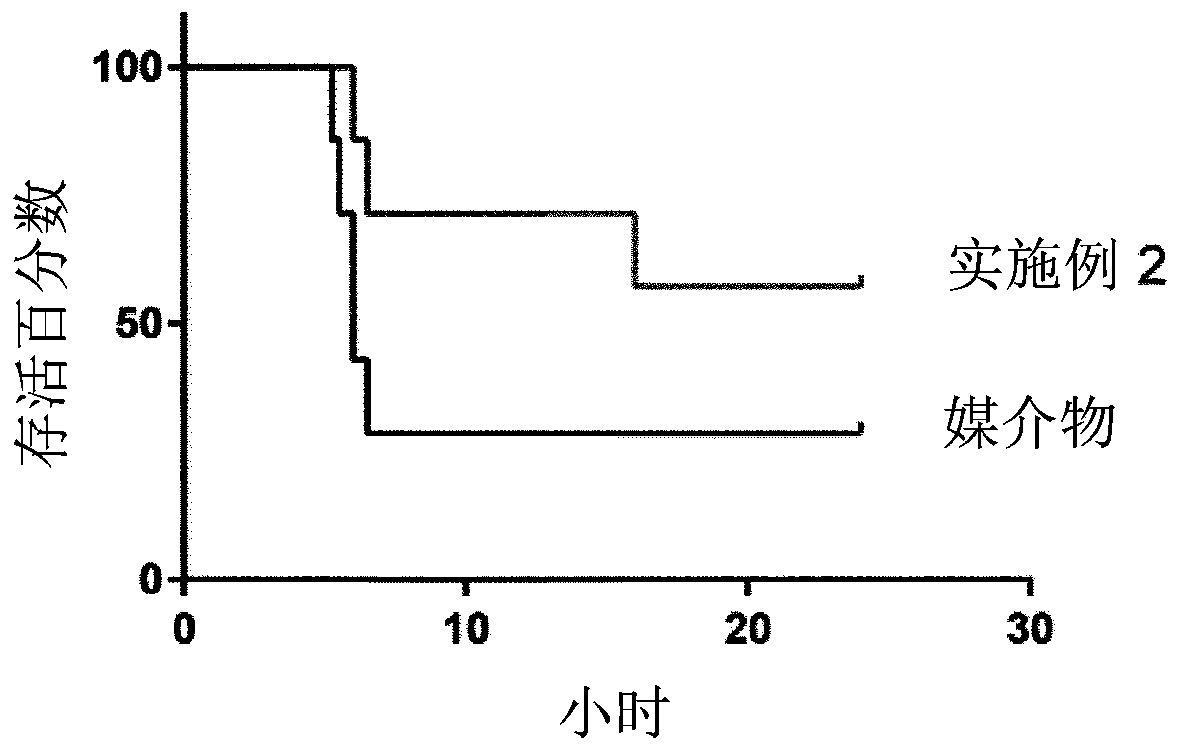
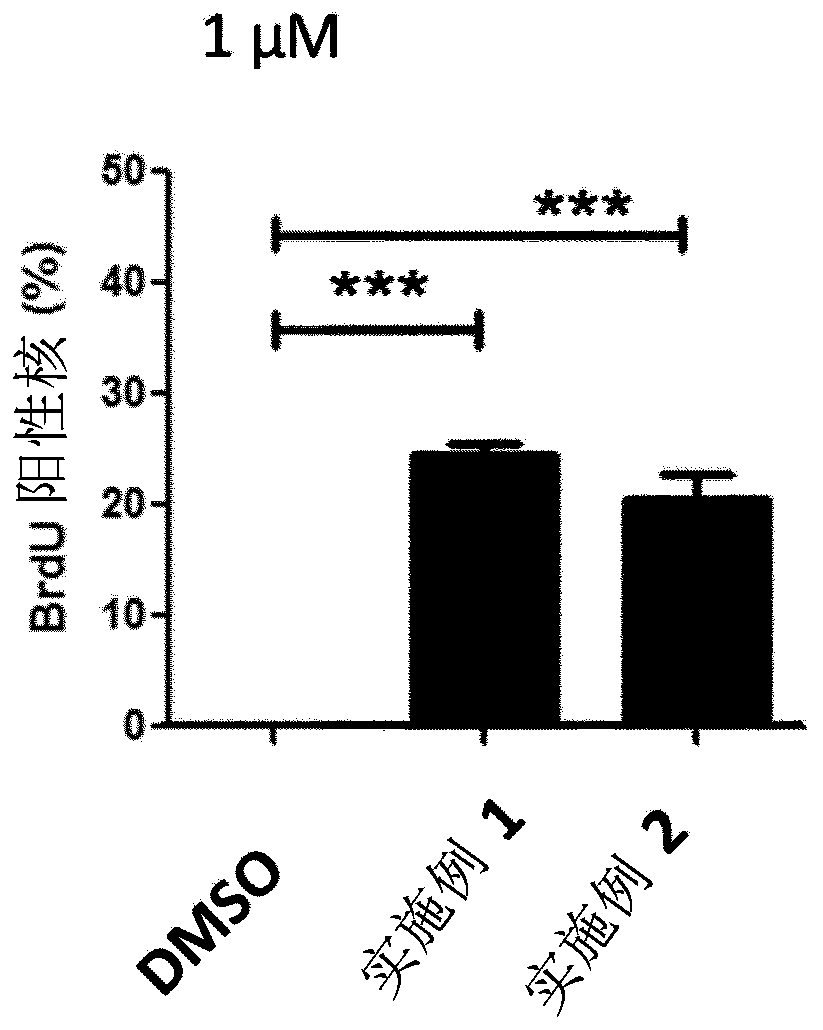
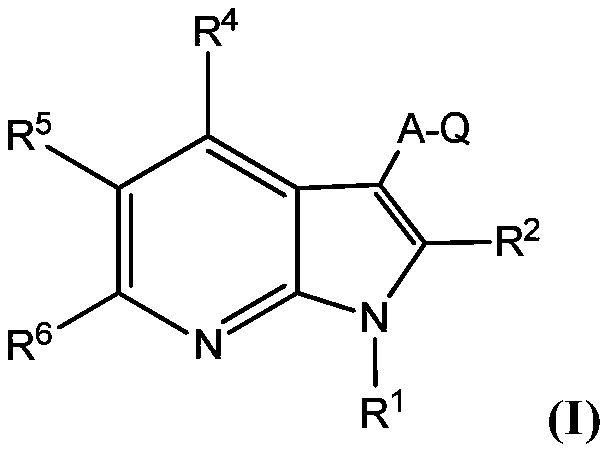
Protein kinase inhibitors for promoting liver regeneration or reducing or preventing hepatocyte death
一种蛋白激酶、抑制剂的技术,应用在含有效成分的医用配制品、药物组合、有机化学等方向,能够解决再生能力受损、不足以补偿肝细胞和器官功能、丧失等问题
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0386] Example 1: Propane-1-sulfonic acid [3-(5-chloro-1H-pyrrolo[2,3-b]pyridine-3-carbonyl)-2,4-difluoro-phenyl}amide, such as WO The preparation is disclosed in 2007 / 002433:
[0387]
Embodiment 2
[0388] Example 2: Propane-1-sulfonic acid {3-[5-(4-chloro-phenyl)-1H-pyrrolo[2,3-b]pyridine-3-carbonyl]-2,4-difluoro- Phenyl}-amides, prepared as disclosed in WO 2007 / 002433:
[0389]
[0390] For the preparation of compounds of the invention, the general procedures (GP) aa to ae are used:
[0391] General process aa:
[0392] Oxalyl chloride (1.1 equiv) was added to a suspension of the carboxylic acid (1.0 equiv) in anhydrous DCM (0.5m). A few drops of DMF were added and the resulting mixture was stirred at room temperature until gas formation was complete. Excess MeOH was added to the solution, and the solvent was evaporated under reduced pressure. The residue was dried in vacuo and the product was used without further purification.
