Drug composite containing calcium 2,5-dihydroxysulfonate and ADP receptor antagonist
A technology of calcium hydroxybenzenesulfonate and receptor antagonists, applied in the field of new pharmaceutical compositions, can solve the problems of reducing patient compliance and rebound of cardiovascular events, and achieve the effect of reducing bleeding rate and inhibiting platelet aggregation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
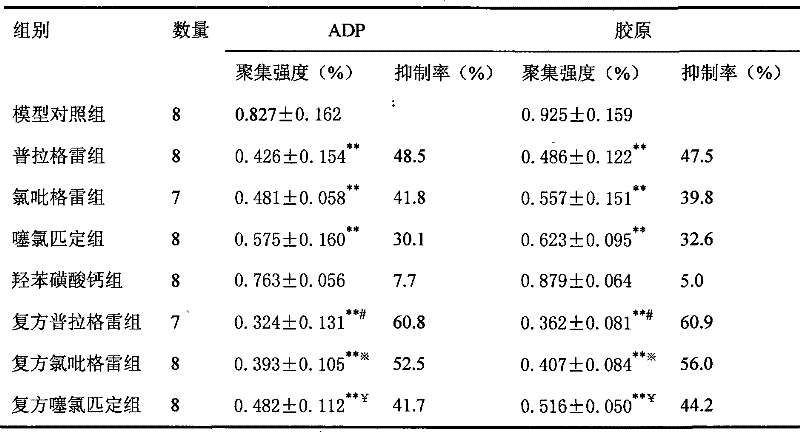
Image
Examples
Embodiment 1
[0013] Embodiment 1 common tablet
[0014] Calcium 2,5-dihydroxybenzenesulfonate 200g
[0015] Prasugrel Maleate 2g
[0016] Microcrystalline Cellulose 500g
[0017] Lactose 40g
[0018] 10% starch slurry appropriate amount
[0019] Magnesium stearate 8g
[0020] Preparation process: Weigh the prescribed amount of calcium 2,5-dihydroxybenzenesulfonate, prasugrel maleate, microcrystalline cellulose, and lactose and mix evenly. In addition, add an appropriate amount of 10% starch slurry to the mixed powder, mix evenly, make soft material, pass through a 18-mesh nylon sieve to make wet granules, and dry at about 60°C. The moisture content of the dry granules should be controlled below 1.5%. Sieve through a 20-mesh sieve, mix with magnesium stearate, and press into tablets.
Embodiment 2
[0021] Embodiment 2 common tablet
[0022] Calcium 2,5-dihydroxybenzenesulfonate 50g
[0023] Clopidogrel Bisulfate 2.5g
[0024] Starch 140g
[0025] Dextrin 120g
[0026] 50% ethanol appropriate amount
[0027] Magnesium Stearate 1.0g
[0028] Preparation process: Weigh the prescribed amount of calcium 2,5-dihydroxybenzenesulfonate, clopidogrel sulfate, starch, and dextrin and mix evenly. In addition, add an appropriate amount of 50% ethanol to the mixed powder, mix evenly, make soft material, pass through a 18-mesh nylon sieve to make wet granules, and dry at about 60°C. The moisture content of the dry granules should be controlled below 1.5%. Sieve through a 20-mesh sieve, mix with magnesium stearate, and press into tablets.
Embodiment 3
[0029] Embodiment 3 common tablet
[0030] Calcium 2,5-dihydroxybenzenesulfonate 200g
[0031] Ticlopidine hydrochloride 50g
[0032] Microcrystalline Cellulose 500g
[0033] Lactose 40g
[0034] 10% starch slurry appropriate amount
[0035] Magnesium stearate 8g
[0036] Preparation process: Weigh the prescribed amount of calcium 2,5-dihydroxybenzenesulfonate, ticlopidine hydrochloride, microcrystalline cellulose, and lactose and mix evenly. In addition, add an appropriate amount of 10% starch slurry to the mixed powder, mix evenly, make soft material, pass through a 18-mesh nylon sieve to make wet granules, and dry at about 60°C. The moisture content of the dry granules should be controlled below 1.5%. Sieve through a 20-mesh sieve, mix with magnesium stearate, and press into tablets.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap