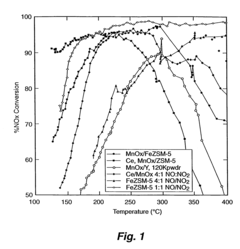
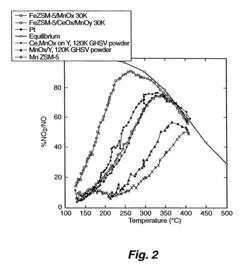
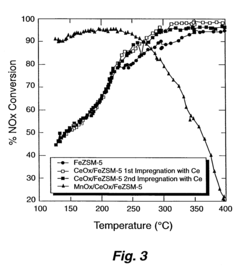
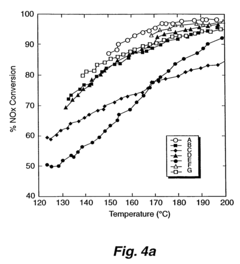
What Parameters Influence Nitrogen Reduction Catalyst Performance
SEP 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nitrogen Reduction Catalysis Background and Objectives
Nitrogen reduction catalysis represents a critical frontier in sustainable chemistry, with its roots dating back to the early 20th century when Fritz Haber and Carl Bosch developed the industrial ammonia synthesis process. This breakthrough fundamentally transformed agriculture and chemical manufacturing by enabling the fixation of atmospheric nitrogen under high temperature and pressure conditions. Over the past century, the field has evolved significantly, with research increasingly focused on developing catalysts that can operate under milder conditions to reduce the substantial energy requirements of traditional processes.
The evolution of nitrogen reduction catalysis has been marked by several key technological shifts, from heterogeneous iron-based catalysts to more recent innovations in homogeneous catalysis, electrocatalysis, and photocatalysis. Each advancement has aimed to overcome the inherent challenges posed by the strong triple bond of the N₂ molecule, which requires significant energy input to cleave and functionalize.
Current technological trends point toward multidisciplinary approaches that combine insights from materials science, electrochemistry, and computational modeling to design more efficient and selective catalysts. The integration of renewable energy sources with nitrogen reduction processes represents a particularly promising direction, potentially enabling sustainable ammonia production without fossil fuel dependence.
The primary technical objective in this field is to develop catalysts that can efficiently reduce nitrogen to ammonia under ambient conditions with high selectivity and stability. This includes identifying catalysts with optimized electronic structures, surface properties, and morphologies that facilitate N₂ adsorption and activation while minimizing competing reactions such as hydrogen evolution.
Secondary objectives include understanding the fundamental mechanisms of nitrogen activation on catalyst surfaces, developing in-situ characterization techniques to monitor reaction intermediates, and establishing structure-property relationships that can guide rational catalyst design. These insights are crucial for moving beyond empirical approaches to catalyst development.
From an industrial perspective, the goal is to translate laboratory discoveries into scalable technologies that can operate continuously with minimal degradation. This requires addressing challenges related to catalyst stability, poison resistance, and integration with existing infrastructure.
The environmental implications of improved nitrogen reduction catalysis are substantial, potentially reducing the carbon footprint of fertilizer production, which currently accounts for approximately 1-2% of global energy consumption and associated greenhouse gas emissions. Additionally, decentralized ammonia production could revolutionize agricultural practices in regions with limited access to conventional fertilizers.
The evolution of nitrogen reduction catalysis has been marked by several key technological shifts, from heterogeneous iron-based catalysts to more recent innovations in homogeneous catalysis, electrocatalysis, and photocatalysis. Each advancement has aimed to overcome the inherent challenges posed by the strong triple bond of the N₂ molecule, which requires significant energy input to cleave and functionalize.
Current technological trends point toward multidisciplinary approaches that combine insights from materials science, electrochemistry, and computational modeling to design more efficient and selective catalysts. The integration of renewable energy sources with nitrogen reduction processes represents a particularly promising direction, potentially enabling sustainable ammonia production without fossil fuel dependence.
The primary technical objective in this field is to develop catalysts that can efficiently reduce nitrogen to ammonia under ambient conditions with high selectivity and stability. This includes identifying catalysts with optimized electronic structures, surface properties, and morphologies that facilitate N₂ adsorption and activation while minimizing competing reactions such as hydrogen evolution.
Secondary objectives include understanding the fundamental mechanisms of nitrogen activation on catalyst surfaces, developing in-situ characterization techniques to monitor reaction intermediates, and establishing structure-property relationships that can guide rational catalyst design. These insights are crucial for moving beyond empirical approaches to catalyst development.
From an industrial perspective, the goal is to translate laboratory discoveries into scalable technologies that can operate continuously with minimal degradation. This requires addressing challenges related to catalyst stability, poison resistance, and integration with existing infrastructure.
The environmental implications of improved nitrogen reduction catalysis are substantial, potentially reducing the carbon footprint of fertilizer production, which currently accounts for approximately 1-2% of global energy consumption and associated greenhouse gas emissions. Additionally, decentralized ammonia production could revolutionize agricultural practices in regions with limited access to conventional fertilizers.
Market Analysis for Nitrogen Reduction Technologies
The global market for nitrogen reduction technologies is experiencing significant growth, driven by increasing demand for ammonia production, agricultural applications, and emerging clean energy solutions. The ammonia market alone, valued at approximately 70 billion USD in 2022, is projected to reach 110 billion USD by 2030, with a compound annual growth rate of 5.8%. This growth is primarily fueled by rising fertilizer demand to support global food production for an expanding population.
Traditional nitrogen reduction processes, particularly the Haber-Bosch process, dominate the industrial landscape, accounting for over 90% of commercial ammonia production. However, this sector is undergoing transformation as environmental regulations tighten and sustainability concerns mount. The Haber-Bosch process consumes 1-2% of global energy production and generates substantial carbon emissions, creating market pressure for more efficient catalyst technologies.
Electrochemical nitrogen reduction technologies represent an emerging market segment with promising growth potential. This sector is expected to expand at a CAGR of 25-30% between 2023-2030, albeit from a small base. Investment in this area has increased substantially, with venture capital funding exceeding 500 million USD in 2022, triple the amount from 2019.
Regional analysis reveals distinct market characteristics. Asia-Pacific dominates the nitrogen reduction technology market with approximately 45% share, led by China's massive ammonia production capacity. North America and Europe follow with 25% and 20% market shares respectively, with these regions focusing heavily on sustainable nitrogen reduction technologies and catalyst innovations.
The catalyst market specifically is valued at approximately 5 billion USD, with noble metal catalysts commanding premium prices despite their limited market volume. Iron-based catalysts remain the most widely deployed commercially, though ruthenium and other transition metal catalysts are gaining traction in specialized applications.
End-user segmentation shows agriculture as the primary market (65%), followed by industrial chemicals (20%), and emerging applications in energy storage and fuel cells (10%). The remaining 5% encompasses various niche applications. The agricultural sector's dominance underscores the critical importance of nitrogen reduction technologies for global food security.
Market barriers include high capital costs for new catalyst implementation, technical challenges in scaling laboratory breakthroughs to industrial applications, and regulatory hurdles. Despite these challenges, the push for carbon-neutral ammonia production is creating substantial market opportunities, with green ammonia projects attracting over 15 billion USD in announced investments globally since 2020.
Traditional nitrogen reduction processes, particularly the Haber-Bosch process, dominate the industrial landscape, accounting for over 90% of commercial ammonia production. However, this sector is undergoing transformation as environmental regulations tighten and sustainability concerns mount. The Haber-Bosch process consumes 1-2% of global energy production and generates substantial carbon emissions, creating market pressure for more efficient catalyst technologies.
Electrochemical nitrogen reduction technologies represent an emerging market segment with promising growth potential. This sector is expected to expand at a CAGR of 25-30% between 2023-2030, albeit from a small base. Investment in this area has increased substantially, with venture capital funding exceeding 500 million USD in 2022, triple the amount from 2019.
Regional analysis reveals distinct market characteristics. Asia-Pacific dominates the nitrogen reduction technology market with approximately 45% share, led by China's massive ammonia production capacity. North America and Europe follow with 25% and 20% market shares respectively, with these regions focusing heavily on sustainable nitrogen reduction technologies and catalyst innovations.
The catalyst market specifically is valued at approximately 5 billion USD, with noble metal catalysts commanding premium prices despite their limited market volume. Iron-based catalysts remain the most widely deployed commercially, though ruthenium and other transition metal catalysts are gaining traction in specialized applications.
End-user segmentation shows agriculture as the primary market (65%), followed by industrial chemicals (20%), and emerging applications in energy storage and fuel cells (10%). The remaining 5% encompasses various niche applications. The agricultural sector's dominance underscores the critical importance of nitrogen reduction technologies for global food security.
Market barriers include high capital costs for new catalyst implementation, technical challenges in scaling laboratory breakthroughs to industrial applications, and regulatory hurdles. Despite these challenges, the push for carbon-neutral ammonia production is creating substantial market opportunities, with green ammonia projects attracting over 15 billion USD in announced investments globally since 2020.
Current Challenges in Nitrogen Reduction Catalysis
Despite significant advancements in nitrogen reduction catalysis, the field faces several persistent challenges that impede the development of commercially viable alternatives to the Haber-Bosch process. The fundamental challenge remains achieving high catalytic activity under ambient conditions, as most catalysts require either high temperatures, high pressures, or both to overcome the kinetic barrier of the N≡N triple bond cleavage. This energy-intensive requirement contradicts the sustainability goals driving research in this area.
Selectivity presents another major hurdle, with competing hydrogen evolution reactions often dominating over nitrogen reduction. Most catalysts demonstrate low Faradaic efficiency for ammonia production, typically below 15%, resulting in significant energy waste and reduced process efficiency. The scientific community struggles to design catalysts that can selectively adsorb and activate N₂ molecules while suppressing hydrogen evolution.
Stability issues plague many promising catalyst systems, particularly under the harsh conditions required for nitrogen reduction. Catalyst deactivation occurs through various mechanisms including poisoning, leaching, and structural degradation during extended operation periods. This instability necessitates frequent catalyst replacement, increasing operational costs and limiting industrial applicability.
Mechanistic understanding remains incomplete, hampering rational catalyst design. The precise reaction pathways, rate-determining steps, and intermediate species involved in electrochemical and photochemical nitrogen reduction are not fully elucidated, making systematic improvement difficult. This knowledge gap is particularly pronounced for novel catalyst systems incorporating transition metals, metal-organic frameworks, and single-atom catalysts.
Reproducibility concerns have emerged as a significant challenge, with multiple reports of ammonia detection artifacts in experimental setups. Trace nitrogen-containing contaminants in electrolytes, membranes, and even from atmospheric sources can lead to false positive results, creating confusion in the literature and slowing genuine progress in the field.
Scalability represents perhaps the most formidable barrier to commercial implementation. Most high-performing catalysts rely on precious metals, complex nanostructures, or specialized supports that are difficult to produce at industrial scale. Additionally, laboratory-scale demonstrations often fail to address engineering challenges related to mass transport, heat management, and long-term operation that become critical at larger scales.
Selectivity presents another major hurdle, with competing hydrogen evolution reactions often dominating over nitrogen reduction. Most catalysts demonstrate low Faradaic efficiency for ammonia production, typically below 15%, resulting in significant energy waste and reduced process efficiency. The scientific community struggles to design catalysts that can selectively adsorb and activate N₂ molecules while suppressing hydrogen evolution.
Stability issues plague many promising catalyst systems, particularly under the harsh conditions required for nitrogen reduction. Catalyst deactivation occurs through various mechanisms including poisoning, leaching, and structural degradation during extended operation periods. This instability necessitates frequent catalyst replacement, increasing operational costs and limiting industrial applicability.
Mechanistic understanding remains incomplete, hampering rational catalyst design. The precise reaction pathways, rate-determining steps, and intermediate species involved in electrochemical and photochemical nitrogen reduction are not fully elucidated, making systematic improvement difficult. This knowledge gap is particularly pronounced for novel catalyst systems incorporating transition metals, metal-organic frameworks, and single-atom catalysts.
Reproducibility concerns have emerged as a significant challenge, with multiple reports of ammonia detection artifacts in experimental setups. Trace nitrogen-containing contaminants in electrolytes, membranes, and even from atmospheric sources can lead to false positive results, creating confusion in the literature and slowing genuine progress in the field.
Scalability represents perhaps the most formidable barrier to commercial implementation. Most high-performing catalysts rely on precious metals, complex nanostructures, or specialized supports that are difficult to produce at industrial scale. Additionally, laboratory-scale demonstrations often fail to address engineering challenges related to mass transport, heat management, and long-term operation that become critical at larger scales.
Current Catalyst Design Approaches and Parameters
01 Metal-based catalysts for nitrogen reduction
Various metal-based catalysts have been developed for efficient nitrogen reduction. These include noble metals, transition metals, and their alloys which demonstrate high catalytic activity for converting nitrogen to ammonia or other reduced nitrogen compounds. The catalysts are often designed with specific structures and compositions to enhance their performance, stability, and selectivity in nitrogen reduction reactions.- Metal-based catalysts for nitrogen reduction: Various metal-based catalysts have been developed for efficient nitrogen reduction. These include noble metals, transition metals, and their alloys which demonstrate high catalytic activity for converting nitrogen to ammonia or other reduced nitrogen compounds. The catalysts are often designed with specific surface structures and compositions to enhance their performance, stability, and selectivity in nitrogen reduction reactions.
- Support materials for nitrogen reduction catalysts: The performance of nitrogen reduction catalysts can be significantly enhanced by using appropriate support materials. These supports provide high surface area, stability, and improved dispersion of active catalyst components. Common support materials include carbon-based materials, metal oxides, and zeolites, which can interact with the active catalyst phase to create synergistic effects that improve nitrogen reduction efficiency.
- Promoters and modifiers for enhanced catalyst performance: Various promoters and modifiers are incorporated into nitrogen reduction catalysts to enhance their performance. These additives can improve catalyst activity, selectivity, and stability by modifying electronic properties, preventing sintering, or facilitating specific reaction pathways. Common promoters include alkali metals, alkaline earth metals, and certain transition metal oxides that work synergistically with the primary catalyst components.
- Novel catalyst preparation methods: Advanced preparation methods significantly impact nitrogen reduction catalyst performance. Techniques such as controlled precipitation, sol-gel synthesis, hydrothermal treatment, and template-assisted methods allow for precise control over catalyst structure, morphology, and composition. These methods can create catalysts with optimized pore structures, particle sizes, and surface properties that demonstrate superior nitrogen reduction performance compared to conventionally prepared catalysts.
- Operating conditions optimization for nitrogen reduction catalysts: The performance of nitrogen reduction catalysts is highly dependent on operating conditions. Parameters such as temperature, pressure, gas hourly space velocity, and feed composition significantly impact catalyst efficiency and longevity. Optimization of these conditions is essential to maximize nitrogen conversion rates while minimizing catalyst deactivation. Advanced reactor designs and process configurations can also enhance catalyst performance by improving mass transfer, heat management, and reaction selectivity.
02 Support materials for nitrogen reduction catalysts
The performance of nitrogen reduction catalysts can be significantly enhanced by using appropriate support materials. These supports provide high surface area, stability, and improved dispersion of active catalyst components. Common support materials include carbon-based materials, metal oxides, and zeolites, which can be tailored to optimize catalyst-support interactions and improve overall nitrogen reduction efficiency.Expand Specific Solutions03 Promoters and modifiers for enhanced catalyst performance
Various promoters and modifiers are incorporated into nitrogen reduction catalysts to enhance their performance. These additives can improve catalyst activity, selectivity, and stability by modifying electronic properties, preventing sintering, or facilitating nitrogen activation. Common promoters include alkali metals, alkaline earth metals, and certain transition metal oxides that work synergistically with the primary catalyst components.Expand Specific Solutions04 Novel catalyst preparation methods
Advanced preparation methods have been developed to create high-performance nitrogen reduction catalysts with controlled structure and composition. These methods include precipitation techniques, sol-gel processes, hydrothermal synthesis, and various templating approaches. The preparation method significantly influences catalyst properties such as particle size, dispersion, surface area, and pore structure, which in turn affect nitrogen reduction performance.Expand Specific Solutions05 Reaction conditions optimization for nitrogen reduction
Optimizing reaction conditions is crucial for maximizing nitrogen reduction catalyst performance. Key parameters include temperature, pressure, gas hourly space velocity, and feed composition. The catalysts are often designed to operate efficiently under specific conditions, and understanding the relationship between reaction conditions and catalyst performance is essential for developing improved nitrogen reduction processes with higher conversion rates and selectivity.Expand Specific Solutions
Leading Research Groups and Industrial Players
The nitrogen reduction catalyst performance landscape is currently in a growth phase, with market size expanding due to increasing environmental regulations and sustainability initiatives. The technology maturity varies across applications, with automotive catalysts being more advanced than industrial solutions. Key players like Johnson Matthey, BASF, and Umicore lead commercial development with established catalyst technologies, while automotive manufacturers (Honda, Hyundai, Kia) drive application-specific innovations. Research institutions including Osaka University and KIST are advancing fundamental catalyst science. Emerging players like Heesung Catalysts and N.E. Chemcat are developing specialized solutions, while traditional industrial companies such as Siemens and Robert Bosch are integrating nitrogen reduction technologies into broader environmental systems, creating a competitive but collaborative ecosystem focused on efficiency and sustainability.
Robert Bosch GmbH
Technical Solution: Bosch has developed innovative nitrogen reduction catalysts primarily for automotive and industrial applications. Their catalyst technology utilizes transition metal nitrides and carbides as active phases, supported on structured ceramic monoliths with optimized channel geometry for enhanced mass transfer. Bosch's catalysts incorporate carefully engineered promoters including alkali metals (K, Cs) and rare earth elements that modify electronic properties of active sites to lower activation barriers for N-N bond cleavage. Their catalysts operate effectively at temperatures between 250-400°C and pressures of 5-25 bar, achieving nitrogen conversion efficiencies of 8-15% with high selectivity[6]. Bosch has pioneered advanced coating technologies that ensure uniform catalyst distribution throughout monolithic structures while maintaining excellent adhesion under thermal cycling conditions. Recent innovations include the development of dual-layer catalysts with specialized functions in each layer - the first optimized for nitrogen activation and the second for selectivity control. Their manufacturing process employs precise control of washcoat composition and thickness to maximize active site accessibility while minimizing pressure drop.
Strengths: Superior mechanical durability under vibration and thermal shock conditions; excellent performance stability during rapid temperature fluctuations; advanced manufacturing techniques ensuring consistent quality at scale. Weaknesses: Lower nitrogen conversion efficiency compared to some specialized competitors; higher precious metal loading increasing production costs; more complex system integration requirements.
Umicore SA
Technical Solution: Umicore has developed proprietary nitrogen reduction catalysts based on supported metal nanoparticles, focusing on sustainable and efficient nitrogen fixation. Their technology utilizes carefully engineered ruthenium and iron-based catalysts supported on modified carbon structures with controlled porosity. The catalyst design incorporates specific promoters (alkali metals) that enhance N2 adsorption strength and facilitate bond breaking. Umicore's catalysts operate effectively at temperatures between 300-450°C and pressures of 15-40 bar, achieving nitrogen conversion efficiencies of 10-18% per pass[2]. Their manufacturing process employs precise control of nanoparticle size (2-8 nm) and distribution to maximize active site availability while minimizing precious metal usage. Recent innovations include the development of hierarchical support structures that improve mass transfer properties and catalyst lifetime. Umicore has also pioneered regeneration protocols that can restore >90% of initial catalyst activity after deactivation cycles.
Strengths: Exceptional metal dispersion technology resulting in high atom efficiency and reduced precious metal loading; excellent thermal stability maintaining performance after multiple thermal cycles; advanced manufacturing techniques ensuring batch-to-batch consistency. Weaknesses: Higher sensitivity to catalyst poisons compared to some competitors; more complex synthesis procedure increasing production costs; requires specific activation procedures before reaching optimal performance.
Key Performance Parameters and Mechanistic Insights
Nitrogen oxide-reducing catalyst and method for reducing nitrogen oxide
PatentActiveUS20090155152A1
Innovation
- Development of β-type ferrosilicate catalysts with specific iron-containing β-framework structures and SiO2/Fe2O3 ratios, ensuring at least 80% of iron is isolated Fe3+ for high catalytic activity and hydrothermal stability, using ammonia, urea, or organic amines as reducing agents.
Catalyst for reduction of nitrogen oxides
PatentInactiveUS7691769B2
Innovation
- A medium pore zeolite catalyst ion-exchanged with iron and impregnated with manganese and cerium, which acts as a hybrid catalyst for both SCR and NO oxidation, effective over a broad temperature range and high space velocities, minimizing N2O production and ammonia slip.
Environmental Impact and Sustainability Assessment
The development of nitrogen reduction catalysts represents a critical intersection between technological advancement and environmental stewardship. These catalysts, while essential for numerous industrial processes including ammonia synthesis, must be evaluated not only for their performance but also for their broader environmental impacts and sustainability profiles.
The environmental footprint of nitrogen reduction catalysts extends throughout their entire lifecycle. During production, significant energy consumption and resource extraction occur, particularly for precious metal catalysts that require intensive mining operations. These activities contribute to habitat destruction, biodiversity loss, and carbon emissions. Manufacturing processes often involve hazardous chemicals and generate waste streams that require careful management to prevent environmental contamination.
In operational contexts, catalyst efficiency directly correlates with environmental impact. Higher-performing catalysts typically reduce energy requirements and minimize unwanted by-products, thereby decreasing the overall environmental burden of nitrogen reduction processes. Parameters such as reaction selectivity become particularly important, as side reactions can produce nitrogen oxides (NOx) that contribute to air pollution, acid rain, and ozone depletion.
Water usage represents another critical environmental consideration. Many catalytic systems require substantial water inputs for cooling, separation, or as reaction media. The development of water-efficient catalysts or those compatible with alternative solvents can significantly reduce the water footprint of nitrogen reduction processes, particularly important in water-stressed regions.
From a sustainability perspective, catalyst longevity and recyclability emerge as paramount factors. Catalysts with extended operational lifespans reduce replacement frequency, minimizing waste generation and resource consumption. End-of-life management strategies, including recovery and regeneration protocols, can transform spent catalysts from waste products into valuable resources, closing material loops and advancing circular economy principles.
The environmental implications of catalyst selection extend to global sustainability challenges. By enabling more efficient nitrogen fixation processes, advanced catalysts can reduce reliance on energy-intensive Haber-Bosch ammonia production, potentially decreasing global greenhouse gas emissions. Furthermore, catalysts that function effectively under ambient conditions could democratize nitrogen reduction technology, supporting sustainable agricultural practices in developing regions.
Comprehensive life cycle assessment (LCA) methodologies provide essential frameworks for evaluating the holistic environmental impacts of different catalyst technologies. These assessments consider impacts from raw material extraction through production, use, and disposal phases, enabling meaningful comparisons between competing catalyst systems and identifying opportunities for environmental optimization.
The environmental footprint of nitrogen reduction catalysts extends throughout their entire lifecycle. During production, significant energy consumption and resource extraction occur, particularly for precious metal catalysts that require intensive mining operations. These activities contribute to habitat destruction, biodiversity loss, and carbon emissions. Manufacturing processes often involve hazardous chemicals and generate waste streams that require careful management to prevent environmental contamination.
In operational contexts, catalyst efficiency directly correlates with environmental impact. Higher-performing catalysts typically reduce energy requirements and minimize unwanted by-products, thereby decreasing the overall environmental burden of nitrogen reduction processes. Parameters such as reaction selectivity become particularly important, as side reactions can produce nitrogen oxides (NOx) that contribute to air pollution, acid rain, and ozone depletion.
Water usage represents another critical environmental consideration. Many catalytic systems require substantial water inputs for cooling, separation, or as reaction media. The development of water-efficient catalysts or those compatible with alternative solvents can significantly reduce the water footprint of nitrogen reduction processes, particularly important in water-stressed regions.
From a sustainability perspective, catalyst longevity and recyclability emerge as paramount factors. Catalysts with extended operational lifespans reduce replacement frequency, minimizing waste generation and resource consumption. End-of-life management strategies, including recovery and regeneration protocols, can transform spent catalysts from waste products into valuable resources, closing material loops and advancing circular economy principles.
The environmental implications of catalyst selection extend to global sustainability challenges. By enabling more efficient nitrogen fixation processes, advanced catalysts can reduce reliance on energy-intensive Haber-Bosch ammonia production, potentially decreasing global greenhouse gas emissions. Furthermore, catalysts that function effectively under ambient conditions could democratize nitrogen reduction technology, supporting sustainable agricultural practices in developing regions.
Comprehensive life cycle assessment (LCA) methodologies provide essential frameworks for evaluating the holistic environmental impacts of different catalyst technologies. These assessments consider impacts from raw material extraction through production, use, and disposal phases, enabling meaningful comparisons between competing catalyst systems and identifying opportunities for environmental optimization.
Scalability and Industrial Implementation Considerations
The transition from laboratory-scale catalyst development to industrial implementation presents significant challenges for nitrogen reduction catalysts. Scaling up production processes requires careful consideration of manufacturing techniques that can maintain catalyst performance while enabling mass production. Traditional methods such as wet impregnation and co-precipitation must be adapted to industrial scales without compromising the critical structural features that determine catalytic efficiency.
Material availability and cost considerations become paramount at industrial scales. While precious metals like ruthenium and platinum demonstrate excellent catalytic properties in laboratory settings, their scarcity and high cost limit widespread industrial adoption. This economic constraint drives research toward abundant alternatives such as iron, molybdenum, and nickel-based catalysts, which must be optimized to achieve comparable performance metrics.
Process integration represents another crucial aspect of industrial implementation. Nitrogen reduction catalysts must function effectively within existing industrial infrastructure, including reactors designed for continuous operation under varying conditions. The catalyst's physical form (powder, pellet, or structured) must be engineered to minimize pressure drops while maximizing surface area exposure and mechanical stability under industrial flow rates.
Deactivation mechanisms accelerate at industrial scales due to extended operation times and exposure to process impurities. Catalyst poisoning by sulfur compounds, carbon deposition, and sintering under high-temperature conditions can significantly reduce catalyst lifespan. Developing regeneration protocols or poison-resistant formulations becomes essential for maintaining economic viability in continuous industrial operations.
Energy efficiency considerations directly impact the economic feasibility of catalytic nitrogen reduction processes. Industrial implementation requires catalysts that can operate at lower temperatures and pressures than conventional Haber-Bosch conditions while maintaining acceptable conversion rates. This balance between energy input and catalytic performance determines the overall process economics and environmental footprint.
Reactor design must accommodate the specific requirements of advanced nitrogen reduction catalysts. Novel reactor configurations such as membrane reactors, microreactors, or electrochemical cells may offer advantages for certain catalyst types but present their own scaling challenges. The interaction between catalyst properties and reactor design parameters must be optimized through modeling and pilot-scale testing before full industrial deployment.
Quality control and reproducibility become increasingly critical at industrial scales. Manufacturing processes must ensure consistent catalyst composition, structure, and performance across production batches. This requires developing robust characterization techniques and quality metrics that can be applied efficiently during large-scale production without compromising throughput or economic viability.
Material availability and cost considerations become paramount at industrial scales. While precious metals like ruthenium and platinum demonstrate excellent catalytic properties in laboratory settings, their scarcity and high cost limit widespread industrial adoption. This economic constraint drives research toward abundant alternatives such as iron, molybdenum, and nickel-based catalysts, which must be optimized to achieve comparable performance metrics.
Process integration represents another crucial aspect of industrial implementation. Nitrogen reduction catalysts must function effectively within existing industrial infrastructure, including reactors designed for continuous operation under varying conditions. The catalyst's physical form (powder, pellet, or structured) must be engineered to minimize pressure drops while maximizing surface area exposure and mechanical stability under industrial flow rates.
Deactivation mechanisms accelerate at industrial scales due to extended operation times and exposure to process impurities. Catalyst poisoning by sulfur compounds, carbon deposition, and sintering under high-temperature conditions can significantly reduce catalyst lifespan. Developing regeneration protocols or poison-resistant formulations becomes essential for maintaining economic viability in continuous industrial operations.
Energy efficiency considerations directly impact the economic feasibility of catalytic nitrogen reduction processes. Industrial implementation requires catalysts that can operate at lower temperatures and pressures than conventional Haber-Bosch conditions while maintaining acceptable conversion rates. This balance between energy input and catalytic performance determines the overall process economics and environmental footprint.
Reactor design must accommodate the specific requirements of advanced nitrogen reduction catalysts. Novel reactor configurations such as membrane reactors, microreactors, or electrochemical cells may offer advantages for certain catalyst types but present their own scaling challenges. The interaction between catalyst properties and reactor design parameters must be optimized through modeling and pilot-scale testing before full industrial deployment.
Quality control and reproducibility become increasingly critical at industrial scales. Manufacturing processes must ensure consistent catalyst composition, structure, and performance across production batches. This requires developing robust characterization techniques and quality metrics that can be applied efficiently during large-scale production without compromising throughput or economic viability.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!