthrough came in the 2010s with the "resolution revolution," driven by the introduction of direct electron detectors that dramatically improved signal-to-noise ratios and enabled single-particle analysis at near-atomic resolutions.
Parallel to hardware developments, software advancements in image processing algorithms allowed researchers to extract maximum information from noisy cryo-EM images. Machine learning approaches further revolutionized particle picking and classification, enabling the analysis of heterogeneous samples that would be impossible with conventional methods.
For soft matter specifically, the evolution has been particularly impactful. Traditional polymer science relied heavily on bulk characterization techniques, with TEM providing limited structural insights due to contrast and beam damage issues. Cryo-EM techniques adapted specifically for soft materials have enabled visualization of dynamic assemblies, phase separations, and interfacial phenomena previously inaccessible to direct imaging.
Recent developments have focused on time-resolved cryo-EM, allowing researchers to capture transient states in soft matter systems. Techniques like cryo-electron tomography have further expanded capabilities by providing three-dimensional views of complex soft assemblies without the need for crystallization or averaging.
The technical evolution continues with innovations in sample preparation, including focused ion beam milling for examining thick specimens and microfluidic approaches for capturing dynamic processes. These developments represent a continuous refinement of the fundamental approach established by early cryo-EM pioneers, each iteration expanding the range of soft matter systems that can be effectively studied at the nanoscale.
The 1980s marked a pivotal turning point with the development of cryo-electron microscopy (cryo-EM), pioneered by Jacques Dubochet and colleagues. This technique involved rapid freezing of samples in vitreous ice, preserving their native structure while minimizing radiation damage. This innovation fundamentally changed how researchers could examine soft matter, enabling visualization of hydrated specimens in near-native states.
Throughout the 1990s, incremental improvements in electron detectors and computational methods gradually enhanced image quality, though resolution remained limited compared to crystallographic techniques. The real breakthrough came in the 2010s with the "resolution revolution," driven by the introduction of direct electron detectors that dramatically improved signal-to-noise ratios and enabled single-particle analysis at near-atomic resolutions.
Parallel to hardware developments, software advancements in image processing algorithms allowed researchers to extract maximum information from noisy cryo-EM images. Machine learning approaches further revolutionized particle picking and classification, enabling the analysis of heterogeneous samples that would be impossible with conventional methods.
For soft matter specifically, the evolution has been particularly impactful. Traditional polymer science relied heavily on bulk characterization techniques, with TEM providing limited structural insights due to contrast and beam damage issues. Cryo-EM techniques adapted specifically for soft materials have enabled visualization of dynamic assemblies, phase separations, and interfacial phenomena previously inaccessible to direct imaging.
Recent developments have focused on time-resolved cryo-EM, allowing researchers to capture transient states in soft matter systems. Techniques like cryo-electron tomography have further expanded capabilities by providing three-dimensional views of complex soft assemblies without the need for crystallization or averaging.
The technical evolution continues with innovations in sample preparation, including focused ion beam milling for examining thick specimens and microfluidic approaches for capturing dynamic processes. These developments represent a continuous refinement of the fundamental approach established by early cryo-EM pioneers, each iteration expanding the range of soft matter systems that can be effectively studied at the nanoscale.
Market Demand for Advanced Soft Matter Imaging Solutions
The soft matter imaging market is experiencing significant growth driven by increasing research activities in materials science, biology, and nanotechnology. The global electron microscopy market, valued at approximately $4.1 billion in 2021, is projected to reach $7.3 billion by 2028, with advanced imaging solutions for soft matter representing a rapidly expanding segment within this market.
Pharmaceutical and biotechnology sectors demonstrate particularly strong demand for high-resolution imaging technologies capable of visualizing soft matter samples without structural damage. This demand stems from the critical need to understand protein structures, drug-target interactions, and biomolecular assemblies at near-atomic resolution, which directly impacts drug discovery and development processes.
Academic and research institutions constitute another major market segment, with substantial funding directed toward advanced microscopy facilities. The ability to visualize soft materials in their native state has become essential for cutting-edge research in fields ranging from polymer science to cellular biology, driving investment in technologies that preserve sample integrity.
Industrial applications represent an emerging market opportunity, particularly in sectors developing advanced materials, nanomaterials, and biomaterials. Companies in these fields increasingly require sophisticated imaging capabilities to characterize material properties, validate manufacturing processes, and ensure quality control of soft matter products.
Geographically, North America and Europe currently dominate the market for advanced soft matter imaging solutions, accounting for approximately 65% of global demand. However, the Asia-Pacific region is witnessing the fastest growth rate, with China, Japan, and South Korea making substantial investments in research infrastructure and advanced microscopy capabilities.
The market shows a clear preference shift from conventional TEM toward cryo-EM technologies for soft matter applications. This transition is evidenced by the increasing allocation of research budgets toward cryo-EM facilities, with many institutions reporting 30-40% higher spending on cryo-EM compared to conventional TEM systems over the past five years.
Cost remains a significant market constraint, with high-end cryo-EM systems typically requiring investments of $5-7 million, plus substantial operational and maintenance expenses. This has created a market segment for shared facilities and service providers offering access to advanced imaging capabilities on a fee-for-service basis, particularly benefiting smaller research organizations and companies.
Market forecasts indicate continued strong growth for soft matter imaging solutions, with cryo-EM technologies expected to see compound annual growth rates of 12-15% through 2030, significantly outpacing the broader electron microscopy market's growth rate of 8-10%.
Pharmaceutical and biotechnology sectors demonstrate particularly strong demand for high-resolution imaging technologies capable of visualizing soft matter samples without structural damage. This demand stems from the critical need to understand protein structures, drug-target interactions, and biomolecular assemblies at near-atomic resolution, which directly impacts drug discovery and development processes.
Academic and research institutions constitute another major market segment, with substantial funding directed toward advanced microscopy facilities. The ability to visualize soft materials in their native state has become essential for cutting-edge research in fields ranging from polymer science to cellular biology, driving investment in technologies that preserve sample integrity.
Industrial applications represent an emerging market opportunity, particularly in sectors developing advanced materials, nanomaterials, and biomaterials. Companies in these fields increasingly require sophisticated imaging capabilities to characterize material properties, validate manufacturing processes, and ensure quality control of soft matter products.
Geographically, North America and Europe currently dominate the market for advanced soft matter imaging solutions, accounting for approximately 65% of global demand. However, the Asia-Pacific region is witnessing the fastest growth rate, with China, Japan, and South Korea making substantial investments in research infrastructure and advanced microscopy capabilities.
The market shows a clear preference shift from conventional TEM toward cryo-EM technologies for soft matter applications. This transition is evidenced by the increasing allocation of research budgets toward cryo-EM facilities, with many institutions reporting 30-40% higher spending on cryo-EM compared to conventional TEM systems over the past five years.
Cost remains a significant market constraint, with high-end cryo-EM systems typically requiring investments of $5-7 million, plus substantial operational and maintenance expenses. This has created a market segment for shared facilities and service providers offering access to advanced imaging capabilities on a fee-for-service basis, particularly benefiting smaller research organizations and companies.
Market forecasts indicate continued strong growth for soft matter imaging solutions, with cryo-EM technologies expected to see compound annual growth rates of 12-15% through 2030, significantly outpacing the broader electron microscopy market's growth rate of 8-10%.
Current Capabilities and Limitations of Cryo-EM vs TEM
Cryo-electron microscopy (Cryo-EM) and conventional transmission electron microscopy (TEM) represent two distinct approaches to visualizing soft matter at the nanoscale level. While both techniques utilize electron beams to generate high-resolution images, they differ significantly in their sample preparation methods, imaging conditions, and resultant capabilities.
Conventional TEM typically operates at room temperature and requires extensive sample preparation, including chemical fixation, dehydration, embedding in resin, and heavy metal staining. These procedures, while established and widely accessible, often introduce artifacts and structural alterations to soft matter specimens. The high vacuum environment and electron beam exposure during imaging further exacerbate these issues, causing radiation damage and sample deformation.
Cryo-EM, by contrast, employs rapid vitrification of samples in their native hydrated state, preserving their structural integrity without chemical fixation or staining. This approach maintains specimens in a near-native environment, significantly reducing preparation artifacts. The cryogenic temperatures (-180°C or lower) also mitigate radiation damage by limiting the mobility of free radicals generated during electron beam exposure.
Resolution capabilities represent another critical distinction between these techniques. While conventional TEM can achieve resolutions of 0.5-2 nm, it often struggles with soft matter due to low contrast and preparation-induced artifacts. Cryo-EM, particularly with recent technological advancements, can now routinely achieve sub-nanometer resolution (0.3-0.5 nm) for soft matter specimens, enabling visualization of molecular details previously inaccessible.
Sample thickness limitations also differentiate these approaches. Conventional TEM typically requires ultrathin sections (50-100 nm) for optimal imaging, necessitating physical sectioning that can disrupt delicate structures. Cryo-EM can accommodate slightly thicker specimens (up to 500 nm), allowing for more comprehensive structural analysis of intact soft matter assemblies.
Contrast generation presents unique challenges in both techniques. Conventional TEM relies heavily on heavy metal stains to enhance contrast, potentially obscuring native structures. Cryo-EM generates phase contrast from the specimen itself, offering more authentic structural information, though often with lower signal-to-noise ratios that require sophisticated image processing.
Time and resource requirements further distinguish these methods. Conventional TEM sample preparation is time-consuming (days) but utilizes widely available equipment. Cryo-EM sample preparation is rapid (minutes) but requires specialized vitrification equipment and cryogenic handling expertise. Additionally, cryo-EM facilities are less accessible and more expensive to operate, limiting widespread adoption despite their superior capabilities for soft matter analysis.
Conventional TEM typically operates at room temperature and requires extensive sample preparation, including chemical fixation, dehydration, embedding in resin, and heavy metal staining. These procedures, while established and widely accessible, often introduce artifacts and structural alterations to soft matter specimens. The high vacuum environment and electron beam exposure during imaging further exacerbate these issues, causing radiation damage and sample deformation.
Cryo-EM, by contrast, employs rapid vitrification of samples in their native hydrated state, preserving their structural integrity without chemical fixation or staining. This approach maintains specimens in a near-native environment, significantly reducing preparation artifacts. The cryogenic temperatures (-180°C or lower) also mitigate radiation damage by limiting the mobility of free radicals generated during electron beam exposure.
Resolution capabilities represent another critical distinction between these techniques. While conventional TEM can achieve resolutions of 0.5-2 nm, it often struggles with soft matter due to low contrast and preparation-induced artifacts. Cryo-EM, particularly with recent technological advancements, can now routinely achieve sub-nanometer resolution (0.3-0.5 nm) for soft matter specimens, enabling visualization of molecular details previously inaccessible.
Sample thickness limitations also differentiate these approaches. Conventional TEM typically requires ultrathin sections (50-100 nm) for optimal imaging, necessitating physical sectioning that can disrupt delicate structures. Cryo-EM can accommodate slightly thicker specimens (up to 500 nm), allowing for more comprehensive structural analysis of intact soft matter assemblies.
Contrast generation presents unique challenges in both techniques. Conventional TEM relies heavily on heavy metal stains to enhance contrast, potentially obscuring native structures. Cryo-EM generates phase contrast from the specimen itself, offering more authentic structural information, though often with lower signal-to-noise ratios that require sophisticated image processing.
Time and resource requirements further distinguish these methods. Conventional TEM sample preparation is time-consuming (days) but utilizes widely available equipment. Cryo-EM sample preparation is rapid (minutes) but requires specialized vitrification equipment and cryogenic handling expertise. Additionally, cryo-EM facilities are less accessible and more expensive to operate, limiting widespread adoption despite their superior capabilities for soft matter analysis.
Comparative Methodologies and Protocols for Soft Matter Imaging
01 Resolution enhancement techniques in cryo-EM
Various techniques have been developed to enhance the resolution of cryo-electron microscopy (cryo-EM) imaging compared to conventional transmission electron microscopy (TEM). These include advanced image processing algorithms, noise reduction methods, and specialized sample preparation protocols that preserve the native structure of biological specimens. These enhancements allow researchers to achieve near-atomic resolution in structural biology studies, enabling visualization of macromolecular complexes in their native states.- Resolution enhancement techniques in cryo-EM: Various techniques have been developed to enhance the resolution of cryo-electron microscopy (cryo-EM) imaging compared to conventional transmission electron microscopy (TEM). These include advanced image processing algorithms, improved sample preparation methods, and specialized detectors that can capture high-resolution structural details of biological specimens under cryogenic conditions. These enhancements allow researchers to visualize macromolecular structures at near-atomic resolution while preserving the native state of biological samples.
- Sample preparation differences between cryo-EM and conventional TEM: Sample preparation methods significantly impact imaging quality in both cryo-EM and conventional TEM. Cryo-EM involves rapid freezing of samples in their native hydrated state, avoiding chemical fixation and heavy metal staining typically used in conventional TEM. This vitrification process preserves the structural integrity of biological specimens and prevents the formation of ice crystals that could damage the sample. The differences in sample preparation directly affect contrast, resolution, and the ability to visualize biological structures in their native conformations.
- Detector technology advancements for electron microscopy: Modern detector technologies have revolutionized both cryo-EM and conventional TEM imaging quality. Direct electron detectors with improved detective quantum efficiency (DQE) and faster frame rates have significantly enhanced signal-to-noise ratios compared to traditional CCD cameras. These technological advancements allow for better contrast, higher resolution, and reduced beam-induced damage to sensitive biological specimens. The development of specialized detectors optimized for different imaging conditions has expanded the capabilities of both cryo-EM and conventional TEM systems.
- Contrast mechanisms and image formation differences: Cryo-EM and conventional TEM utilize different contrast mechanisms that affect image quality. Conventional TEM often relies on heavy metal staining to enhance contrast, while cryo-EM depends on phase contrast from unstained specimens. The low-dose imaging techniques necessary in cryo-EM to minimize radiation damage result in inherently lower contrast compared to conventional TEM. Various computational methods and optical configurations have been developed to optimize contrast while maintaining high resolution in both techniques, including phase plates, energy filters, and contrast transfer function correction algorithms.
- 3D reconstruction and computational processing methods: Advanced computational methods play a crucial role in extracting high-quality structural information from both cryo-EM and conventional TEM data. Single-particle analysis, tomographic reconstruction, and machine learning approaches have dramatically improved the ability to generate 3D models from 2D projections. These computational techniques can compensate for imaging artifacts, enhance contrast, and improve resolution beyond what is directly visible in raw micrographs. The integration of sophisticated algorithms with hardware improvements has enabled researchers to achieve near-atomic resolution structures from cryo-EM data that were previously only possible with X-ray crystallography.
02 Sample preparation differences between cryo-EM and conventional TEM
Sample preparation methods significantly impact imaging quality in both cryo-EM and conventional TEM. Cryo-EM involves rapid freezing of samples in their native state without staining, while conventional TEM often requires chemical fixation, dehydration, and heavy metal staining. The vitrification process in cryo-EM preserves biological structures in their near-native state, avoiding artifacts associated with conventional sample preparation, resulting in more accurate structural information but requiring specialized handling techniques.Expand Specific Solutions03 Detector technology advancements for electron microscopy
Modern detector technologies have revolutionized both cryo-EM and conventional TEM imaging quality. Direct electron detectors with improved detective quantum efficiency (DQE) and signal-to-noise ratio have particularly benefited cryo-EM by enabling better detection of weak signals from radiation-sensitive biological specimens. These detectors allow for electron counting and super-resolution imaging modes, significantly improving the quality of structural data obtained from biological samples compared to traditional scintillator-based cameras used in conventional TEM.Expand Specific Solutions04 Image processing and computational methods
Advanced computational methods play a crucial role in enhancing image quality for both cryo-EM and conventional TEM. Single-particle analysis, tomographic reconstruction, and machine learning algorithms help overcome limitations in raw imaging data. For cryo-EM specifically, motion correction algorithms compensate for beam-induced movement, while classification and averaging techniques enhance signal-to-noise ratio by combining information from multiple particles. These computational approaches have been instrumental in achieving high-resolution structural information from electron microscopy data.Expand Specific Solutions05 Applications and limitations comparison
Cryo-EM and conventional TEM have distinct applications and limitations regarding imaging quality. Cryo-EM excels in visualizing biological macromolecules in their native hydrated state at near-atomic resolution but requires specialized equipment and expertise. Conventional TEM offers higher contrast for certain applications and is more accessible but may introduce artifacts during sample preparation. The choice between these techniques depends on the specific research question, with cryo-EM being preferred for high-resolution structural biology and conventional TEM for applications where sample preparation artifacts are less critical.Expand Specific Solutions
Leading Manufacturers and Research Institutions in EM Technology
Cryo-EM technology for soft matter analysis is currently in a growth phase, with the market expanding rapidly due to its superior capabilities over conventional TEM in preserving delicate structures. The global market is estimated to reach several billion dollars by 2025, driven by pharmaceutical and materials science applications. Technologically, major academic institutions (Tsinghua University, Peking University, Harvard) are advancing fundamental research, while commercial players like FEI Co. and Protochips are developing specialized instrumentation. Companies such as NanoSoft and Hysitron are focusing on cryogenic sample preparation and handling solutions. The ecosystem shows a balanced mix of established microscopy leaders and innovative startups, with significant collaboration between academic and commercial entities driving technological maturity.
FEI Co.
Technical Solution: FEI Co. (now part of Thermo Fisher Scientific) has developed advanced cryo-electron microscopy (cryo-EM) solutions specifically optimized for soft matter analysis. Their Titan Krios platform integrates direct electron detectors with automated data collection workflows to achieve near-atomic resolution imaging of biological specimens. The system employs phase plates and energy filters to enhance contrast in radiation-sensitive samples without requiring heavy metal staining. FEI's technology utilizes sophisticated motion correction algorithms to compensate for beam-induced movement, allowing for higher resolution structural determination of proteins, viruses, and other macromolecular assemblies in their native states[1][3]. Their integrated workflow solutions encompass sample preparation through automated vitrification processes, ensuring specimens remain hydrated and structurally preserved during imaging.
Strengths: Superior resolution capabilities for visualizing soft matter in near-native states; automated workflows reduce user intervention and improve reproducibility; advanced motion correction algorithms enhance image quality. Weaknesses: Significantly higher cost compared to conventional TEM systems; requires specialized infrastructure including liquid nitrogen handling; complex operation requiring highly trained personnel.
New York Structural Biology Center, Inc.
Technical Solution: The New York Structural Biology Center (NYSBC) has developed an integrated approach to soft matter imaging that leverages both cryo-EM and conventional TEM methodologies. Their technology platform incorporates high-end Titan Krios cryo-electron microscopes with specialized sample preparation protocols optimized for different classes of soft materials. NYSBC has pioneered correlative light and electron microscopy (CLEM) workflows that allow researchers to identify regions of interest using fluorescence microscopy before transitioning to high-resolution cryo-EM analysis. Their automated data collection and processing pipeline employs machine learning algorithms to identify optimal imaging conditions for different soft matter specimens, maximizing resolution while minimizing beam damage[4]. NYSBC has also developed specialized grid preparation techniques that improve particle distribution and ice quality for challenging soft matter samples, including membrane proteins, polymeric materials, and hydrogels.
Strengths: Comprehensive workflow from sample preparation to data analysis; correlative imaging capabilities enhance contextual understanding; specialized protocols for different soft material classes improve success rates. Weaknesses: Centralized facility model limits accessibility; requires significant expertise to fully utilize capabilities; sample preparation remains a bottleneck for novel soft materials.
Breakthrough Innovations in Cryo-EM for Soft Matter Visualization
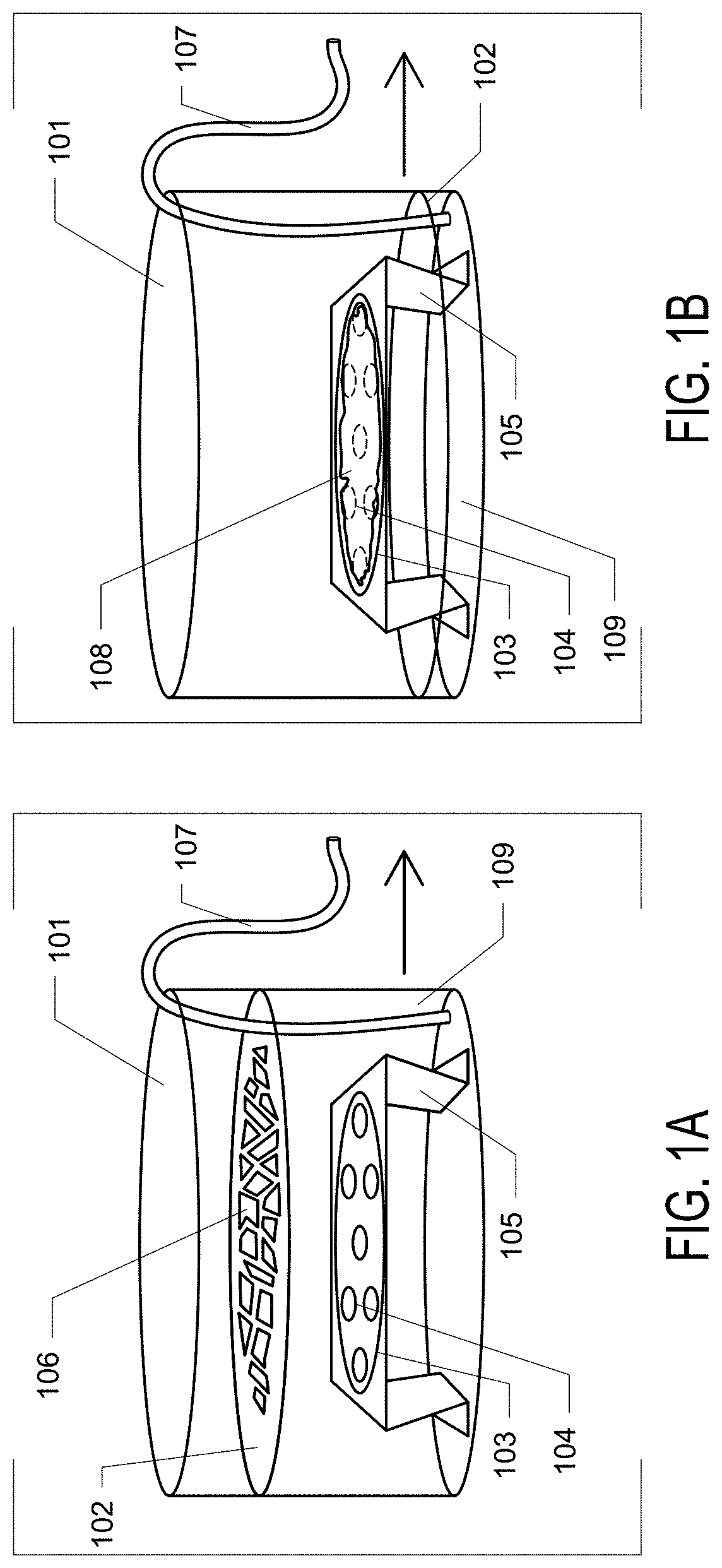
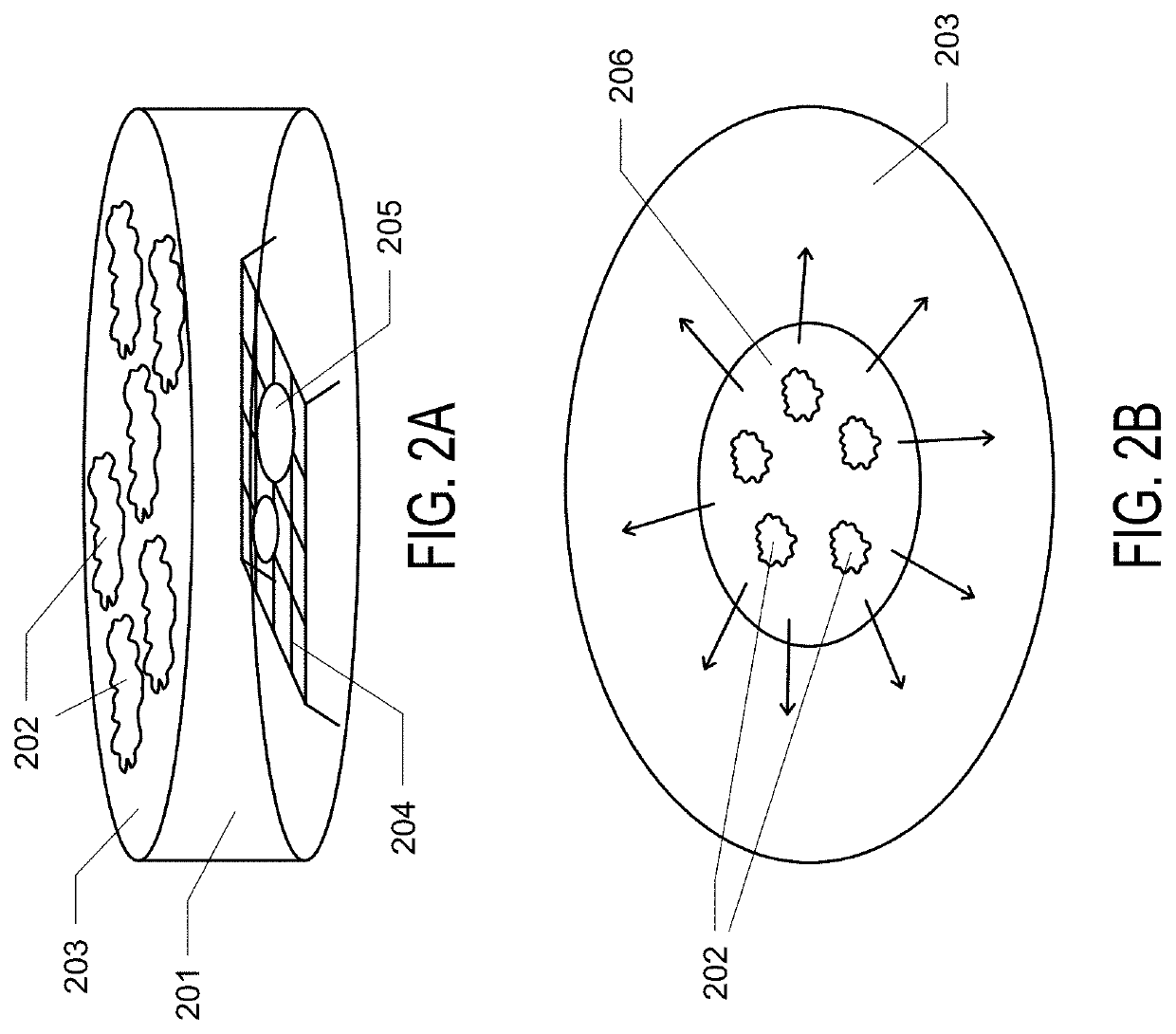
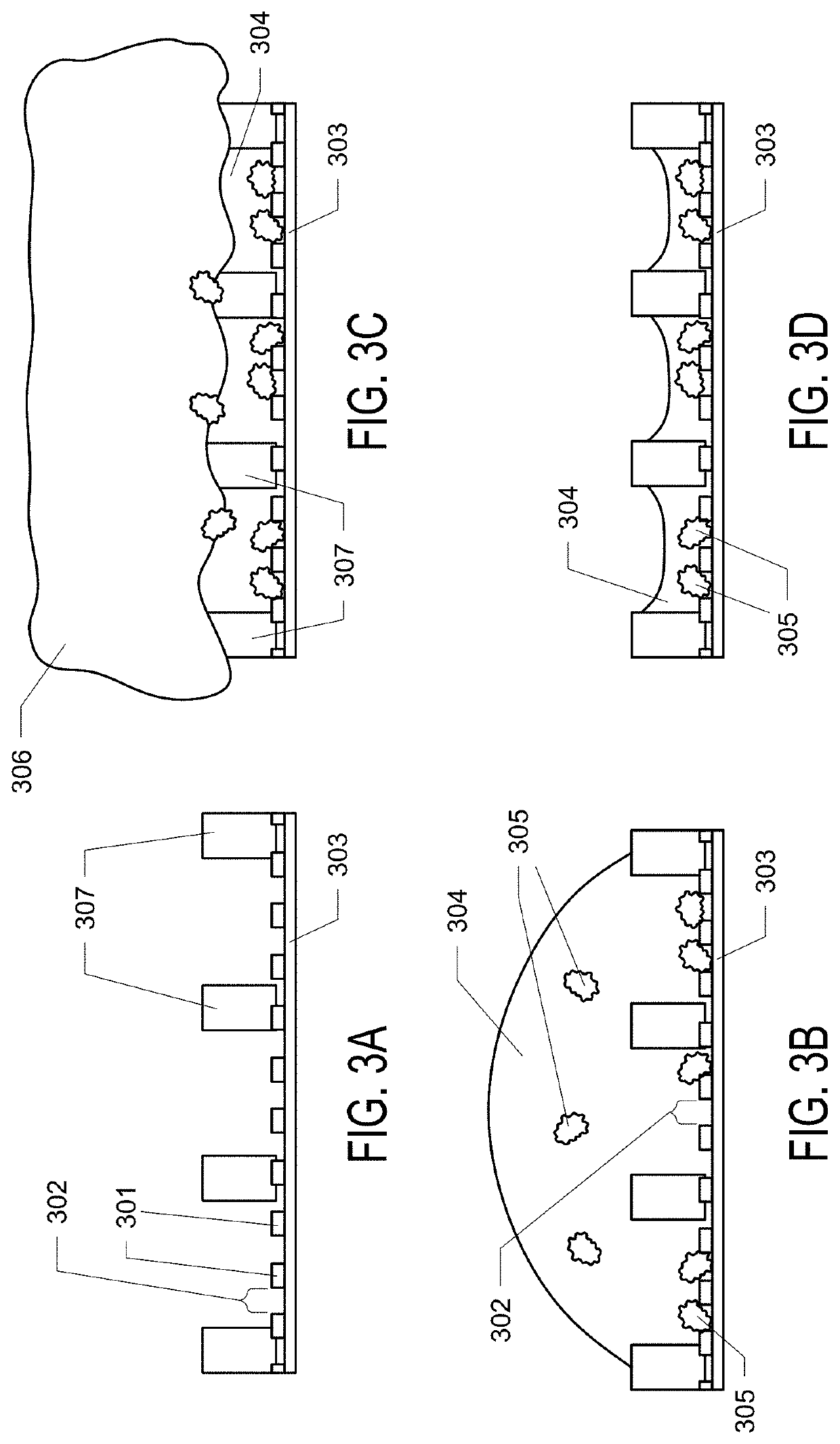
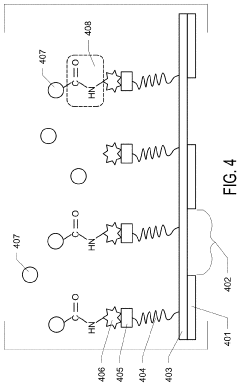
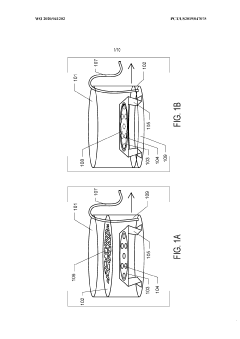
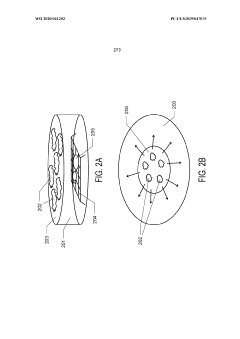
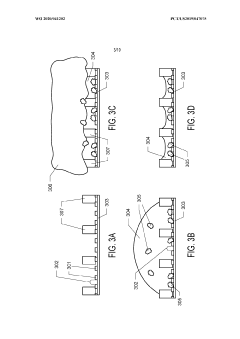
Graphene Oxide Affinity Sample Grids for Cyro-EM
PatentActiveUS20210310910A1
Innovation
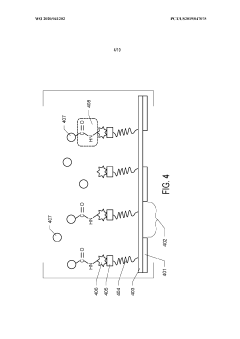
- The use of graphene oxide films functionalized with various chemistries for immobilizing diverse target species on cryo-EM grids, combined with polyethylene glycol linkers to position samples optimally away from the air-water interface and substrate, enabling efficient and versatile sample preparation for high-quality cryo-EM imaging.
Graphene oxide affinity sample grids for cryo-em
PatentWO2020041202A1
Innovation
- The use of graphene oxide affinity grids with functionalized surfaces, including polyethylene glycol spacers, to immobilize samples away from the air-water interface and substrate interactions, enabling diverse target species capture and storage, and facilitating high-quality sample preparation for cryo-EM imaging.
Sample Preparation Challenges and Solutions for Soft Materials
Sample preparation represents one of the most critical challenges in electron microscopy of soft materials. Soft matter, including polymers, biological specimens, and colloidal systems, presents unique difficulties due to their inherent sensitivity to electron beam damage, low contrast, and structural instability. These challenges are magnified when comparing cryo-electron microscopy (cryo-EM) with conventional transmission electron microscopy (TEM) approaches.
In conventional TEM, sample preparation typically involves chemical fixation, dehydration, and heavy metal staining, which can introduce artifacts and alter the native structure of soft materials. The vacuum environment and electron beam exposure often cause significant damage to these delicate specimens, resulting in structural collapse or molecular rearrangement that compromises data integrity.
Cryo-EM addresses many of these challenges through vitrification—a process where samples are rapidly frozen in a thin layer of amorphous ice, preserving their native hydrated state. This technique eliminates the need for chemical fixation and staining, thereby reducing preparation artifacts. However, cryo-EM introduces its own set of challenges, including the formation of crystalline ice, uneven ice thickness, and difficulties in maintaining the vitreous state throughout imaging.
The plunge-freezing technique represents a significant advancement for soft matter sample preparation in cryo-EM. This method involves rapidly immersing the specimen in liquid ethane cooled by liquid nitrogen, achieving cooling rates of approximately 10,000°C/second. Such rapid freezing prevents ice crystal formation that would otherwise disrupt molecular structures. Nevertheless, optimizing parameters such as blotting time, humidity, and temperature remains challenging and often requires extensive experimentation.
For larger soft matter specimens, high-pressure freezing offers an alternative approach by applying pressure (approximately 2,100 bar) during freezing, allowing vitrification of samples up to 200 μm thick. This technique has proven valuable for studying complex soft matter systems such as hydrogels and tissue-engineered constructs.
Recent innovations in sample preparation include the development of specialized grids with nanopatterned surfaces that improve ice thickness uniformity and specimen distribution. Additionally, automated sample preparation systems have emerged to enhance reproducibility by precisely controlling environmental conditions and freezing parameters.
Despite these advances, challenges persist in preparing beam-sensitive soft materials for electron microscopy. Researchers continue to develop novel approaches, including correlative light and electron microscopy techniques, which allow pre-identification of regions of interest before electron beam exposure, thereby minimizing radiation damage while maximizing information yield.
In conventional TEM, sample preparation typically involves chemical fixation, dehydration, and heavy metal staining, which can introduce artifacts and alter the native structure of soft materials. The vacuum environment and electron beam exposure often cause significant damage to these delicate specimens, resulting in structural collapse or molecular rearrangement that compromises data integrity.
Cryo-EM addresses many of these challenges through vitrification—a process where samples are rapidly frozen in a thin layer of amorphous ice, preserving their native hydrated state. This technique eliminates the need for chemical fixation and staining, thereby reducing preparation artifacts. However, cryo-EM introduces its own set of challenges, including the formation of crystalline ice, uneven ice thickness, and difficulties in maintaining the vitreous state throughout imaging.
The plunge-freezing technique represents a significant advancement for soft matter sample preparation in cryo-EM. This method involves rapidly immersing the specimen in liquid ethane cooled by liquid nitrogen, achieving cooling rates of approximately 10,000°C/second. Such rapid freezing prevents ice crystal formation that would otherwise disrupt molecular structures. Nevertheless, optimizing parameters such as blotting time, humidity, and temperature remains challenging and often requires extensive experimentation.
For larger soft matter specimens, high-pressure freezing offers an alternative approach by applying pressure (approximately 2,100 bar) during freezing, allowing vitrification of samples up to 200 μm thick. This technique has proven valuable for studying complex soft matter systems such as hydrogels and tissue-engineered constructs.
Recent innovations in sample preparation include the development of specialized grids with nanopatterned surfaces that improve ice thickness uniformity and specimen distribution. Additionally, automated sample preparation systems have emerged to enhance reproducibility by precisely controlling environmental conditions and freezing parameters.
Despite these advances, challenges persist in preparing beam-sensitive soft materials for electron microscopy. Researchers continue to develop novel approaches, including correlative light and electron microscopy techniques, which allow pre-identification of regions of interest before electron beam exposure, thereby minimizing radiation damage while maximizing information yield.
Cost-Benefit Analysis of Cryo-EM vs Conventional TEM Implementation
When evaluating the implementation of Cryo-Electron Microscopy (Cryo-EM) versus Conventional Transmission Electron Microscopy (TEM) for soft matter research, a comprehensive cost-benefit analysis is essential for informed decision-making.
Initial investment represents the most significant cost differential between these technologies. Cryo-EM systems typically require an investment of $5-7 million, approximately 2-3 times higher than conventional TEM systems. This substantial difference stems from the specialized equipment needed for cryogenic sample preparation, including vitrification devices, cryo-transfer systems, and advanced direct electron detectors.
Operational expenses also differ markedly between the two technologies. Cryo-EM demands specialized consumables such as liquid nitrogen and ethane, adding approximately $50,000-$100,000 annually to operational costs. Additionally, Cryo-EM facilities require dedicated technical staff with specialized training, increasing personnel expenses by 30-40% compared to conventional TEM operations.
Maintenance considerations further differentiate the technologies. Cryo-EM systems typically incur annual maintenance costs of $200,000-$300,000, significantly higher than the $80,000-$150,000 for conventional TEM systems. The complex cryogenic components require more frequent servicing and specialized expertise for maintenance.
Despite higher costs, Cryo-EM offers substantial benefits for soft matter research. The preservation of native hydrated states enables visualization of delicate structures that would be destroyed or distorted by conventional TEM sample preparation. This capability translates to more accurate structural data and fewer artifacts, potentially reducing the need for repeated experiments.
Resolution advantages provide another significant benefit. Modern Cryo-EM systems can achieve near-atomic resolution (2-3Å) for soft matter samples, compared to the 5-10Å typically achievable with conventional TEM for similar specimens. This resolution improvement can accelerate research timelines and enable discoveries not possible with conventional methods.
Return on investment calculations must consider research productivity gains. Studies indicate that Cryo-EM implementation can increase publication output in soft matter research by 30-50% within three years, potentially offsetting the higher initial investment through enhanced research impact and increased funding opportunities.
Institutional considerations also affect the cost-benefit equation. Shared facility models can distribute costs across multiple research groups or institutions, making Cryo-EM more financially viable. Additionally, the competitive advantage in securing grants and attracting top talent represents an intangible but significant benefit that should factor into long-term strategic planning.
Initial investment represents the most significant cost differential between these technologies. Cryo-EM systems typically require an investment of $5-7 million, approximately 2-3 times higher than conventional TEM systems. This substantial difference stems from the specialized equipment needed for cryogenic sample preparation, including vitrification devices, cryo-transfer systems, and advanced direct electron detectors.
Operational expenses also differ markedly between the two technologies. Cryo-EM demands specialized consumables such as liquid nitrogen and ethane, adding approximately $50,000-$100,000 annually to operational costs. Additionally, Cryo-EM facilities require dedicated technical staff with specialized training, increasing personnel expenses by 30-40% compared to conventional TEM operations.
Maintenance considerations further differentiate the technologies. Cryo-EM systems typically incur annual maintenance costs of $200,000-$300,000, significantly higher than the $80,000-$150,000 for conventional TEM systems. The complex cryogenic components require more frequent servicing and specialized expertise for maintenance.
Despite higher costs, Cryo-EM offers substantial benefits for soft matter research. The preservation of native hydrated states enables visualization of delicate structures that would be destroyed or distorted by conventional TEM sample preparation. This capability translates to more accurate structural data and fewer artifacts, potentially reducing the need for repeated experiments.
Resolution advantages provide another significant benefit. Modern Cryo-EM systems can achieve near-atomic resolution (2-3Å) for soft matter samples, compared to the 5-10Å typically achievable with conventional TEM for similar specimens. This resolution improvement can accelerate research timelines and enable discoveries not possible with conventional methods.
Return on investment calculations must consider research productivity gains. Studies indicate that Cryo-EM implementation can increase publication output in soft matter research by 30-50% within three years, potentially offsetting the higher initial investment through enhanced research impact and increased funding opportunities.
Institutional considerations also affect the cost-benefit equation. Shared facility models can distribute costs across multiple research groups or institutions, making Cryo-EM more financially viable. Additionally, the competitive advantage in securing grants and attracting top talent represents an intangible but significant benefit that should factor into long-term strategic planning.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!