[0393] General process ab:
[0394] Pd / C (0.1 equiv) was added to a solution of nitrobenzene (1.0 equiv) in EtOH (0.2m). The suspension was washed with H 2 The gas was degassed, and the reaction was stirred at room temperature to ...
Embodiment 3
[0437] Example 3: The above N-(2,4-difluoro-3-(5-(4-methoxyphenyl)-1H-pyrrolo[2,3-b]pyridine-3-carbonyl base) phenyl) propane-1-sulfonamide)
[0438]
[0439] Process: follow GP ad The title compound was obtained.
[0440] Yield: 22.7 mg, 47 μmol, 36% (white solid)
[0441] TLC: DCM / MeOH 5%
[0442] 1 H NMR (DMSO-d 6 ,400MHz,ppm):δ12.96(s,1H),9.78(s,1H),8.67(d,J=1.7Hz,1H),8.57(s,1H),8.21(s,1H),7.68( d,J=8.5Hz,2H),7.59(dd,J=14.8,8.9Hz,1H),7.28(t,J=8.5Hz,1H),7.08(d,J=8.6Hz,2H),3.82( s,3H),3.19–3.06(m,2H),1.74(dq,J=14.7,7.2Hz,2H),0.96(t,J=7.4Hz,3H); 13 C NMR (DMSO-d 6 ,101Hz,ppm): δ180.5,159.0,156.0(dd,J=246.6,6.9Hz),152.4(dd,J=258.5,8.9Hz),148.5,143.7,138.4,131.3,130.4,128.7(d,J= 8.7Hz), 128.2, 126.4, 121.9(dd, J=13.1, 3.6Hz), 118.2(dd, J=25.0, 23.0Hz), 117.5, 115.6, 114.6, 112.2(dd, J=22.5, 3.3Hz), 55.2, 53.5, 16.74, 12.5. TLC-MS: C 24 h 21 f 2 N 3 o 4 S([M-H] - ) calculated value of m / z: 484.1, found value: 484.2.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap