Novel compounds for the treatment, alleviation or prevention of disorders associated with tau aggregates
A compound and disease technology, applied in the direction of drug combination, organic chemistry, nervous system diseases, etc., to achieve the effect of reducing the spread of pathology
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
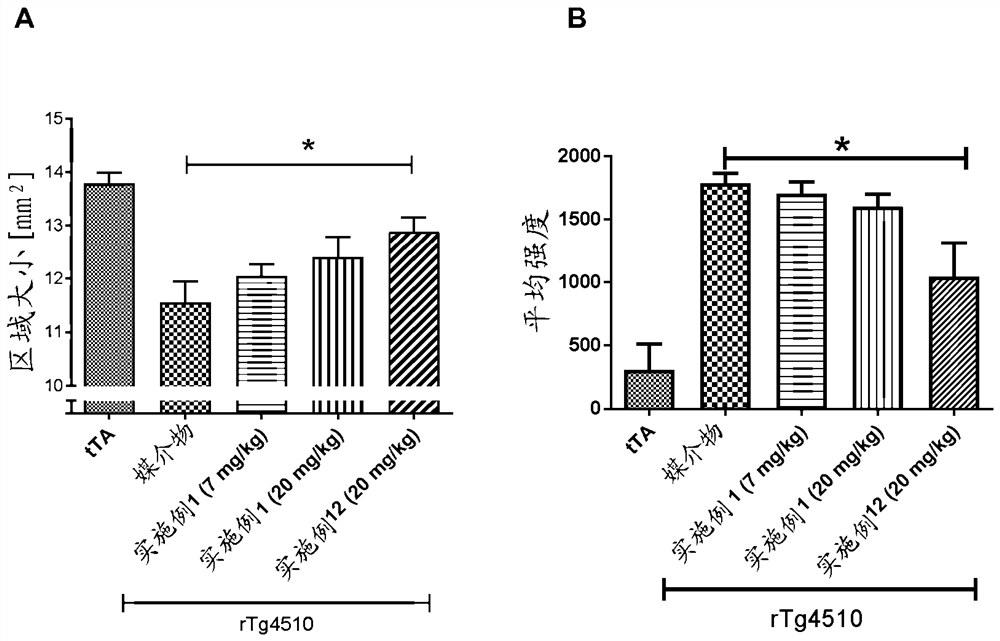
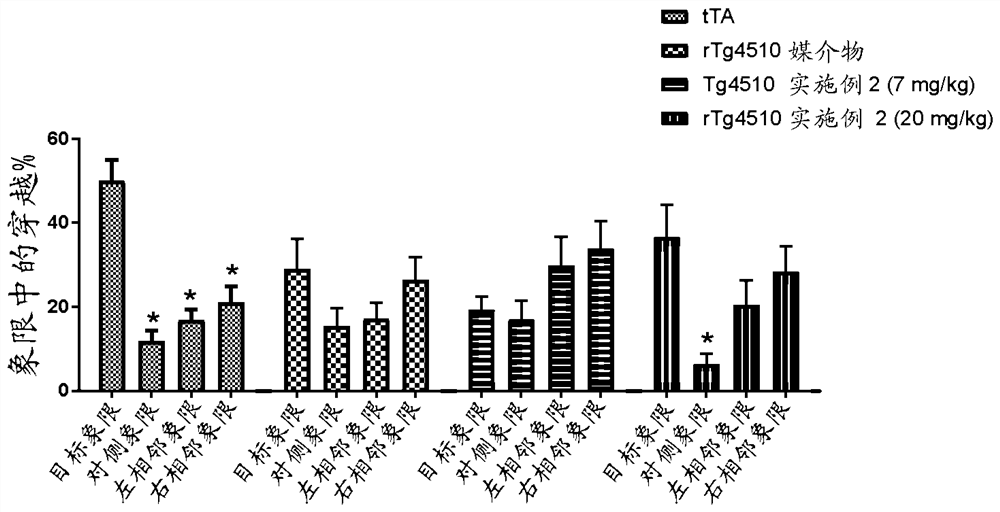
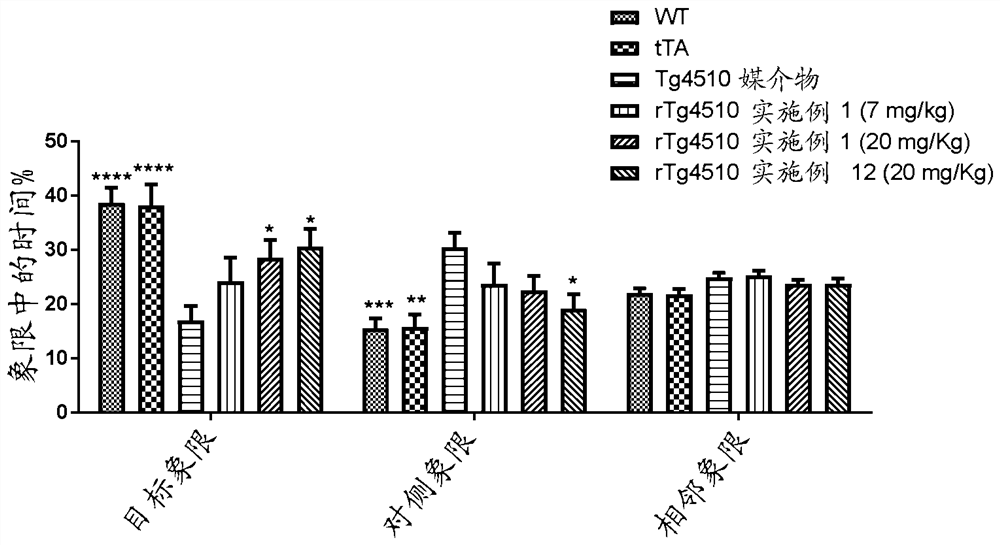
Image
Examples
preparation Embodiment 1
[0164]
[0165] Step A
[0166] To a suspension of commercially available 2,6-dibromopyridine (10 g, 42.2 mmol) in ethanol (50 mL) was added commercially available N - Methylhydrazine (11.11 mL, 211 mmol). The mixture was heated at 80°C (reaction mixture temperature) for 48 hours. The reaction mixture was concentrated to dryness and the residue was purified by chromatography on silica using a Biotage Isolera One purification system using an ethyl acetate / n-heptane gradient (15 / 75 -> 35 / 65) to afford a reddish oil The title compound of , which became a solid upon standing at room temperature (7.6 g, 89 %).
[0167]
[0168] Step B
[0169] 4-(methylamino)cyclohexanone (2,2-dimethyl-1,3-propanediol) hydrochloride (4-(methylamino)cyclohexanone-2,2-dimethyltrimethylene ketal hydrochloride) (3.7 g, 14.8 mmol) and a suspension of the title compound from Step A above (3 g, 14.8 mmol) in dioxane (30 mL) was placed in an ice bath. Concentrated sulfuric acid (3 mL) was sl...
preparation Embodiment 2
[0180]
[0181] Step A
[0182] The chiral starting material (2 g, 5.67 mmol, chemical purity: 99.2%; chiral purity: 99.2% ee) (prepared in a similar manner to that reported in WO2011 / 128455 Preparation Example 337 by using DMF as solvent instead of MeOH ) was dissolved in dichloromethane (80 mL) and 40 mL of a 4 M solution of hydrogen chloride in 1,4-dioxane was added. The reaction mixture was stirred at room temperature for 18 hours and the solvent was removed under reduced pressure. The residue was dissolved in dichloromethane (250 mL) and water (120 mL) and the pH of the aqueous phase was adjusted to pH~12 by adding sodium hydroxide pellets. The organic phase was separated and the aqueous phase was extracted with dichloromethane (120 mL). The combined organic phases were washed with Na 2 SO 4 Dry, filter and remove solvent under reduced pressure to afford the title compound as a colorless oil (1.46 g, 98%; TLC matches racemic material).
[0183]
[0184] Ste...
preparation Embodiment 3
[0188]
[0189] Step A
[0190] Commercially available 1-isopropylpiperidin-4-one (500 g, 3.541 mol), hydroxylamine hydrochloride (369.1 g, 5.315 mol) and potassium carbonate (1221 g, 8.855 mol) were dissolved in ethanol (2.5 L) in Heat at 80° C. for 3 hours in an oil bath. After completion of the reaction was determined by TLC, the reaction mixture was cooled to 25 °C and the solid was filtered, washed with water (5 L) and diethyl ether (2.5 L). The compound was dried under line vacuum for 24 hours to obtain 1-isopropylpiperidin-4-one oxime (525 g, 95%) as a white solid.
[0191]
[0192] Step B
[0193] To a stirred solution of the title compound from Step A above (500 g, 3.201 mol) in DMF (5 L) was added NaH 60% in mineral oil (192 g, 4.801 mol) portionwise at 0 °C. After the addition, the reaction mixture was stirred at 25°C for 1 hour, and then the reaction mixture was cooled to 0°C. Commercially available 2-bromo-6-fluoropyridine (563 g, 3.201 mol) dissolve...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap