Compositions and methods for detection of epstein barr virus (EBV)
a technology of epstein barr virus and composition method, applied in the field of in vitro diagnostics, can solve the problems of complex actual diversity characterization of the virus, particular risk to immunosuppressed transplant patients, and a threat to immunosuppressed patients
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
Design of Primers and Probes for Detection of EBV by Real-Time PCR
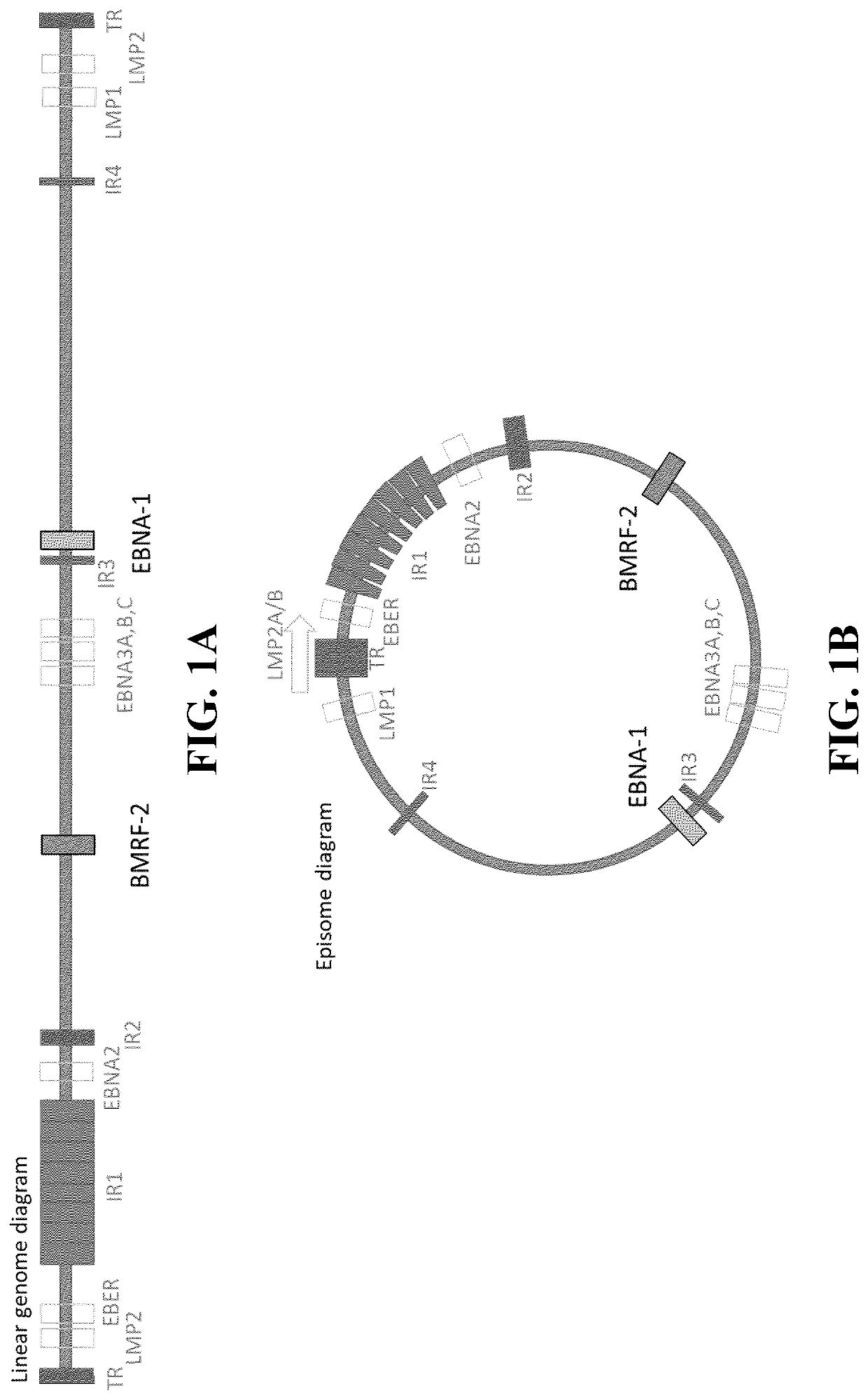
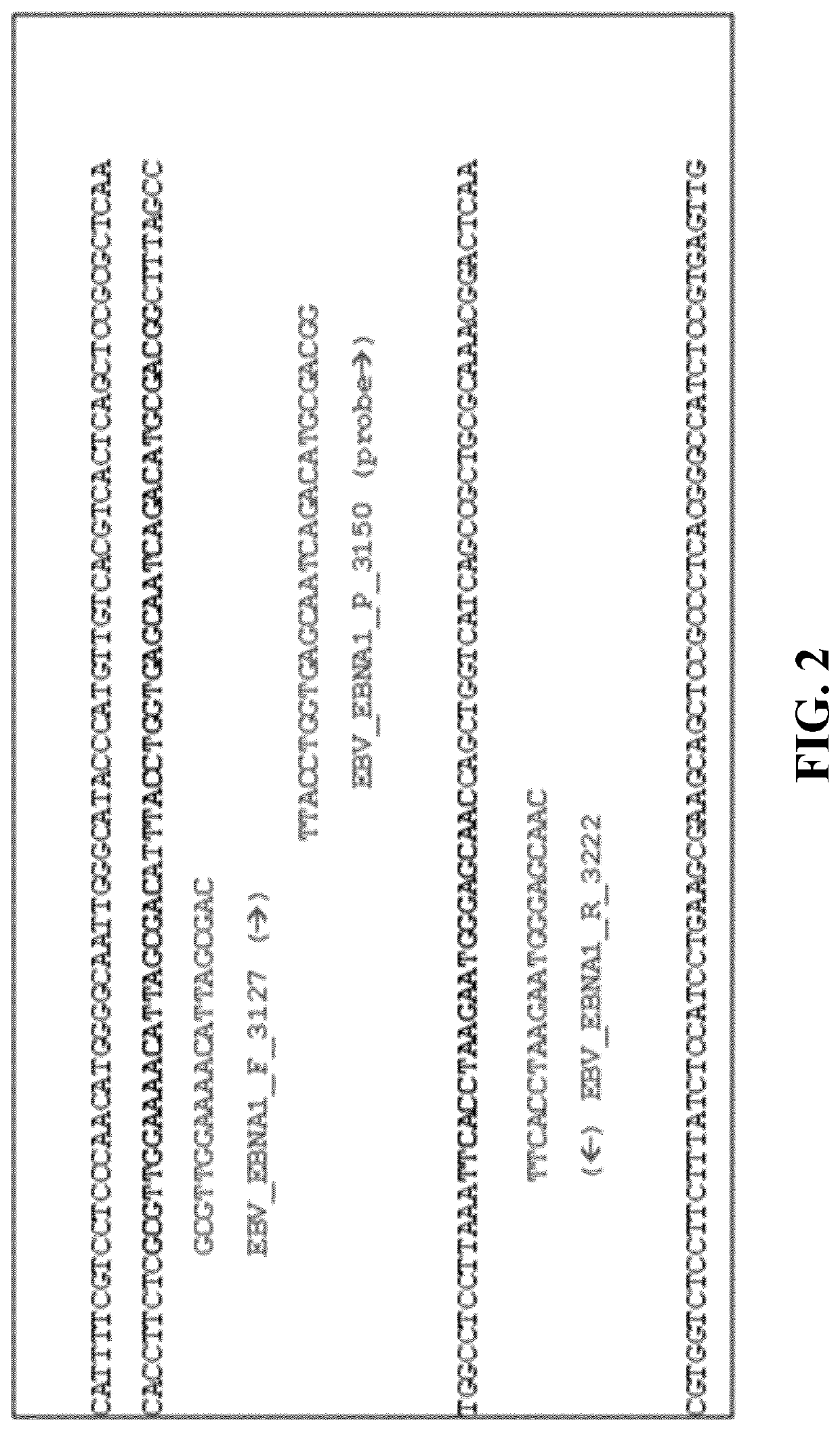
[0080]The EBV nucleic acid test was designed with two targets in mind. The two targets chosen were BMRF2 and EBNA1, and are shown in FIG. 1. These candidates could be used individually or together duplexed in a dual target assay. If used as a dual target assay, two sets of primers and probe are employed (each set detecting either EBNA1 or BMRF2). As shown in Table 1, above, the primers for BMRF2 target have the nucleic acid sequence SEQ ID NOs:1 and 3, and the probe for BMRF2 target has the nucleic acid sequence of SEQ ID NO:2. Also, as shown in Table 1, above, the primers for EBNA1 target have the nucleic acid sequence SEQ ID NOs:4 and 6, and the probe for EBNA1 target has the nucleic acid sequence of SEQ ID NO:5. The amplicon generated by the primers targeting BMRF2 is a 96 base pair long amplicon, and is shown in FIG. 2 (along with locations where the primers and probes overlap the amplicon). The amplicon generated...
