How Do Innovations in Biocompatible Materials Impact Microfluidic Chips?
OCT 10, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Biocompatible Materials Evolution and Objectives
Biocompatible materials have undergone significant evolution since the inception of microfluidic technology in the early 1990s. Initially, microfluidic chips relied heavily on silicon and glass substrates, which offered excellent chemical stability but presented challenges in terms of fabrication complexity and cost. The late 1990s witnessed a paradigm shift with the introduction of polydimethylsiloxane (PDMS), which revolutionized the field due to its optical transparency, gas permeability, and ease of fabrication through soft lithography techniques.
The 2000s marked the exploration of a wider range of polymeric materials including polymethyl methacrylate (PMMA), polycarbonate (PC), and cyclic olefin copolymer (COC), each offering specific advantages for different biological applications. This diversification addressed various limitations of PDMS, particularly its hydrophobicity and tendency to absorb small hydrophobic molecules, which could interfere with biological assays.
Recent years have seen accelerated innovation in biocompatible materials for microfluidic applications, driven by the growing demands of organ-on-chip systems, point-of-care diagnostics, and personalized medicine. Hydrogel-based materials have emerged as promising candidates due to their tunable mechanical properties and structural similarity to extracellular matrices, enabling more physiologically relevant cell culture environments within microfluidic devices.
The integration of stimuli-responsive materials represents another significant advancement, allowing for dynamic control of surface properties and fluid flow within microchannels. These "smart" materials can change their characteristics in response to external stimuli such as temperature, pH, or light, enabling unprecedented control over microenvironments for biological studies.
Nanomaterial-enhanced composites have also gained traction, with the incorporation of nanoparticles, carbon nanotubes, and graphene derivatives improving the mechanical, electrical, and thermal properties of base materials while introducing novel functionalities such as antimicrobial properties or enhanced cell adhesion.
The primary objective in biocompatible material development for microfluidic applications is to create platforms that can faithfully recapitulate in vivo conditions while maintaining compatibility with biological entities. This includes minimizing non-specific protein adsorption, supporting cell adhesion and proliferation where desired, and ensuring no release of cytotoxic compounds during operation.
Additional objectives include developing materials with improved optical properties for enhanced imaging capabilities, extended shelf life for commercial viability, and compatibility with sterilization techniques. The ultimate goal remains the creation of materials that bridge the gap between in vitro testing and in vivo reality, thereby accelerating drug development processes and enabling more accurate disease modeling and personalized treatment approaches.
The 2000s marked the exploration of a wider range of polymeric materials including polymethyl methacrylate (PMMA), polycarbonate (PC), and cyclic olefin copolymer (COC), each offering specific advantages for different biological applications. This diversification addressed various limitations of PDMS, particularly its hydrophobicity and tendency to absorb small hydrophobic molecules, which could interfere with biological assays.
Recent years have seen accelerated innovation in biocompatible materials for microfluidic applications, driven by the growing demands of organ-on-chip systems, point-of-care diagnostics, and personalized medicine. Hydrogel-based materials have emerged as promising candidates due to their tunable mechanical properties and structural similarity to extracellular matrices, enabling more physiologically relevant cell culture environments within microfluidic devices.
The integration of stimuli-responsive materials represents another significant advancement, allowing for dynamic control of surface properties and fluid flow within microchannels. These "smart" materials can change their characteristics in response to external stimuli such as temperature, pH, or light, enabling unprecedented control over microenvironments for biological studies.
Nanomaterial-enhanced composites have also gained traction, with the incorporation of nanoparticles, carbon nanotubes, and graphene derivatives improving the mechanical, electrical, and thermal properties of base materials while introducing novel functionalities such as antimicrobial properties or enhanced cell adhesion.
The primary objective in biocompatible material development for microfluidic applications is to create platforms that can faithfully recapitulate in vivo conditions while maintaining compatibility with biological entities. This includes minimizing non-specific protein adsorption, supporting cell adhesion and proliferation where desired, and ensuring no release of cytotoxic compounds during operation.
Additional objectives include developing materials with improved optical properties for enhanced imaging capabilities, extended shelf life for commercial viability, and compatibility with sterilization techniques. The ultimate goal remains the creation of materials that bridge the gap between in vitro testing and in vivo reality, thereby accelerating drug development processes and enabling more accurate disease modeling and personalized treatment approaches.
Market Analysis for Biocompatible Microfluidic Applications
The global market for biocompatible microfluidic applications is experiencing robust growth, driven by increasing demand in healthcare, pharmaceuticals, and life sciences sectors. Current market valuations indicate that the microfluidic devices market reached approximately $15 billion in 2022, with biocompatible applications representing a significant segment of this market. Industry analysts project a compound annual growth rate of 18-20% through 2028, potentially pushing the market beyond $40 billion.
Healthcare applications dominate the biocompatible microfluidic market, accounting for nearly 45% of total demand. Point-of-care diagnostics represents the fastest-growing segment within healthcare, as innovations in biocompatible materials enable more sensitive, reliable, and cost-effective diagnostic platforms. The COVID-19 pandemic has further accelerated this trend, with unprecedented demand for rapid diagnostic solutions.
Pharmaceutical research and drug discovery applications constitute the second-largest market segment at approximately 30%. The ability of advanced biocompatible microfluidic platforms to mimic in vivo conditions while requiring minimal sample volumes has revolutionized drug screening processes. This has resulted in significant cost reductions and accelerated development timelines for pharmaceutical companies.
Regional analysis reveals North America as the current market leader with approximately 40% market share, followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is expected to demonstrate the highest growth rate over the next five years due to increasing healthcare expenditure, expanding research infrastructure, and growing manufacturing capabilities in countries like China, Japan, and South Korea.
Consumer preferences are increasingly favoring sustainable and environmentally friendly biocompatible materials. This shift is creating new market opportunities for biodegradable polymers and naturally derived materials in microfluidic applications. Companies that can effectively integrate sustainability into their product development strategies are likely to gain competitive advantages.
Market barriers include high initial development costs, complex regulatory approval processes, and technical challenges in scaling production. Despite these challenges, venture capital investment in biocompatible microfluidic startups has increased by over 200% in the past three years, indicating strong investor confidence in the sector's growth potential.
End-user analysis shows academic and research institutions currently represent the largest customer segment, though clinical and industrial applications are growing at a faster rate. This shift suggests a maturing market moving from research-focused applications toward widespread commercial and clinical implementation, driven by innovations in biocompatible materials that enhance performance, reliability, and cost-effectiveness.
Healthcare applications dominate the biocompatible microfluidic market, accounting for nearly 45% of total demand. Point-of-care diagnostics represents the fastest-growing segment within healthcare, as innovations in biocompatible materials enable more sensitive, reliable, and cost-effective diagnostic platforms. The COVID-19 pandemic has further accelerated this trend, with unprecedented demand for rapid diagnostic solutions.
Pharmaceutical research and drug discovery applications constitute the second-largest market segment at approximately 30%. The ability of advanced biocompatible microfluidic platforms to mimic in vivo conditions while requiring minimal sample volumes has revolutionized drug screening processes. This has resulted in significant cost reductions and accelerated development timelines for pharmaceutical companies.
Regional analysis reveals North America as the current market leader with approximately 40% market share, followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is expected to demonstrate the highest growth rate over the next five years due to increasing healthcare expenditure, expanding research infrastructure, and growing manufacturing capabilities in countries like China, Japan, and South Korea.
Consumer preferences are increasingly favoring sustainable and environmentally friendly biocompatible materials. This shift is creating new market opportunities for biodegradable polymers and naturally derived materials in microfluidic applications. Companies that can effectively integrate sustainability into their product development strategies are likely to gain competitive advantages.
Market barriers include high initial development costs, complex regulatory approval processes, and technical challenges in scaling production. Despite these challenges, venture capital investment in biocompatible microfluidic startups has increased by over 200% in the past three years, indicating strong investor confidence in the sector's growth potential.
End-user analysis shows academic and research institutions currently represent the largest customer segment, though clinical and industrial applications are growing at a faster rate. This shift suggests a maturing market moving from research-focused applications toward widespread commercial and clinical implementation, driven by innovations in biocompatible materials that enhance performance, reliability, and cost-effectiveness.
Current Landscape and Barriers in Microfluidic Chip Materials
The microfluidic chip materials landscape has evolved significantly over the past decade, with traditional materials like polydimethylsiloxane (PDMS), glass, and silicon still dominating commercial applications. PDMS remains the most widely used material due to its optical transparency, gas permeability, and ease of fabrication through soft lithography techniques. However, its limitations—including hydrophobicity, absorption of small molecules, and poor long-term stability—have driven research toward alternative materials.
Glass and silicon offer excellent chemical resistance and thermal stability but present challenges in rapid prototyping and complex structure fabrication. These materials typically require cleanroom facilities and specialized equipment, increasing production costs and limiting accessibility for many research groups.
Thermoplastics such as polymethyl methacrylate (PMMA), polycarbonate (PC), and cyclic olefin copolymer (COC) have gained traction for commercial applications due to their compatibility with mass production techniques like injection molding and hot embossing. These materials offer improved chemical resistance compared to PDMS but often lack the same level of gas permeability crucial for cell-based applications.
A significant barrier in current microfluidic materials is biocompatibility. Many conventional materials exhibit surface properties that can trigger protein adsorption, cell adhesion, or immune responses when in contact with biological samples. This non-specific adsorption can alter analyte concentrations and compromise assay performance, particularly in diagnostic applications requiring high sensitivity.
Surface modification techniques have emerged as temporary solutions, including plasma treatment, chemical functionalization, and coating with biocompatible layers. However, these approaches often provide only short-term improvements and may alter the material's bulk properties or introduce additional fabrication complexity.
Another critical challenge is the integration of different materials within a single microfluidic platform. Multi-material devices are increasingly necessary to combine optimal properties—such as optical clarity in detection regions with chemical resistance in reaction chambers—but bonding dissimilar materials remains technically challenging due to differences in thermal expansion coefficients and surface chemistries.
Manufacturing scalability presents another significant barrier. While academic prototyping has advanced rapidly, transitioning novel biocompatible materials to industrial-scale production faces hurdles in standardization, quality control, and cost-effectiveness. The disconnect between laboratory innovation and commercial implementation has slowed the adoption of next-generation materials in practical applications.
Regulatory considerations further complicate material selection, particularly for clinical applications. Materials must meet stringent safety standards and demonstrate consistent performance under various conditions, creating a high barrier to entry for novel biocompatible materials lacking extensive validation data.
Glass and silicon offer excellent chemical resistance and thermal stability but present challenges in rapid prototyping and complex structure fabrication. These materials typically require cleanroom facilities and specialized equipment, increasing production costs and limiting accessibility for many research groups.
Thermoplastics such as polymethyl methacrylate (PMMA), polycarbonate (PC), and cyclic olefin copolymer (COC) have gained traction for commercial applications due to their compatibility with mass production techniques like injection molding and hot embossing. These materials offer improved chemical resistance compared to PDMS but often lack the same level of gas permeability crucial for cell-based applications.
A significant barrier in current microfluidic materials is biocompatibility. Many conventional materials exhibit surface properties that can trigger protein adsorption, cell adhesion, or immune responses when in contact with biological samples. This non-specific adsorption can alter analyte concentrations and compromise assay performance, particularly in diagnostic applications requiring high sensitivity.
Surface modification techniques have emerged as temporary solutions, including plasma treatment, chemical functionalization, and coating with biocompatible layers. However, these approaches often provide only short-term improvements and may alter the material's bulk properties or introduce additional fabrication complexity.
Another critical challenge is the integration of different materials within a single microfluidic platform. Multi-material devices are increasingly necessary to combine optimal properties—such as optical clarity in detection regions with chemical resistance in reaction chambers—but bonding dissimilar materials remains technically challenging due to differences in thermal expansion coefficients and surface chemistries.
Manufacturing scalability presents another significant barrier. While academic prototyping has advanced rapidly, transitioning novel biocompatible materials to industrial-scale production faces hurdles in standardization, quality control, and cost-effectiveness. The disconnect between laboratory innovation and commercial implementation has slowed the adoption of next-generation materials in practical applications.
Regulatory considerations further complicate material selection, particularly for clinical applications. Materials must meet stringent safety standards and demonstrate consistent performance under various conditions, creating a high barrier to entry for novel biocompatible materials lacking extensive validation data.
Current Biocompatible Material Solutions for Microfluidics
01 Biocompatible materials for medical implants
Biocompatible materials are used in medical implants to minimize adverse reactions in the body. These materials are designed to integrate with biological tissues while maintaining their structural integrity. Various polymers, metals, and ceramics are engineered to have properties that reduce inflammation and promote tissue healing when implanted. The impact of these materials includes improved patient outcomes, reduced rejection rates, and longer implant lifespans.- Biocompatible materials for medical implants: Biocompatible materials are used in medical implants to minimize adverse reactions in the body. These materials include polymers, metals, and ceramics that are designed to be compatible with human tissue. The impact of these materials is significant in improving patient outcomes, reducing rejection rates, and extending the lifespan of implants. The biocompatibility ensures that the implant integrates well with surrounding tissues and performs its intended function without causing inflammation or immune responses.
- Drug delivery systems using biocompatible materials: Biocompatible materials are utilized in drug delivery systems to control the release of therapeutic agents in the body. These materials can be formulated as hydrogels, nanoparticles, or coatings that protect drugs and ensure their targeted delivery. The impact of these biocompatible delivery systems includes improved drug efficacy, reduced side effects, and enhanced patient compliance. These systems can be designed to respond to specific physiological conditions, allowing for precise control over when and where drugs are released.
- Tissue engineering applications of biocompatible materials: Biocompatible materials serve as scaffolds in tissue engineering to support cell growth and tissue regeneration. These materials provide structural support while allowing cells to proliferate and differentiate. The impact of these scaffolds is crucial in regenerative medicine, enabling the repair or replacement of damaged tissues and organs. The materials can be designed with specific mechanical properties, porosity, and degradation rates to match the requirements of different tissue types, facilitating the development of functional tissue constructs.
- Environmental impact of biocompatible materials: The environmental impact of biocompatible materials is an important consideration in their development and use. Biodegradable biocompatible materials can reduce waste and environmental pollution compared to traditional materials. These materials can be designed to break down into non-toxic components after their useful life. The sustainability aspects of biocompatible materials include their source (renewable vs. non-renewable), production methods, energy consumption during manufacturing, and end-of-life disposal options.
- Surface modifications of biocompatible materials: Surface modifications of biocompatible materials can enhance their performance and functionality. Techniques such as coating, grafting, and plasma treatment can alter surface properties like wettability, charge, and roughness. The impact of these modifications includes improved cell adhesion, reduced bacterial colonization, and enhanced integration with surrounding tissues. Surface modifications can also incorporate bioactive molecules that promote specific cellular responses, making the materials more effective for their intended applications.
02 Drug delivery systems using biocompatible materials
Biocompatible materials are formulated into drug delivery systems that can release therapeutic agents in a controlled manner. These systems utilize biodegradable polymers and hydrogels that break down safely in the body while delivering medication. The impact includes targeted drug delivery, reduced systemic side effects, and improved therapeutic efficacy. These materials can be engineered to respond to specific physiological conditions, allowing for smart drug release mechanisms.Expand Specific Solutions03 Tissue engineering applications of biocompatible materials
Biocompatible materials serve as scaffolds for tissue engineering, providing structural support for cell growth and tissue regeneration. These materials can be designed with specific porosity, surface chemistry, and mechanical properties to mimic natural tissues. The impact includes the ability to repair or replace damaged tissues and organs, reducing the need for donor organs and improving regenerative medicine outcomes. These scaffolds gradually degrade as new tissue forms, allowing for complete biological integration.Expand Specific Solutions04 Environmental impact of biocompatible materials
The environmental impact of biocompatible materials extends beyond medical applications. These materials are often biodegradable and can be derived from renewable resources, reducing reliance on petroleum-based products. The development of eco-friendly biocompatible materials has implications for sustainable manufacturing and waste reduction. However, the production processes and disposal methods for some biocompatible materials may still have environmental consequences that need to be addressed through improved manufacturing techniques and end-of-life management.Expand Specific Solutions05 Biocompatible materials in wound healing and surgical applications
Biocompatible materials are used in wound healing products and surgical applications to promote tissue repair and prevent complications. These materials include bioactive dressings, surgical meshes, and adhesives that interact positively with the wound environment. The impact includes faster healing times, reduced scarring, and decreased infection rates. Advanced biocompatible materials can also incorporate antimicrobial properties or growth factors to further enhance the healing process and improve surgical outcomes.Expand Specific Solutions
Leading Organizations in Biocompatible Microfluidics
The biocompatible materials innovation landscape in microfluidic chips is currently in a growth phase, with an estimated market size exceeding $10 billion and expanding at 15-20% annually. The technology demonstrates moderate maturity with significant advancements from key players across academia and industry. Leading research institutions like MIT, Tsinghua University, and Fudan University are pioneering fundamental innovations, while commercial entities including Philips, Samsung, and 10X Genomics are driving practical applications. The competitive landscape features a balanced ecosystem where academic-industrial partnerships accelerate development, particularly in healthcare applications where biocompatibility requirements are most stringent. Recent breakthroughs in polymer science and surface modification techniques are enabling new functionalities in microfluidic devices.
The Charles Stark Draper Laboratory, Inc.
Technical Solution: Draper Laboratory has developed advanced biocompatible material solutions for microfluidic chips focused on long-term implantable and wearable biomedical applications. Their proprietary approach combines silicon-based microfluidics with specialized biocompatible coatings that significantly reduce foreign body responses and protein fouling, enabling extended functionality in vivo. Draper has pioneered the integration of medical-grade thermoplastics with sophisticated surface modification techniques to create microfluidic platforms that maintain structural integrity while interfacing seamlessly with biological tissues. Their innovation in hemocompatible microfluidic materials stands out for blood-contacting applications, utilizing specialized phosphorylcholine-based surface modifications that mimic cell membrane components to minimize thrombosis and inflammatory responses. Draper has also developed novel composite materials that combine the precision manufacturing capabilities of traditional microfluidic substrates with the biocompatibility of advanced hydrogels, creating gradient interfaces that reduce mechanical mismatch between implanted devices and surrounding tissues.
Strengths: Exceptional expertise in materials for implantable and long-term biomedical applications; strong focus on hemocompatibility for blood-contacting microfluidic devices; extensive experience with regulatory pathways for medical materials. Weaknesses: Higher production costs associated with specialized biocompatible materials; longer development cycles due to rigorous biocompatibility testing requirements; some proprietary materials have limited availability outside of Draper collaborations.
10X Genomics, Inc.
Technical Solution: 10X Genomics has revolutionized microfluidic chip technology through their proprietary Gel Bead-In-EMulsion (GEM) technology, which utilizes advanced biocompatible hydrogel materials specifically engineered for single-cell analysis applications. Their microfluidic platforms incorporate specialized surface treatments that minimize biomolecule adsorption and cellular damage during processing, critical for maintaining sample integrity in genomic applications. The company has developed proprietary biocompatible surfactants that stabilize droplet interfaces while preserving cellular viability and molecular integrity, enabling high-throughput single-cell RNA sequencing with minimal bias. Their Chromium platform utilizes microfluidic chips with precisely engineered biocompatible channel geometries and surface chemistries that prevent sample degradation and cross-contamination. 10X Genomics has also pioneered the integration of specialized biocompatible optical materials within their microfluidic chips that enhance detection sensitivity while maintaining compatibility with biological samples, allowing for multiplexed analysis of cellular properties alongside genomic information.
Strengths: Industry-leading expertise in biocompatible materials optimized specifically for genomic applications; highly integrated systems approach combining materials science with microfluidic engineering; extensive commercial experience in scaling production of biocompatible microfluidic devices. Weaknesses: Proprietary materials and designs create platform dependency; higher cost compared to academic research platforms; primarily focused on genomic applications rather than broader microfluidic uses.
Key Patents and Research in Microfluidic Biocompatibility
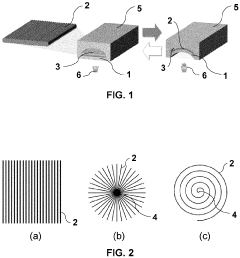
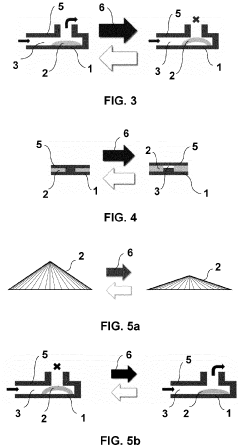
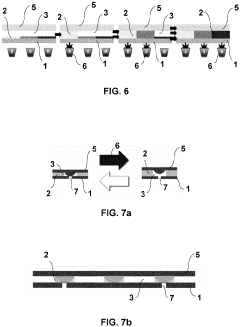
Microfluidic valve, method for its manufacture, and uses thereof
PatentActiveEP4170212A1
Innovation
- The use of liquid crystal networks or elastomers (LCN/LCE) with controlled polymer chain cross-linking, enabled by additive layer manufacturing, to create microfluidic valves that respond to stimuli such as light, temperature, and pH, allowing for precise regulation of fluid flow through 4D printing processes.
Microfluidic chip frame
PatentWO2005084808A1
Innovation
- A frame designed for microfluidic chips that includes a spring mechanism for easy handling and precise positioning, with integrated features such as patterned recesses for cost-effective production and a radio frequency chip for tracking, ensuring the chip's safety and accurate alignment with laboratory apparatuses.
Regulatory Framework for Biomedical Microfluidic Devices
The regulatory landscape governing biomedical microfluidic devices has evolved significantly in response to innovations in biocompatible materials. In the United States, the Food and Drug Administration (FDA) classifies microfluidic devices based on their intended use and risk profile, with most falling under Class II (moderate risk) or Class III (high risk) categories. The 510(k) premarket notification pathway is commonly utilized for devices demonstrating substantial equivalence to legally marketed devices, while novel technologies often require the more rigorous Premarket Approval (PMA) process.
European regulation has undergone substantial transformation with the implementation of the Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR), replacing the previous directives. These regulations impose stricter requirements for clinical evidence, post-market surveillance, and traceability, particularly emphasizing biocompatibility assessment for materials in direct contact with biological samples or tissues.
International standards play a crucial role in harmonizing regulatory approaches across different jurisdictions. ISO 10993 series specifically addresses the biological evaluation of medical devices, with ISO 10993-1 providing a framework for biocompatibility assessment. For microfluidic applications, ISO 18562 offers guidance on the evaluation of gas pathway components in healthcare applications.
The regulatory framework increasingly recognizes the unique challenges posed by novel biocompatible materials in microfluidic chips. Regulatory bodies now require comprehensive characterization of material properties, degradation profiles, and potential leachables or extractables that might interact with biological samples. The FDA's guidance on biocompatibility testing emphasizes the importance of considering the specific context of use when evaluating material safety.
Emerging regulatory considerations include the development of specialized frameworks for organ-on-chip technologies and point-of-care diagnostic platforms. These applications often incorporate advanced biocompatible materials with unique properties that traditional regulatory pathways may not adequately address. Regulatory science initiatives, such as the FDA's Medical Device Development Tools (MDDT) program, aim to qualify new methodologies for evaluating the safety and effectiveness of innovative materials and technologies.
Global harmonization efforts, including the International Medical Device Regulators Forum (IMDRF), are working to establish consistent approaches to regulating microfluidic devices incorporating novel biocompatible materials. These initiatives seek to reduce regulatory barriers while maintaining rigorous safety standards, ultimately accelerating the translation of innovative technologies from laboratory research to clinical application.
European regulation has undergone substantial transformation with the implementation of the Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR), replacing the previous directives. These regulations impose stricter requirements for clinical evidence, post-market surveillance, and traceability, particularly emphasizing biocompatibility assessment for materials in direct contact with biological samples or tissues.
International standards play a crucial role in harmonizing regulatory approaches across different jurisdictions. ISO 10993 series specifically addresses the biological evaluation of medical devices, with ISO 10993-1 providing a framework for biocompatibility assessment. For microfluidic applications, ISO 18562 offers guidance on the evaluation of gas pathway components in healthcare applications.
The regulatory framework increasingly recognizes the unique challenges posed by novel biocompatible materials in microfluidic chips. Regulatory bodies now require comprehensive characterization of material properties, degradation profiles, and potential leachables or extractables that might interact with biological samples. The FDA's guidance on biocompatibility testing emphasizes the importance of considering the specific context of use when evaluating material safety.
Emerging regulatory considerations include the development of specialized frameworks for organ-on-chip technologies and point-of-care diagnostic platforms. These applications often incorporate advanced biocompatible materials with unique properties that traditional regulatory pathways may not adequately address. Regulatory science initiatives, such as the FDA's Medical Device Development Tools (MDDT) program, aim to qualify new methodologies for evaluating the safety and effectiveness of innovative materials and technologies.
Global harmonization efforts, including the International Medical Device Regulators Forum (IMDRF), are working to establish consistent approaches to regulating microfluidic devices incorporating novel biocompatible materials. These initiatives seek to reduce regulatory barriers while maintaining rigorous safety standards, ultimately accelerating the translation of innovative technologies from laboratory research to clinical application.
Sustainability Aspects of Advanced Microfluidic Materials
The sustainability of microfluidic materials has become a critical consideration as these technologies gain wider adoption in healthcare, environmental monitoring, and industrial applications. Traditional microfluidic chips often rely on petroleum-based polymers like polydimethylsiloxane (PDMS) and polymethyl methacrylate (PMMA), which present significant end-of-life disposal challenges and contribute to plastic pollution.
Recent innovations in biocompatible materials are driving a paradigm shift toward more sustainable microfluidic platforms. Biodegradable polymers such as polylactic acid (PLA), polyhydroxyalkanoates (PHAs), and cellulose derivatives are emerging as environmentally responsible alternatives that maintain the necessary biocompatibility for medical and biological applications while reducing environmental impact.
Life cycle assessment (LCA) studies indicate that biobased microfluidic materials can reduce carbon footprints by 30-65% compared to conventional petroleum-based counterparts. This reduction stems from renewable feedstocks, less energy-intensive manufacturing processes, and biodegradable end-of-life scenarios that avoid persistent environmental contamination.
Water consumption represents another critical sustainability metric for microfluidic chip production. Novel fabrication techniques utilizing biocompatible materials have demonstrated potential water savings of up to 40% compared to traditional manufacturing methods. These improvements are particularly significant given the water-intensive nature of semiconductor and microfluidic device production.
Circular economy principles are increasingly being applied to microfluidic chip design, with materials selected not only for their performance but also for their recoverability and reusability. Some innovative approaches include designing chips with separable components made from different biocompatible materials that can be individually recycled or composted after use.
Regulatory frameworks are evolving to address the sustainability aspects of advanced materials in medical devices and diagnostic tools. The European Union's Medical Device Regulation (MDR) and similar initiatives worldwide are beginning to incorporate sustainability criteria alongside safety and performance requirements, creating market drivers for environmentally responsible biocompatible materials.
Research institutions and industry leaders are collaborating on green chemistry approaches to synthesize biocompatible materials with reduced toxicity and environmental persistence. These efforts include developing solvent-free processing methods, utilizing enzymatic polymerization, and incorporating naturally derived crosslinking agents that eliminate the need for potentially harmful chemical catalysts.
The economic viability of sustainable microfluidic materials continues to improve as scale increases and manufacturing processes mature. While currently commanding a premium of approximately 15-30% over conventional materials, this gap is narrowing as consumer demand for sustainable healthcare products grows and production efficiencies improve.
Recent innovations in biocompatible materials are driving a paradigm shift toward more sustainable microfluidic platforms. Biodegradable polymers such as polylactic acid (PLA), polyhydroxyalkanoates (PHAs), and cellulose derivatives are emerging as environmentally responsible alternatives that maintain the necessary biocompatibility for medical and biological applications while reducing environmental impact.
Life cycle assessment (LCA) studies indicate that biobased microfluidic materials can reduce carbon footprints by 30-65% compared to conventional petroleum-based counterparts. This reduction stems from renewable feedstocks, less energy-intensive manufacturing processes, and biodegradable end-of-life scenarios that avoid persistent environmental contamination.
Water consumption represents another critical sustainability metric for microfluidic chip production. Novel fabrication techniques utilizing biocompatible materials have demonstrated potential water savings of up to 40% compared to traditional manufacturing methods. These improvements are particularly significant given the water-intensive nature of semiconductor and microfluidic device production.
Circular economy principles are increasingly being applied to microfluidic chip design, with materials selected not only for their performance but also for their recoverability and reusability. Some innovative approaches include designing chips with separable components made from different biocompatible materials that can be individually recycled or composted after use.
Regulatory frameworks are evolving to address the sustainability aspects of advanced materials in medical devices and diagnostic tools. The European Union's Medical Device Regulation (MDR) and similar initiatives worldwide are beginning to incorporate sustainability criteria alongside safety and performance requirements, creating market drivers for environmentally responsible biocompatible materials.
Research institutions and industry leaders are collaborating on green chemistry approaches to synthesize biocompatible materials with reduced toxicity and environmental persistence. These efforts include developing solvent-free processing methods, utilizing enzymatic polymerization, and incorporating naturally derived crosslinking agents that eliminate the need for potentially harmful chemical catalysts.
The economic viability of sustainable microfluidic materials continues to improve as scale increases and manufacturing processes mature. While currently commanding a premium of approximately 15-30% over conventional materials, this gap is narrowing as consumer demand for sustainable healthcare products grows and production efficiencies improve.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!