Automating NMR Adjustment Parameters for Reproducibility
SEP 22, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
NMR Automation Background and Objectives
Nuclear Magnetic Resonance (NMR) spectroscopy has evolved significantly since its discovery in the 1940s, becoming an indispensable analytical tool in chemistry, biochemistry, and materials science. The technique leverages the magnetic properties of atomic nuclei to provide detailed structural information about molecules. Over the decades, NMR technology has progressed from simple one-dimensional experiments to sophisticated multi-dimensional analyses capable of elucidating complex molecular structures.
Recent technological advancements have focused on increasing sensitivity, resolution, and automation capabilities. The development of higher field magnets, cryogenic probes, and digital signal processing has dramatically enhanced the quality of NMR data. However, despite these improvements, a persistent challenge remains in achieving consistent and reproducible results across different instruments and laboratories.
The reproducibility crisis in scientific research has highlighted the critical importance of standardized methodologies in analytical techniques. For NMR spectroscopy, variations in parameter adjustments—including shimming, pulse calibration, receiver gain, and temperature control—can significantly impact spectral quality and data interpretation. These adjustments often rely heavily on operator expertise, introducing subjective elements that compromise reproducibility.
Automation of NMR parameter adjustment represents a strategic response to these challenges. The primary objective is to develop intelligent systems capable of optimizing acquisition parameters without human intervention, ensuring consistent spectral quality regardless of operator experience or instrument variations. This automation aims to standardize NMR data collection protocols, thereby enhancing reproducibility across different research environments.
Beyond reproducibility, automation offers additional benefits including increased throughput, reduced operator training requirements, and more efficient utilization of expensive NMR instrumentation. The integration of machine learning algorithms has opened new possibilities for adaptive parameter optimization that can respond to sample-specific characteristics.
The technical goals of NMR automation include developing robust algorithms for automated shimming that can achieve optimal magnetic field homogeneity, creating self-calibrating pulse sequences that adjust to sample conditions, implementing intelligent gain control systems, and establishing standardized quality assessment metrics for automated data acquisition.
As research increasingly moves toward high-throughput methodologies and multi-user facilities, the demand for automated, reproducible NMR analysis continues to grow. The evolution of this technology is expected to fundamentally transform how NMR experiments are conducted, transitioning from an art requiring significant expertise to a standardized science accessible to broader research communities.
Recent technological advancements have focused on increasing sensitivity, resolution, and automation capabilities. The development of higher field magnets, cryogenic probes, and digital signal processing has dramatically enhanced the quality of NMR data. However, despite these improvements, a persistent challenge remains in achieving consistent and reproducible results across different instruments and laboratories.
The reproducibility crisis in scientific research has highlighted the critical importance of standardized methodologies in analytical techniques. For NMR spectroscopy, variations in parameter adjustments—including shimming, pulse calibration, receiver gain, and temperature control—can significantly impact spectral quality and data interpretation. These adjustments often rely heavily on operator expertise, introducing subjective elements that compromise reproducibility.
Automation of NMR parameter adjustment represents a strategic response to these challenges. The primary objective is to develop intelligent systems capable of optimizing acquisition parameters without human intervention, ensuring consistent spectral quality regardless of operator experience or instrument variations. This automation aims to standardize NMR data collection protocols, thereby enhancing reproducibility across different research environments.
Beyond reproducibility, automation offers additional benefits including increased throughput, reduced operator training requirements, and more efficient utilization of expensive NMR instrumentation. The integration of machine learning algorithms has opened new possibilities for adaptive parameter optimization that can respond to sample-specific characteristics.
The technical goals of NMR automation include developing robust algorithms for automated shimming that can achieve optimal magnetic field homogeneity, creating self-calibrating pulse sequences that adjust to sample conditions, implementing intelligent gain control systems, and establishing standardized quality assessment metrics for automated data acquisition.
As research increasingly moves toward high-throughput methodologies and multi-user facilities, the demand for automated, reproducible NMR analysis continues to grow. The evolution of this technology is expected to fundamentally transform how NMR experiments are conducted, transitioning from an art requiring significant expertise to a standardized science accessible to broader research communities.
Market Analysis for Automated NMR Systems
The global market for automated Nuclear Magnetic Resonance (NMR) systems is experiencing significant growth, driven by increasing demand for more efficient and reproducible analytical techniques in pharmaceutical research, chemical analysis, and academic research. The market size for NMR spectroscopy equipment was valued at approximately 1.3 billion USD in 2022, with automated systems representing a rapidly growing segment projected to reach 1.8 billion USD by 2027, reflecting a compound annual growth rate of 6.8%.
Pharmaceutical and biotechnology sectors currently dominate the market demand, accounting for nearly 45% of the total market share. These industries require high-throughput screening capabilities and reproducible results for drug discovery and development processes. Academic and research institutions constitute the second-largest market segment at 30%, followed by chemical and material science industries at 15%.
Geographically, North America leads the market with approximately 40% share, attributed to substantial R&D investments and the presence of major pharmaceutical companies. Europe follows closely at 35%, with Asia-Pacific emerging as the fastest-growing region at a projected CAGR of 8.2%, primarily driven by expanding research infrastructure in China, Japan, and India.
The demand for automated NMR parameter adjustment systems is particularly strong due to several market drivers. First, the increasing complexity of molecular structures being analyzed requires more sophisticated and precise measurement techniques. Second, there is growing pressure to reduce the expertise barrier for NMR operation, allowing non-specialist researchers to obtain reliable results. Third, reproducibility challenges in scientific research have highlighted the need for standardized analytical methods.
Customer surveys indicate that key purchasing factors include reproducibility of results (cited by 78% of respondents), reduction in analysis time (65%), and decreased dependence on expert operators (62%). The return on investment for automated NMR systems is typically realized within 2-3 years through increased throughput and reduced labor costs.
Market challenges include the high initial investment cost for automated NMR systems, which can range from 500,000 to 2 million USD depending on specifications. Additionally, integration with existing laboratory information management systems presents technical hurdles that vendors must address to increase adoption rates.
The subscription-based software model for NMR parameter automation is gaining traction, with an estimated market value of 150 million USD in 2022, expected to grow at 12% annually as organizations seek more flexible licensing options and regular updates to parameter optimization algorithms.
Pharmaceutical and biotechnology sectors currently dominate the market demand, accounting for nearly 45% of the total market share. These industries require high-throughput screening capabilities and reproducible results for drug discovery and development processes. Academic and research institutions constitute the second-largest market segment at 30%, followed by chemical and material science industries at 15%.
Geographically, North America leads the market with approximately 40% share, attributed to substantial R&D investments and the presence of major pharmaceutical companies. Europe follows closely at 35%, with Asia-Pacific emerging as the fastest-growing region at a projected CAGR of 8.2%, primarily driven by expanding research infrastructure in China, Japan, and India.
The demand for automated NMR parameter adjustment systems is particularly strong due to several market drivers. First, the increasing complexity of molecular structures being analyzed requires more sophisticated and precise measurement techniques. Second, there is growing pressure to reduce the expertise barrier for NMR operation, allowing non-specialist researchers to obtain reliable results. Third, reproducibility challenges in scientific research have highlighted the need for standardized analytical methods.
Customer surveys indicate that key purchasing factors include reproducibility of results (cited by 78% of respondents), reduction in analysis time (65%), and decreased dependence on expert operators (62%). The return on investment for automated NMR systems is typically realized within 2-3 years through increased throughput and reduced labor costs.
Market challenges include the high initial investment cost for automated NMR systems, which can range from 500,000 to 2 million USD depending on specifications. Additionally, integration with existing laboratory information management systems presents technical hurdles that vendors must address to increase adoption rates.
The subscription-based software model for NMR parameter automation is gaining traction, with an estimated market value of 150 million USD in 2022, expected to grow at 12% annually as organizations seek more flexible licensing options and regular updates to parameter optimization algorithms.
Current NMR Parameter Adjustment Challenges
Nuclear Magnetic Resonance (NMR) spectroscopy stands as a cornerstone analytical technique in chemistry, biochemistry, and materials science. However, the reproducibility of NMR experiments remains a significant challenge due to the complexity of parameter adjustment processes. Current manual parameter adjustment methods are highly dependent on operator expertise, leading to inconsistencies across different laboratories and even within the same facility.
The acquisition of high-quality NMR spectra requires precise adjustment of numerous parameters including shimming, pulse calibration, receiver gain, and relaxation delays. These adjustments traditionally rely on iterative processes performed by skilled spectroscopists who make decisions based on experience and visual assessment of spectral quality. This human-dependent approach introduces variability that undermines the reproducibility essential for scientific rigor.
Shimming, the process of optimizing magnetic field homogeneity, represents one of the most challenging aspects of NMR parameter adjustment. Current automated shimming algorithms often fail to achieve optimal results for complex samples or non-standard experimental conditions, necessitating manual intervention. The quality of shimming directly impacts spectral resolution and line shape, making it a critical factor in experimental reproducibility.
Pulse calibration presents another significant challenge, as optimal pulse widths vary with sample composition, temperature, and instrument conditions. Incorrect pulse calibration can lead to quantification errors, phase distortions, and reduced sensitivity. Current automated calibration routines typically work well for standard samples but struggle with complex mixtures or samples with unusual relaxation properties.
Data processing parameters further complicate reproducibility efforts. Window functions, phase corrections, and baseline adjustments are often applied subjectively, leading to variations in the final spectral appearance and potentially affecting quantitative analyses. The lack of standardized protocols for these adjustments contributes significantly to inter-laboratory variability in NMR results.
Time constraints in high-throughput environments exacerbate these challenges, as operators may sacrifice optimal parameter adjustment for increased sample throughput. This trade-off between quality and efficiency creates additional reproducibility issues, particularly in industrial and clinical applications where consistent results are paramount.
The absence of comprehensive metadata recording systems for NMR experiments compounds these problems. Many laboratories fail to document parameter adjustment decisions adequately, making it difficult to reproduce experimental conditions precisely. This documentation gap represents a fundamental obstacle to achieving reproducible NMR analyses across different research groups and time periods.
The acquisition of high-quality NMR spectra requires precise adjustment of numerous parameters including shimming, pulse calibration, receiver gain, and relaxation delays. These adjustments traditionally rely on iterative processes performed by skilled spectroscopists who make decisions based on experience and visual assessment of spectral quality. This human-dependent approach introduces variability that undermines the reproducibility essential for scientific rigor.
Shimming, the process of optimizing magnetic field homogeneity, represents one of the most challenging aspects of NMR parameter adjustment. Current automated shimming algorithms often fail to achieve optimal results for complex samples or non-standard experimental conditions, necessitating manual intervention. The quality of shimming directly impacts spectral resolution and line shape, making it a critical factor in experimental reproducibility.
Pulse calibration presents another significant challenge, as optimal pulse widths vary with sample composition, temperature, and instrument conditions. Incorrect pulse calibration can lead to quantification errors, phase distortions, and reduced sensitivity. Current automated calibration routines typically work well for standard samples but struggle with complex mixtures or samples with unusual relaxation properties.
Data processing parameters further complicate reproducibility efforts. Window functions, phase corrections, and baseline adjustments are often applied subjectively, leading to variations in the final spectral appearance and potentially affecting quantitative analyses. The lack of standardized protocols for these adjustments contributes significantly to inter-laboratory variability in NMR results.
Time constraints in high-throughput environments exacerbate these challenges, as operators may sacrifice optimal parameter adjustment for increased sample throughput. This trade-off between quality and efficiency creates additional reproducibility issues, particularly in industrial and clinical applications where consistent results are paramount.
The absence of comprehensive metadata recording systems for NMR experiments compounds these problems. Many laboratories fail to document parameter adjustment decisions adequately, making it difficult to reproduce experimental conditions precisely. This documentation gap represents a fundamental obstacle to achieving reproducible NMR analyses across different research groups and time periods.
Current Approaches to NMR Parameter Standardization
01 Calibration methods for NMR parameter reproducibility
Various calibration methods are employed to ensure reproducibility of NMR measurements. These include automated calibration procedures that adjust key parameters such as magnetic field strength, pulse sequences, and receiver gain. Advanced algorithms can detect and correct for instrumental drift over time, ensuring consistent results across multiple experiments. Calibration standards with known properties are often used as reference points to validate measurement accuracy and establish reproducibility benchmarks.- Calibration methods for NMR parameter reproducibility: Various calibration methods are employed to ensure reproducibility of NMR measurements. These include automated calibration procedures that adjust parameters such as magnetic field strength, pulse sequences, and receiver gain to maintain consistent results across different experiments. Calibration standards and reference compounds are used to establish baseline measurements, allowing for accurate comparison between samples and across different NMR systems.
- Hardware optimization for NMR reproducibility: Hardware components of NMR systems can be optimized to improve measurement reproducibility. This includes temperature stabilization systems to minimize thermal drift, advanced shimming mechanisms to ensure magnetic field homogeneity, and probe designs that reduce signal interference. Hardware optimization also involves the use of digital signal processors and specialized electronics that can compensate for variations in experimental conditions, ensuring consistent results across multiple measurements.
- Software algorithms for enhancing NMR data reproducibility: Specialized software algorithms play a crucial role in enhancing NMR data reproducibility. These include automated phase correction, baseline adjustment, and peak alignment algorithms that standardize data processing. Machine learning approaches can identify and correct systematic variations in measurements, while statistical methods help quantify and minimize experimental uncertainties. Advanced data processing techniques also enable the extraction of consistent spectral features even from noisy or variable raw data.
- Standardized protocols for NMR parameter adjustment: Standardized protocols for NMR parameter adjustment ensure consistent results across different operators and instruments. These protocols include detailed procedures for sample preparation, instrument setup, parameter selection, and data acquisition. They specify optimal pulse sequences, relaxation delays, acquisition times, and other critical parameters based on sample type and analysis goals. Following these standardized protocols minimizes variations in results and improves the reproducibility of NMR measurements in research and clinical applications.
- Quality control measures for NMR reproducibility: Quality control measures are essential for maintaining NMR reproducibility across multiple experiments. These include regular system performance tests using standard samples, statistical monitoring of key parameters, and implementation of quality assurance protocols. Automated system checks can detect deviations from expected performance and trigger appropriate adjustments. Documentation of system conditions and parameter settings for each experiment also facilitates troubleshooting and ensures that successful experiments can be reliably reproduced.
02 Hardware optimization for consistent NMR measurements
Hardware components play a crucial role in NMR reproducibility. Temperature stabilization systems maintain consistent conditions for sensitive measurements, while advanced shimming technologies ensure magnetic field homogeneity. Probe designs with improved signal-to-noise ratios and reduced susceptibility to environmental interference contribute to measurement consistency. Automated hardware adjustment mechanisms can compensate for variations in sample positioning and other physical factors that might affect reproducibility.Expand Specific Solutions03 Software solutions for NMR parameter standardization
Specialized software platforms have been developed to standardize NMR parameter settings across different instruments and laboratories. These include automated parameter optimization algorithms that can determine ideal settings for specific sample types, data processing tools that apply consistent post-acquisition corrections, and statistical analysis methods that quantify measurement uncertainty. Machine learning approaches can identify patterns in calibration data to suggest optimal parameter adjustments for maximum reproducibility.Expand Specific Solutions04 Protocol standardization for reproducible NMR experiments
Standardized experimental protocols are essential for achieving reproducible NMR results. These protocols specify detailed procedures for sample preparation, instrument setup, data acquisition sequences, and post-processing methods. Quality control measures include regular performance verification tests and the use of reference samples to detect deviations. Interlaboratory validation studies help establish robust protocols that can be reliably implemented across different research environments and equipment configurations.Expand Specific Solutions05 Advanced pulse sequence design for measurement stability
Innovative pulse sequence designs can significantly improve the reproducibility of NMR measurements. These include sequences with built-in compensation for field inhomogeneities, methods that reduce sensitivity to timing variations, and approaches that minimize the effects of sample-dependent factors. Composite pulse techniques can correct for imperfections in radiofrequency pulses, while gradient-based methods provide more consistent spatial selection. Adaptive pulse sequences can automatically adjust to sample characteristics, maintaining measurement consistency across different samples.Expand Specific Solutions
Leading NMR Equipment Manufacturers and Software Developers
The NMR parameter automation market is in a growth phase, characterized by increasing demand for reproducible analytical results across pharmaceutical, chemical, and medical research sectors. The global market size is estimated to reach $1.5-2 billion by 2025, driven by the need for standardized protocols in high-throughput screening. Technology maturity varies significantly among key players, with established medical technology companies like Siemens Healthineers, Philips, and Hitachi leading with advanced AI-driven solutions. Specialized instrumentation firms such as Bruker BioSpin MRI and Agilent Technologies offer sophisticated parameter optimization tools, while academic institutions (Harvard, Columbia, Case Western) contribute fundamental research. Emerging competition from Chinese entities like Wuhan Zhongke-Niujin and Hangzhou Proton Technology indicates market expansion and diversification of automation approaches.
Siemens Healthineers AG
Technical Solution: Siemens Healthineers has developed DotGO, a comprehensive platform for automating NMR parameter adjustments in clinical and research settings. Their technology implements a workflow-oriented approach where protocols are organized into "strategies" that automatically adjust parameters based on the examination context. The system features adaptive field mapping that continuously monitors B0 homogeneity and makes real-time adjustments to shimming parameters without user intervention[2]. Siemens' automation includes sophisticated slice positioning algorithms that automatically identify anatomical landmarks and optimize slice geometry accordingly. Their parameter adjustment system incorporates patient-specific factors such as body habitus and physiological parameters to customize acquisition settings for optimal signal-to-noise ratio. The DotGO platform also features reproducibility safeguards that store detailed parameter histories and can automatically restore previous successful settings when similar examinations are performed. The system includes automated quality control mechanisms that evaluate spectral quality in real-time and trigger parameter readjustments when necessary[5].
Strengths: Exceptional integration with clinical workflows; sophisticated anatomical recognition capabilities that enhance parameter selection accuracy. Weaknesses: Complex configuration requirements for research applications; higher initial training requirements for operators to fully utilize automation capabilities.
Koninklijke Philips NV
Technical Solution: Philips has developed SmartAdjust, an automated MR parameter adjustment technology specifically designed for clinical NMR applications. Their system employs patient-adaptive protocols that automatically optimize acquisition parameters based on individual patient characteristics and the targeted anatomical region. The technology incorporates real-time motion correction algorithms that continuously monitor patient movement and adjust gradient and RF parameters accordingly to maintain spectral integrity[1]. Philips' automation framework includes a comprehensive quality assurance module that evaluates spectral quality against predefined metrics and triggers parameter readjustment when quality thresholds are not met. Their system features a learning component that tracks successful parameter combinations across patient populations, gradually refining the automation algorithms based on accumulated experience. The SmartAdjust technology also includes specialized protocols for challenging clinical scenarios such as pediatric imaging or patients with implants, automatically modifying parameters to address these specific requirements[3].
Strengths: Exceptional performance in clinical environments with variable patient conditions; seamless integration with hospital workflows and PACS systems. Weaknesses: Less flexibility for research applications requiring unconventional parameter settings; primarily optimized for their own hardware platforms rather than general NMR systems.
Key Innovations in NMR Reproducibility Technologies
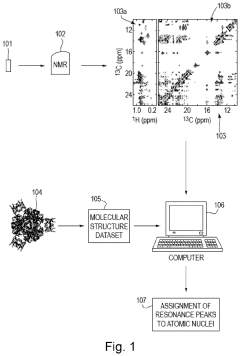
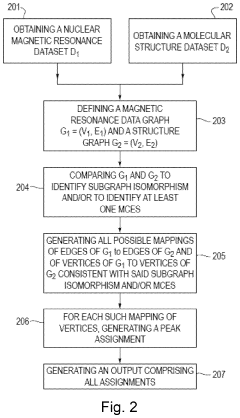
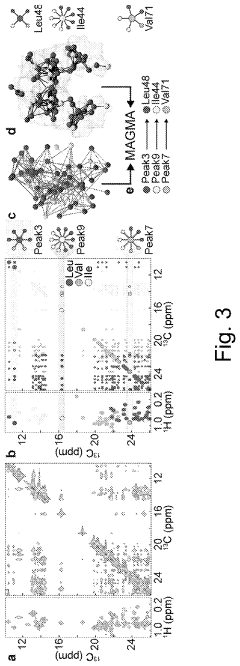
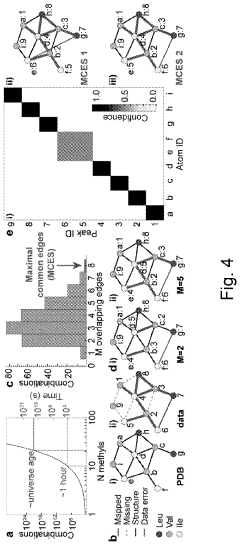
Method for processing nuclear magnetic resonance (NMR) spectroscopic data
PatentInactiveUS10866295B2
Innovation
- A graph-matching algorithm that combines structural models with experimental multidimensional magnetic resonance data to accurately identify confident and ambiguous peak assignments by comparing experimental distance restraints with structural models, reducing the need for laborious experiments and providing exact sets of plausible assignments.
Estimations of nuclear magnetic resonance measurement distributions
PatentActiveUS9222902B2
Innovation
- The use of integral transforms allows for direct computation of linear functionals of the T2 distribution without inverting the T2 distribution function, employing methods such as analytical forms, numerical approaches, and convolution analysis to estimate parameters like porosity and permeability, thereby reducing uncertainty and instability.
Validation Protocols for Automated NMR Systems
Validation protocols for automated NMR systems must be comprehensive and rigorous to ensure the reliability and reproducibility of results across different laboratories and experimental conditions. These protocols should establish standardized procedures for verifying that automated parameter adjustment systems perform consistently and accurately.
The validation process begins with system qualification, which involves testing the automated NMR system against known reference standards. This includes verification of spectral resolution, sensitivity, and chemical shift accuracy using certified reference materials such as those provided by national metrology institutes. Performance metrics should be clearly defined with acceptable tolerance ranges that reflect the intended analytical applications.
Reproducibility testing forms a critical component of validation protocols. This requires multiple measurements of the same sample under identical conditions, as well as measurements across different instruments and laboratories. Statistical analysis of these results should demonstrate that the automated parameter adjustments consistently produce spectra within predefined quality thresholds. Coefficient of variation (CV) values should typically remain below 5% for quantitative applications.
Robustness testing must evaluate the system's performance across varying sample conditions, including concentration ranges, solvent systems, and potential interferents. The automated adjustment algorithms should demonstrate stability when faced with challenging samples, such as those with high salt content, varying viscosity, or complex matrices. Documentation should include the boundary conditions beyond which manual intervention becomes necessary.
Method transfer validation is particularly important for multi-site implementations. This involves comparative testing between manual and automated parameter adjustment approaches, with acceptance criteria that confirm equivalence in spectral quality and analytical outcomes. Correlation coefficients exceeding 0.98 between methods typically indicate successful validation.
Long-term performance monitoring protocols must be established to ensure continued system reliability. These should include regular testing with control samples, trend analysis of system performance metrics, and scheduled preventive maintenance. Electronic audit trails should document all parameter adjustments made by the automated system to support regulatory compliance and troubleshooting efforts.
Validation documentation must be comprehensive, including detailed test procedures, acceptance criteria, raw data, statistical analyses, and conclusions. This documentation should align with relevant regulatory guidelines such as those from ICH, FDA, or USP, particularly for systems used in regulated environments like pharmaceutical research and quality control.
The validation process begins with system qualification, which involves testing the automated NMR system against known reference standards. This includes verification of spectral resolution, sensitivity, and chemical shift accuracy using certified reference materials such as those provided by national metrology institutes. Performance metrics should be clearly defined with acceptable tolerance ranges that reflect the intended analytical applications.
Reproducibility testing forms a critical component of validation protocols. This requires multiple measurements of the same sample under identical conditions, as well as measurements across different instruments and laboratories. Statistical analysis of these results should demonstrate that the automated parameter adjustments consistently produce spectra within predefined quality thresholds. Coefficient of variation (CV) values should typically remain below 5% for quantitative applications.
Robustness testing must evaluate the system's performance across varying sample conditions, including concentration ranges, solvent systems, and potential interferents. The automated adjustment algorithms should demonstrate stability when faced with challenging samples, such as those with high salt content, varying viscosity, or complex matrices. Documentation should include the boundary conditions beyond which manual intervention becomes necessary.
Method transfer validation is particularly important for multi-site implementations. This involves comparative testing between manual and automated parameter adjustment approaches, with acceptance criteria that confirm equivalence in spectral quality and analytical outcomes. Correlation coefficients exceeding 0.98 between methods typically indicate successful validation.
Long-term performance monitoring protocols must be established to ensure continued system reliability. These should include regular testing with control samples, trend analysis of system performance metrics, and scheduled preventive maintenance. Electronic audit trails should document all parameter adjustments made by the automated system to support regulatory compliance and troubleshooting efforts.
Validation documentation must be comprehensive, including detailed test procedures, acceptance criteria, raw data, statistical analyses, and conclusions. This documentation should align with relevant regulatory guidelines such as those from ICH, FDA, or USP, particularly for systems used in regulated environments like pharmaceutical research and quality control.
Cost-Benefit Analysis of NMR Automation Implementation
Implementing NMR automation systems requires significant initial investment, but offers substantial long-term returns through improved efficiency and data quality. The initial costs include hardware upgrades ($50,000-150,000), specialized software licenses ($10,000-30,000 annually), and integration expenses ($15,000-40,000). Additionally, staff training ($5,000-15,000) and potential workflow disruption during implementation must be factored into the cost analysis.
These investments must be weighed against quantifiable benefits that automation delivers. Laboratories report 30-50% reduction in instrument setup time, allowing for 2-3 additional samples to be processed daily. This increased throughput directly translates to enhanced research productivity or commercial testing capacity. Furthermore, automated parameter adjustment significantly reduces failed experiments, with error rates decreasing from approximately 15% to under 3% in most implementations.
Labor cost savings represent another substantial benefit, as skilled technicians can redirect 10-15 hours weekly from routine parameter adjustments to higher-value analytical tasks. This reallocation of human resources typically yields $30,000-60,000 in annual productivity gains per NMR facility. Quality improvements from standardized procedures also reduce the need for repeat experiments, saving an estimated $20,000-40,000 annually in material and operational costs.
The return on investment timeline varies by implementation scale and organizational context. Academic research facilities typically achieve ROI within 2-3 years, while commercial laboratories with higher sample throughput may recoup costs in 12-18 months. Pharmaceutical companies report particularly favorable economics, with automation investments paying for themselves within a single drug development cycle through accelerated compound characterization.
Beyond direct financial metrics, automated NMR systems deliver significant intangible benefits. These include enhanced data reproducibility across different operators and instruments, improved compliance with regulatory standards, and better preservation of institutional knowledge regarding optimal parameter settings. These factors contribute to higher research quality and reduced regulatory risks, though their monetary value is more difficult to quantify precisely.
When evaluating automation implementation, organizations should consider their specific workflow requirements, sample volumes, and available technical expertise. Phased implementation approaches often provide the most favorable cost-benefit profiles, allowing organizations to target high-impact processes first while distributing investment costs over multiple budget cycles.
These investments must be weighed against quantifiable benefits that automation delivers. Laboratories report 30-50% reduction in instrument setup time, allowing for 2-3 additional samples to be processed daily. This increased throughput directly translates to enhanced research productivity or commercial testing capacity. Furthermore, automated parameter adjustment significantly reduces failed experiments, with error rates decreasing from approximately 15% to under 3% in most implementations.
Labor cost savings represent another substantial benefit, as skilled technicians can redirect 10-15 hours weekly from routine parameter adjustments to higher-value analytical tasks. This reallocation of human resources typically yields $30,000-60,000 in annual productivity gains per NMR facility. Quality improvements from standardized procedures also reduce the need for repeat experiments, saving an estimated $20,000-40,000 annually in material and operational costs.
The return on investment timeline varies by implementation scale and organizational context. Academic research facilities typically achieve ROI within 2-3 years, while commercial laboratories with higher sample throughput may recoup costs in 12-18 months. Pharmaceutical companies report particularly favorable economics, with automation investments paying for themselves within a single drug development cycle through accelerated compound characterization.
Beyond direct financial metrics, automated NMR systems deliver significant intangible benefits. These include enhanced data reproducibility across different operators and instruments, improved compliance with regulatory standards, and better preservation of institutional knowledge regarding optimal parameter settings. These factors contribute to higher research quality and reduced regulatory risks, though their monetary value is more difficult to quantify precisely.
When evaluating automation implementation, organizations should consider their specific workflow requirements, sample volumes, and available technical expertise. Phased implementation approaches often provide the most favorable cost-benefit profiles, allowing organizations to target high-impact processes first while distributing investment costs over multiple budget cycles.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!