example 2
EBV Primers and Probes Detect BMRF2 and EBNA-1 of EBV in Real-Time PCR Assay
[0082]The EBV nucleic acid test was tested using primers / probes for detecting BMRF2 (SEQ ID NOs:1-3) and EBNA1 (SEQ ID NOs:4-6). A full process of the EBV assay was run. Four types of EBV-containing samples were employed: (1) EBV-infected Raji cells extracts spiked into EBV-negative plasma; (2) extracted EBV DNA (extracted from a B95-8 cell line from Advanced Biotechnologies (Catalog No. 17-926-500)); (3) Qnostics EBV Analytic Panel (EBV1604009C); and (4) the 1st WHO International Standard for EBV. Reagents used include cobas® 6800 / 8800 generic PCR Master Mix, with the profile and conditions for use with the cobas® 6800 / 8800, and using TaqMan® amplification and detection technology. The final concentration of oligonucleotides in the master mix was 0.3 μM for primers and 0.1 μM for probes. The cobas® 6800 / 8800 PCR Profile employed is depicted in Table 2, below:
TABLE 2cobas ® 6800 / 8800 PCR ProfileTargetHold ti...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Acidity | aaaaa | aaaaa |
| Biological properties | aaaaa | aaaaa |
| Nucleic acid sequence | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap