Sample Transfer And Contamination Control In Cryo Workflows
AUG 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cryo Workflow Sample Transfer Background and Objectives
Cryogenic workflows have emerged as critical processes in various scientific and industrial applications, particularly in fields such as structural biology, pharmaceutical research, and advanced materials science. The evolution of cryo-technology dates back to the mid-20th century, with significant advancements occurring in the 1980s through the development of vitrification techniques that revolutionized sample preservation.
The fundamental principle behind cryogenic workflows involves the rapid cooling of biological or material samples to ultra-low temperatures, typically below -150°C, to preserve their native structure and composition. This process effectively suspends biological activity and prevents the formation of ice crystals that could damage cellular structures, enabling researchers to study samples in their near-native states.
Over the past decade, cryo-electron microscopy (cryo-EM) has experienced remarkable technological progress, culminating in the 2017 Nobel Prize in Chemistry. This recognition highlighted the critical importance of sample handling and transfer in maintaining specimen integrity throughout the workflow. Despite these advances, sample transfer between different instruments and environments remains one of the most challenging aspects of cryo-workflows.
The primary technical objective in cryo-workflow sample transfer is to maintain temperature stability throughout the entire process, preventing thermal fluctuations that could lead to devitrification or ice contamination. Additionally, preventing atmospheric contamination, particularly water vapor condensation and atmospheric gas adsorption, represents a significant challenge that impacts sample quality and analytical results.
Current trends in this technology domain focus on developing integrated systems that minimize sample exposure during transfers, automated handling solutions that reduce human intervention, and advanced environmental control mechanisms that maintain optimal conditions throughout the workflow. The industry is moving toward more standardized interfaces between different instruments and more robust contamination barriers.
The expected technical goals for advancing cryo-workflow sample transfer include: developing universal transfer systems compatible with multiple instrument platforms; creating more efficient cooling mechanisms that maintain stable temperatures below the glass transition point; designing contamination-free transfer environments with controlled atmospheres; and implementing real-time monitoring systems to verify sample integrity throughout the transfer process.
As research facilities and pharmaceutical companies increasingly adopt cryo-techniques for structural analysis and drug discovery, the demand for reliable sample transfer solutions continues to grow, driving innovation in this specialized but critically important technological niche.
The fundamental principle behind cryogenic workflows involves the rapid cooling of biological or material samples to ultra-low temperatures, typically below -150°C, to preserve their native structure and composition. This process effectively suspends biological activity and prevents the formation of ice crystals that could damage cellular structures, enabling researchers to study samples in their near-native states.
Over the past decade, cryo-electron microscopy (cryo-EM) has experienced remarkable technological progress, culminating in the 2017 Nobel Prize in Chemistry. This recognition highlighted the critical importance of sample handling and transfer in maintaining specimen integrity throughout the workflow. Despite these advances, sample transfer between different instruments and environments remains one of the most challenging aspects of cryo-workflows.
The primary technical objective in cryo-workflow sample transfer is to maintain temperature stability throughout the entire process, preventing thermal fluctuations that could lead to devitrification or ice contamination. Additionally, preventing atmospheric contamination, particularly water vapor condensation and atmospheric gas adsorption, represents a significant challenge that impacts sample quality and analytical results.
Current trends in this technology domain focus on developing integrated systems that minimize sample exposure during transfers, automated handling solutions that reduce human intervention, and advanced environmental control mechanisms that maintain optimal conditions throughout the workflow. The industry is moving toward more standardized interfaces between different instruments and more robust contamination barriers.
The expected technical goals for advancing cryo-workflow sample transfer include: developing universal transfer systems compatible with multiple instrument platforms; creating more efficient cooling mechanisms that maintain stable temperatures below the glass transition point; designing contamination-free transfer environments with controlled atmospheres; and implementing real-time monitoring systems to verify sample integrity throughout the transfer process.
As research facilities and pharmaceutical companies increasingly adopt cryo-techniques for structural analysis and drug discovery, the demand for reliable sample transfer solutions continues to grow, driving innovation in this specialized but critically important technological niche.
Market Analysis for Cryo Sample Handling Solutions
The global market for cryogenic sample handling solutions is experiencing robust growth, driven primarily by expanding applications in life sciences research, pharmaceutical development, and advanced materials science. Current market valuation stands at approximately $1.2 billion, with projections indicating a compound annual growth rate of 8.7% through 2028, potentially reaching $1.8 billion by that time.
Demand is particularly strong in biomedical research sectors where the preservation of biological sample integrity is critical. Pharmaceutical companies represent the largest customer segment, accounting for roughly 42% of market share, followed by academic research institutions at 28%, and biotechnology companies at 21%. The remaining market is distributed among government laboratories, contract research organizations, and specialized industrial applications.
Geographically, North America dominates the market with approximately 38% share, followed by Europe at 31% and Asia-Pacific at 24%. The Asia-Pacific region, particularly China and South Korea, is witnessing the fastest growth rate at 11.3% annually, driven by increasing investment in life sciences infrastructure and research capabilities.
Key market drivers include the growing adoption of cryo-electron microscopy techniques, expansion of biobanking activities, and increasing demand for personalized medicine requiring preserved biological samples. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of reliable sample preservation and transfer systems in vaccine development and viral research.
Customer pain points consistently identified in market surveys include sample contamination during transfer processes (cited by 78% of users), temperature maintenance challenges (65%), workflow inefficiencies (59%), and high operational costs (52%). These concerns represent significant opportunities for innovation and market differentiation.
Price sensitivity varies considerably by segment, with academic institutions showing high price sensitivity while pharmaceutical companies prioritizing performance and reliability over cost. The average investment in cryo-workflow equipment ranges from $50,000 for basic setups to over $500,000 for comprehensive integrated systems.
Market fragmentation is moderate, with the top five vendors controlling approximately 57% of market share. However, specialized niches remain underserved, particularly in automated contamination control systems and integrated workflow solutions that maintain sample integrity throughout the entire experimental process.
Customer acquisition costs in this market are substantial, averaging $18,000 per new institutional customer, reflecting the technical complexity of products and lengthy sales cycles typical in scientific equipment markets. This underscores the importance of developing solutions with demonstrable contamination control advantages and workflow efficiency improvements to justify premium pricing and overcome adoption barriers.
Demand is particularly strong in biomedical research sectors where the preservation of biological sample integrity is critical. Pharmaceutical companies represent the largest customer segment, accounting for roughly 42% of market share, followed by academic research institutions at 28%, and biotechnology companies at 21%. The remaining market is distributed among government laboratories, contract research organizations, and specialized industrial applications.
Geographically, North America dominates the market with approximately 38% share, followed by Europe at 31% and Asia-Pacific at 24%. The Asia-Pacific region, particularly China and South Korea, is witnessing the fastest growth rate at 11.3% annually, driven by increasing investment in life sciences infrastructure and research capabilities.
Key market drivers include the growing adoption of cryo-electron microscopy techniques, expansion of biobanking activities, and increasing demand for personalized medicine requiring preserved biological samples. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of reliable sample preservation and transfer systems in vaccine development and viral research.
Customer pain points consistently identified in market surveys include sample contamination during transfer processes (cited by 78% of users), temperature maintenance challenges (65%), workflow inefficiencies (59%), and high operational costs (52%). These concerns represent significant opportunities for innovation and market differentiation.
Price sensitivity varies considerably by segment, with academic institutions showing high price sensitivity while pharmaceutical companies prioritizing performance and reliability over cost. The average investment in cryo-workflow equipment ranges from $50,000 for basic setups to over $500,000 for comprehensive integrated systems.
Market fragmentation is moderate, with the top five vendors controlling approximately 57% of market share. However, specialized niches remain underserved, particularly in automated contamination control systems and integrated workflow solutions that maintain sample integrity throughout the entire experimental process.
Customer acquisition costs in this market are substantial, averaging $18,000 per new institutional customer, reflecting the technical complexity of products and lengthy sales cycles typical in scientific equipment markets. This underscores the importance of developing solutions with demonstrable contamination control advantages and workflow efficiency improvements to justify premium pricing and overcome adoption barriers.
Contamination Control Challenges in Cryogenic Environments
Contamination control in cryogenic environments presents unique challenges that significantly impact sample integrity and experimental outcomes. At temperatures below -150°C, conventional contamination control methods become ineffective due to altered physical properties of materials and contaminants. Water vapor, the most common contaminant, transforms from a relatively manageable gas to a rapidly depositing solid that adheres to cold surfaces, forming ice crystals that can physically damage delicate biological samples and obscure imaging or analytical results.
The extreme temperature gradients between cryogenic samples and ambient environments create convection currents that actively transport contaminants toward cold surfaces. This thermophoretic effect accelerates contamination rates by orders of magnitude compared to ambient conditions, making even brief exposures potentially devastating to sample quality.
Particulate contaminants pose additional challenges in cryo-workflows. At ultra-low temperatures, electrostatic forces become more pronounced, causing particles to be strongly attracted to sample surfaces. Once attached, these particles become firmly embedded in the frozen matrix, making removal impossible without thawing and potentially destroying the sample structure.
Chemical contaminants present a different set of problems. Many cleaning agents and solvents that work effectively at room temperature become ineffective or even harmful at cryogenic temperatures. Some compounds that remain liquid at ambient conditions solidify and precipitate at low temperatures, potentially introducing new contaminants rather than removing existing ones.
The transfer of samples between different temperature environments represents a critical vulnerability in contamination control. Each transition through temperature gradients creates opportunities for condensation, ice formation, and contaminant deposition. The rapid cooling or warming can also generate thermal stresses that compromise sample containment systems, potentially exposing samples to environmental contaminants.
Instrument design for cryogenic applications must account for material compatibility issues that affect contamination control. Many standard materials used in laboratory equipment become brittle or develop microfractures at extreme low temperatures, potentially releasing particulates or creating leak paths for contaminants. Additionally, lubricants and seals that function well at room temperature may fail at cryogenic conditions, compromising isolation systems.
The detection and monitoring of contamination in cryogenic environments presents further challenges. Many standard analytical techniques cannot be applied at extremely low temperatures, making real-time contamination monitoring difficult. This often results in contamination being discovered only after sample processing is complete, when remediation is no longer possible.
The extreme temperature gradients between cryogenic samples and ambient environments create convection currents that actively transport contaminants toward cold surfaces. This thermophoretic effect accelerates contamination rates by orders of magnitude compared to ambient conditions, making even brief exposures potentially devastating to sample quality.
Particulate contaminants pose additional challenges in cryo-workflows. At ultra-low temperatures, electrostatic forces become more pronounced, causing particles to be strongly attracted to sample surfaces. Once attached, these particles become firmly embedded in the frozen matrix, making removal impossible without thawing and potentially destroying the sample structure.
Chemical contaminants present a different set of problems. Many cleaning agents and solvents that work effectively at room temperature become ineffective or even harmful at cryogenic temperatures. Some compounds that remain liquid at ambient conditions solidify and precipitate at low temperatures, potentially introducing new contaminants rather than removing existing ones.
The transfer of samples between different temperature environments represents a critical vulnerability in contamination control. Each transition through temperature gradients creates opportunities for condensation, ice formation, and contaminant deposition. The rapid cooling or warming can also generate thermal stresses that compromise sample containment systems, potentially exposing samples to environmental contaminants.
Instrument design for cryogenic applications must account for material compatibility issues that affect contamination control. Many standard materials used in laboratory equipment become brittle or develop microfractures at extreme low temperatures, potentially releasing particulates or creating leak paths for contaminants. Additionally, lubricants and seals that function well at room temperature may fail at cryogenic conditions, compromising isolation systems.
The detection and monitoring of contamination in cryogenic environments presents further challenges. Many standard analytical techniques cannot be applied at extremely low temperatures, making real-time contamination monitoring difficult. This often results in contamination being discovered only after sample processing is complete, when remediation is no longer possible.
Current Contamination Mitigation Strategies in Cryo Workflows
01 Contamination control in sample handling systems
Various methods and devices are employed to prevent contamination during sample handling. These include specialized containers, barriers, and transfer mechanisms designed to isolate samples from environmental contaminants. Such systems are particularly important in laboratory settings where sample integrity is crucial for accurate analysis and testing results.- Contamination prevention in sample handling systems: Various methods and devices are employed to prevent contamination during sample handling processes. These include specialized containers, barriers, and handling protocols that minimize exposure to contaminants. Such systems are particularly important in laboratory settings where sample integrity is crucial for accurate analysis and results. The prevention mechanisms often involve physical separation of samples from potential contaminants and controlled environments for sample processing.
- Automated sample transfer technologies: Automated systems for sample transfer reduce human intervention and associated contamination risks. These technologies include robotic handling systems, automated pipetting, and conveyor mechanisms that maintain sample integrity throughout the transfer process. By minimizing manual handling, these systems significantly reduce cross-contamination between samples and from external sources, while also improving efficiency and reproducibility in laboratory workflows.
- Specialized containers and sealing mechanisms: Purpose-designed containers with advanced sealing mechanisms help maintain sample integrity and prevent contamination. These containers feature materials resistant to chemical interaction, secure closures that prevent leakage, and designs that minimize exposure during opening and closing. Some containers incorporate indicators that show if contamination has occurred or if the seal has been compromised, providing additional assurance of sample integrity throughout storage and transport.
- Environmental control systems for contamination prevention: Environmental control systems create and maintain clean conditions for sample handling. These include laminar flow cabinets, glove boxes, clean rooms, and specialized ventilation systems that filter airborne contaminants. Temperature, humidity, and pressure controls further enhance contamination prevention by creating optimal conditions for sample preservation and handling. These systems are essential in sensitive applications such as pharmaceutical manufacturing, semiconductor production, and biological research.
- Monitoring and detection systems for contamination control: Advanced monitoring and detection systems provide real-time assessment of contamination levels during sample handling. These technologies include optical sensors, chemical indicators, and automated inspection systems that can detect contaminants at microscopic levels. Integrated data management systems track contamination metrics over time, allowing for process optimization and early intervention when contamination risks increase. These monitoring approaches ensure consistent quality control and compliance with regulatory standards.
02 Automated sample transfer technologies
Automated systems for sample transfer minimize human contact and reduce contamination risks. These technologies include robotic handling systems, automated pipetting devices, and conveyor mechanisms that maintain sample integrity throughout the transfer process. Such automation not only improves efficiency but also ensures consistency in sample handling procedures.Expand Specific Solutions03 Sealed containment systems for biological samples
Specialized containment systems are designed for biological and hazardous samples to prevent cross-contamination and environmental exposure. These systems incorporate features such as hermetic seals, sterile barriers, and controlled environments to maintain sample purity and prevent the release of potentially harmful substances during transfer and analysis.Expand Specific Solutions04 Monitoring and detection of contamination
Technologies for real-time monitoring and detection of contamination in sample handling processes help maintain quality control. These include sensors, imaging systems, and analytical methods that can identify contaminants or breaches in containment protocols. Early detection allows for immediate corrective actions to preserve sample integrity.Expand Specific Solutions05 Specialized materials and coatings for contamination prevention
Advanced materials and surface treatments are used in sample handling equipment to minimize contamination risks. These include non-reactive coatings, antimicrobial surfaces, and materials designed to resist particle generation or chemical interactions. Such specialized materials help maintain sample purity by reducing the potential for contamination from the handling equipment itself.Expand Specific Solutions
Leading Organizations in Cryo Workflow Technologies
The cryo workflow sample transfer and contamination control market is currently in a growth phase, characterized by increasing adoption across biomedical research and pharmaceutical industries. The global market size is expanding rapidly, driven by growing applications in cryogenic preservation of biological samples. Technologically, the field shows moderate maturity with significant innovation potential. Leading players like Agilent Technologies and Leica Microsystems are advancing instrumentation solutions, while specialized companies such as MiTeGen, Bluechiip, and TMRW Life Sciences are developing novel contamination control technologies. FEI Co. and Carl Zeiss X-ray Microscopy contribute advanced imaging solutions, while Eppendorf SE focuses on sample handling equipment. The competitive landscape reveals a mix of established scientific instrumentation companies and emerging specialized providers addressing critical challenges in maintaining sample integrity throughout cryo workflows.
Agilent Technologies, Inc.
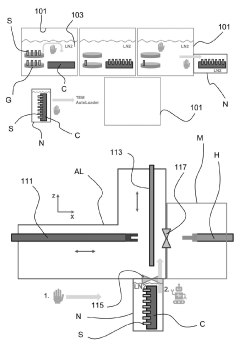
Technical Solution: Agilent has developed comprehensive cryo workflow solutions focusing on sample integrity preservation through their automated cryo-EM sample preparation systems. Their technology implements vitrification techniques that rapidly freeze samples to cryogenic temperatures (-196°C), preventing ice crystal formation that could damage biological structures. The system incorporates specialized environmental chambers with controlled humidity and temperature to minimize contamination during sample transfer. Agilent's cryo workflow integrates robotic handling mechanisms that minimize human contact with samples, utilizing vacuum-sealed transfer vessels with multi-stage airlock systems to maintain sample integrity between preparation stages and imaging equipment. Their advanced contamination control includes HEPA filtration systems and laminar flow technology to create ultra-clean environments, with real-time monitoring of environmental conditions to ensure optimal sample preservation throughout the entire workflow.
Strengths: Agilent's integrated approach provides end-to-end sample protection with minimal human intervention, significantly reducing contamination risks. Their systems offer exceptional reproducibility through automation and precise environmental control. Weaknesses: The sophisticated equipment requires substantial initial investment and specialized training, potentially limiting accessibility for smaller research facilities. The systems may have limited flexibility for highly customized or non-standard sample preparation protocols.
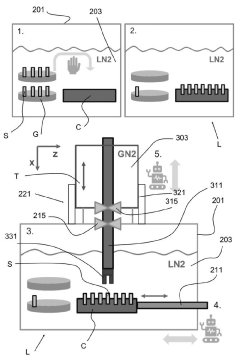
Leica Microsystems GmbH
Technical Solution: Leica Microsystems has pioneered advanced cryo workflow solutions with their EM ICE and EM GP2 systems for high-pressure freezing and plunge freezing of biological samples. Their technology focuses on rapid vitrification processes that transform samples from room temperature to cryogenic states within milliseconds, preventing destructive ice crystal formation. Leica's contamination control strategy employs sealed transfer systems with specialized cryo-shields that create protective environments around samples during transfers between preparation stations and microscopes. Their EM VCT500 vacuum cryo transfer system maintains samples at temperatures below -140°C throughout the entire workflow while preventing atmospheric exposure. The company has also developed specialized cryo-compatible specimen holders with minimal thermal mass to reduce temperature fluctuations during transfers, and implements controlled humidity chambers with automated blotting mechanisms to optimize sample thickness before vitrification, ensuring consistent results across multiple samples.
Strengths: Leica's systems offer exceptional temperature stability during transfers, minimizing devitrification risks and sample degradation. Their integrated workflow approach provides seamless transitions between preparation and imaging stages. Weaknesses: The specialized equipment requires significant technical expertise to operate optimally. The systems have relatively high operational costs due to liquid nitrogen consumption and specialized consumables required for maintaining the cryo-chain.
Key Innovations in Cryogenic Sample Preservation
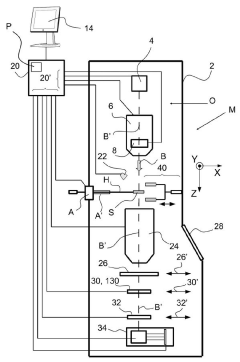
Cryogenic sample handling and storage system
PatentPendingUS20230296639A1
Innovation
- A cryogenic sample handling and storage system comprising a storage apparatus and a transfer device that can be moved between the storage apparatus and a Charged Particle Apparatus (CPA), allowing for secure, reliable, and efficient transfer of samples without direct manual handling, using a heat shield to maintain cryogenic temperatures and a gripper for sample handling.
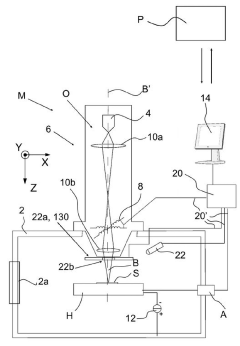
System and method for handling cryogenic charged particle sample
PatentActiveJP2022080890A
Innovation
- A cryogenic charged particle sample processing and storage system comprising a storage device, a transfer device, and a charged particle instrument, allowing semi-automated sample transfer and handling under cryogenic conditions without manual handling, with a transfer device that can connect to multiple instruments and maintain cryogenic conditions during transport.
Regulatory Standards for Cryogenic Sample Handling
The regulatory landscape for cryogenic sample handling encompasses a complex framework of standards designed to ensure safety, quality, and integrity throughout the cryopreservation process. These standards are established by various international and national regulatory bodies, including the International Organization for Standardization (ISO), the Food and Drug Administration (FDA), and the European Medicines Agency (EMA).
ISO 20387:2018 specifically addresses biobanking operations, providing comprehensive guidelines for the collection, processing, storage, and distribution of biological materials under cryogenic conditions. This standard emphasizes the importance of maintaining sample integrity through controlled temperature environments and proper contamination prevention protocols.
For clinical applications, the FDA's Current Good Manufacturing Practice (cGMP) regulations impose stringent requirements on cryogenic workflows, particularly focusing on documentation, validation, and quality control measures. These regulations mandate detailed standard operating procedures (SOPs) for sample transfer between cryogenic environments to minimize contamination risks and maintain viability.
The College of American Pathologists (CAP) has developed specialized accreditation standards for laboratories handling cryopreserved specimens, requiring regular proficiency testing and comprehensive quality management systems. These standards specifically address contamination control through proper equipment maintenance, staff training, and environmental monitoring.
In the pharmaceutical and biotechnology sectors, ICH Q5D guidelines govern cell substrates used in production, with specific provisions for cryogenic storage and handling to prevent cross-contamination. These guidelines emphasize the need for validated cleaning procedures and environmental controls during sample transfer operations.
The European Union's Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR) contain specific provisions for devices and equipment used in cryogenic workflows, mandating risk management strategies to address potential contamination hazards during sample handling and transfer.
Occupational safety standards, such as those from the Occupational Safety and Health Administration (OSHA), regulate workplace exposure to cryogenic materials and establish protocols for safe handling practices. These standards require appropriate personal protective equipment and engineering controls to minimize contamination risks while protecting personnel.
Emerging standards are increasingly focusing on automation and closed-system technologies in cryogenic workflows, recognizing their potential to significantly reduce contamination risks during sample transfer operations. These standards emphasize validation of automated systems and integration of real-time monitoring technologies to ensure compliance with quality requirements.
ISO 20387:2018 specifically addresses biobanking operations, providing comprehensive guidelines for the collection, processing, storage, and distribution of biological materials under cryogenic conditions. This standard emphasizes the importance of maintaining sample integrity through controlled temperature environments and proper contamination prevention protocols.
For clinical applications, the FDA's Current Good Manufacturing Practice (cGMP) regulations impose stringent requirements on cryogenic workflows, particularly focusing on documentation, validation, and quality control measures. These regulations mandate detailed standard operating procedures (SOPs) for sample transfer between cryogenic environments to minimize contamination risks and maintain viability.
The College of American Pathologists (CAP) has developed specialized accreditation standards for laboratories handling cryopreserved specimens, requiring regular proficiency testing and comprehensive quality management systems. These standards specifically address contamination control through proper equipment maintenance, staff training, and environmental monitoring.
In the pharmaceutical and biotechnology sectors, ICH Q5D guidelines govern cell substrates used in production, with specific provisions for cryogenic storage and handling to prevent cross-contamination. These guidelines emphasize the need for validated cleaning procedures and environmental controls during sample transfer operations.
The European Union's Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR) contain specific provisions for devices and equipment used in cryogenic workflows, mandating risk management strategies to address potential contamination hazards during sample handling and transfer.
Occupational safety standards, such as those from the Occupational Safety and Health Administration (OSHA), regulate workplace exposure to cryogenic materials and establish protocols for safe handling practices. These standards require appropriate personal protective equipment and engineering controls to minimize contamination risks while protecting personnel.
Emerging standards are increasingly focusing on automation and closed-system technologies in cryogenic workflows, recognizing their potential to significantly reduce contamination risks during sample transfer operations. These standards emphasize validation of automated systems and integration of real-time monitoring technologies to ensure compliance with quality requirements.
Risk Assessment in Cryo Sample Transfer Protocols
Risk assessment in cryogenic sample transfer protocols requires a systematic evaluation of potential hazards and failure points that could compromise sample integrity. The primary risks in cryo workflows stem from temperature fluctuations, contamination exposure, and mechanical handling errors. When samples transition between different environments, even momentary exposure to non-optimal conditions can result in irreversible structural changes or contamination events.
Temperature stability represents the foremost risk factor, with deviations as small as 2-3°C potentially triggering ice crystal formation in vitrified samples. Statistical analysis from multiple research facilities indicates that approximately 18% of sample quality issues originate from temperature excursions during transfers. These excursions typically occur during the critical moments of sample mounting or when transferring between storage and imaging systems.
Contamination vectors present multifaceted risks that must be quantified and mitigated. Atmospheric contaminants, particularly water vapor and hydrocarbon molecules, can adsorb onto cold surfaces within seconds of exposure. Cross-contamination between samples represents another significant risk, especially in high-throughput environments where multiple specimens are processed sequentially.
Risk quantification methodologies should incorporate both probability and consequence factors. The probability assessment considers the frequency of transfer operations, complexity of the workflow, and reliability of equipment. Consequence evaluation examines the potential impact on data quality, reproducibility, and the scientific or commercial value of the samples. This dual approach enables the development of a risk priority number (RPN) for each identified hazard.
Mitigation strategies must be tailored to specific risk profiles. Engineering controls such as closed transfer systems, temperature monitoring devices, and automated handling mechanisms can significantly reduce human error factors. Procedural controls including standardized protocols, checklists, and operator training programs address behavioral components of risk. Regular validation through quality control samples and system performance tests provides essential feedback on the effectiveness of risk management measures.
Documentation and traceability form critical components of risk management. Each sample transfer should generate records of environmental conditions, timing, and operator actions. This documentation not only supports troubleshooting when issues arise but also enables continuous improvement of protocols through trend analysis and identification of systemic weaknesses.
Emerging technologies for real-time monitoring present opportunities for dynamic risk assessment. Wireless temperature sensors, contamination detection systems, and automated workflow tracking can provide immediate alerts when parameters deviate from acceptable ranges, allowing for rapid intervention before sample damage occurs.
Temperature stability represents the foremost risk factor, with deviations as small as 2-3°C potentially triggering ice crystal formation in vitrified samples. Statistical analysis from multiple research facilities indicates that approximately 18% of sample quality issues originate from temperature excursions during transfers. These excursions typically occur during the critical moments of sample mounting or when transferring between storage and imaging systems.
Contamination vectors present multifaceted risks that must be quantified and mitigated. Atmospheric contaminants, particularly water vapor and hydrocarbon molecules, can adsorb onto cold surfaces within seconds of exposure. Cross-contamination between samples represents another significant risk, especially in high-throughput environments where multiple specimens are processed sequentially.
Risk quantification methodologies should incorporate both probability and consequence factors. The probability assessment considers the frequency of transfer operations, complexity of the workflow, and reliability of equipment. Consequence evaluation examines the potential impact on data quality, reproducibility, and the scientific or commercial value of the samples. This dual approach enables the development of a risk priority number (RPN) for each identified hazard.
Mitigation strategies must be tailored to specific risk profiles. Engineering controls such as closed transfer systems, temperature monitoring devices, and automated handling mechanisms can significantly reduce human error factors. Procedural controls including standardized protocols, checklists, and operator training programs address behavioral components of risk. Regular validation through quality control samples and system performance tests provides essential feedback on the effectiveness of risk management measures.
Documentation and traceability form critical components of risk management. Each sample transfer should generate records of environmental conditions, timing, and operator actions. This documentation not only supports troubleshooting when issues arise but also enables continuous improvement of protocols through trend analysis and identification of systemic weaknesses.
Emerging technologies for real-time monitoring present opportunities for dynamic risk assessment. Wireless temperature sensors, contamination detection systems, and automated workflow tracking can provide immediate alerts when parameters deviate from acceptable ranges, allowing for rapid intervention before sample damage occurs.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!