Process for the preparation of aryl alcohols and heteroaryl alcohols
A kind of aryl and alkyl technology, applied in the field of preparation of aryl alcohol and heteroaryl alcohol, can solve the problems such as yield loss and complexity
Active Publication Date: 2022-01-18
MERCK PATENT GMBH
View PDF4 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0009] However, these syntheses are often complex and / or performed in multiple reaction steps, which leads to loss of yield
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 11
[0098] Embodiment 1.1, the preparation of 2-oxaborolane and 1,2-oxaborolane
Embodiment 11
[0099] Example 1.1.2-butoxy-1,2-oxaborolane
[0100]
[0101] The title compound was prepared according to WO 2009 / 046098 A1 by hydroboration and transesterification with butanol.
Embodiment 12
[0102] Example 1.2.2-Ethoxy-1,2-oxaborolane
[0103]
[0104] The starting material 2-butoxy-1,2-oxaborolane (1 g) was stirred in 10 ml EtOH at 60° C. for 120 minutes and after 4-8 hours was subjected to vacuum at 100 mbar distilled. First, EtOH and BuOH are separated off, and then the product 2-ethoxy-1,2-oxaborolane is distilled off.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
Login to View More
Abstract
The present invention relates to a process for the preparation of aryl and heteroaryl alcohols by coupling aromatic halogen or pseudohalogen compounds to cyclic boronic esters.
Description
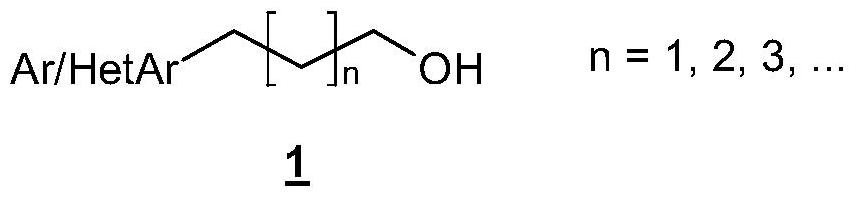
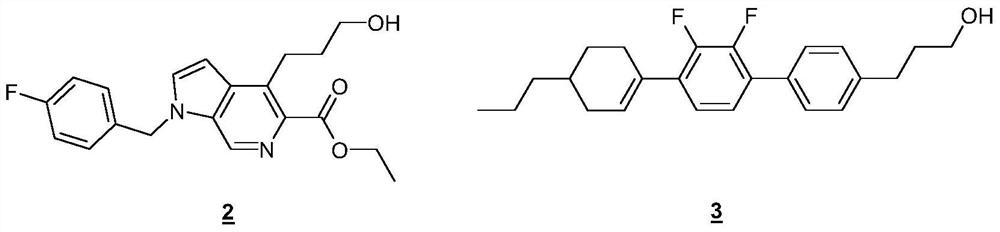
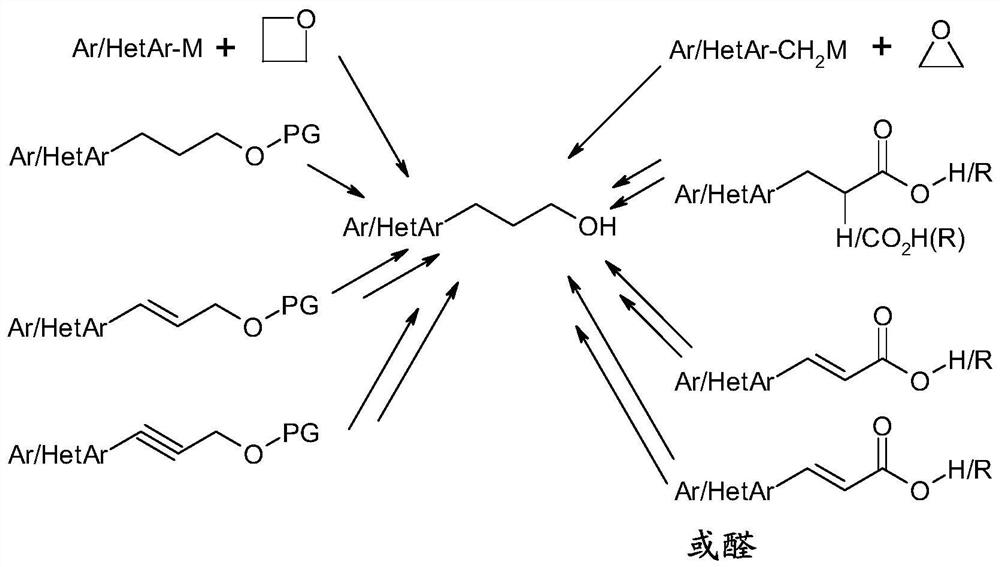
technical field [0001] The present invention relates to a process for the preparation of aryl and heteroaryl alcohols by coupling aromatic halogen or pseudohalogen compounds to cyclic boronic esters. Background technique [0002] Types of 1 Aryl and Heteroaryl Alcohols [0003] [0004] are known, inter alia, as pharmaceutically active compounds or precursors thereof, or also as performance chemicals, such as liquid crystals or precursors thereof. compound 2 and 3 Can be referenced by way of example. [0005] [0006] Efficient and feasible access to struct types 1 Necessary for economical preparation. So far, type 1 The synthesis of derivatives has mainly proceeded by reaction of precursor structures, usually in a reductive manner, as outlined in Scheme 1 by the example of C3 alcohols. [0007] [0008] Scheme 1. Ar = aryl, HetAr = heteroaryl, M = metal, PG = protecting group, R = alkyl. [0009] However, these syntheses are often complex and / or performed ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Patents(China)
IPC IPC(8): C07D311/14C07C39/11C07C33/46C07C33/26C07C33/20C07C33/24C07C29/36
CPCC07C29/36C07C33/20C07C33/24C07C33/26C07C33/46C07C39/11C07D311/14B01J23/44C07C29/32C07F7/1804C07C33/50C07C33/22C07C33/18
Inventor A·巴特M·赫尔姆雷驰
Owner MERCK PATENT GMBH